- Home
- »
- Medical Devices
- »
-
Medical Instrument Washing & Disinfection Market 2033GVR Report cover
![Medical Instrument Washing & Disinfection Market Size, Share & Trends Report]()
Medical Instrument Washing & Disinfection Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Chemistries, Equipment), By Process Stage, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-743-2
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
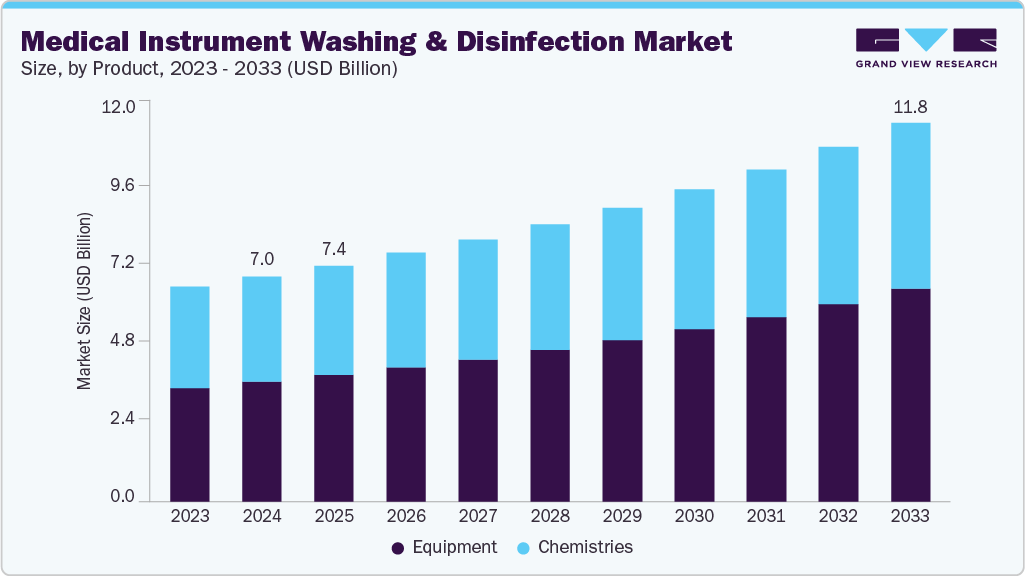
Medical Instrument Washing & Disinfection Market Summary
The global medical instrument washing & disinfection market size was estimated at USD 7.01 billion in 2024 and is projected to reach USD 11.82 billion by 2033, growing at a CAGR of 6.1% from 2025 to 2033. The growth of the market is attributed to the rising surgical and endoscopy procedure volumes, growing focus on reducing hospital-acquired infections (HAIs), and stricter global standards such as ISO 15883 and AAMI guidelines that mandate validated, automated reprocessing.
Key Market Trends & Insights
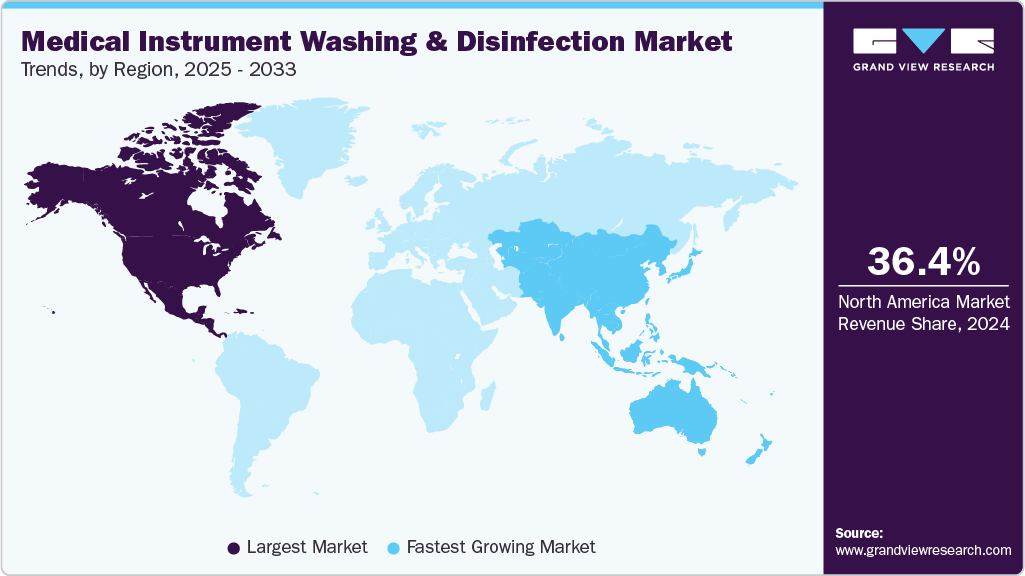
- North America dominated the medical instrument washing & disinfection market with the largest revenue share of 36.39% in 2024.
- The medical instrument washing & disinfection market in the U.S. accounted for the largest market revenue share of 83.43% in North America in 2024.
- By product, the equipment segment led the market with the largest revenue share of 53.22% in 2024.
- By process stage, the automated disinfection segment led the market with the largest revenue share of 38.56% in 2024.
- By end use, the hospitals segment led the market with the largest revenue share of 59.42% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 7.01 Billion
- 2033 Projected Market Size: USD 11.82 Billion
- CAGR (2025-2033): 6.1%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
Increasing complexity of surgical instruments, including robotic and lumened devices, further accelerates demand for advanced washer-disinfectors, ultrasonic cleaners, and automated endoscope reprocessors (AERs). Hospitals are also investing in digital connectivity, traceability, and energy-efficient systems to meet compliance, improve workflow efficiency, and lower operating costs. At the same time, recurring demand for specialized chemistries and service contracts supports sustained market growth.The global rise in surgical procedures, from everyday elective procedures to major organ transplants, the need for effective cleaning and disinfection of medical instruments has never been higher. Hospitals and surgical centers are seeing rising volumes of procedures, which means a constant flow of reusable instruments that need to be washed, disinfected, and ready for use day after day. This puts added pressure on facilities to keep up with demand and strictly follow safety protocols. To tackle these challenges, healthcare providers are turning to automated cleaning and disinfection systems that make the process faster, more consistent, and safer for patients and staff. Recent procedural data backs up this trend: in 2024, about 1,585,878 cosmetic surgeries took place, a 1% jump versus the previous year, and reconstructive surgeries saw a 2% increase, showing steady growth in surgical activity and further driving the need for advanced instrument reprocessing solutions.
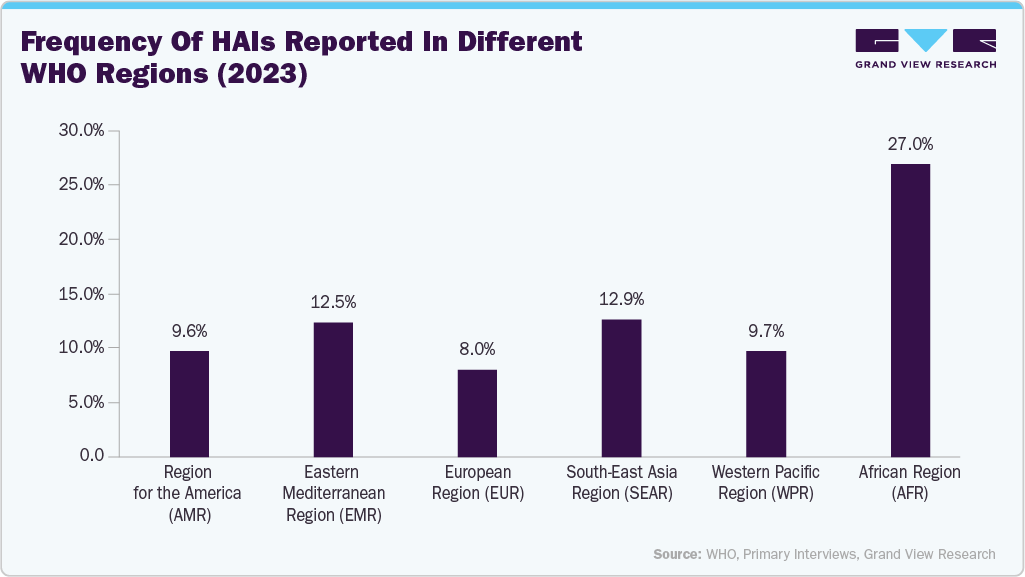
The rising incidence of hospital-acquired infections (HAIs) is a key driver of market growth, as healthcare facilities are increasingly prioritizing strong infection control measures, thereby fueling demand for advanced medical instrument washing and disinfection systems. According to WHO’s 2024 Global Report on Infection Prevention and Control, a large-scale systematic review of nearly 400 studies conducted worldwide in 2023 estimated the global prevalence of HAIs at approximately 14%. Furthermore, the European point prevalence survey conducted in 2022/2023 across 28 EU/EEA nations and three Western Balkan countries reported an adjusted HAI prevalence of 8.0% among patients. This growing burden of HAIs strengthens the focus on infection prevention, drives healthcare providers to adopt automated and validated reprocessing solutions, and is expected to significantly support the market expansion in the coming years.
The rising need for high-level disinfection (HLD) is emerging as a critical growth driver for the medical instrument washing and disinfection market, as healthcare facilities face mounting scrutiny over infection control standards. There is an increasing push toward adopting consistent, evidence-based disinfection practices across all care settings. Supporting this shift, the Society for Healthcare Epidemiology of America (SHEA) and eight partner organizations released new multisociety guidance in April 2025 to strengthen HLD and sterilize reusable medical devices to reduce healthcare-associated infections. The guidance underscores persistent challenges in device processing, particularly when complex or ambiguous manufacturer instructions and resource limitations within healthcare systems compromise protocols.
"Sterilization and HLD are some of the most challenging practices for health care facilities to get right due to their highly technical aspects and the dedicated resources required. This guidance provides health care facilities with concrete infection prevention recommendations to ensure best practices are in place to support safe patient care and reduce risk to patients." Said Dr. Erica Shenoy, co-chair of the guidelines panel and Chief of Infection Control at Mass General Brigham.
The increasing regulatory approvals of high-level disinfection (HLD) consumables and systems are creating attractive opportunities for product portfolio expansion among industry players. Regulatory bodies such as the U.S. FDA, the European Medicines Agency (EMA), and various regional health authorities actively support the development of new HLD formulations designed to combat a wider range of pathogens, including bacterial spores. As these advanced formulations enter the market, hospitals and outpatient surgical centers are more likely to adopt upgraded disinfection protocols to meet accreditation standards and comply with evolving infection control guidelines.
Recent Regulatory Approvals and Product Launches in High-Level Disinfection (HLD)
Date
Organization / Authority
Product / Standard Update
Application Area
Key Highlights
Feb-25
AAMI & ANSI
Updated ST58:2024 standard
Medical devices (general)
Recognized chlorine dioxide foam as an HLD; includes Parker Laboratories’ FDA-cleared Tristel ULT system with enhanced ultrasound probe disinfection and residue removal.
Jun-23
U.S. FDA
Tristel ULT (Parker Laboratories)
Ultrasound probes (endocavity, transrectal, transvaginal, and surface transducers)
First FDA-cleared HLD foam for ultrasound probes; 2-minute disinfection time (fastest approved); ready-to-use application with residue removal wipes.
Apr-25
Arxada
NUGEN HLD-CD
Medical devices (broad-spectrum, Europe)
Novel HLD with 5-minute contact time, effective against resilient pathogens including Clostridium difficile; launched to enhance infection control in critical healthcare.
May-25
U.S. FDA
Tristel OPH
Ophthalmic devices
FDA-approved HLD for eye-care instruments, expanding portfolio into ophthalmology.
Source: Grand View Research
“More than 215 million ultrasound scans are performed in the U.S. each year, with approximately 20% of them requiring high-level disinfection. Tristel ULT is the first product of its kind in the US, a high-level disinfectant foam for reprocessing ultrasound probes. As the exclusive manufacturer of Tristel ULT and Tristel DUO in the US, Parker Laboratories now offers a complete line of disinfecting solutions for ultrasound departments”. Neal Buchalter, President of Parker Laboratories
The growing adoption of automated disinfection systems creates substantial opportunities for medical instrument washing and disinfection market manufacturers. Rising surgical volumes, workforce shortages, and stricter compliance requirements drive healthcare facilities to implement automated workflows that minimize human error and ensure consistent reprocessing outcomes. Automated washers and disinfectors equipped with programmable cycles and integrated chemical dosing are increasingly vital for Central Sterile Supply Departments (CSSDs) and outpatient surgical centers. For instance, in July 2025, Nanosonics introduced trophon3, an advanced ultrasound probe disinfection system that delivers over 40% faster performance than earlier models. Initially launched across Europe, the UK, Australia, and New Zealand, the device offers enhanced digital connectivity and traceability, including integration with DICOM imaging systems.
Key Opinions of Leaders
Company Name
KOLs
Product Launch
Nanosonics
“The FDA clearance and USA launch of trophon3 and trophon2 Plus mark important milestones for Nanosonics. These innovations set a new benchmark in automated high-level disinfection and unlock significant growthopportunity through both new installed base and upgrades,” said Michael Kavanagh, CEO and President.”
In August 2025, Nanosonics has received approval from the U.S. Food and Drug Administration (FDA) for its latest ultrasound probe disinfection technologies-trophon3 and the trophon2 Plus software upgrade-paving the way for their commercial launch across the US healthcare sector. This regulatory approval enables Nanosonics to expand into US hospitals and private clinics, which collectively represent approximately 30,000 potential new device installations. The new trophon3 system offers several improvements over previous models, including a disinfection cycle that is over 40% faster, enhanced digital integration capabilities, and expanded traceability features.
Getinge
"XEN represents a significant advancement in cleaning chemistries, offering comprehensive documentation, performance verification, and a reduced environmental footprint. XEN provides a cost-effective and sustainable solution that benefits both our customers and the environment.” Said Erin Maxwell, Senior Product Manager at Getinge.
In August 2024, Getinge announced the U.S. launch of XEN, a new line of 16 advanced cleaning chemistries designed to optimize medical instrument cleaning and sterile processing workflows. This product is useful in every stage, including pretreatment, manual cleaning, automated cleaning, ultrasonic cleaning, final rinsing, and maintenance. XEN improved instrument lifespan, enhanced safety, supported efficiency, and offered environmentally friendly, thoroughly documented solutions for sterile processing departments nationwide.
Getinge
“Our new washer-disinfector is designed to adapt to unique needs while maintaining high performance,” says Marcus Samuelsson, Product Manager Cleaning and Disinfection at Getinge. “With a top modern multi-chamber technology, it offers high capacity, throughput, and low utility consumption without leaving any compromises on cleaning efficacy. It’s fully connected, allowing CSSD staff to quickly maximize uptime and ensure that the washer-disinfector is always ready to perform.”
In March 2024, Getinge's launch of a new, intuitive, high-performance multi-chamber washer-disinfector is a significant development in the infection control sector, aimed at addressing the need for efficient, high-capacity cleaning in healthcare facilities. Multi-chamber washer disinfectors are particularly valuable in hospitals and large medical centers where large volumes of surgical instruments and medical equipment must be sterilized quickly and effectively.
Source: Nanosonics, Getinge, Grand View Research
Market Concentration & Characteristics
Degree of Innovation: The degree of innovation in the medical instrument washing and disinfection market is high, driven by integrating advanced automation, digital traceability, and sustainable technologies into reprocessing workflows. Manufacturers are introducing multi-chamber washer-disinfectors, automated endoscope reprocessors, and high-level disinfection (HLD) systems with faster cycle times, programmable settings, and smart chemical dosing to enhance efficiency and compliance. For instance, in 2024 Germitec received the first-ever U.S. FDA De Novo classification for its Chronos UV-C HLD device, offering a chemical-free and eco-friendly alternative for disinfecting ultrasound probes. Similarly, Nanosonics’ trophon3, launched in 2025, provides a 40% faster disinfection cycle, enhanced digital connectivity, and traceability. These innovations, along with advancements in ergonomics, energy and water efficiency, and predictive maintenance, are elevating safety, consistency, and cost-effectiveness, positioning the sector as a rapidly evolving and innovation-intensive segment of infection prevention.
Impact of Regulations: Regulations play a critical role in shaping the medical instrument washing and disinfection market by enforcing stringent infection prevention standards and driving consistent adoption of validated reprocessing practices. Guidelines issued by global and regional authorities such as the U.S. FDA, European Medicines Agency (EMA), WHO, AAMI, and ISO mandate strict adherence to high-level disinfection (HLD) protocols, validated cleaning cycles, and thorough documentation for compliance and audit purposes. These regulatory requirements compel healthcare facilities to upgrade from manual or semi-automated processes to advanced automated systems that ensure safety, traceability, and standardization. As a result, regulations safeguard patient safety, reduce hospital-acquired infections (HAIs), and create continuous demand for technologically advanced washing and disinfection solutions, significantly influencing purchasing decisions and market growth.

Level of M&A Activities: Industry mergers and acquisitions are transforming the market, with top medical device companies growing bigger, reaching new markets, and rolling out advanced automation, all at a faster speed, especially in emerging regions. As these large players join forces, they lead with better innovations and greater efficiency, but this tighter market can be tough on smaller businesses facing stricter regulations and financial hurdles. Many of those businesses either look to team up or get acquired themselves, making partnerships and consolidation an ongoing part of how the sector evolves.
End User Concentration: The end-user concentration in the medical instrument washing and disinfection market centers primarily on hospitals, followed by ambulatory surgical centers, diagnostic laboratories, and specialty clinics. Hospitals account for the largest share due to their high patient inflow, complex surgical procedures, and stringent infection control requirements, driving consistent demand for automated washers and disinfectors. Ambulatory centers and diagnostic labs are emerging as important contributors as they increasingly adopt advanced disinfection solutions to meet regulatory standards and ensure patient safety. This concentration reflects the critical role of infection prevention across diverse healthcare settings.
Product Insights
The equipment segment held the largest share in 2024 and is expected to witness the fastest growth over the forecast period. This growth can be attributed to delivering automated, standardized, and highly reliable systems that ensure thorough cleaning and disinfection of reusable medical instruments. These solutions minimize human error, enhance compliance with global infection control guidelines, and support the growing shift toward centralized sterilization units in hospitals and clinics. Rising concerns over hospital-acquired infections (HAIs) and the need for scalable, efficient disinfection processes further strengthen the dominance of this segment. For instance, a 2025 report by the European Centre for Disease Prevention and Control (ECDC), over 3.5 million cases of HAIs occur annually in the EU/EEA, leading to more than 90,000 deaths, with up to 50% of these infections preventable through proper infection prevention measures, highlighting the critical role of automated disinfection equipment in reducing infection risks.
Process Stage Insights
The automated disinfection segment held the largest share in 2024 and is expected to witness the fastest growth over the forecast period. Automated disinfection and cleaning systems deliver reliable, high-quality reprocessing of reusable instruments in a way that manual methods cannot match. By reducing human error and limiting staff exposure to infectious agents, these systems protect healthcare workers and ensure that every cycle meets strict regulatory standards for sterilization and hygiene. As hospitals handle more surgeries and increasingly complex reusable devices, the demand for faster, safer, and more efficient reprocessing has made automation necessary. In fact, a 2024 WHO report noted that effective sterilization and disinfection protocols can cut hospital-acquired infections by up to 50%, highlighting just how vital automated systems are in protecting patients and improving outcomes in modern healthcare settings.
End Use Insights
The general surgery segment held the largest share in 2024. Hospitals face immense pressure from the persistent burden of healthcare-associated infections (HAIs), which remain among the most pressing global patient safety challenges. Despite advancements in infection control, HAIs continue to cause severe clinical and economic consequences, including higher morbidity, increased healthcare costs, and the spread of antimicrobial resistance. According to a July 2025 report by the NIH, roughly one in every 31 hospitalized patients has at least one HAI on any given day. The World Health Organization further estimates that out of 421 million hospital admissions worldwide each year, more than 42.7 million patients contract an HAI. Similarly, about 4.3 million hospitalized patients in the EU/EEA and nearly 687,000 patients in U.S. acute care facilities are affected annually, with the latter leading to approximately 72,000 related deaths. These alarming figures have accelerated the adoption of automated washer-disinfectors in hospitals, as they provide validated, repeatable, and traceable reprocessing cycles, helping facilities ensure compliance, sterility, and enhanced patient safety.
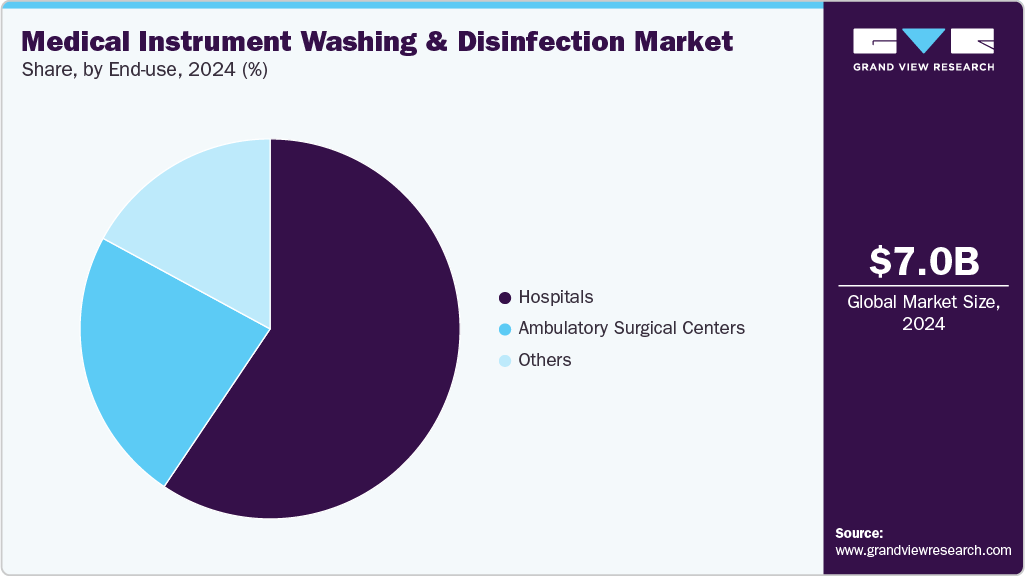
Ambulatory surgical centers segment is expected to witness the fastest CAGR over the forecast period. This growth is driven by the rapid rise in outpatient procedures and the growing shift of surgeries from hospitals to cost-efficient, specialized centers. The increasing procedure volume in ASCs fuels demand for compact, high-throughput washer-disinfectors, endoscope reprocessors, and specialty detergents that align with their space-constrained infrastructure while meeting stringent infection-control and accreditation requirements. Technological innovations such as benchtop and mid-capacity automated washers with shorter cycles, IoT-enabled compliance tracking, and energy-efficient designs are accelerating adoption in these facilities. For instance, in July 2025, several U.S. ASCs integrated single-chamber endoscope reprocessors to improve traceability and reduce turnaround time, underscoring the segment’s shift toward streamlined, digitally connected reprocessing solutions tailored to outpatient surgical workflows.
Regional Insights
North America medical instrument washing & disinfection market dominated the global market with the largest revenue share of 36.39% in 2024. The growth is driven by rising healthcare-associated infection rates, stringent infection control regulations, and the continuous modernization of healthcare infrastructure. The U.S. leads the region due to its robust regulatory framework from agencies such as the FDA, CDC, and OSHA, along with reimbursement policies from CMS that incentivize hospitals to reduce preventable infections through improved sterilization practices.
U.S. Medical Instrument Washing & Disinfection Market Trends
Medical instrument washing & disinfection market in the U.S. is primarily driven by the rising prevalence of healthcare-associated infections, increasing surgical procedure volumes, and stringent infection control regulations enforced by agencies such as the CDC and the Occupational Safety and Health Administration (OSHA). The U.S. healthcare sector faces a significant burden from HAIs, including Clostridioides difficile infections, MRSA bloodstream infections, and multidrug-resistant organisms-requiring advanced cleaning and disinfection technologies. Furthermore, the U.S. market benefits from substantial investments in healthcare infrastructure, high adoption rates of innovative solutions, and growing awareness among healthcare professionals about occupational safety, accelerating the adoption of medical instrument washing and disinfection systems. According to the National Foundation for Infectious Diseases 2023, on the overall burden of CDI in the U.S., there are an estimated 500,000 C. diff infections annually, 30,000 of which are fatal.
Europe Medical Instrument Washing & Disinfection Market Trends
The European medical instrument washing and disinfection market is witnessing steady growth, fueled by strict infection prevention protocols, a well-established regulatory framework, and the rising incidence of healthcare-associated infections. The demand for reliable and efficient disinfection systems is increasing across the region, with an aging population and a growing number of complex surgical procedures. Countries such as Germany, France, the UK, and the Nordics are actively investing in advanced washer-disinfectors to meet EN ISO 15883 standards and comply with EU MDR regulations. Ongoing hospital infrastructure upgrades and heightened awareness of patient safety are further accelerating adoption, reinforcing Europe’s strong position in the market.
Medical instrument washing and disinfection market in the UK is largely driven by stringent regulatory requirements, rising surgical volumes, and rapid technological advancements. National guidelines such as NHS Health Technical Memorandum (HTM) 01-01, Care Quality Commission (CQC) infection prevention standards, and the Health & Social Care Act’s Code of Practice on infection prevention and control mandate healthcare providers to follow validated, standardized, and auditable decontamination processes. Regular inspections and compliance audits further reinforce these requirements, prompting hospitals and independent healthcare providers to invest in advanced washer-disinfectors, ultrasonic cleaners, and high-level disinfection systems to ensure safety and regulatory adherence.
Asia Pacific Medical Instrument Washing & Disinfection Market Trends
Medical instrument washing & disinfection market in APAC is experiencing the fastest CAGR of 8.8% from 2025 to 2033. Fueled by expanding healthcare infrastructure, rising patient volumes, and increasing emphasis on infection prevention. The surge in surgical procedures, endoscopic examinations, and diagnostic interventions drives strong demand for effective reprocessing of reusable medical devices across hospitals, clinics, and diagnostic centers. Governments in countries such as India, China, Japan, and Thailand are actively modernizing healthcare systems, with investments directed toward upgrading central sterile services departments (CSSDs) and aligning with international disinfection and sterilization standards.
Medical instrument washing & disinfection market in China is driven by major healthcare infrastructure upgrades and a stronger focus on infection control. With growing emphasis on patient safety and hygiene, especially in surgical units and intensive care departments, hospitals and clinics are increasingly adopting automated washer-disinfectors to meet both national and international standards. These systems improve workflow efficiency, minimize cross-contamination risks, and promote standardized reprocessing practices, aligning with the country’s broader public health and quality-care objectives.
Latin America Medical Instrument Washing & Disinfection Market Trends
Medical instrument washing & disinfection market in Latin America is showing steady growth, driven by rising investments in healthcare infrastructure and an increasing focus on infection control. Countries such as Brazil and Argentina are modernizing their healthcare systems, leading to wider adoption of automated washer-disinfectors and standardized cleaning protocols across hospitals and clinics. The growing incidence of healthcare-associated infections, coupled with a rising volume of surgical procedures, are further accelerating the demand for reliable and effective disinfection solutions.
Middle East Africa Medical Instrument Washing & Disinfection Market Trends
Medical instrument washing & disinfection market in MEA miis experiencing moderate growth, driven by expanding healthcare infrastructure, a high burden of chronic diseases such as diabetes, and a greater emphasis on infection prevention. The region records some of the world’s highest diabetes prevalence rates, with countries such as Saudi Arabia, Kuwait, the UAE, and South Africa reporting adult rates between 17% and over 20%. As diabetic patients face higher risks of infections, surgical complications, and extended hospital stays, the demand for stringent instrument reprocessing and advanced disinfection standards to curb healthcare-associated infections (HAIs) is rising sharply.
Key Medical Instrument Washing & Disinfection Company Insights
The medical instrument washing & disinfection market is extremely fragmented, with both major and local market competitors. Due to the fact that the current market players are stepping up their efforts to grab the majority in medical instrument washing & disinfection market, fierce competition is anticipated, with the degree of competitiveness perhaps rising even higher. Many market participants are engaging in various strategic activities, such as product launches, mergers and acquisitions, and geographic growth, in an effort to gain a competitive edge over rivals. Thus, with various strategies adopted by the market players, the medical instrument washing & disinfection market is predicted to impel during the forecast period.
Key Medical Instrument Washing & Disinfection Companies:
The following are the leading companies in the medical instrument washing & disinfection market. These companies collectively hold the largest market share and dictate industry trends.
- ASP (Fortive)
- STERIS
- MELAG Medizintechnik GmbH & Co. KG
- Tuttnauer
- Olympus
- ECOLAB
- Shinva Medical Instrument Co., Ltd.
- Getinge
- Skytron, LLC
- AT-OS S.r.l.
- COLTENE Group
- Map Industries
- Spire Integrated Solutions
- Steelco S.p.A.
- Belimed
- Smeg
- TBT Medical
- DEKO MedTech Oy.
- MMM Group
- Nanosonics
- SCHLUMBOHM Medizin-Labor-Technologie-Hamburg GmbH
- KEN Hygiene Systems
- Germitec
- CS Medical LLC
- Metrex Research, LLC
- CIVCO Medical Solutions
- Tristel Plc
- IMA-X (EDM Medical Solutions)
- Lumicare
- UV Smart
- Ruhof
- Sklar
- Oro Clean Chemie AG
- Solventum
Recent Developments
-
In February 2025, Germitec, one of the prominent MedTech companies specializing in UV-C High-Level Disinfection solutions, announced the successful completion of a USD 30 million financing round. This funding is expected to support the company's efforts to accelerate growth in the U.S. market and further advance innovations in UV-C infection prevention technologies.
-
In March 2024, Getinge's launch of a new, intuitive, high-performance multi-chamber washer-disinfector is a significant development in the infection control sector, aimed at addressing the need for efficient, high-capacity cleaning in healthcare facilities. Multi-chamber washer disinfectors are particularly valuable in hospitals and large medical centers where large volumes of surgical instruments and medical equipment must be sterilized quickly and effectively.
-
In June 2024, Metall Zug and Miele officially finalized their joint venture, merging Steelco and Belimed in the fields of Infection Control and Life Science. This significant industry milestone has resulted in the creation of a new brand: SteelcoBelimed. This strategic alliance offers a unique opportunity for both companies to strengthen their market presence and accelerate innovation, aiming to become the most customer-focused providers in Infection Control and Life Science. The joint venture between Belimed and Steelco also marks an important step in meeting the rising demands of the global Healthcare and Life Sciences sectors. Complementing each other in markets, products, and expertise, both companies manufacture cleaning, disinfection, and sterilization solutions for hospitals, biomedical research, and pharmaceutical and biotechnological applications.
-
In April 2024, SHINVA Medical Instrument Co., Ltd. signed a Memorandum of Understanding (MOU) with Genoray, a South Korean company specializing in imaging systems, to expand its cooperation in the Chinese healthcare market. This MOU helps SHINVA establish a stronger foothold in healthcare facilities, positioning the company as a trusted partner for medical imaging and infection control solutions. This can drive the adoption of SHINVA’s washer-disinfectors.
Medical Instrument Washing & Disinfection Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 7.36 billion
Revenue forecast in 2033
USD 11.82 billion
Growth rate
CAGR of 6.1% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, process stage, end use, and region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa (MEA)
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; Kuwait; UAE.
Key companies profiled
ASP (Fortive); STERIS; MELAG Medizintechnik GmbH & Co. KG; Tuttnauer; Olympus; Ecolab; Shinva Medical Instrument Co. Ltd.; Getinge; Skytron LLC; AT-OS S.r.l.; COLTENE Group; Map Industries; Spire Integrated Solutions; Steelco S.p.A.; Belimed; Smeg; TBT Medical; DEKO MedTech Oy.; MMM Group; Nanosonics; SCHLUMBOHM Medizin-Labor-Technologie-Hamburg GmbH; KEN Hygiene Systems; Germitec; CS Medical LLC; Metrex Research LLC; CIVCO Medical Solutions; Tristel Plc; IMA-X (EDM Medical Solutions); Lumicare; UV Smart; Ruhof; Sklar; Oro Clean Chemie AG; Solventum
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Medical Instrument Washing & Disinfection Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global medical instrument washing & disinfection market report based on product, process stage, end use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Chemistries
-
Enzymatic Cleaners
-
Detergents
-
HLD Chemicals
-
-
Equipment
-
Washer-disinfectors
-
Ultrasonic Cleaners
-
UV-C Disinfection Systems
-
Other Non-chemical Disinfection Systems
-
-
-
Process Stage Outlook (Revenue, USD Million, 2021 - 2033)
-
Manual Cleaning
-
Pre-cleaning
-
Automated Disinfection
-
UV-C Disinfection
-
Thermal Disinfection
-
Chemical Disinfection
-
-
Rinse/Drying
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Ambulatory Surgical Centers
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global medical instrument washing & disinfection market size was estimated at USD 7.01 billion in 2024 and is expected to reach USD 7.36 billion in 2025.
b. The global medical instrument washing & disinfection market is expected to grow at a compound annual growth rate of 6.10% from 2025 to 2033 to reach USD 11.82 billion by 2033.
b. North America dominated the medical instrument washing & disinfection market with a share of 36.39% in 2024. This dominance is attributed to the well-established healthcare infrastructure, high adoption of advanced sterilization and disinfection technologies, and stringent infection control regulations set by agencies such as the U.S. FDA and CDC.
b. Some key players operating in the medical instrument washing & disinfection market include ASP (Fortive), STERIS, MELAG Medizintechnik GmbH & Co. KG, Tuttnauer, Olympus, Ecolab, Shinva Medical Instrument Co., Ltd., Getinge, Skytron LLC, AT-OS S.r.l., COLTENE Group, Map Industries, Spire Integrated Solutions, Steelco S.p.A., Belimed, Smeg, TBT Medical, DEKO MedTech Oy., MMM Group, Nanosonics, SCHLUMBOHM Medizin-Labor-Technologie-Hamburg GmbH, KEN Hygiene Systems, Germitec, CS Medical LLC, Metrex Research LLC, CIVCO Medical Solutions, Tristel Plc, IMA-X (EDM Medical Solutions), Lumicare, UV Smart, Ruhof, Sklar, Oro Clean Chemie AG, and Solventum.
b. Key factors that are driving the market growth include the increasing prevalence of hospital-acquired infections, rising surgical procedure volumes, and the growing adoption of advanced automated reprocessing systems that enhance efficiency and compliance with stringent infection control standards. Factors such as expanding healthcare infrastructure, technological advancements in washer-disinfectors and sterilization systems, and supportive regulatory frameworks across major markets fuel demand.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










