- Home
- »
- Medical Devices
- »
-
Middle East Medical Device Testing Services Market Report, 2033GVR Report cover
![Middle East Medical Device Testing Services Market Size, Share & Trends Report]()
Middle East Medical Device Testing Services Market (2025 - 2033) Size, Share & Trends Analysis Report By Service (Biocompatibility Tests, Chemistry Test, Microbiology & Sterility Testing, Package Validation), By Phase, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-725-0
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
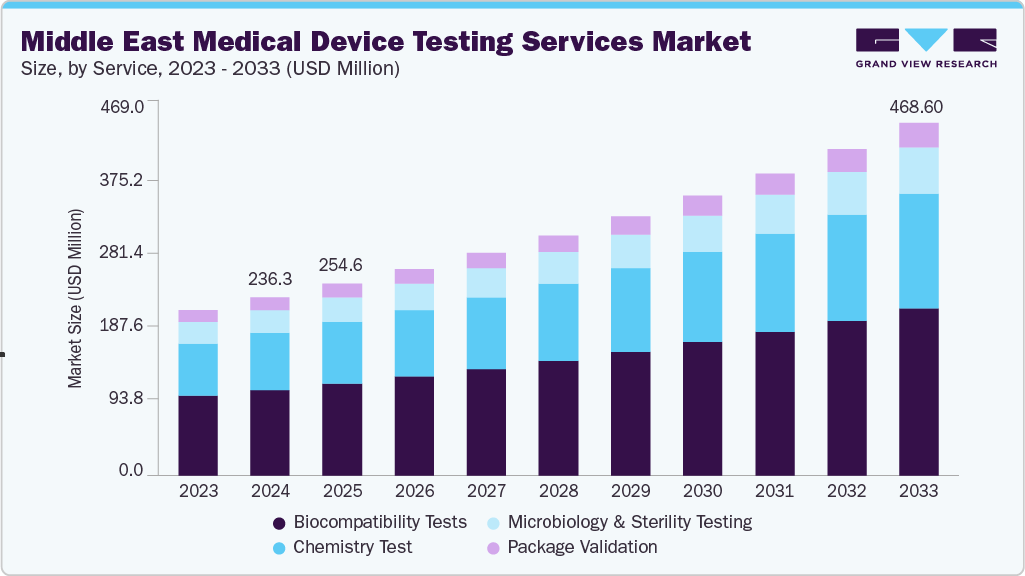
Middle East Medical Device Testing Services Market Summary
The Middle East medical device testing services market size was estimated at USD 236.31 million in 2024 and is expected to grow at a CAGR of 7.93% from 2025 to 2023. The growth is driven by the tightening of regulatory frameworks such as Saudi Arabia’s SFDA Medical Devices Interim Regulation and the UAE’s updated conformity assessment procedures, which are increasing the volume and complexity of testing requirements. Expansion of regional healthcare infrastructure, particularly in Saudi Arabia and the UAE, is boosting the demand for certified and compliant medical devices.
In addition, rising healthcare infrastructure investments and increasing localization of manufacturing is also one of the factors contributing to the market growth. Countries such as Saudi Arabia and the UAE have been reinforcing device approval pathways through more stringent conformity assessments, pushing manufacturers to seek accredited testing for safety, efficacy, and biocompatibility. The expansion of domestic production capabilities in Saudi Arabia, Turkey, and Egypt is also amplifying the need for in-region testing to reduce turnaround times and comply with both domestic and export market requirements. Moreover, the growing prevalence of chronic diseases such as cardiovascular disorders and diabetes is spurring demand for advanced devices, which typically require complex analytical validation before market entry.
The Middle East medical device testing services industry’s growth is further driven by the region’s strategic role as a medical technology distribution hub for Africa and South Asia, making rapid and compliant testing essential for cross-border trade. Increasing adoption of cutting-edge technologies, including in-vitro diagnostics, wearable health monitors, and minimally invasive surgical devices, is raising the bar for performance and safety evaluation. Moreover, the emergence of specialized testing facilities within free zones, coupled with public-private partnerships, is streamlining regulatory approvals and fostering investor confidence. These factors position the Middle East as a growing center for medical device testing services, offering both regional and international players a faster route to commercialization.
Technological Advancements
Advances in automation, artificial intelligence, and simulation modeling are transforming medical device testing efficiency in the region. High-throughput analytical systems are enabling faster turnaround for chemical and mechanical tests, while AI-based image analysis is improving the accuracy of wear-and-tear assessments. Non-destructive testing (NDT) technologies, such as advanced imaging and digital twin simulations, are reducing device failure risks before clinical deployment. Cloud-based data management systems are streamlining regulatory submissions to authorities like the Saudi SFDA and UAE MOHAP. Moreover, the introduction of portable test equipment is expanding point-of-origin testing, allowing smaller manufacturers and importers to conduct preliminary checks before engaging full laboratory certification.
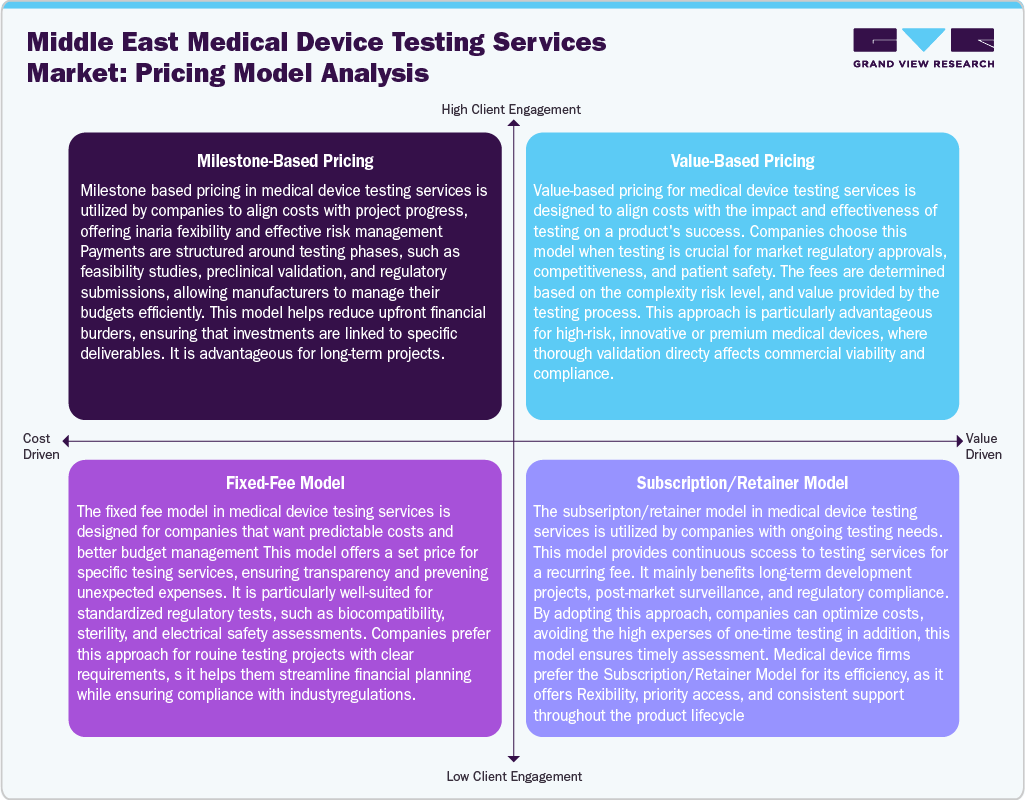
Pricing Model Analysis
Pricing in the Middle East medical device testing services market varies by test complexity, regulatory pathway, and turnaround time. Basic safety and performance tests are typically charged on a per-test basis. At the same time, more specialized evaluations, such as biocompatibility or EMC testing, often follow a project-based quotation due to equipment and expertise requirements. Accelerated testing timelines—especially for urgent market launches—command premium rates, sometimes 25-40% higher than standard lead times. Subscription-based models, though rare, are emerging for repeat clients with large annual test volumes, providing cost predictability and priority access. Free-zone testing facilities in the UAE and Oman occasionally offer reduced fees to attract regional clients, though international accreditation costs can offset these savings. Overall, pricing competition is moderate, as the limited number of accredited facilities allows service providers to maintain healthy margins, particularly in high-specialization segments.
Service Insights
The biocompatibility tests segment led the Middle East medical device testing services industry with the largest revenue share of 47.95 % in 2024. The biocompatibility test is crucial in the medical devices industry for bringing devices to market. The test is a critical step for medical device development where an assessment of biocompatibility helps medical device manufacturers ensure the device’s safety for patients & provides data that can be used to support regulatory compliance. In addition, stringent regulations, technological advancements, and a focus on personalized healthcare are expected to boost the growth of biocompatibility testing within the medical device testing services market.
The chemistry test segment is expected to grow at a lucrative CAGR during the forecast period. Chemistry tests support the medical device industry throughout the value chain. These tests ensure that medical devices do not cause reactions when in contact with the human body. The tests check if any solutes and/or chemicals are present on the surface of medical devices, which may leach out into the surroundings when used with the intended liquids.
Phase Insights
The clinical segment accounted for the largest share of the Middle East medical device testing services market in 2024. The segment growth is driven by a rise in the product pipeline and continuous upgradation of regulatory standards. Growing emphasis on clinical evidence for new device approvals accelerates the demand for robust testing services during clinical trials. Moreover, the growing preference for market participants that offer advanced testing methodologies with affordable cost & reduced turnaround time for diverse clinical studies is expected to drive segment growth.
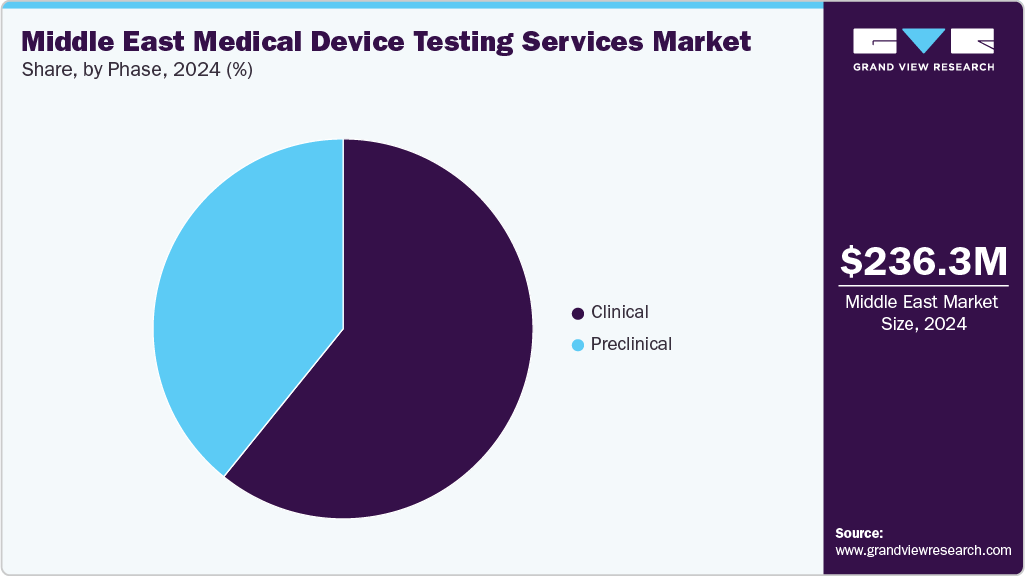
The preclinical market segment is expected to grow at the fastest CAGR over the forecast period. The preclinical is further segment into small animals, large animals, and others. Preclinical testing is crucial in the development & evaluation of medical devices before they are approved for human use. Leading CROs like Charles River Laboratories provide biocompatibility, safety testing, and pathology services during this stage. For devices and high-risk medical devices that lack appropriate clinical data, preclinical tests are conducted to demonstrate compliance with the requirements for CE marking. These tests determine the efficacy, safety, and performance of the devices to mitigate potential risks & enhance patient outcomes.
Country Insights
The medical device testing services market in Saudi Arabia held the largest share of the Middle East in 2024. This can be attributed to the country’s robust regulatory framework, expanding healthcare infrastructure, and increasing domestic manufacturing capabilities. The Saudi Food and Drug Authority (SFDA) has established comprehensive medical device regulations that require stringent conformity assessment, driving demand for accredited testing services within the country. Large-scale investments under Vision 2030, including the development of specialized healthcare cities and manufacturing zones, have created a strong base for local and foreign manufacturers to conduct in-region testing rather than relying on overseas facilities.
The UAE medical device testing services market is anticipated to grow significantly over the forecast period. The country's market growth is due to due to the country’s strong positioning as a regional hub for medical technology trade and regulatory compliance. The presence of free zones such as Dubai Science Park and Dubai Healthcare City, coupled with favorable business policies, has attracted multinational device manufacturers and testing service providers.The Ministry of Health and Prevention (MOHAP) and the Department of Health Abu Dhabi (DoH) have been aligning regulatory standards with benchmarks, increasing the demand for advanced performance, safety, and quality testing.
The Medical Device Testing Services market in Kuwait held a significant share in 2024. The country's growth is due to the country’s high healthcare expenditure per capita, strong reliance on imported medical technologies, and a well-developed hospital network that demands rigorous compliance checks. Kuwait’s Ministry of Health mandates that all imported medical devices meet international safety and performance standards, which drives the need for accredited testing and certification services.
Key Middle East Medical Device Testing Services Company Insights
Market players are undertaking various strategic initiatives, such as the launch of new product partnerships, collaborations, and mergers & acquisitions, to strengthen their service portfolio and provide a competitive advantage. For instance, in January 2025, STEMart announced the expansion of its medical device testing capabilities by introducing Balloon Catheter Testing Services. These new testing services are designed to help ensure the development of safe medical devices, meeting the industry's compliance requirements and assisting manufacturers in minimizing compliance risks.
Key Middle East Medical Device Testing Services Companies:
- SGS SA
- Laboratory Corporation of America Holdings
- Nelson Laboratories, LLC
- TÜV SÜD
- Charles River Laboratories
- Element Minnetonka
- Eurofins Scientific
- Pace Analytical Services LLC
- Intertek Group Plc
Recent Developments
-
In February 2025,Intertek announced the opening of its regional headquarters in Riyadh, Saudi Arabia. This new hub will play a key role in supporting Intertek's expanding portfolio of services and solidifying its commitment to customers in the region, aligning with Saudi Arabia’s Vision 2030.
-
In March 2024, Element Materials Technology launched its Rapid Response Pharmaceutical Testing Service from its Manchester Life Sciences laboratory. This offering provides expedited and comprehensive testing solutions for customers worldwide, including those in the Middle East.
Middle East Medical Device Testing Services Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 254.57 million
Revenue forecast in 2033
USD 468.60 million
Growth rate
CAGR of 7.93% from 2025 to 2033
Historical year
2021 - 2024
Forecast period
2025 - 2023
Quantitative units
Revenue in USD million and CAGR from 2025 to 2023
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Service, phase, region
Country scope
Saudi Arabia; UAE; Kuwait; Oman; Qatar
Key companies profiled
SGS SA; Nelson Laboratories, LLC; Laboratory Corporation of America Holdings; TÜV SÜD; Charles River Laboratories; Eurofins Scientific; Element Minnetonka; Pace Analytical Services LLC; Intertek Group Plc
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Middle East Medical Device Testing Services Market Report Segmentation
This report forecasts revenue growth at the regional and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Middle East medical device testing services market report based on service, phase, and region:
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Biocompatibility Tests
-
Cardiovascular Device's Biocompatibility Tests
-
Orthopedic Device's Biocompatibility Tests
-
Dental Implant Devices' Biocompatibility Tests
-
Dermal Filler's Biocompatibility Tests
-
General Surgery Implantation Devices Biocompatibility Tests
-
Neurosurgical Implantation Devices Biocompatibility Tests
-
Ophthalmic Implantation Device's Biocompatibility Tests
-
Others
-
-
Chemistry Test
-
Chemical characterization (E&L)
-
Analytical method development and validation
-
Toxicological Risk Assessment and consulting
-
-
Microbiology & Sterility Test
-
Bioburden Determination
-
Pyrogen & Endotoxin Testing
-
Sterility Test & Validation
-
Antimicrobial Testing
-
Others
-
-
Package Validation
-
-
Phase Outlook (Revenue, USD Million, 2021 - 2033)
-
Preclinical
-
Large animal research
-
Biocompatibility Tests
-
Chemistry Test
-
Microbiology & Sterility Test
-
-
Small animal research
-
Biocompatibility Tests
-
Chemistry Test
-
Microbiology & Sterility Test
-
-
-
Clinical
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
Middle East
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
-
Frequently Asked Questions About This Report
b. The Middle East medical device testing services market size was estimated at USD 236.31 billion in 2024 and is expected to reach USD 254.57 billion in 2025.
b. The Middle East medical device testing services market is expected to grow at a compound annual growth rate of 7.93% from 2025 to 2033 to reach USD 468.60 billion by 2033.
b. Biocompatibility Tests captured the highest market in the Middle East medical device testing services market with a share of 47.95% in 2024. This is attributable to the rising demand for safe and reliable implantable devices, increasing regulatory emphasis on ISO 10993 standards, and growing adoption of advanced biomaterials across medical applications.
b. Some key players operating in the Middle East medical device testing services market include SGS SA, Nelson Laboratories, LLC, Laboratory Corporation of America Holdings, TÜV SÜD, Charles River Laboratories, Eurofins Scientific, Element Minnetonka, Pace Analytical Services LLC, Intertek Group Plc
b. Key factors that are driving the market growth include tightening of regulatory frameworks such as Saudi Arabia’s SFDA Medical Devices Interim Regulation and the UAE’s updated conformity assessment procedures, which are increasing the volume and complexity of testing requirements.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










