- Home
- »
- Clinical Diagnostics
- »
-
Latin America Clinical Laboratory Services Market ReportGVR Report cover
![Latin America Clinical Laboratory Services Market Size, Share & Trends Report]()
Latin America Clinical Laboratory Services Market (2023 - 2030) Size, Share & Trends Analysis Report By Test Type (Human & Tumor Genetics), By Service Provider (Hospital-based Laboratories), By Application, By Country, And Segment Forecasts
- Report ID: GVR-2-68038-093-4
- Number of Report Pages: 125
- Format: PDF
- Historical Range: 2018 - 2020
- Forecast Period: 2023 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Report Overview
The Latin America clinical laboratory services market size was estimated at USD 12.68 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 3.6% from 2023- 2030. The increasing burden of chronic diseases, technological advancements in the field of clinical testing, and growing demand for early diagnostic tests are expected to drive the market. Moreover, rapid advancements in data management, automation, and sample preparation due to growing volumes of testing samples are anticipated to boost market growth during the forecast period.For instance, in February 2020, MGI expanded the availability of its laboratory automation and DNA sequencing products in Latin America with top distributors in Brazil, Peru, Ecuador, Argentina, Mexico, and Colombia.
Advancements in laboratory testing technology through incremental and breakthrough developments are high-impact rendering drivers of the Latin America clinical laboratory services market. Incremental changes are introduced to make the testing processes simpler and enhance their efficiency. The introduction of novel solutions to maximize efficiency and minimize errors is anticipated to further boost the market. For instance, in September 2019, SCIEX received ANVISA approval for a suite of medical devices, including QTRAP 4500MD LC-MS/MS, Topaz and Citrine systems, SCIEX Triple Quad 4500MD LC-MS/MS, which is anticipated to enhance the quality standards of diagnostic testing
Due to the COVID-19 pandemic, there has been an increase in the approval of tests for diagnosis of the novel coronavirus. In February 2020, PAHO took efforts to ensure that countries in Latin America are prepared for COVID-19 laboratory diagnosis. This step was taken to ensure the timely response and identification of the disease in the region. Moreover, during this time, PAHO in collaboration with Fiocruz and the Ministry of Health led training in Brazil for nine countries including Argentina, Chile, Bolivia, Panama, Colombia, Paraguay, Ecuador, Uruguay, and Peru to enhance the preparedness for the COVID-19 diagnosis.
Personalized medicine involves tailoring medical treatments to individual patients based on their genetic information, lifestyle factors, and other personal data. Clinical laboratory services will play a crucial role in providing the genetic testing and analysis necessary for personalized medicine. For instance, genetic testing can help determine the optimal dosage of a medication for a particular patient based on their genetic makeup.
Unclear regulatory frameworks/guidelines for diagnostics in developing economies are anticipated to restrain the Latin America clinical laboratory services market growth. The presence of such discrete and uncertain scenarios in regulations for molecular diagnostic products can create confusion among manufacturers regarding commercialization. However, a number of initiatives are being undertaken in the region to enhance the acceptability and adoption of clinical laboratory tests and solutions with developed regulatory scenario.
Test Type Insights
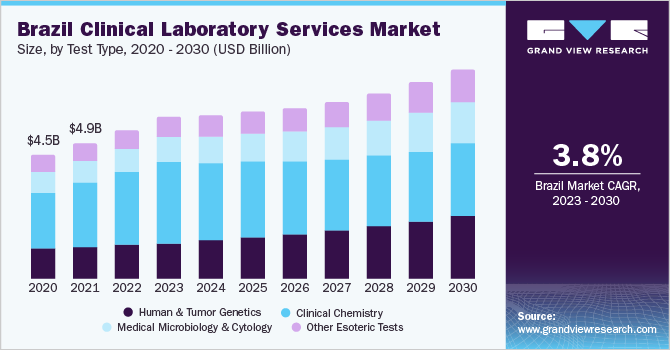
The clinical chemistry segment accounted for the largest revenue share of 47.69% in 2022owing to the presence of several clinical chemistry tests involved in pathology analysis of body fluids, including analysis of plasma, serum, urine, and other body fluids. In addition, the introduction of new technologies, alternative sampling methods, and the emergence of point-of-care testing methods are likely to further boost the Latin America clinical laboratory services market growth. In February 2020, Ortho Clinical Diagnostics launched a clinical chemistry system to complete its integrated Vitros XT line, designed to cover most typical lab tests.
The human and tumor genetics tests segment is expected to show the fastest growth during the forecast period. This can be attributed to rise in presence of intensive research activities on genetic and proteomic studies, in context of hereditary & gene-mutation-related disorders and rising demand for personalized medicine. Growing need for efficient tests in the early diagnosis of cancer and major infections is expected to drive the segment, primarily due to the precision and accuracy offered by genetic tests. Federal agencies such as the FDA, CMS, and FTC among others regulate these genetic tests.
Service Provider Insights
The hospital-based laboratories segment held the largest revenue share of the Latin America clinical laboratory services market of 53.91% in 2022. The growing number of outreach programs by hospitals, coupled with the high turnaround of patients suffering from complex and major diseases, is expected to drive the segment. Based on an article published in Forbes Magazine in May 2020, Hospital Israelita Albert Einstein of Brazil announced the development of a highly scalable type of COVID-19 test with the help of Varstation, a startup under the hospital’s incubation program.
The clinic-based laboratories segment is expected to show lucrative growth over the forecast period. One significant trend in the market is the adoption of Point-Of-Care Testing (POCT), which allows medical diagnostic testing to be performed at or near a patient's location. This trend has been driven by the benefits of POCT, such as faster turnaround time, reduced need for sample transportation, and improved patient outcomes. POCT has also been crucial amid the COVID-19 pandemic, with rapid antigen & molecular tests enabling widespread testing and early detection of infected individuals.
Application Insights
The bioanalytical and lab chemistry services segment accounted for the largest revenue share of 52.60% in 2022 attributed to the increasing number of investigational drugs undergoing clinical trials and the growing number of novel drug candidates being developed for clinical trials. The FDA (U.S.), ANVISA (Brazil), COFEPRIS (Mexico), ANMAT (Argentina), ISP (Chile), and other regulatory bodies provide bioanalytical method development & validation guidelines that help enforce the values required for reliable bioanalytical & lab chemistry methods. The Global Bioanalysis Consortium and other initiatives can help LATAM countries set up their guidelines and move forward more rationally.
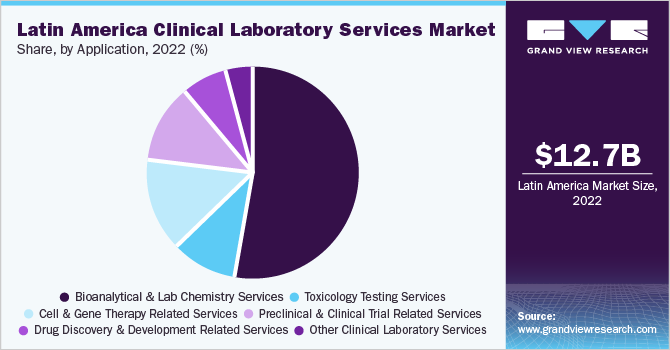
The toxicology testing segment is expected to grow at a significant rate. Toxicology testing is in high demand as most of the startups or smaller laboratories find it financially and clinically feasible. However, the confirmation of toxicity may require the use of advanced testing technologies. Larger laboratories that serve a broader population need to consider the turnaround time when outsourcing. This highlights the need of a cost-benefit analysis for labs considering advanced and confirmatory toxicology services. Moreover, Pharmacogenetic (PGx) testing can be performed to observe adverse drug reactions in individual patients. Therapeutic drug management can help optimize the dose of a therapeutic drug and is complementary to PGx testing
Country Insights
Brazil dominated the Latin America clinical laboratory services market with a revenue share of 41.92% in 2022owing to the increasing prevalence of chronic diseases, its developing economy, and various reforms that are being initiated by the government. Furthermore, the market in Brazil started to grow owing to the increased private investment, but the market growth is hampered by ethical reasons and a lack of improved infrastructure. Recently, Brazil replaced its Committee of Ethics with an electronic submission system, which helps accelerate the process of test assessment and clinical trials as well. This is expected to significantly boost market growth.
Mexico is estimated to show the fastest growth over the forecast period driven by improvements in healthcare infrastructure, rise in consumer awareness regarding diagnosis, and an increase in the prevalence of chronic diseases. Moreover, laboratories in the country are focusing on expanding their presence across various places in the country. For instance, in October 2022, TriCore Reference Laboratories opened a new facility in southern New Mexico to bring fast medical testing.
Key Companies & Market Share Insights
The key players in the Latin America clinical laboratory services market are investing in new technologies to improve the accuracy and speed of testing, expand their geographic presence, and offer new services to existing customers. For Instance, in January 2023, Unilabs partnered with Ambry Genetics to improve its high-quality genetic testing services for biopharmaceutical companies conducting clinical trials in Europe, Latin America, and Middle East. Some of the key players in the Latin America clinical laboratory services market include:
-
Abbott
-
OPKO Health, Inc.
-
Fresenius Medical Care AG & Co. KGaA
-
QIAGEN
-
Quest Diagnostics Incorporated
-
Siemens Healthcare GmbH
-
Charles River Laboratories
-
Laboratory Corporation of America Holdings (LabCorp)
-
DaVita Inc.
-
Myriad Genetics, Inc.
-
Genomic Health (Exact Sciences Corporation)
-
DASA
-
Sysmex Corporation
Latin America Clinical Laboratory Services Market Report Scope
Report Attribute
Details
Market size value in 2023
USD 13.74 billion
Revenue forecast in 2030
USD 17.59 billion
Growth rate
CAGR of 3.6% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2020
Forecast period
2023 - 2030
Report updated
June 2023
Quantitative units
Revenue in USD billion and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Test type, service provider, application, country
Country scope
Brazil, Mexico, Argentina, Colombia, Peru, Chile
Uruguay, Dominican Republic, Costa Rica, Panama
Key companies profiled
Abbott; OPKO Health, Inc.; Fresenius Medical Care AG & Co. KgaA; QIAGEN; Quest Diagnostics Incorporated; Siemens Healthcare GmbH; Charles River Laboratories; Laboratory Corporation of America Holdings (LabCorp); DaVita Inc.; Myriad Genetics, Inc.; Genomic Health (Exact Sciences Corporation); DASA; Sysmex Corporation
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Latin America Clinical Laboratory Services Market Report Segmentation
This report forecasts revenue growth at regional and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For the purpose of this report, Grand View Research has segmented the Latin America clinical laboratory services market report on the basis of test type, service provider, application, and country:
-
Test Type Outlook (Revenue, USD Billion, 2018 - 2030)
-
Human & Tumor Genetics
-
Clinical Chemistry
-
Medical Microbiology & Cytology
-
Other Esoteric Tests
-
-
Service Provider Outlook (Revenue, USD Billion, 2018 - 2030)
-
Hospital-Based Laboratories
-
Stand-Alone Laboratories
-
Clinic-Based Laboratories
-
-
Application Outlook (Revenue, USD Billion, 2018 - 2030)
-
Bioanalytical & Lab Chemistry Services
-
Toxicology Testing Services
-
Cell & Gene Therapy Related Services
-
Preclinical & Clinical Trial Related Services
-
Drug Discovery & Development Related Services
-
Other Clinical Laboratory Services
-
-
Country Outlook (Revenue, USD Billion, 2018 - 2030)
-
Brazil
-
Mexico
-
Argentina
-
Colombia
-
Peru
-
Chile
-
Uruguay
-
Dominican Republic
-
Costa Rica
-
Panama
-
Frequently Asked Questions About This Report
b. The Latin America clinical laboratory services market size was estimated at USD 12.68 billion in 2022 and is expected to reach USD 13.74 billion in 2023.
b. The Latin America clinical laboratory services market is expected to grow at a compound annual growth rate of 3.6% from 2023 to 2030 and is expected to reach USD 17.59 billion by 2030.
b. The clinical chemistry segment accounted for the largest revenue share of 47.69% in 2022 owing to the presence of several clinical chemistry tests involved in pathology analysis of body fluids, including analysis of plasma, serum, urine, and other body fluids.
b. Some key players operating in the Latin America clinical laboratory services market include QIAGEN, Abbott, Quest Diagnostics Incorporated, Laboratory Corporation of America Holdings, and OPKO Health, Inc. among others.
b. The increasing burden of infectious & chronic diseases, technological advancements, and increasing demand for early diagnostic tests are the major factors driving the Latin America clinical laboratory services market growth over the forecast period.
b. Hospital-based laboratories segment held the largest share of the Latin America clinical laboratory services market of 53.91% in 2022 owing to the growing number of outreach programs by hospitals, coupled with the high turnaround of patients suffering from complex and major diseases.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










