- Home
- »
- Medical Devices
- »
-
Middle East Pharmaceutical Contract Development & Manufacturing Organization Market Report, 2033GVR Report cover
![Middle East Pharmaceutical Contract Development & Manufacturing Organization Market Size, Share & Trends Report]()
Middle East Pharmaceutical Contract Development & Manufacturing Organization Market (2025 - 2033) Size, Share & Trends Analysis Report By Type, By Product, By Service, By Workflow, By Therapeutic Area, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-756-9
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Middle East Pharmaceutical Contract Development & Manufacturing Organization Market Summary
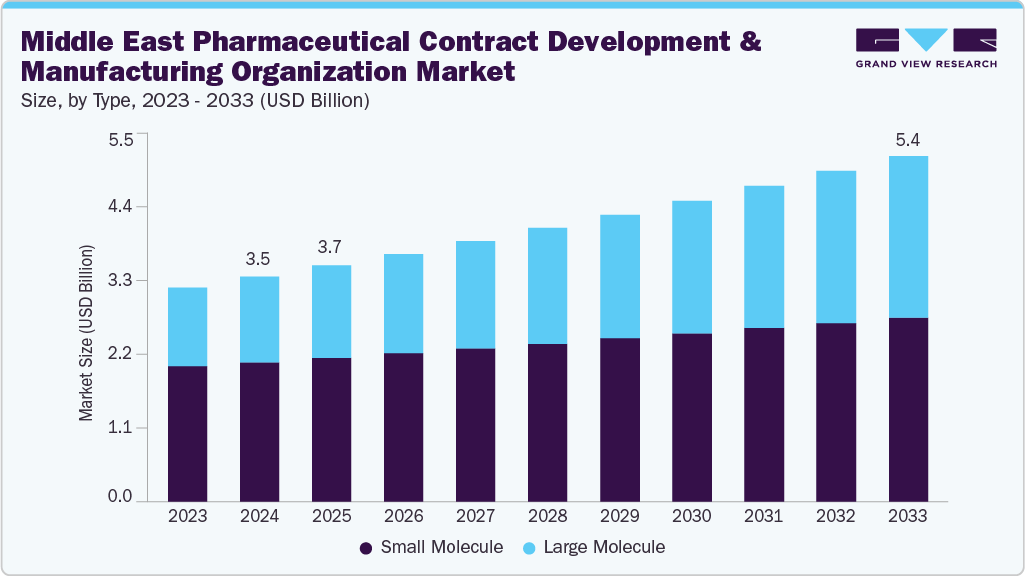
The Middle East pharmaceutical contract development and manufacturing organization market size was estimated at USD 3.50 billion in 2024 and is projected to reach USD 5.39 billion by 2033, growing at a CAGR of 4.90% from 2025 to 2033. The market growth is driven by increasing healthcare spending, rising prevalence of chronic diseases, and a growing demand for cost-effective drug development and manufacturing facilities.
Key Market Trends & Insights
- The pharmaceutical contract development and manufacturing organization in the UAE is expected to grow significantly from 2025 to 2033.
- By workflow, the commercial segment held the highest market share in 2024.
- By type, the small molecule segment led the market with the largest revenue share of 61.92% in 2024.
- By product, the API segment led the market with the largest revenue share in 2024.
- By service, the contract manufacturing segment led the market with the largest revenue share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 3.50 Billion
- 2033 Projected Market Size: USD 5.39 Billion
- CAGR (2025-2033): 4.90%
In addition, growing government investments in healthcare infrastructure and the presence of strategic locations in the Middle East as a trade hub that connects Asia, Europe, and Africa have further contributed to market growth. Moreover, most of the established pharmaceutical companies across the globe are expanding operations in the Gulf and broader Middle East with the shifting trends of outsourcing towards contract development & manufacturing organization (CDMO) to optimize costs, accelerate time-to-market, and enhance supply chain resilience.Moreover, rising technological advancements are enhancing both efficiency & quality within pharmaceutical development and manufacturing is expected to drive the market. Besides, the integration of automation, digital platforms, and advanced analytical tools has streamlined operations further, minimizing errors. For instance, processes such as continuous manufacturing and single-use technologies are increasingly used to shorten production timelines and provide flexibility for both small and large molecule products. In addition, data-driven monitoring systems & real-time analytics have been implemented to ensure greater precision, compliance, and productivity across the entire value chain. Furthermore, in the Middle East, pharmaceutical innovation is driven by an expanding pipeline of biologics, biosimilars, and specialty medicines. CDMOs are stepping up to offer their expertise in complex formulations, sterile manufacturing, and the production of HPAPIs.
In addition, growing collaborative partnerships among global and emerging market players are expected to enhance the capabilities in the Middle East for biologics, cell and gene therapies, and specialized therapeutic areas. In addition, the growing expansion of R&D activities in the region and increasing requirement for outsourcing facilities due to evolving demands of the pharmaceutical pipeline are expected to drive the market over the estimated time period.
Also, the regulatory landscape in the region is more aligned with the international standards to attract investment and prioritize patient safety. Authorities such as the Saudi Food and Drug Authority & the UAE Ministry of Health and Prevention are further enhancing quality assurance and compliance frameworks. Besides, increasing requirements to harmonize regulatory guidelines with those from the U.S. FDA and EMA are underway to facilitate global market access for regionally manufactured drugs. Furthermore, growing approval processes and transparent regulatory pathways that are established to support CDMOs in maintaining competitiveness and building credibility within global pharmaceutical supply chains are expected to drive the market over the estimated time period.
Opportunity Analysis
Growing healthcare investments drive the Middle East pharmaceutical contract development and manufacturing organization market’s growth, the rising prevalence of diseases, and the emerging demand for therapies. Moreover, in the Middle East, pharmaceutical companies are increasingly shifting towards outsourcing facilities such as CDMOs to minimize costs, accelerate product development, and ensure regulatory compliance.
In addition, as the GCC countries establish themselves as regional healthcare hubs, the outsourcing model is expected to gain further traction, particularly in biologics, biosimilars, and specialty drugs.
Moreover, the emerging technological opportunity involves advanced manufacturing platforms, single-use systems, and digitalized quality monitoring, which are expected to enhance market productivity. Besides, CDMOs in the Middle East further embrace automation, continuous manufacturing, and data-driven solutions, and are well-positioned to meet the rising demand for efficiency & scalability, creating a competitive edge in the region. In addition, increasing the drug pipeline, especially in oncology, rare diseases, and personalized medicines, further strengthens the role of CDMOs. Furthermore, growing collaborations with pharmaceutical and local companies will create new growth opportunities for complex formulations and high-value therapies. Regulatory reforms, harmonization with global standards, and government support for local manufacturing create a favorable environment. These developments position the Middle East as an attractive destination for CDMO partnerships and long-term investments.
Impact of U.S. Tariffs on the Middle East Pharmaceutical Contract Development And Manufacturing Organization Market
The imposition of U.S. tariffs has influenced the global market, with effects extending to the Middle East. Trade barriers in the region have increased costs for raw materials, APIs, & equipment sourced from the U.S. and other regions. This has led pharmaceutical companies to focus on cost optimization by outsourcing CDMOs in the Middle East. Moreover, the tariff impact has created new opportunities for market players by offering competitive pricing and localized production. Furthermore, at the same time, the supply chain diversification strategies have been accelerated to reduce dependency on U.S.-linked imports, positioning the Middle East as a strategic alternative for global pharmaceutical manufacturing and development partnerships. Thus, the tariff impact is expected to offer new opportunities for the Middle East pharmaceutical contract development and manufacturing organization industry.
Technological Advancements

The market is driven by rising technological innovations that support enhancing operational efficiency, quality standards, & competitive positioning. Continuous manufacturing plays a crucial role in the market, allowing the manufacturer to achieve shorter production timelines, scalability, and consistent quality, making regional CDMOs attractive partners for both small- and large-molecule products. In addition, adopting single-use technologies increases flexibility, reduces cleaning validation costs, and minimizes contamination risks, specifically for biologics and sterile manufacturing areas, further contributing to market growth. Moreover, integrating advanced analytical tools, including chromatography, spectroscopy, and MS, further strengthens compliance frameworks while enabling real-time monitoring & improved decision-making for the development lifecycle. In addition, automation & digitalization are emerging in the region as they streamline workflows, reduce human error, and enhance supply chain visibility, improving productivity and lowering costs. Furthermore, growing investments in biologics and HPAPI capabilities are broadening the region's capacity to deliver high-value therapeutics for oncology, rare diseases, and personalized medicine. Thus, these advancements are expected to strengthen the Middle East's pharmaceutical outsourcing market.
Pricing Model Analysis
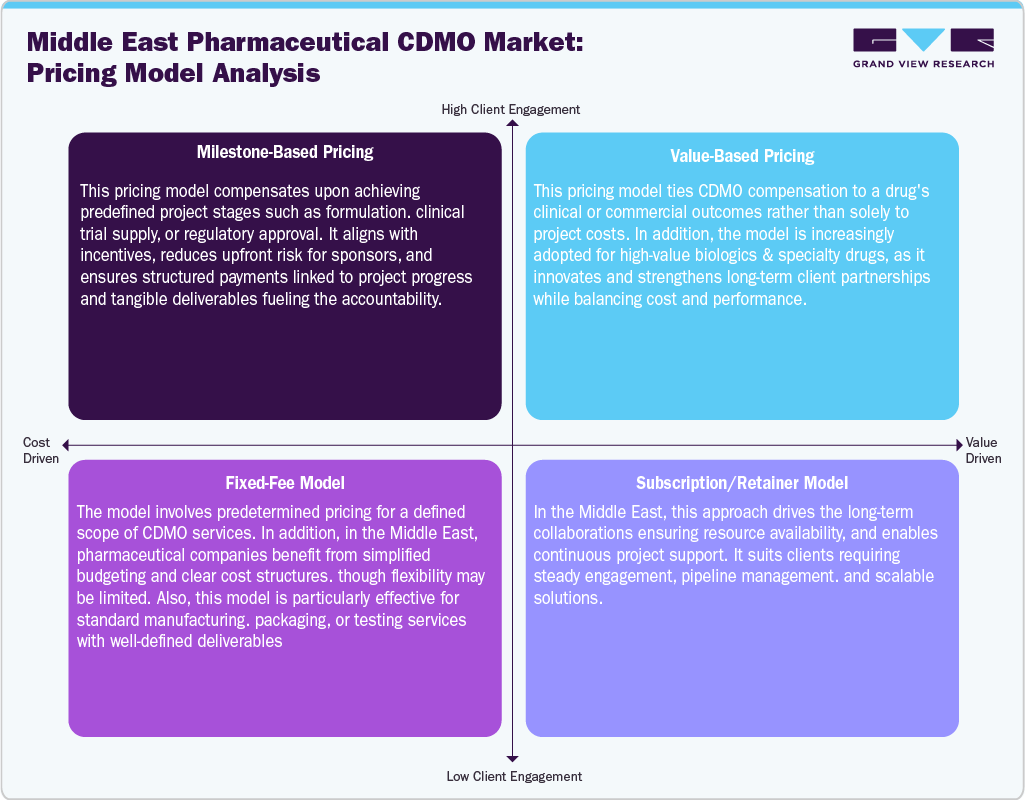
The Middle East pharmaceutical contract development and manufacturing organization market adopts diverse pricing models to meet the project and client requirements. Milestone-based pricing focuses on cost linked to formulation, clinical trials, & regulatory approval, ensuring accountability & risk-sharing. Besides, value-based pricing is emerging due to the growing need for biologics and specialty drugs, wherein the costs are aligned with clinical or commercial outcomes, rewarding innovation, and strengthening long-term partnerships. In addition, the fixed-fee model offers transparency & cost predictability, making it suitable for standardized services like manufacturing, packaging, and quality testing. However, it provides less flexibility for complex projects. Furthermore, pipeline development projects increasingly prefer the subscription model as it offers consistent support, resource availability, and scalable collaboration. Thus, these pricing models support meeting the client expectations, improving cost efficiency, and reinforcing their role as strategic outsourcing partners.
Market Concentration & Characteristics
The Middle East pharmaceutical CDMO market growth stage is moderate, and growth is accelerating. The market is characterized by the degree of innovation, level of M&A activities, regulatory impact, service expansion, and regional expansion.
The Middle East pharmaceutical CDMO industry is experiencing a rising degree of innovation, driven by biologics, biosimilars, and high-potency drug manufacturing investments. Adopting advanced technologies such as continuous manufacturing, single-use systems, and real-time analytics transforms efficiency and quality standards. In addition, growing collaboration, R&D initiatives, and global partnerships further drive the Middle East CDMOs as competitive players in addressing complex therapeutic needs and supporting evolving pharmaceutical pipelines.

Mergers and acquisitions in the Middle East pharmaceutical CDMO market are expected to be moderate; however, they are gradually increasing as global players seek regional entry and local companies pursue scale. In addition, cross-border alliances and equity investments are expanding service portfolios and technical capabilities. These activities enhance competitiveness, enable knowledge transfer, and position the region as an emerging outsourcing hub.
The regulatory environment in the Middle East is evolving with international standards ensuring compliance and patient safety. In addition, authorities such as the SFDA and the UAE’s Ministry of Health are streamlining the processes in the region. Harmonization with U.S. FDA and EMA guidelines facilitates global market access, enhances credibility, and encourages CDMO investment in high-quality manufacturing.
CDMOs in the Middle East are broadening their service portfolios to meet rising demand across the drug development lifecycle. Besides, biologics, sterile injectables, and HPAPI manufacturing capabilities are being prioritized, contributing to the market. In addition, expanded clinical trial support, packaging, and analytical testing offerings are also being integrated, enabling regional CDMOs to deliver end-to-end solutions and attract global pharmaceutical partnerships.
Substantial investments from GCC countries in healthcare infrastructure and manufacturing support regional expansion within the Middle East market. Saudi Arabia & the UAE are emerging as key hubs attracting collaborations and fueling the local production. In addition, growing expansion in the market is expected to enhance manufacturing, diversify services, and build strategic partnerships across the Asia Pacific, Europe, and Africa.
Type Insights
In 2024, the small molecule segment held the largest share of the Middle East pharmaceutical contract development and manufacturing organization market. The market for small molecules is driven by increasing demand for cost-effective manufacturing and faster time-to-market. Besides, regional CDMOs are expanding formulation, chemical synthesis, and analytical testing capabilities, further supporting the range of therapeutic areas. In addition, growing adoption of advanced process technologies, automation, and regulatory compliance frameworks has further enabled higher efficiency and quality. Moreover, the presence of established and local pharmaceutical companies has further facilitated access to specialized expertise, while government initiatives & infrastructure investments promote local production. These developments position the region as a competitive hub for small-molecule drug development and manufacturing.
The large molecule segment is expected to grow at the fastest CAGR during the forecast period. In the Middle East, large molecules are driven by increasing focus on biologics, biosimilars, and high-potency therapeutics. Besides, regional players are increasingly investing in single-use systems, bioreactors, and advanced analytical platforms to ensure process scalability, product quality, and regulatory compliance. In addition, growing collaboration with established pharmaceutical companies further supports innovation in complex formulations and specialized therapies, positioning the Middle East as a growing hub for biologics and large-molecule development and manufacturing solutions. Such factors are anticipated to drive the segment over the estimated time period.
Product Insights
The API segment dominated the Middle East pharmaceutical contract development and manufacturing organization industry with the largest revenue share in 2024. Rising healthcare investments, local manufacturing initiatives, and a growing demand for generic and specialty drugs drive the segment’s growth. In addition, the growing emphasis of governments across the region, specifically in Saudi Arabia and the UAE, to create the region as a pharmaceutical self-sufficiency, further fuels partnerships with global CDMOs for technology transfer and high-quality API production. Moreover, with the increasing prevalence of chronic diseases & supportive regulatory frameworks, the region is anticipated to present strong opportunities for CDMOs to establish API capabilities, further enhance regional supply security, and minimize the import dependency. Such factors are expected to drive the segment over the estimated time period.
The drug product segment is expected to grow at the fastest CAGR during the forecast period. The drug segment is driven by growing new opportunities for outsourcing domains and rising interest from multinational and local companies for drug product innovations in the region, which is expected to drive the market. In the region, many global pharmaceutical companies are implementing strategies to strengthen the drug product manufacturing and partnerships, further contributing to segment growth. Moreover, global companies are implementing strategies to enhance localized drug product manufacturing & partnerships by adapting business models, tailoring capabilities to local requirements, and committing to long-term localization.
Furthermore, growing investments in the drug products value chain enhance infrastructure, establish local production of innovative and essential medicines, upgrade technology and workforce skills, and expand clinical trial activities, which are anticipated to drive the market over the estimated time period. In addition, growing opportunities in public-private partnerships for generics and branded generics, aimed at meeting the needs of a growing middle class and underserved rural populations, support the market. The region’s large patient base, including those with genetic and hereditary diseases, further highlights unmet demand and creates new growth avenues for drug product companies to deliver targeted, affordable, and innovative therapeutic solutions.
Service Insights
The contract manufacturing segment accounted for the largest share of the Middle East pharmaceutical CDMO industry in 2024. In the Middle East, contract manufacturing has enabled pharmaceutical and biotechnology companies to outsource drug production, reduce capital expenditure, and accelerate time-to-market, further contributing to market growth. In addition, the segment is driven by a surge in government spending for healthcare initiatives and the growing burden of diseases such as obesity, heart disease, and chronic conditions due to an ageing population, urbanization, and lifestyle changes, which has led to a rising need for a range of therapies in the region. In addition, contract manufacturing offers small and large molecule synthesis, formulation, sterile fill-finish, and packaging services under strict quality and regulatory standards, which further drives the segment's growth. Furthermore, CDMOs support established and local companies in meeting growing regional healthcare demand while ensuring compliance, reliability, and cost-effectiveness in domestic and international markets.
The contract development segment is expected to grow at the fastest CAGR during the forecast period. The segment is driven by rising innovative drug development, supported by advanced research facilities and strong intellectual property protection. In addition, growing adoption of contract development in the region is driven by an evolving ecosystem, where localized partnerships, regulatory maturity, and infrastructure investments drive innovation in the market. Moreover, with the expansion of CDMOs, the pharmaceutical companies are optimizing costs, accelerating market entry, and enhancing pipelines, further enabling sustainable growth, ensuring improved access to advanced therapeutics across regional and global markets.
Workflow Insights
The commercial segment dominated the Middle East pharmaceutical CDMO market with the largest revenue share in 2024. The segment’s growth is driven by increasing pharmaceutical outsourcing, growing investments in production facilities, technological advancements, and rising advanced operations techniques, which support the segment's growth. Besides, the increasing number of small- and medium-sized pharmaceutical companies and growing adoption of CDMOs in the region have further led to rising pharmaceutical commercial drug production, leading to higher profitability in the market.
In addition, rising end-to-end solutions, including quality control, regulatory compliance, and supply chain management, have led CDMOs in the region to enable efficient market entry and product availability. Moreover, investments in automation, continuous manufacturing, modern facilities, and adherence to international standards are anticipated to support market growth. Furthermore, growing pharmaceutical companies' collaboration further facilitates technology advancements and scale-up, contributing to rising demand for generics, specialty drugs, and biologics, positioning the Middle East as a strategic hub for pharmaceutical commercialization.
On the other hand, the clinical segment is projected to grow at a significant CAGR during the forecast period. The segment growth is primarily attributed to a rising requirement for clinical services supporting formulation development, GMP manufacturing for clinical trials, and analytical testing. Besides, growing expertise for efficient clinical trials has led to accelerated timelines, reducing costs and producing high-quality, reliable outputs. These services enhance the Middle East's role in global drug development, further facilitating faster clinical trial completion and contributing to therapeutic innovations. Such factors are expected to drive the market over the estimated time period.
Therapeutic Area Insights
The oncology segment accounted for the largest market share in 2024. Oncology-focused CDMO services are driven by rising cancer prevalence and demand for advanced therapies. CDMOs provide end-to-end solutions, including small and large molecule synthesis, biologics manufacturing, sterile fill-finish, and analytical support. In addition, rising adoption of single-use bioreactors, automation, and AI-driven analytics further drives the high-quality production. Partnerships with global pharmaceutical companies enable clinical and commercial supply of oncology drugs. Middle Eastern CDMOs are positioning themselves as critical players in regional and international oncology drug development and manufacturing by supporting innovative cancer therapies. Such factors are expected to drive the market over the estimated time period.
The autoimmune diseases segment is expected to grow at the fastest CAGR during the forecast period. The CDMOs in the region are strategically positioning themselves to support the development and production of therapies targeting autoimmune diseases. Their service offerings encompass biologics, small molecule manufacturing, and comprehensive analytical testing and regulatory compliance support. In addition, rising investments in continuous manufacturing and automation further enhance operational efficiency and product quality. Moreover, collaborations with global pharmaceutical companies drive production timelines, ensuring the timely availability of clinical and commercial products. Thus, the growing capabilities in the region support CDMOs to effectively address autoimmune diseases and enhance the Middle East's reputation as an emerging hub for specialized therapeutic manufacturing. Such factors are expected to drive the market.
End Use Insights
The large pharmaceutical companies segment accounted for the largest share of the Middle East pharmaceutical contract development and manufacturing organization industry in 2024. The segment is experiencing new growth opportunities as most of the pharmaceutical companies are increasingly opting to outsource their production needs. Besides, the rising focus on cost optimization fuels this trend, helping to speed up time-to-market and providing access to specialized expertise that is not available in-house. In addition, with the growing complexity of biologics, cell and gene therapies, and advanced formulations, the CDMOs are anticipated to offer flexibility and scalability to their partners. Moreover, global pharmaceutical leaders are forming strategic alliances with CDMOs to concentrate on their core research and development initiatives while taking advantage of external manufacturing capabilities. In addition, growing consolidation among CDMOs, the adoption of digital technologies, and substantial investments in high-value biologics capabilities are expected to support the market over the estimated period. Thus, CDMOs support large pharmaceutical companies as critical partners in maintaining competitiveness and driving innovation.
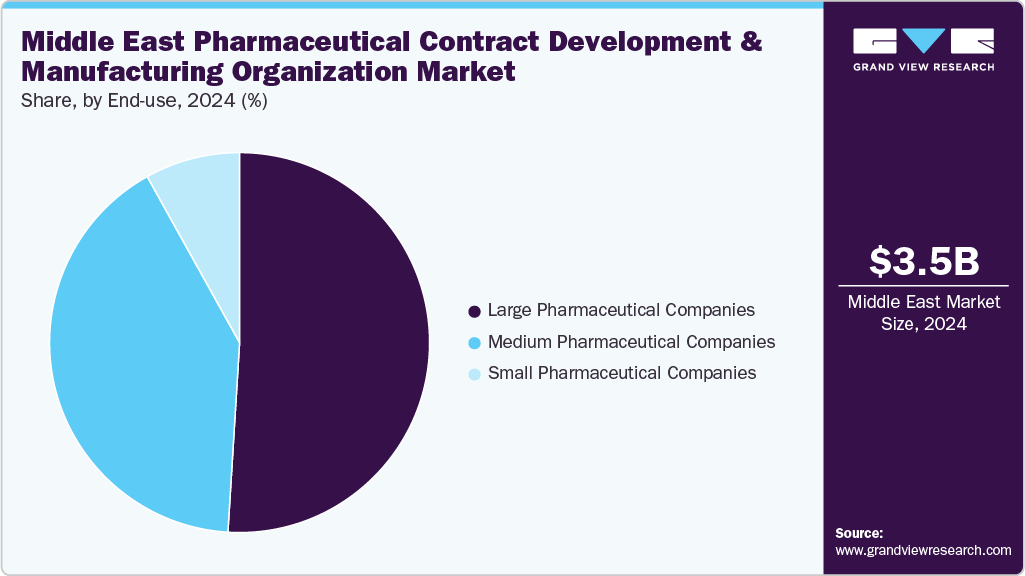
The medium pharmaceutical companies segment is expected to grow at the second-highest CAGR during the forecast period. Medium-sized pharmaceutical companies increasingly rely on CDMOs to bridge infrastructure, technology, and global reach gaps. In addition, with the limited in-house capacity for large-scale production, these companies are shifting towards CDMOs for end-to-end support from drug development and regulatory compliance to commercial-scale manufacturing. Moreover, outsourcing enables medium-sized companies to remain competitive, reduce capital expenditure, and access advanced biologics and specialty drug capabilities. Further, current trends of CDMOs and drug production development support the market growth, such as demand for flexible, cost-effective partnerships, faster product launches, and regional CDMO collaborations that enhance agility and market penetration for mid-sized pharma players. Such factors are expected to drive the market over the estimated time period.
Regional Insights
The Middle East pharmaceutical CDMO market is experiencing steady growth, driven by rising healthcare investments, expanding local manufacturing initiatives, and increasing demand for cost-effective drug development solutions. Besides, the governments across the Gulf and Africa prioritize pharmaceutical localization to strengthen supply chains and reduce reliance on imports. In addition, the established and regional companies are increasingly collaborating with CDMOs to access advanced technologies, regulatory expertise, and scalable production capacity. Moreover, factors such as increasing focus on biologics, sterile injectables, and generics, along with digital integration and quality compliance, are expected to drive new growth opportunities in the market.
UAE Pharmaceutical Contract Development And Manufacturing Organization Market Trends
The UAE pharmaceutical CDMO market is driven by the country's growing focus on strengthening its life sciences hub position. Besides, the government is strongly supported by government-led healthcare investments, favorable regulations, and strategic initiatives such as “Make it in the Emirates”. The market is attracting established and regional market players. In addition, the pharmaceutical companies in the region increasingly rely on CDMOs for end-to-end services and advanced expertise in biologics, generics, and specialty formulations. Moreover, growing demand for high-quality, compliant, scalable manufacturing to meet domestic needs and export opportunities is expected to drive the market over the estimated period.
Key Middle East Pharmaceutical Contract Development And Manufacturing Organization Company Insights
The key players operating across the market are adopting strategic initiatives such as service launches, mergers & acquisitions, partnerships & agreements, and expansions to gain a competitive edge in the market. For instance, in January 2025, Gaelic Laboratories, an Ireland-based CMO, mentioned the expansion in the UAE by setting up a new subsidiary, Gaelic Drug Store LLC. This strategy will strengthen the company's ability to comply with regional regulations while addressing the rising demand in the Middle East pharmaceutical sector, forecasted to reach USD 32 billion by 2027. In addition, the Dubai-based subsidiary is equipped with infrastructure for medicine importation, registration, & distribution, which a temperature-controlled warehouse supports. Further, the Gaelic Laboratories also position themselves as a Marketing Authorization Holder offering collaboration opportunities to global pharmaceutical companies entering the region.
Key Middle East Pharmaceutical Contract Development And Manufacturing Organization Companies:
- Thermo Fisher Scientific
- Eurofins CDMO
- Piramal Pharma Solutions
- Lonza
- Recipharm AB
- Catalent, Inc.
- WuXi AppTec, Inc.
- Samsung Biologics
- Siegfried Holding AG
- Corden Pharma International
- Cambrex Corporation
- Delpharm
- Jubilant Pharmova / HollisterStier
- Almac Pharma Service
Recent Developments
-
In August 2025, MS Pharma launched a biologics manufacturing facility in Saudi Arabia. This facility represents a USD 50 million investment, enhancing MS Pharma's role as a partner for pharmaceutical companies aiming to reach the Saudi Arabian and MENA market through local manufacturing and contract services for broader international distribution. The facility is built to comply with regulatory standards such as those of the EMA and the U.S. FDA. Also, it focuses on the production of monoclonal antibodies and complex peptides. Further, the plant is equipped with the region's first in-house bioanalytical testing laboratories, reducing the dependency on overseas testing and fueling the market introduction of essential treatments.
Middle East Pharmaceutical Contract Development And Manufacturing Organization Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 3.68 billion
Revenue forecast in 2033
USD 5.39 billion
Growth rate
CAGR of 4.90% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, product, service, workflow, therapeutic area, end use, region
Regional scope
Middle East
Country scope
UAE; Saudi Arabia; Kuwait; Qatar; Oman
Key companies profiled
Thermo Fisher Scientific; Eurofins CDMO; Piramal Pharma Solutions; Lonza; Recipharm AB; Catalent, Inc.; WuXi AppTec, Inc.; Samsung Biologics; Siegfried Holding AG; Corden Pharma International; Cambrex Corporation; Delpharm; Jubilant Pharmova / HollisterStier; Almac Pharma Service
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Middle East Pharmaceutical Contract Development And Manufacturing Organization Market Report Segmentation
This report forecasts revenue growth at the regional & country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Middle East pharmaceutical contract development and manufacturing organization market report based on type, product, service, workflow, therapeutic area, end use, and region:
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Small Molecule
-
Branded
-
Generic
-
-
Large Molecule
-
Biologics
-
Biosimilar
-
-
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
API
-
Traditional Active Pharmaceutical Ingredient (Traditional API)
-
Highly Potent Active Pharmaceutical Ingredient (HP-API)
-
Biologics
-
Others
-
-
Drug Product
-
Oral solid dose
-
Semi-solid dose
-
Liquid dose
-
Others
-
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Contract Development
-
Pre-formulation & Formulation Development Service
-
Process Development & Optimization
-
Analytical Testing & Method Validation
-
Scale-up & Tech Transfer
-
-
Contract Manufacturing
-
API Manufacturing
-
Finished Drug Products Manufacturing
-
-
Packaging and Labelling
-
Regulatory Affairs
-
Others
-
-
Workflow Outlook (Revenue, USD Million, 2021 - 2033)
-
Clinical
-
Commercial
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
Infectious Diseases
-
Neurological Disorders
-
Cardiovascular Disease
-
Metabolic Disorders
-
Autoimmune Diseases
-
Respiratory Diseases
-
Ophthalmology
-
Gastrointestinal Disorders
-
Orthopedic Diseases
-
Dental Diseases
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Small Pharmaceutical Companies
-
Medium Pharmaceutical Companies
-
Large Pharmaceutical Companies
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
Middle East
-
UAE
-
Saudi Arabia
-
Kuwait
-
Qatar
-
Oman
-
-
Frequently Asked Questions About This Report
b. The Middle East pharmaceutical contract development and manufacturing organization market size was estimated at USD 3.50 billion in 2024 and is expected to reach USD 3.68 billion in 2025.
b. The Middle East pharmaceutical contract development and manufacturing organization market is expected to grow at a compound annual growth rate of 4.90% from 2025 to 2033 to reach USD 5.39 billion by 2033.
b. The small molecule accounted for the largest revenue share of 61.92% in 2024. The segment growth is driven by increasing demand for cost-effective manufacturing and faster time-to-market. Besides, regional CDMOs are expanding formulation, chemical synthesis, and analytical testing capabilities, further supporting the market growth.
b. Some key players operating in the Middle East pharmaceutical contract development and manufacturing organization market include Thermo Fisher Scientific , Eurofins CDMO, Piramal Pharma Solutions, Lonza, Recipharm AB, Catalent, Inc., WuXi AppTec, Inc., Samsung Biologics, Siegfried Holding AG, Corden Pharma International, Cambrex Corporation, Delpharm, Jubilant Pharmova / HollisterStier, and Almac Pharma Service among others.
b. Key factors driving the Middle East pharmaceutical contract development and manufacturing organization market growth include increasing healthcare spending, rising prevalence of chronic diseases, and a growing demand for cost-effective drug development and manufacturing facilities. In addition, growing government investments in healthcare infrastructure and strategic locations in the Middle East as a trade hub connecting Asia, Europe, and Africa have further contributed to market growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










