- Home
- »
- Medical Devices
- »
-
Medical Device Contract Manufacturing Market Report, 2033GVR Report cover
![Medical Device Contract Manufacturing Market Size, Share & Trends Report]()
Medical Device Contract Manufacturing Market (2026 - 2033) Size, Share & Trends Analysis Report Product (Class I, Class II, Class III), By Services (Accessories Manufacturing, Assembly Manufacturing, Component Manufacturing), By Therapeutic Area, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-498-9
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Medical Device Contract Manufacturing Market Summary
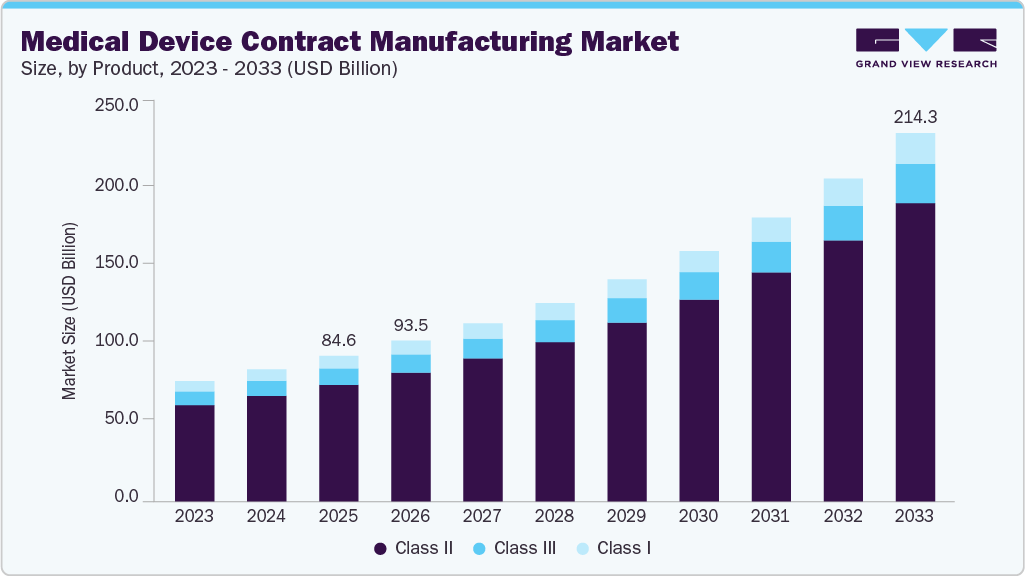
The global medical device contract manufacturing market size was valued at USD 84.61 billion in 2025 and is projected to reach USD 214.33 billion by 2033, growing at a CAGR of 12.58% from 2026 to 2033. The market is experiencing growth driven by growing demand for complex and customized devices, increasing outsourcing trends owing to cost affordability, rising investment in healthcare infrastructure and upsurging off-shoring medical device manufacturing to the emerging economies.
Key Market Trends & Insights
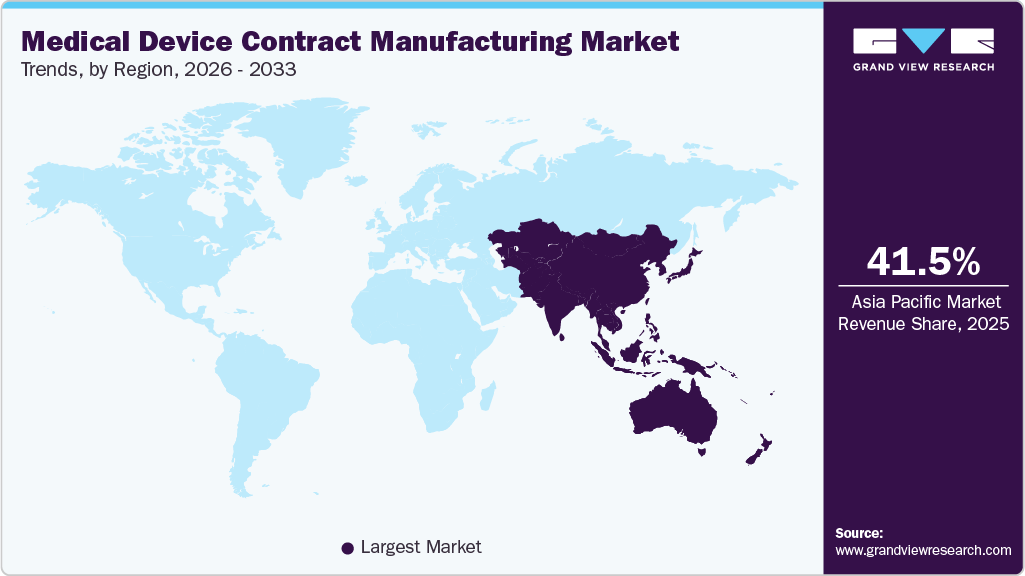
- Asia Pacific medical device contract manufacturing market held the largest share of 41.54% of the global market in 2025.
- The medical device contract manufacturing in the India is expected to grow significantly over the forecast period.
- Based on product, the class II segment held the largest market share of 80.10% in 2025.
- Based on service, accessories manufacturing segment held the largest market share in 2025.
- Based on therapeutic area, the cardiovascular devices segment held the highest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 84.61 Billion
- 2033 Projected Market Size: USD 214.33 Billion
- CAGR (2026-2033): 12.58%
- Asia Pacific: Largest market in 2025
In addition, rising demand for complex, customized devices is fueled by chronic diseases and growth in minimally invasive and wearable technologies. Increasing shift toward personalized medicine is accelerating adoption of advanced manufacturing solutions such as automation, precision engineering, and additive manufacturing. Besides, OEMs are outsourcing to full-service partners in large numbers offering comprehensive capabilities from design to commercialization, reducing time-to-market. In addition, growing regulatory complexity and high compliance costs further boost demand for specialized CMOs.Furthermore, cost affordability is the major factor driving the medical device contract manufacturing market, as OEMs shift from capital-intensive in-house production to outsourcing. High costs of advanced equipment, cleanrooms, compliance, and skilled labor are pushing companies toward CMOs that offer established infrastructure and expertise. The low-cost manufacturing regions enhance price competitiveness without compromising quality. In addition, CMOs also provide technical expertise and support innovation, enabling OEMs to focus on core activities.
Moreover, offshoring to developing economies is a key driver of the market, as OEMs seek cost efficiency, scalability, and regulatory compliance. Lower labor and operational costs, along with the expansion of manufacturing infrastructure, have encouraged nearly 60% of contract manufacturers to establish operations in emerging regions. In addition, regulatory improvements, including streamlined approvals and supportive policies, have reduced time-to-market and compliance risks. Furthermore, rising healthcare demand, aging populations, and the prevalence of chronic diseases are driving the need for localized, high-volume production. Similarly, favorable macroeconomic factors, such as export incentives and currency advantages, further enhance the appeal of outsourcing.
Opportunity Analysis
The medical device contract manufacturing market presents significant growth opportunities driven by increasing demand for advanced, personalized, and digitally integrated devices. The shift toward minimally invasive procedures, wearable technologies, and drug-device combination products is creating demand for specialized manufacturing capabilities, including micro-precision engineering, automation, and additive manufacturing. Contract manufacturers offering end-to-end services from design and prototyping to regulatory support and commercialization are well-positioned to capture higher-value opportunities as OEMs increasingly prefer integrated partners.
In addition, emerging markets offer strong growth potential driven by improving healthcare infrastructure, favorable government policies, and rising local demand for medical devices. In addition, growing adoption of digital health, AI-enabled diagnostics, and connected devices is opening new avenues for collaboration between CMOs, technology firms, and OEMs. Furthermore, increasing regulatory complexity is encouraging outsourcing to experienced partners with established compliance systems. Investments in smart manufacturing, modular production, and sustainable practices are also expected to unlock long-term competitive advantages and revenue growth opportunities.
Impact of U.S. Tariffs on Medical Device Contract Manufacturing Market
Tariffs and import/export duties are transforming the medical device contract manufacturing market by increasing input costs and disrupting global supply chains. CMOs rely on internationally sourced components such as sensors, polymers, and specialty metals, and rising tariffs across major trade routes have elevated production expenses. These cost increases are often passed on to OEMs, leading to higher contract values and margin pressures. As a result, CMOs with expanded sourcing strategies and multi-country manufacturing footprints are gaining a competitive advantage by mitigating tariff exposure through localized production.
In transatlantic trade, rising tariffs between the U.S. and Europe have increased the cost of imported components, despite relatively low direct duties on finished devices. This has encouraged production localization and contract restructuring. In North America, trade agreements maintain tariff-free movement but impose stricter compliance requirements, increasing operational complexity while supporting nearshoring trends.
Technological Advancements
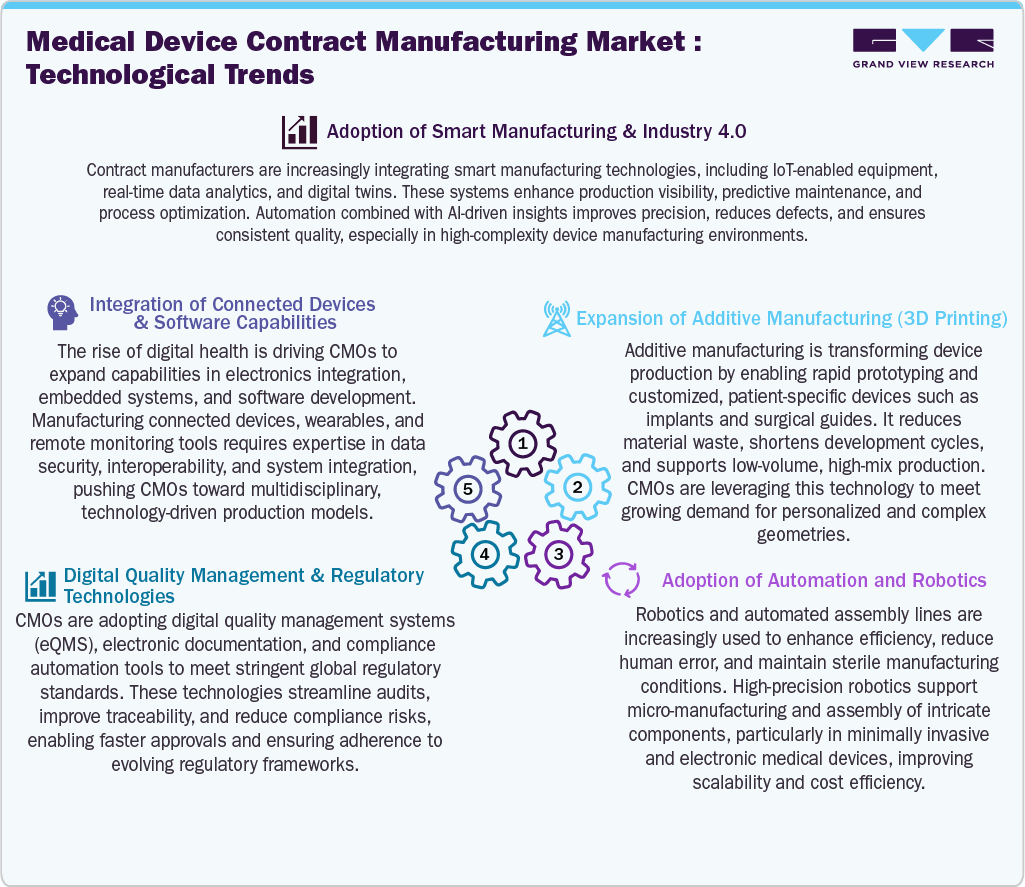
Technological advancements are transforming the medical device contract manufacturing market, driven by the increasing adoption of smart manufacturing and Industry 4.0 solutions. CMOs are integrating IoT, AI-driven analytics, and digital twins to enhance production efficiency, predictive maintenance, and quality control. In addition, additive manufacturing is gaining traction for rapid prototyping and production of patient-specific devices, supporting customization and reducing time-to-market. In addition, advanced automation and robotics are improving precision, scalability, and sterility, particularly in the manufacturing of complex, minimally invasive devices. Furthermore, digital quality management systems and regulatory technologies streamline compliance, improving traceability and accelerating approval timelines. The growing demand for connected medical devices and digital health solutions is also driving CMOs to expand capabilities in electronics integration, embedded systems, and software development.
Pricing Model Analysis
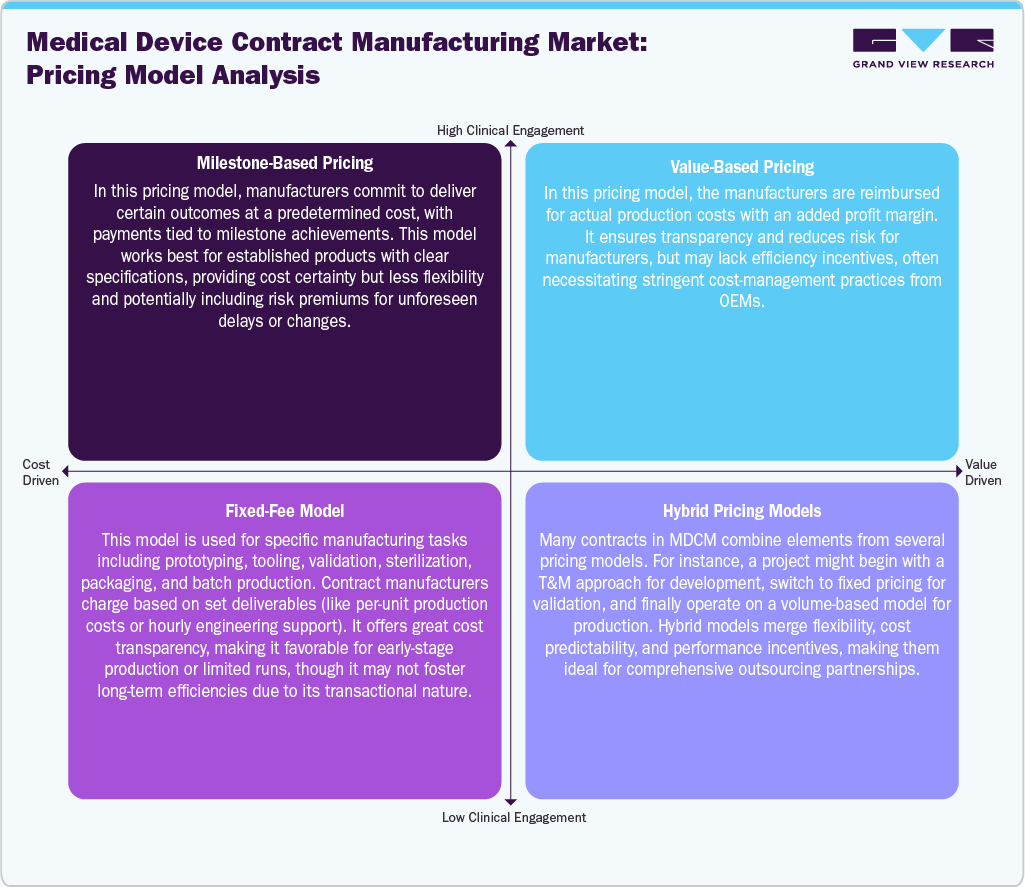
The medical device contract manufacturing employs diverse pricing models to balance cost control, flexibility, and risk-sharing. Milestone-based pricing ties payments to predefined deliverables, offering cost certainty for well-defined projects but limited flexibility and potential risk premiums. Whereas value-based pricing reimburses actual production costs plus a margin, ensuring transparency and reducing manufacturer risk, though it may require strict oversight to maintain efficiency. The fixed-fee model is commonly used for specific tasks such as prototyping, tooling, validation, and packaging, with charges based on set deliverables or unit costs, making it suitable for early-stage or low-volume production despite its transactional nature. Furthermore, hybrid pricing models are adopted, combining elements of multiple approaches across project phases, for instance, time-and-materials during development, fixed pricing for validation, and volume-based pricing for production. These models provide a balanced framework that align cost predictability, operational flexibility, and performance incentives in long-term outsourcing partnerships.
Market Concentration & Characteristics
Market growth stage is high and is expected to accelerate over the estimated period. The medical device contract manufacturing market is characterized by technologies, regulatory considerations, and globalization & outsourcing of product processes to influence advantages and specialized capabilities.
The market is highly innovation-driven, shaped by rising device complexity, personalization, and digital integration. Manufacturers are investing in automation, 3D printing, precision machining, and advanced molding to support customized and high-performance devices. Innovation is also evident in new branded component offerings. For instance, in June 2025, Phillips-Medisize announced TheraVolt medical connectors, its first branded offering of connectors, which are aimed at improving integration, performance, and reliability in sophisticated medical devices, especially in electrophysiology applications.

Regulatory requirements are a defining market characteristic, with strict quality systems, compliance standards, and validation expectations transforming supplier selection. Contract manufacturers must operate within frameworks such as ISO and FDA-aligned processes, making regulatory expertise a major differentiator. Compliance adds cost and complexity, but it also strengthens barriers to entry and increases OEM reliance on established, audit-ready partners.
M&A activity is moderate to high, as larger players expand capabilities, technology platforms, and geographic reach through acquisitions and partnerships. Consolidation helps manufacturers build comprehensive service portfolios and strengthen specialized expertise in areas like combination products, precision components, and device assembly. For instance, in January 2026, Tecomet and Orchid Orthopedic Solutions merged resulting in the establishment of an orthopedic CDMO with more robust manufacturing footprint and greater capacity to invest in advanced manufacturing, automation, and capacity expansion in line with customer demand.
Service expansion is a key feature of the market, with manufacturers moving beyond basic assembly into design, prototyping, testing, packaging, labeling, and full lifecycle support. Comprehensive manufacturing models are preferred more by OEMs seeking faster development and simpler supply chains.
Regional expansion is accelerating as manufacturers build global footprints to serve local and cross-border demand more efficiently. Companies are expanding into North America, Europe, and Asia to support supply resilience, lower costs, and improve market access. Nearshoring and multi-region manufacturing are becoming important strategies, especially as OEMs seek faster delivery, reduced risk, and better regulatory alignment.
Product Insights
On the basis of product segment, the class II segment led the medical device contract manufacturing market and accounted for 80.10% of global revenue in 2025. The segment is growing significantly due to frequent regulatory clearances, continuous product upgrades, and well-defined approval pathways. More than 90% of devices reviewed by the FDA follow the 510(k) route, with Class II devices accounting for around 43% of the market, ensuring consistent manufacturing demand. Layered regulatory requirements, including general and special controls, necessitate rigorous engineering, validation, and documentation, increasing production complexity. Furthermore, modification-based pathways such as Special 510(k) enable faster updates, encouraging iterative innovation and repeated production cycles, thereby sustaining long-term growth in this segment.
The class III segment is projected to experience the second fastest growth over the forecast period. The increasing technical complexity and high-risk nature of Class III medical devices are driving significant growth in the market. Some of the devices, such as implantable pacemakers, heart valves, and neurostimulators, require stringent regulatory compliance, extensive clinical validation, and precise engineering, creating a highly demanding production environment. Their multi-stage development processes, including prototyping, clinical trials, and sterilization validation, add operational complexity and encourage outsourcing to specialized manufacturers.
Service Insights
On the basis of the service, the accessories manufacturing segment dominated the market in 2025, due to rising demand for highly customized surgical instruments, precision-driven designs, and efficient development-to-production workflows. Products such as spinal instruments, rod holders, reducers, and silicone handles require tight tolerances and application-specific customization, driving adoption of Design for Manufacturing (DFM) and rapid prototyping. In addition, strong R&D capabilities and flexible production support customized solutions, enabling manufacturers to meet evolving surgical requirements while ensuring reliability, scalability, and faster product development cycles.
The device manufacturing is expected to grow significantly during the forecast period. The advancement of integrated manufacturing models, coupled with the growing deployment of automation technologies and the rising need for consistent, high-volume production, is substantially driving the expansion of device manufacturing in the medical device contract manufacturing market. In addition, increasing pressure to accelerate delivery timelines, optimize production costs, and enhance supply chain efficiency is pushing manufacturers to consolidate multiple production stages into unified, end-to-end manufacturing platforms. For instance, in March 2026, TANAKA PRECIOUS METAL TECHNOLOGIES Co. Ltd, a contract manufacturer. has developed a comprehensive total solution system for test kit contract manufacturing, enabling seamless execution of processes from initial development through to final product realization.
Therapeutic Area Insights
On the basis of the therapeutic area, the cardiovascular device segment dominated the market in 2025. The adoption of robotic-assisted Coronary Artery Bypass Grafting (CABG) is driving a shift toward minimally invasive cardiovascular procedures, increasing demand for advanced devices such as robotic instruments, precision catheters, and specialized implants. These procedures require highly engineered, miniaturized components that ensure accuracy and safety, encouraging OEMs to partner with contract manufacturers for efficient, compliant production. In addition, the rising prevalence of cardiovascular diseases and faster recovery times are boosting procedure volumes and patient adoption. This, in turn, is increasing demand for cardiovascular devices and consumables, supporting strong growth in the medical device contract manufacturing market through innovation and scalable manufacturing capabilities.
The surgical instruments segment is expected to grow significantly during the forecast period. The integration of robotics and artificial intelligence in surgical procedures is driving strong growth in surgical instruments within the medical device contract manufacturing market. AI-assisted robotic surgeries enhance precision and reduce complications, increasing demand for advanced tools such as robotic end-effectors, minimally invasive instruments, and sensor-enabled devices. These require complex designs, high manufacturing accuracy, and strict regulatory compliance, encouraging OEMs to outsource production.
End Use Insights
On the basis of end use, the OEM segment held the largest revenue share in 2025, driven by its central role in innovation, commercialization, and large-scale device production. OEMs generate significant outsourcing demand by relying on specialized manufacturers to manage complex development processes, regulatory compliance, and cost pressures. In addition, high-volume production requirements for therapeutic, diagnostic, and combination devices strengthen this dominance. In addition, expanding OEM partnerships with contract manufacturers continues to drive demand for advanced capabilities, including precision manufacturing, cleanroom operations, and robust quality systems.
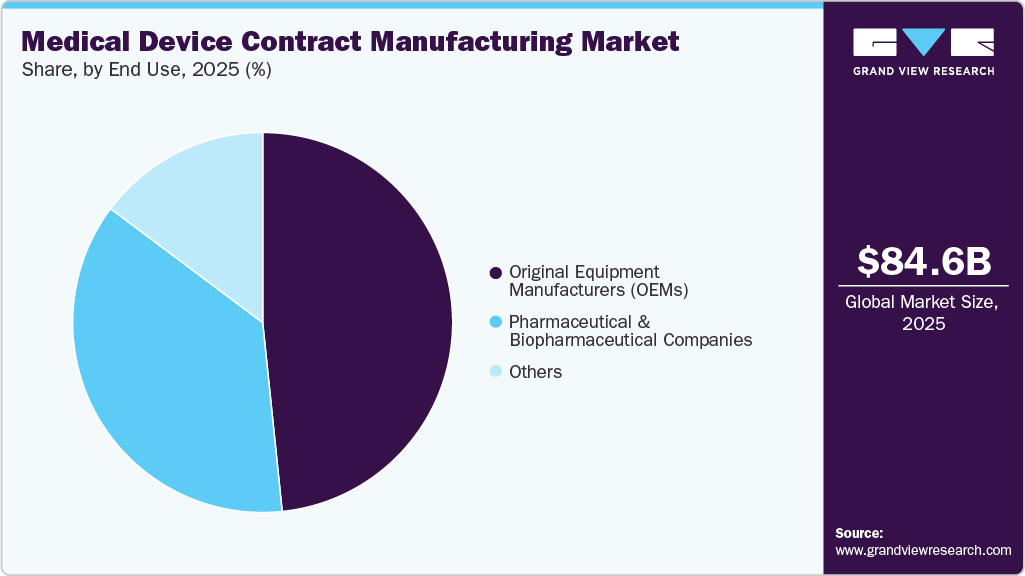
The pharmaceutical and biopharmaceutical companies segment in the market is the second fastest growing due to increasing demand for combination products such as pre-filled injectors, inhalers, and drug-eluting systems. These firms often lack specialized capabilities in precision engineering, clean-room assembly, sterilization, and regulatory compliance, driving reliance on contract manufacturers. In addition, outsourcing enables them to focus on core activities such as research and commercialization while ensuring high-quality production and compliance. This collaboration accelerates time-to-market and supports scalable manufacturing without heavy capital investment. Industry participation in global events further highlights ongoing partnerships and expanding opportunities in this segment.
Regional Insights
North America medical device contract manufacturing market is driven by combination of regulatory sophistication, deep OEM ecosystems, and accelerating capital deployment across U.S., Canada, and Mexico. U.S. is anchored by a high density of FDA-registered, ISO 13485-certified contract manufacturers operating across therapeutic segments including cardiovascular, orthopedic, and surgical instruments. Private equity-led consolidation is a defining feature of the competitive landscape, with platform-building strategies driving multi-facility acquisitions and capability expansions across silicone molding, sterile assembly, precision component manufacturing, and vertically integrated device production.
U.S. Medical Device Contract Manufacturing Market Trends
U.S. medical device contract manufacturing market supported by a robust regulatory framework, strong OEM presence, and a well-established network of FDA- and ISO 13485-compliant manufacturers. Private equity-driven consolidation is transforming the landscape. For instance, in October 2025, Los Angeles-based Vance Street Capital partnered with Molded Rubber and Plastic Corporation (MRPC), a Butler, Wisconsin-headquartered ISO 13485-certified manufacturer with additional facilities in Hudson, New Hampshire and Largo, Florida. Furthermore, vertically integrated players such as Viant Medical are gaining traction, particularly across cardiovascular and orthopedic segments.
Canada medical device contract manufacturing market is driven by presence of strong government support, infrastructure investments, and a growing base of certified partners. The CAMEDA program, backed by FedDev Ontario, provides non-dilutive funding and access to ISO 13485-certified CDMOs offering end-to-end capabilities. In addition, institutions such as McMaster Manufacturing Research Institute enable Industry 4.0 adoption, including AI and IoT integration. In addition, initiatives such as NIICO aim to boost isotope production for diagnostics and sterilization. Furthermore, combined with Health Canada-aligned regulations and collaborative ecosystems, Canada is emerging as a key outsourcing hub for advanced medical devices.
Europe Medical Device Contract Manufacturing Market Trends
Europe held a significant market share in 2025. The region is expanding due to consolidation, capacity investments, and the rise of vertically integrated CDMO platforms. in addition, stringent regulations under EU MDR 2017/745 and ISO 13485 are driving OEMs to outsource to specialized partners for compliance and post-market management. Furthermore, private equity is accelerating platform creation across Germany, France, and the UK through acquisitions and carve-outs. In addition, investments in advanced manufacturing such as biologics, precision molding, 3D printing, and biomaterials are strengthening technological capabilities and regional competitiveness.
The market for medical device contract manufacturing in Germany is expanding due to its position as Europe’s largest MedTech hub and a strong base of SMEs relying on CDMOs for advanced technologies, cost efficiency, and faster development. Rising demand for diagnostic, dental, and surgical devices is boosting outsourcing. For instance, in January 2026, Gilde Healthcare acquired Rudischhauser, strengthening its capabilities. In addition, stringent EU MDR requirements and expansions such as Nelson Labs’ Wiesbaden facility in August 2025 are increasing demand for regulatory, testing, and post-market support services.
The UK medical device contract manufacturing market is considered a lucrative market for pharmaceutical companies due rising demand for precision components, and a strong regulatory ecosystem. Strategic M&A and partnerships are enhancing scalability and capabilities. For instance, in November 2025, Elevaris Medical Devices, formed through Spectra Medical Devices' acquisition of UK-based XL Precision Technologies, launched its CDMO services and procedural needle portfolio across the EMEA region. Furthermore, investments in advanced facilities, and collaborations strengthen the UK’s position as an innovation-driven, high-value contract manufacturing hub.
Asia Pacific Medical Device Contract Manufacturing Market Trends
Asia Pacific dominated the global medical device contract manufacturing market in 2025, holding a revenue share of 41.54%. The growth of this region is due to high-volume production and high-value device development outsourcing dynamics across its major economies. Strategic cross-border alliances in regenerative medicine and combination products are expanding roles beyond traditional manufacturing into advanced therapeutic development. In addition, rapid digital regulation, expanding production capacity, and multi-market certifications are supporting international growth. Furthermore, government incentives and trade agreements enhance competitiveness, while emerging hubs benefit from cost efficiency, raw material access, and supply chain proximity, alongside strong clinical trial infrastructure and innovation-driven partnerships.
The market for medical device contract manufacturing in Japan is expanding due to rising technological complexity, high healthcare spending, and strong government support for innovation. OEMs increasingly outsource design, prototyping, precision manufacturing, testing, and regulatory functions to CDMO/CMO partners to accelerate timelines and manage compliance costs. Some of the strategic partnerships highlight end-to-end service expansion. For instance, in October 2025, SanyoSeiko Co., Ltd., a Japan-based precision engineering and contract manufacturing company, expanded its role with Bluejay Diagnostics to provide end-to-end CMO support. Furthermore, cross-border alliances in regenerative medicine and advanced diagnostics are strengthening capabilities, enabling faster development, scalable production, and broader commercialization opportunities in high-value segments.
The medical device contract manufacturing market in India is expected to experience significant growth at a significant CAGR during the forecast period. The country is witnessing considerable growth due to regulatory strengthening, expanding domestic capabilities, and rising global demand for cost-efficient, quality-compliant production. Government initiatives like Make in India, PLI schemes, and increased healthcare spending are boosting infrastructure and investment. In addition, regulatory standardization is enhancing OEM confidence, while private equity funding supports capacity expansion and automation. Furthermore, trade agreements are improving global access, and India’s cost advantage and skilled workforce continue to attract international OEMs seeking diversified manufacturing and outsourcing partners.
Latin America Medical Device Contract Manufacturing Market Trends
Latin America is emerging as a key hub for medical device contract manufacturing market, driven by increasing demand for cost-effective, high-quality medical device manufacturing and regulatory support, particularly from multinational OEMs looking to expand their regional presence. Some of the countries such as Brazil and Argentina are emerging as key manufacturing hubs due to a combination of growing domestic healthcare needs, near-shoring owing to the present geopolitical environment, expanding healthcare infrastructure, regional prioritization, and favorable labor and manufacturing costs for OEMs.
The medical device contract manufacturing market in Brazil is experiencing growth driven by rising healthcare investment, and supportive policies. In addition, increasing prevalence of chronic diseases and demand for complex Class II and III devices are encouraging OEMs to partner with CMOs for precision manufacturing and compliance. Furthermore, infrastructure expansion and friend-shoring trends are boosting local production. In addition, high import dependence highlights opportunities for domestic manufacturers to adopt advanced technologies, enhance capacity, and strengthen supply chain resilience, supporting long-term market growth.
Middle East And Africa Medical Device Contract Manufacturing Market Trends
The MEA accounted for a significant revenue share in the global medical device contract manufacturing market in 2025. The growth is driven by expanding healthcare infrastructure, rising prevalence of chronic and lifestyle diseases, and increasing government focus on domestic healthcare capabilities. Strategic national initiatives, such as Saudi Arabia’s Vision 2030 and the UAE’s Vision 2031, emphasize healthcare localization, advanced manufacturing, and medical technology self-sufficiency, creating favorable conditions for both domestic and international medical device contract manufacturers. These initiatives provide incentives for infrastructure investment, technology transfer, and workforce development, encouraging global OEMs to collaborate with local partners for outsourcing manufacturing services.
South Africa medical device contract manufacturing market is witnessing growth due to public health reforms, localization policies, and high import dependence. In addition, government tenders and B-BBEE requirements encourage OEMs to establish local partnerships. Expanding healthcare access under national insurance initiatives is increasing demand for cost-effective devices. Furthermore, collaborations between local CMOs and global OEMs, aligned with international standards such as ISO 13485 and GMP, are strengthening the ecosystem and supporting sustainable market growth.
Saudi Arabia medical device contract manufacturing market is emerging as a competitive hub, driven by Vision 2030 initiatives promoting healthcare modernization, localization, and technology adoption. Regulations by the Local Content and Government Procurement Authority (LCGPA) and centralized procurement through NUPCO encourage global OEMs to establish local manufacturing to access government tenders. In addition, rising population, chronic disease burden, and demand for advanced medical technologies are further boosting outsourcing needs. Furthermore, investments in automation, cleanroom infrastructure, and integrated R&D are strengthening capabilities. In addition, large-scale projects like Lenovo and Alat’s USD 2 billion manufacturing facility highlight efforts to enhance regional production capacity and supply chain resilience.
Key Medical Device Contract Manufacturing Company Insights
The medical device contract manufacturing market is highly competitive, driven by stringent regulatory requirements, quality standards, and demand for advanced manufacturing capabilities. Large, globally integrated players dominate through comprehensive service offerings spanning component manufacturing, assembly, device production, and packaging across multiple regions. Meanwhile, specialized manufacturers compete by leveraging deep expertise in drug-device combination products, consumables, and complex devices, supported by robust quality systems and regulatory compliance. For instance, in January 2026, Jabil Inc. signed a manufacturing agreement with TxSphere, enabling the production of reusable on-body injector drug delivery devices, including fill-finish services. This partnership will strengthen Jabil’s presence in the drug-device combination products space, enabling end-to-end CDMO services, as well as the scalable production of innovative wearable drug delivery devices.
Key Medical Device Contract Manufacturing Companies:
The following key companies have been profiled for this study on the medical device contract manufacturing market.
- Jabil Inc.
- Thermo Fisher Scientific Inc.
- Integer Holdings Corporation
- FLEX LTD.
- Sanmina Corporation
- Celestica Inc.
- Phillips-Medisize
- Plexus Corp.
- Nipro Corporation
- Viant Technology LLC
- West Pharmaceutical Services, Inc.
- Mack Molding
- Cirtec Medical
- Cogmedix
- Synecco Ltd
- TE Connectivity
- Keller Technology Corp.
- HDA Technology, Inc.
Recent Developments
-
In November 2025, Sanmina announced the expansion of its Fermoy, Ireland, medical manufacturing facility, thereby adding advanced automation lines, an ISO 8 cleanroom, and high-volume production capabilities for wearable medical devices, strengthening its position in finished medical device manufacturing in Europe.
-
In June 2025, Phillips-Medisize launched TheraVolt medical connectors, its first branded offering of connectors, which are aimed at improving integration, performance, and reliability in sophisticated medical devices, especially in electrophysiology applications. This announcement strengthened its portfolio by helping speed development of devices, improve device functionality, and reduce costs associated with customizations for device manufacturers
-
In February 2025, Mack Molding invested over USD 3 Mn in new hybrid injection molding press, automation, and auxiliary equipment across its facilities. This investment improved precision, efficiency, and capacity of the injection molding of medical device components.
Medical Device Contract Manufacturing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 93.52 billion
Revenue forecast in 2033
USD 214.33 billion
Growth rate
CAGR of 12.58% from 2026 to 2033
Historical year
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD billion/million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, Services, Therapeutic Area, End Use and Region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Thailand; South Korea; Australia; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait; Oman; Qatar
Key companies profiled
Jabil Inc.; Thermo Fisher Scientific Inc.; Integer Holdings Corporation; FLEX LTD.; Sanmina Corporation; Celestica Inc.; Phillips-Medisize; Plexus Corp.; Nipro Corporation; Viant Technology LLC; West Pharmaceutical Services, Inc.; Mack Molding; Cirtec Medical; Cogmedix; Synecco Ltd; TE Connectivity; Keller Technology Corp.; HDA Technology, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Medical Device Contract Manufacturing Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global medical device contract manufacturing market based on product, services, therapeutic area, end use and region.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Class I
-
Class II
-
Class III
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Accessories Manufacturing
-
Assembly Manufacturing
-
Component Manufacturing
-
Device Manufacturing
-
Packaging and Labeling
-
Others
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Cardiovascular Devices
-
Orthopedic Devices
-
Ophthalmic Devices
-
Diagnostic Devices
-
Respiratory Devices
-
Surgical Instruments
-
Dental
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Original Equipment Manufacturers (OEMs)
-
Pharmaceutical & Biopharmaceutical Companies
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
-
Frequently Asked Questions About This Report
b. The global medical device contract manufacturing market size was estimated at USD 84.61 billion in 2025 and is expected to reach USD 93.52 billion in 2026.
b. The global medical device contract manufacturing market is expected to grow at a compound annual growth rate of 12.58% from 2026 to 2033 to reach USD 214.33 billion in 2033.
b. Based on product, the Class II segment dominated the medical device contract manufacturing market and accounted for the largest revenue share of 80.10% in 2025.
b. Some key players operating in the medical device contract manufacturing market include Jabil Inc., Thermo Fisher Scientific Inc., Integer Holdings Corporation, FLEX LTD., Sanmina Corporation, Celestica Inc., Phillips-Medisize, Plexus Corp., Nipro Corporation, Viant Technology LLC, West Pharmaceutical Services, Inc., Mack Molding, Cirtec Medical, Cogmedix, Synecco Ltd, TE Connectivity, Keller Technology Corp., and HDA Technology, Inc., among others.
b. Key factors driving the medical device contract manufacturing market growth include growing demand for complex and customized devices, increasing outsourcing trends owing to cost affordability, rising investment in healthcare infrastructure and upsurging off-shoring medical device manufacturing to the emerging economies.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










