- Home
- »
- Medical Devices
- »
-
Middle East Sterile Barrier Systems Market Size Report, 2033GVR Report cover
![Middle East Sterile Barrier Systems Market Size, Share & Trends Report]()
Middle East Sterile Barrier Systems Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Wraps, Rigid Containers, Pouches & Reels, Indicators Embedded in Packaging), By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-721-5
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
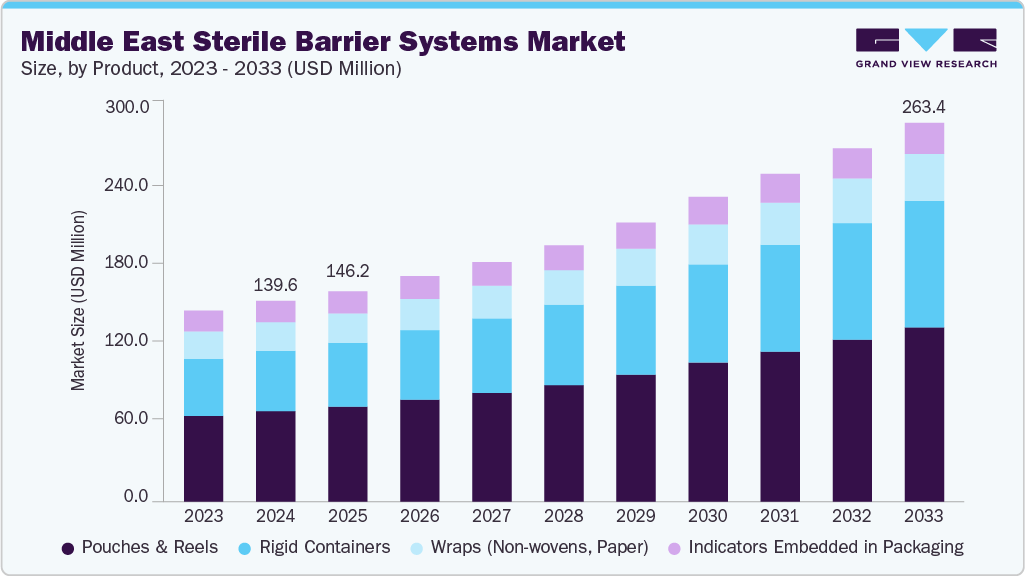
Middle East Sterile Barrier Systems Market Summary
The Middle East sterile barrier systems market size was estimated at USD 139.63 million in 2024 and is projected to reach USD 263.41 million by 2033, growing at a CAGR of 7.64% from 2025 to 2033. The growth of the market is attributed to the rising awareness of hospital-acquired infections (HAIs) and stringent regulatory requirements for surgical sterilization.
Key Market Trends & Insights
- Saudi Arabia dominated the Middle East sterile barrier systems market with the largest revenue share of 21.36% in 2024.
- Based on product, the pouches & reels segment led the market with the largest revenue share of 45.07% in 2024.
- Based on end use, the hospitals segment led the market with the largest revenue share of 49.54% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 139.63 Million
- 2033 Projected Market Size: USD 263.41 Million
- CAGR (2025-2033): 7.64%
- Saudi Arabia: Largest market in 2024
Additionally, the region is witnessing an expansion of healthcare infrastructure, including new hospitals and surgical centers, which increases the adoption of sterile barrier products. Technological advancements, such as improved packaging materials, customized surgical kits, and integration with digital tracking systems, further support market growth.The rising demand for sterile pharmaceuticals and biologics is fueling growth in the Middle East sterile barrier systems market, as these products are susceptible to contamination and require manufacturing under the highest sterility standards. Injectable and parenteral drugs bypass the body’s natural defenses and must remain completely free from microbial contamination. This critical requirement drives pharmaceutical companies to adopt advanced sterilization technologies that maintain product integrity while ensuring sterility. For instance, the large-scale production of COVID-19 vaccines highlighted the rapid deployment of solutions such as vaporized hydrogen peroxide (VH₂O₂) sterilization and aseptic filling systems to meet unprecedented demand for sterile medications.
The rising popularity of cosmetic and plastic surgery in the Middle East is a key driver for the Middle East sterile barrier systems market, as these procedures demand stringent sterility to prevent infections and ensure patient safety. The UAE, particularly Dubai, is leading the region’s medical aesthetics growth, with statistics showing the Middle East and North Africa (MENA) region as the world’s second fastest-growing wellness travel market.
Dubai is emerging as a medical tourism hotspot, aiming to attract 1.3 million medical tourists by 2021. Initiatives like the Dubai Health Experience (DHA) program, launched to provide comprehensive electronic medical tourism services, including wellness, cosmetic and dental procedures, ophthalmology, orthopedics, physiotherapy, and specialized medical tests, further increase procedural volumes. This surge in elective and specialized treatments is boosting demand for advanced sterile barrier systems to ensure safety and maintain sterility throughout surgical and therapeutic interventions.
Governments across the Middle East invest heavily in modern hospitals, specialized clinics, and pharmaceutical manufacturing facilities, creating higher demand for sterile packaging solutions that comply with global quality and safety standards. For instance, Saudi Arabia has earmarked USD 65 billion under its Vision 2030 healthcare transformation plan, including new hospitals and digital health infrastructure. In July 2024, the UAE launched a milestone initiative to bridge global healthcare gaps under the Zayed Humanitarian Legacy Initiative, first announced in March 2024 by President His Highness Sheikh Mohamed bin Zayed Al Nahyan. With a planned investment of USD 150 million (AED 550 million) over the next decade, the UAE Global Hospitals Program will establish up to 10 hospitals worldwide, each designed to address the unique healthcare needs of underserved populations.
This program aims to strengthen access to essential medical services and improve health outcomes for communities with limited healthcare infrastructure. Similarly, new PPP projects in Kuwait and Bahrain drive private sector participation in hospital and clinic expansion. These infrastructure developments are increasing the procurement of sterile barrier systems for surgical instruments, implants, and single-use medical consumables to meet international infection control and patient safety requirements.
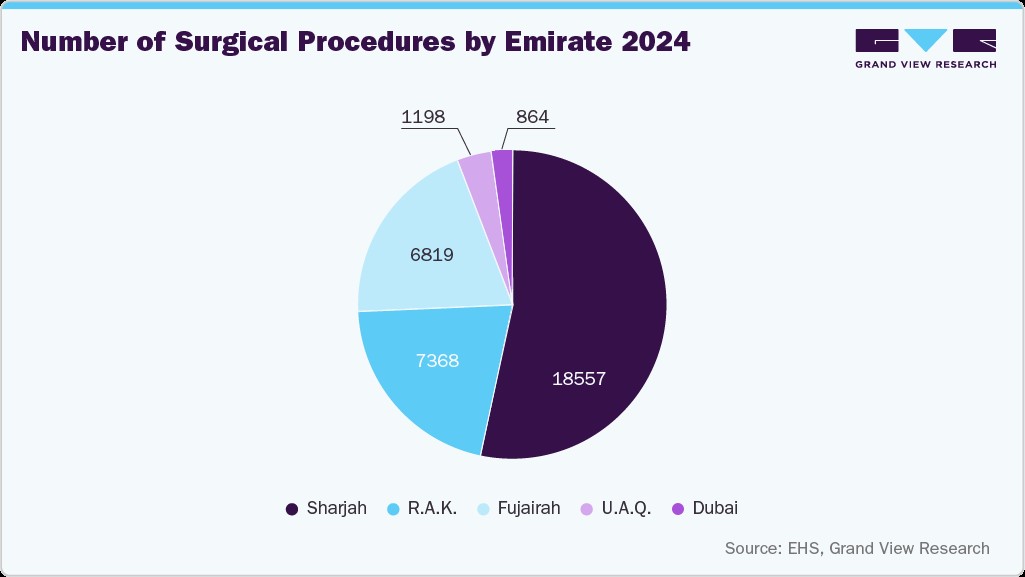
Government investments and policy initiatives under frameworks such as UAE Vision 2031 enhance healthcare quality, accreditation standards, and infection control compliance, further embedding sterile barrier requirements into procurement specifications. For instance, in October 2024, Aster Clinics, part of Aster DM Healthcare GCC, expanded its network with six new clinics across Dubai, Sharjah, Fujairah, and Umm Al Quwain. Such developments increase the volume of medical and surgical procedures performed in the country, directly supporting sustained growth in the sterile barrier systems market. As healthcare modernization accelerates, the UAE market for SBS is poised for consistent and significant expansion.
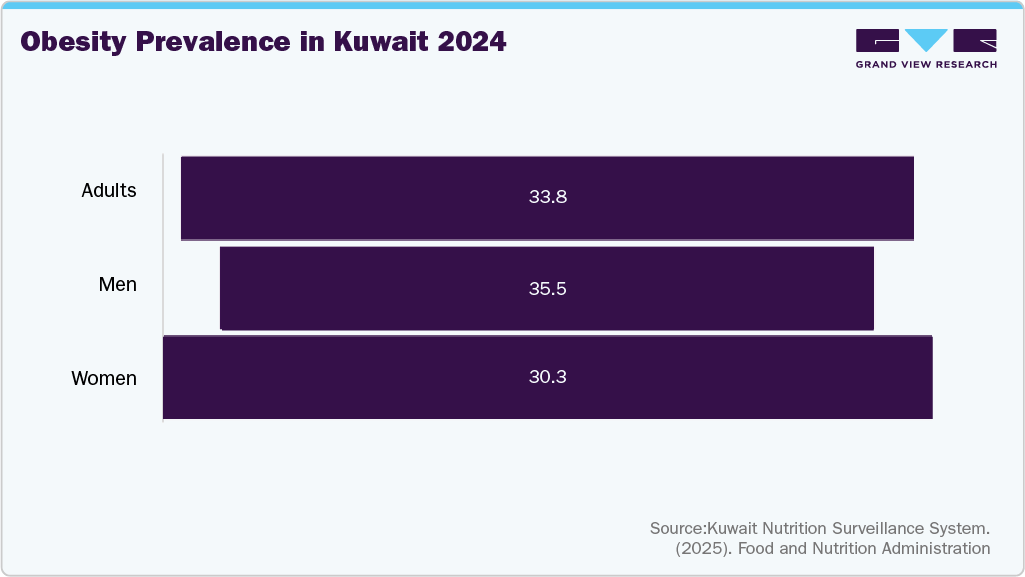
The growing prevalence of chronic diseases such as diabetes, cardiovascular disorders, and obesity is driving higher volumes of surgical interventions and long-term treatments. This trend increases the need for reliable sterile consumables, including sterile barrier systems, to support surgical and procedural safety. Regulatory frameworks are becoming more structured, with the Ministry of Health enforcing tighter controls on product quality, sterilization validation, and supply chain traceability. According to the International Diabetes Federation, as of 2024, Kuwait faces a significant burden of diabetes, with approximately 25.6% of its adult population affected by the disease. Out of a total adult population of 3.2 million, an estimated 908,500 adults live with diabetes. Kuwait is part of the International Diabetes Federation’s Middle East and North Africa (IDF MENA) region, which collectively accounts for 85 million people with diabetes as of 2024-a figure projected to nearly double to 163 million by 2050.
KOLs
-
“When EU GMP Annex 1 was released, we recognized that many of our Customers would need to visually confirm the dryness of a part within its sterile packaging. Verafit Sterilization Bags and Covers make this possible via the viewing window. This enhancement adds to the overall value of our sterilization bags and covers, which provide an easy-to-use, consistent method of wrapping products and maintaining sterility,”- said Elaine Sartain, Vice President, Global Marketing, STERIS Life Sciences Contamination Control Products.
-
“The advancements in sustainability showcased in the winning programs for the 2024 DuPont Tyvek Sustainable Healthcare Packaging Awards are a testament to how these companies are dedicated to making meaningful environmental strides within the healthcare industry. Our DuPont team is dedicated to working alongside our customers and others in the healthcare and healthcare packaging industry to foster continuous collaboration and help them advance sustainability and enable product and patient safety. We join these award winners in viewing advancements in sustainability as an important extension of why we do what we do,” said Kelly Reichert, global business director, DuPont Tyvek & Typar Roll Goods.
Market Concentration and Characteristics
The degree of innovation in the Middle East sterile barrier systems market is high, fueled by the region’s rapid healthcare transformation, increasing medical tourism, and heightened emphasis on infection prevention. Advanced solutions such as peelable films, high-barrier laminates, thermoform trays, and flexible pouches are being adopted to ensure product safety and sterility while accommodating diverse clinical and pharmaceutical needs. In addition, integrating smart packaging technologies, including track-and-trace features and RFID-enabled systems, is gaining traction in alignment with global best practices. Government-driven investments in smart hospitals, pharmaceutical manufacturing plants, and specialized clinics in countries like the UAE, Saudi Arabia, and Qatar further accelerate the demand for next-generation sterile packaging solutions. These advancements reflect a growing shift from conventional wraps to innovative, sustainable, and regulatory-compliant sterile barrier systems, positioning the Middle East as a promising growth hub for advanced packaging technologies.
Regulations play a pivotal role in shaping the sterile barrier systems market in the Middle East, as governments across the region increasingly align healthcare standards with international frameworks such as ISO 11607 and stringent guidelines for infection control, patient safety, and product sterility. Countries like Saudi Arabia, the UAE, and Qatar have tightened their medical device registration and packaging compliance requirements, compelling healthcare providers and manufacturers to adopt barrier systems that meet global sterility, durability, and traceability benchmarks. Regulatory emphasis on single-use sterile packaging, validated sterilization processes, and environmental safety also drives greater innovation and quality assurance within the market. While these regulations increase compliance costs, they ultimately strengthen trust, enhance product safety, and expand opportunities for companies offering high-performance, certified sterile barrier solutions.
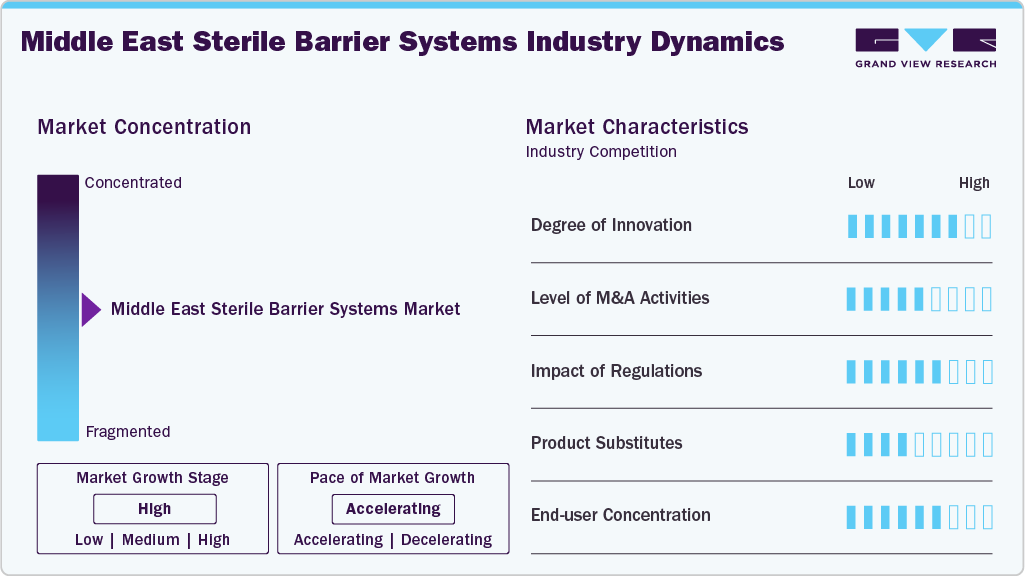
The level of mergers and acquisitions (M&A) activity in the Middle East sterile barrier systems market remains moderate but steadily increasing, driven by the region’s expanding healthcare infrastructure and growing demand for advanced infection prevention solutions. Global packaging and sterilization companies are increasingly exploring strategic alliances, joint ventures, and acquisitions to strengthen their foothold in the Middle East, leveraging local manufacturing and distribution networks to meet rising demand from hospitals, pharmaceutical manufacturers, and medical device companies.
In the Middle East sterile barrier systems market, product substitutes remain limited but present, primarily in the form of reusable sterilization containers, textile-based wraps, and conventional packaging methods. While single-use sterile barrier systems such as pouches, wraps, and trays dominate due to their compliance with international infection prevention standards, some healthcare facilities, particularly in cost-sensitive markets, still rely on reusable stainless-steel containers and cloth drapes as alternatives. However, these substitutes face challenges, including lower sterility assurance, higher risk of contamination, and the need for repeated cleaning and validation, which add operational burdens.
The Middle East sterile barrier systems market exhibits a high end-user concentration, with hospitals and large healthcare networks representing the primary demand segment due to their extensive surgical volumes, advanced infection control protocols, and adherence to international sterilization standards. However, small and medium-sized enterprises (SMEs), including independent clinics, diagnostic centers, and specialty surgical facilities, are also emerging as significant users, particularly in countries like the UAE, Saudi Arabia, and Qatar, where private healthcare investment is expanding. For SMEs, the adoption of sterile barrier systems is often driven by regulatory compliance, accreditation requirements, and the need to attract medical tourists. Still, price sensitivity and limited budgets can influence product choices, occasionally leading to reliance on cost-effective or substitute solutions. Overall, while large hospitals dominate, the growing presence of SMEs as end-users is creating a diversified demand base in the region.
Product Insights
The pouches & reels segment held the largest revenue share in 2024. This growth can be attributed to their versatility, ease of use, and cost-effectiveness in sterilizing and packaging a wide range of medical devices, surgical instruments, and consumables. Their compatibility with various sterilization methods, such as steam, ethylene oxide, and hydrogen peroxide plasma, makes them highly preferred across hospitals, surgical centers, and pharmaceutical manufacturers. Additionally, the rising number of surgical procedures and the expansion of hospital infrastructure in countries like Saudi Arabia and the UAE have fueled the demand for pouches and reels as the most practical sterile packaging format, especially given their ability to ensure barrier protection, maintain sterility until point-of-use, and meet strict regulatory standards.
The rigid containers segment is expected to witness the fastest growth over the forecast period. This growth is driven by their superior durability, reusability, and enhanced protection for surgical instruments compared to flexible packaging formats. With healthcare providers in the region increasingly investing in advanced surgical infrastructure and prioritizing infection prevention, rigid containers are gaining traction as they minimize the risk of sterility breaches and reduce long-term costs through multiple reprocessing cycles. Furthermore, the shift toward sustainable and eco-friendly solutions is encouraging hospitals to adopt rigid containers as an alternative to disposable wraps and pouches, especially in high-volume tertiary care centers across Saudi Arabia, the UAE, and Qatar.
End Use Insights
The hospital segment held the largest revenue share in 2024. This growth can be attributed to the rising volume of surgical procedures, which creates a strong need for efficient and reliable packaging solutions capable of handling high instrument throughput. Growing concerns over healthcare-associated infections (HAIs) have accelerated the adoption of advanced sterile barrier systems, including high-performance wraps, rigid containers, and integrated chemical and biological indicators, to ensure compliance with strict infection control protocols. Moreover, government-led healthcare expansion programs and initiatives to improve surgical access and efficiency are compelling hospitals to strengthen their sterilization workflows, further boosting the demand for SBS solutions in this segment.
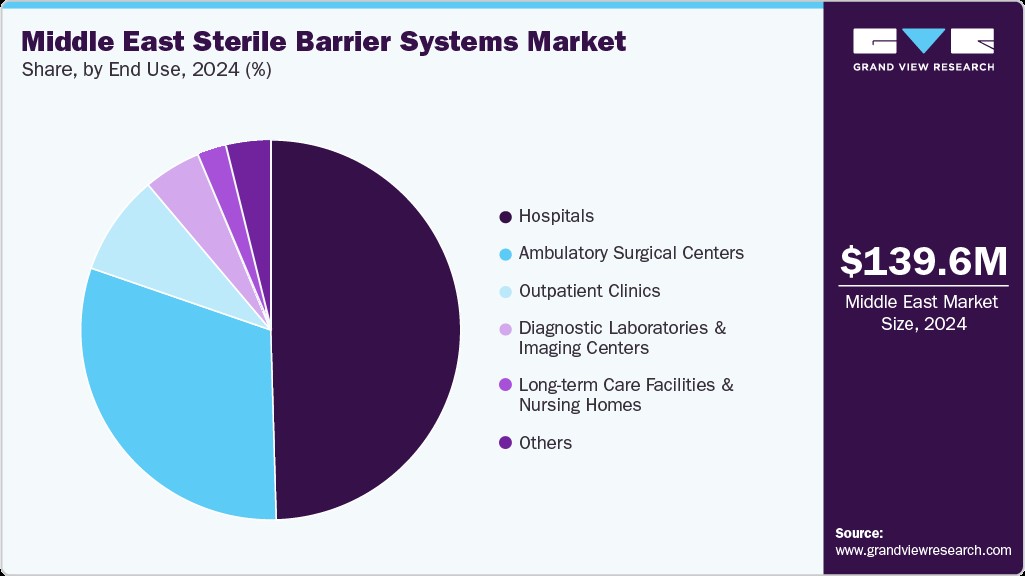
The ambulatory surgical centers segment is expected to witness the fastest CAGR over the forecast period. This growth can be attributed to the region’s growing shift toward day-care and minimally invasive procedures that reduce hospital stays and costs. ASCs rely heavily on sterile packaging solutions that ensure rapid instrument turnaround, consistent sterility assurance, and compliance with stringent infection prevention standards. Rising healthcare reforms promoting cost-efficient care, coupled with increasing investments in specialized outpatient facilities, are further accelerating demand for advanced SBS products in this segment, positioning ASCs as a key growth driver in the market.
Regional Insights
Saudi Arabia Sterile Barrier Systems Market Trends
Saudi Arabia’s sterile barrier systems market is expanding rapidly, driven by Vision 2030 healthcare investments, rising local pharmaceutical and medical device production, and stricter infection control standards. Demand is growing for diverse packaging formats like thermoform trays, pre-filled syringes, and pouches, supported by modern sterilization methods such as autoclaving and radiation. At the same time, a shift toward sustainable materials, including recyclable and degradable options, is shaping procurement trends.
UAE Sterile Barrier Systems Market Trends
The UAE sterile barrier systems market is expanding steadily, driven by its growing role as a regional healthcare and medical tourism hub, strict compliance with global quality standards, and rising demand across pharmaceuticals, diagnostics, and surgical devices. Dominated by high-barrier plastics but with increasing adoption of sustainable paper-based materials, the market leverages multiple sterilization methods (ETO, autoclaving, radiation) to meet diverse needs.
Key Middle East Sterile Barrier Systems Company Insights
The Middle East sterile barrier systems market is extremely fragmented, with both major and local market competitors. Due to the fact that the current market players are stepping up their efforts to grab the majority in the Middle East sterile barrier systems market, fierce competition is anticipated, with the degree of competitiveness perhaps rising even higher. Many market participants are engaging in various strategic activities, such as product launches, mergers and acquisitions, and geographic growth, in an effort to gain a competitive edge over rivals. Thus, with various strategies adopted by the market players, the Middle East sterile barrier systems market is predicted to grow during the forecast period.
Key Middle East Sterile Barrier Systems Companies:
- ASP (Fortive)
- STERIS
- 3M
- Amcor
- Cardinal Health
- DuPont
- Medline Industries, LP.
- Innovia Medical Company (Summit Medical LLC)
- Sklar
Recent Developments
-
In 2024, STERIS launched its Verafit Sterilization bags and covers, with a patent-pending viewing window, enabling operators to visually confirm dryness without breaking the sterile barrier, an enhancement directly addressing EU GMP Annex 1 requirements.
-
In August 2023, STERIS plc has announced the successful completion of its acquisition of surgical instrumentation, laparoscopic instrumentation, and sterilization container assets from BD (Becton, Dickinson and Company).
Middle East Sterile Barrier Systems Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 146.17 million
Revenue forecast in 2033
USD 263.41 million
Growth rate
CAGR of 7.64% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, end use,and region
Regional scope
Middle East
Country scope
Saudi Arabia; Kuwait; Qatar; Oman; UAE
Key companies profiled
ASP (Fortive); STERIS; 3M; Amcor; Cardinal Health; DuPont; Medline Industries, LP.; Innovia Medical Company (Summit Medical LLC); Sklar
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Middle East Sterile Barrier Systems Market Report Segmentation
This report forecasts revenue growth at the country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Middle East sterile barrier systems market report based on product, end use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Wraps (Non-wovens, Paper)
-
Rigid Containers
-
Pouches & Reels
-
Indicators Embedded in Packaging
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Ambulatory Surgical Centers
-
Outpatient Clinics
-
Long-term Care Facilities & Nursing Homes
-
Diagnostic Laboratories & Imaging Centers
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
Middle East
-
Saudi Arabia
-
UAE
-
Oman
-
Qatar
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The Middle East sterile barrier systems market size was estimated at USD 139.63 million in 2024 and is expected to reach USD 146.17 million in 2025.
b. The Middle East sterile barrier systems market is expected to grow at a compound annual growth rate of 7.64% from 2025 to 2033 to reach USD 263.41 million by 2033.
b. Pouches & reels segment held the largest revenue share of 45.07% in 2024, owing to their widespread use as sterile barrier systems across hospitals and surgical centers due to ease of handling, cost-effectiveness, and compatibility with diverse sterilization methods. Their high adoption in packaging single-use medical devices and instruments further reinforced segment dominance.
b. Some key players operating in the Middle East sterile barrier systems market include ASP (Fortive); STERIS; 3M; Amcor; Cardinal Health; DuPont; Medline Industries, LP.; Innovia Medical Company (Summit Medical LLC); Sklar.
b. Key factors driving the market growth include the rising awareness of hospital-acquired infections (HAIs) and stringent regulatory requirements for surgical sterilization. Additionally, the region is witnessing an expansion of healthcare infrastructure, including new hospitals and surgical centers, which increases the adoption of sterile barrier products.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










