- Home
- »
- Clinical Diagnostics
- »
-
Middle East Thyroid Cancer Diagnostics Market Report, 2033GVR Report cover
![Middle East Thyroid Cancer Diagnostics Market Size, Share & Trends Report]()
Middle East Thyroid Cancer Diagnostics Market (2025 - 2033) Size, Share & Trends Analysis Report By Cancer Type (Papillary Carcinoma, Follicular Carcinoma), By Technique (Imaging, Biopsy), By End Use, By Country, And Segment Forecasts
- Report ID: GVR-4-68040-726-3
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Middle East Thyroid Cancer Diagnostics Market Summary
The Middle East thyroid cancer diagnostics market size was estimated at USD 89.64 million in 2024 and is projected to reach USD 142.27 million by 2033, growing at a CAGR of 5.4% from 2025 to 2033. The market shows rising demand, driven by gender-skewed incidence, with female rates notably higher, especially in the UAE. Current practice relies on ultrasound, FNA, and cytology, while molecular testing adoption remains low (~20%), leaving significant growth potential for NGS panels and mutation/fusion assays. Regulatory oversight varies across countries, and reimbursement challenges slow uptake. However, opportunities exist in precision oncology, Bethesda III/IV nodule classifiers, and partnerships with local hospitals and labs to expand access and reduce overtreatment.
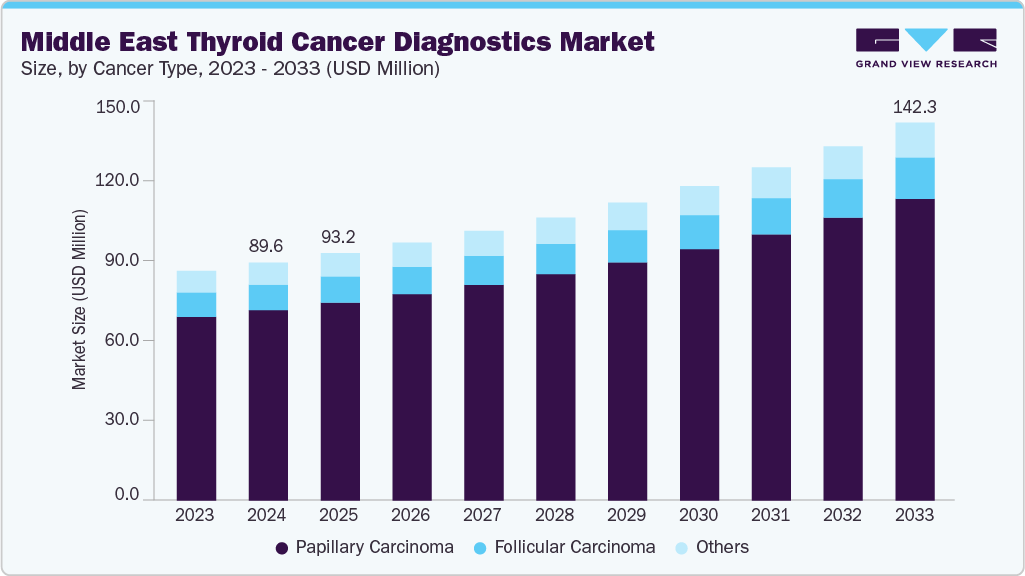
The thyroid cancer burden in the Middle East, particularly across the Gulf Cooperation Council (GCC), is significant and disproportionately affects women, driving demand for diagnostic testing. A 2024 analysis estimates the GCC age-standardized incidence rate (ASR) at ~6.1 per 100,000, with variations by country-highest in Saudi Arabia (~7.5), Kuwait (~5.2), and lowest in Bahrain (~2.4). In the UAE, the female ASR is markedly higher (~13.6/100,000) compared to ~1.9/100,000 in males, underscoring strong gender disparities and informing screening and triage practices in women’s health pathways. Combined with population growth and wider imaging availability, these trends support sustained demand across ultrasound, cytology, and molecular assays.
Current clinical practice continues to rely on neck ultrasound (TI-RADS), fine-needle aspiration (FNA) with Bethesda cytology, and selective IHC. Yet molecular diagnostics remain underutilized; a regional survey found only ~20% of clinicians refer suspicious nodules for molecular testing, while most still default to surgery. This points to significant growth potential for next-generation sequencing (NGS)-based classifiers and mutation/fusion panels, including BRAF, RAS, RET, and NTRK. In addition, guidance from the European Thyroid Association discouraging routine ultrasound in asymptomatic adults is influencing regional practices, shifting emphasis toward risk-adapted triage rather than over-imaging.
The market reflects this pathway structure, encompassing three core areas: imaging (ultrasound and emerging AI triage tools), cytology (FNA kits, liquid-based cytology, IHC), and molecular diagnostics (NGS panels, targeted PCR, fusion assays). Of these, molecular testing is the fastest-growing segment, driven by precision oncology requirements for RET/NTRK fusions and BRAFV600E, alongside tertiary centers establishing in-house NGS capacity or send-out programs. Afirma remains largely U.S.-based as a CLIA LDT, so Middle Eastern uptake generally depends on regional NGS panels or international reference labs.
Regulation and reimbursement are governed nationally, with oversight by agencies such as the SFDA (Saudi Arabia), MOHAP/DHA/DOH (UAE), MoPH (Qatar), NHRA (Bahrain), and EDA (Egypt). Nationals generally access diagnostics through publicly funded systems, while expatriates rely on private insurance, which may only partially cover molecular assays-explaining slower adoption than imaging or cytology. Nonetheless, increasing alignment with international guidelines and the growing need for companion diagnostics in targeted therapies are gradually improving payer acceptance.
The competitive landscape features IVD leaders in imaging, IHC, and immunoassays alongside genomics-focused companies offering NGS instruments and panels. Key opportunities include expanding molecular classifiers for Bethesda III/IV nodules to avoid unnecessary surgeries, deploying mutation/fusion panels to guide advanced treatment, and forging local partnerships with hospitals, labs, and distributors to speed regulatory approval, reduce turnaround times, and strengthen reimbursement viability. Regional clinical data further highlight that recurrent differentiated thyroid cancer is linked to poorer outcomes than persistent disease, reinforcing the importance of longitudinal molecular monitoring and advanced risk stratification solutions.
Market Concentration & Characteristics
The degree of innovation in middle east thyroid cancer diagnostics is advancing rapidly, driven by the need for earlier detection, precise tumor characterization, and improved patient management. Traditional diagnostic pathways relied heavily on ultrasound, fine-needle aspiration (FNA), and histopathology, but these often faced limitations such as indeterminate results and invasive procedures. Recent innovations have introduced molecular and genomic testing (e.g., BRAF, RAS, RET, and NTRK mutation panels) that improve diagnostic accuracy, guide targeted therapies, and reduce unnecessary surgeries. Furthermore, next-generation sequencing (NGS) and liquid biopsy assays are gaining momentum for non-invasive detection of circulating tumor DNA, enabling dynamic monitoring of disease progression. Artificial intelligence (AI) and machine learning algorithms are also being integrated into thyroid ultrasound imaging to enhance diagnostic accuracy and minimize inter-observer variability. Collectively, these advances represent a high degree of innovation, shifting thyroid cancer diagnostics toward more personalized, minimally invasive, and cost-effective approaches.
M&A activities in thyroid cancer diagnostics focus on expanding molecular, genomic, and non-invasive testing capabilities. Large diagnostic firms acquire niche players specializing in NGS panels, mutation analysis (BRAF, RAS, RET, NTRK), and liquid biopsy to enhance precision oncology portfolios. AI-driven imaging startups are also attractive targets, improving ultrasound-based risk stratification. Beyond acquisitions, strategic collaborations are common, enabling companies to access innovative assays, strengthen companion diagnostic offerings, and accelerate market penetration, ultimately driving consolidation and personalized patient care advancements.
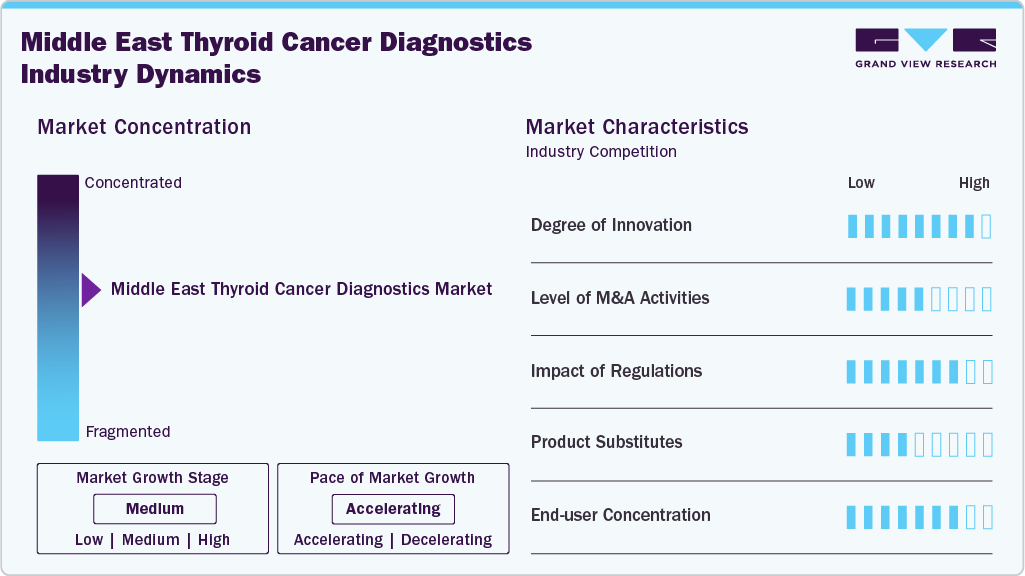
The regulatory landscape in the Middle East significantly influences the adoption of thyroid cancer diagnostics. Stringent product approval processes and country-specific requirements, such as the Saudi Food and Drug Authority’s (SFDA) demand for rigorous clinical validation and local data, often delay market entry for new molecular assays. In contrast, the UAE’s Ministry of Health and Prevention (MOHAP) is streamlining pathways to accelerate access to advanced technologies. Growing government focus on cancer screening programs and harmonization efforts across GCC countries are further creating supportive pathways. Clear reimbursement frameworks and faster regulatory approvals are expected to be critical to drive accessibility, adoption, and sustained growth of thyroid cancer diagnostics in the region.
Product expansion in thyroid cancer diagnostics is driven by the integration of advanced molecular assays, next-generation sequencing (NGS) panels, and liquid biopsy solutions. Companies are broadening portfolios beyond traditional fine-needle aspiration (FNA) to include mutation analysis for BRAF, RAS, RET, and NTRK, as well as companion diagnostics for targeted therapies. AI-enabled imaging tools and digital pathology are also being introduced to improve diagnostic accuracy. These expansions enable more comprehensive, less invasive, and personalized diagnostic pathways, ultimately strengthening competitive positioning in precision oncology.
Regional expansion strategies in thyroid cancer diagnostics emphasize penetrating high-growth markets across the Middle East, where cancer incidence and diagnostic infrastructure are rapidly evolving. Global leaders partner with local laboratories, hospitals, and distributors to accelerate market entry and regulatory approvals. Collaborations with regional health authorities help align diagnostic offerings with local clinical guidelines and reimbursement frameworks. This approach enhances accessibility, addresses unmet needs in early detection, and supports the global shift toward precision oncology and standardized thyroid cancer care.
Cancer Type Insights
In the Middle East, papillary carcinoma dominates the thyroid cancer diagnostics market, contributing the largest revenue share of 80.01% in 2024, driven by its high prevalence across GCC and neighboring countries. Increased awareness, government-led screening initiatives, and the growing availability of advanced imaging and molecular testing have improved early detection rates. In addition, environmental exposures, lifestyle transitions, and the relatively younger average age at diagnosis in the region continue to elevate case numbers, supporting strong demand for accurate diagnostic solutions.
Follicular carcinoma diagnostics in the Middle East are projected to expand at a CAGR of 5.7% over the forecast period, reflecting its position as the region’s second most common thyroid cancer, with a higher malignancy potential than papillary carcinoma. Its aggressive invasion into adjacent tissues and reduced survival outcomes with advancing age underscore the importance of early, precise diagnosis. Greater female susceptibility, rising clinical focus on timely detection, and adoption of molecular and imaging-based technologies are expected to accelerate diagnostic uptake for follicular carcinoma, contributing significantly to market growth across the Middle East.
Technique Insights
In the Middle East, imaging techniques lead the thyroid cancer diagnostics market, accounting for the largest revenue share of 36.90% in 2024, supported by their central role in early detection and evaluation of thyroid nodules. Ultrasound remains the primary tool across GCC and wider regional healthcare systems due to its accessibility, cost-effectiveness, and accuracy; meanwhile, advanced modalities such as CT, MRI, and PET are increasingly used in tertiary centers. The integration of artificial intelligence and improvements in imaging resolution are enhancing diagnostic precision, tumor localization, and monitoring, making imaging indispensable in regional thyroid cancer management.
Biopsy techniques in the Middle East are projected to grow at a significant CAGR of 5.6%, reflecting their critical role in confirming thyroid cancer at the cellular and molecular level. Fine-needle aspiration (FNA) with Bethesda cytology remains standard, but the growing adoption of molecular testing on biopsy samples is enabling personalized treatment decisions. Advances in next-generation sequencing (NGS) and biomarker analysis are gradually entering clinical practice in regional centers, improving diagnostic accuracy for indeterminate nodules. The rising demand for minimally invasive, reliable, and definitive approaches is expected to further drive the uptake of biopsy-based diagnostics across the Middle East market.
End Use Insights
Hospital laboratories held the dominant position in the market and accounted for the largest revenue share of 37.42% in 2024, driven by increasing patient inflow for cancer diagnosis, rising awareness of thyroid disorders, and expanding healthcare infrastructure, especially in developing regions. In addition, hospitals are often the primary point of care, providing comprehensive diagnostic services, advanced technologies, and specialized personnel. Furthermore, investments in modern equipment and integration of molecular and genetic testing capabilities further enhance the accuracy and efficiency of thyroid cancer diagnostics in hospital settings, supporting continued market expansion.
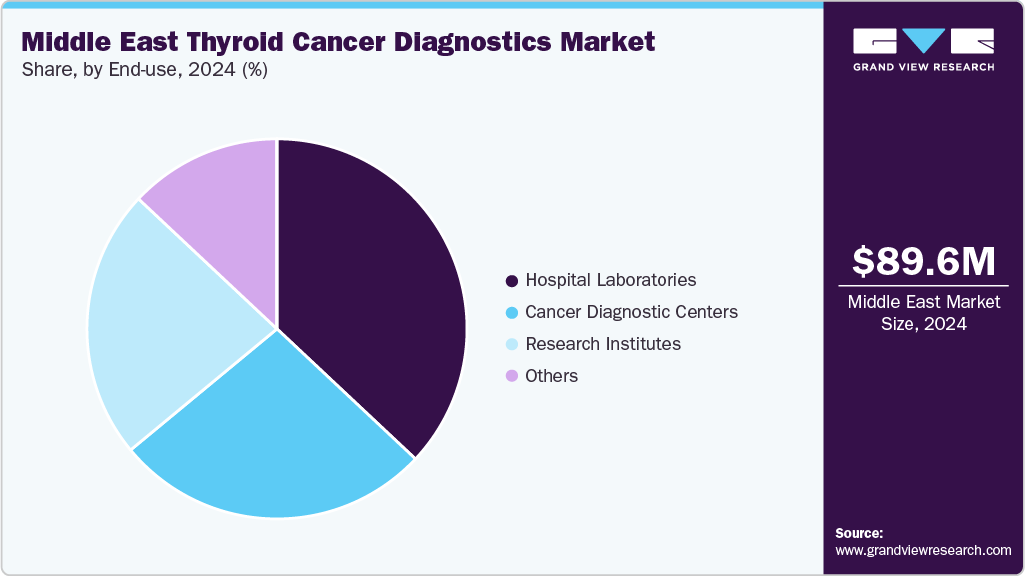
Research institutes are expected to grow at a CAGR of 6.5% from 2025 to 2033, due to increased funding for cancer research and a strong focus on innovation. These institutes play a pivotal role in developing and validating new diagnostic technologies, such as advanced molecular assays and genetic profiling. Furthermore, collaborative efforts with academic and industry partners, access to grant funding, and participation in clinical trials drive breakthroughs in early detection and personalized medicine, positioning research institutes as key contributors to market advancement.
Country Insights
The Middle East thyroid cancer diagnostics market is witnessing steady growth, driven by rising incidence rates, strong gender disparities, and improving healthcare infrastructure across the Gulf Cooperation Council (GCC) and neighboring countries. Women remain disproportionately affected, with female age-standardized incidence rates significantly higher, particularly in the UAE. Current diagnostic practice is dominated by ultrasound, fine-needle aspiration (FNA), and Bethesda cytology, while selective IHC supports confirmation. However, molecular testing remains underpenetrated, with only about 20% of clinicians utilizing NGS-based classifiers or mutation/fusion panels for indeterminate nodules. Despite reimbursement challenges and regulatory complexities, molecular diagnostics are emerging as the fastest-growing segment, fueled by precision oncology needs for RET, NTRK, and BRAFV600E. Increasing payer alignment, strategic hospital-lab partnerships, and investments in localized testing are expected to accelerate adoption and strengthen the region’s diagnostic capabilities.
Saudi Arabia Thyroid Cancer Diagnostics Market Trends
Saudi Arabia holds the highest thyroid cancer incidence in the GCC, with an age-standardized rate (ASR) of ~7.5 per 100,000. The market is supported by robust government healthcare investment and a large public hospital network, ensuring broad access to imaging, FNA, and cytology. However, molecular testing adoption is still limited, with reimbursement barriers slowing uptake. The Saudi Food and Drug Authority (SFDA) oversees IVD approvals, and growing alignment with global guidelines is improving precision diagnostics adoption. Opportunities lie in expanding NGS-based panels for indeterminate nodules and fostering partnerships with tertiary care centers to enable localized molecular testing capacity.
United Arab Emirates (UAE) Thyroid Cancer Diagnostics Market Trends
The UAE has one of the highest gender disparities in thyroid cancer, with female ASR at ~13.6 per 100,000 versus ~1.9 in males. The country’s strong private healthcare sector, alongside public providers (MOHAP, DOH Abu Dhabi, DHA Dubai), drives rapid adoption of advanced diagnostics such as high-resolution ultrasound, AI imaging tools, and FNA. Molecular testing is gaining attention, particularly in leading hospitals, but access still relies heavily on international send-out tests. Expanding insurance coverage and strategic lab partnerships are critical for uptake. The UAE’s advanced infrastructure and medical tourism hub status make it a key growth market for precision diagnostics.
Kuwait Thyroid Cancer Diagnostics Market Trends
Kuwait reports an ASR of ~5.2 per 100,000, with thyroid cancer representing a growing diagnostic priority in women’s health. The healthcare system is dominated by government hospitals, offering strong imaging and cytology services but limited molecular testing infrastructure. Regulatory oversight lies with the Ministry of Health (MOH), which is gradually adopting international diagnostic standards. Rising awareness, younger patient demographics, and increasing investments in oncology services support market growth. Expanding NGS panels for Bethesda III/IV nodules, coupled with collaborations between public hospitals and private labs, present significant opportunities for modernizing thyroid cancer diagnostics and reducing unnecessary surgical interventions.
Qatar Thyroid Cancer Diagnostics Market Trends
Qatar, through its Ministry of Public Health (MoPH) and flagship institutions such as Hamad Medical Corporation, is investing heavily in oncology diagnostics, including thyroid cancer. Though the national incidence rate is moderate compared to Saudi Arabia and the UAE, rising awareness and early detection initiatives are driving demand for ultrasound and cytology. Molecular diagnostics adoption remains limited but is expected to grow, especially in tertiary hospitals and research centers. Reimbursement for advanced assays is still evolving, but precision oncology initiatives are gaining momentum. Opportunities exist in molecular classifiers for indeterminate nodules and partnerships with research institutes to accelerate adoption.
Oman Thyroid Cancer Diagnostics Market Trends
Oman’s thyroid cancer incidence is relatively moderate, but rising awareness, improved imaging availability, and a younger population profile are driving steady demand for diagnostics. The Ministry of Health (MOH) governs regulation and public health delivery, with government hospitals as the primary providers. Current diagnostic approaches rely heavily on ultrasound and FNA, while molecular testing adoption remains minimal, often requiring international reference labs for advanced assays. Investment in healthcare modernization and integration of NGS capabilities within national hospitals likely to be critical to expanding precision diagnostics. Opportunities also lie in public-private collaborations to enhance early detection and reduce overtreatment.
Key Middle East Thyroid Cancer Diagnostics Company Insights
Key players in the ME thyroid cancer diagnostics market include F.Hoffmann-La Roche Ltd., Thermo Fisher Scientific, Inc., Siemens Healthcare GmbH, and others. These companies implement strategies such as investing in research and development, introducing advanced diagnostic technologies, expanding product portfolios, forming collaborations and partnerships, and focusing on regulatory approvals. These approaches help enhance diagnostic accuracy, broaden market reach, and address evolving patient and healthcare provider needs.
-
Siemens Healthcare GmbH offers a comprehensive portfolio of laboratory assays and automated immunoassay systems designed to detect, diagnose, and monitor thyroid disorders, including thyroid cancer. The company manufactures and supplies advanced diagnostic solutions that are used to assess thyroid function, monitor differentiated thyroid cancer, and support clinical decision-making. Siemens operates primarily in the laboratory diagnostics and medical imaging segments.
-
Bio-Rad Laboratories, Inc. develops and markets a wide range of molecular testing kits, reagents, and automated systems that enable the detection and analysis of thyroid cancer biomarkers. Bio-Rad operates in the clinical diagnostics segment, providing tools for genetic, protein, and cellular analysis. It supports both routine diagnostic testing and advanced research in thyroid cancer and related endocrine disorders.
Key Middle East Thyroid Cancer Diagnostics Companies:
- Abbott
- F.Hoffmann-La Roche Ltd.
- Thermo Fisher Scientific, Inc.
- Siemens Healthcare GmbH
- Bio-Rad Laboratories, Inc
- GE HealthCare
- Hologic, Inc.
- Koninklijke Philips N.V.
- Toshiba Corporation
- Agilent Technologies, Inc.
- Illumina, Inc.
Middle East Thyroid Cancer Diagnostics Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 93.15 million
Revenue forecast in 2033
USD 142.27 million
Growth rate
CAGR of 5.4% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Cancer type, technique, end use, and country.
Country scope
Saudi Arabia; UAE; Kuwait; Qatar; Oman
Key companies profiled
Abbott; F.Hoffmann-La Roche Ltd.; Thermo Fisher Scientific, Inc.; Siemens Healthcare GmbH; Bio-Rad Laboratories, Inc; GE HealthCare; Hologic, Inc.; Koninklijke Philips N.V.; Toshiba Corporation; Agilent Technologies, Inc.; Illumina, Inc
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Middle East Thyroid Cancer Diagnostics Market Report Segmentation
This report forecasts revenue growth at regional and country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Middle East thyroid cancer diagnostics market report based on cancer type, technique, end use, and country.
-
Cancer Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Papillary carcinoma
-
Follicular carcinoma
-
Others
-
-
Technique Outlook (Revenue, USD Million, 2021 - 2033)
-
Blood Test
-
Imaging
-
Biopsy
-
Others
-
-
End Use Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospital Laboratories
-
Cancer Diagnostic Centers
-
Research Institutes
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
Middle East
-
Saudi Arabia
-
UAE
-
Kuwait
-
Qatar
-
Oman
-
-
Frequently Asked Questions About This Report
b. The Middle East thyroid cancer diagnostics market size was estimated at USD 89.64 million in 2024 and is expected to reach USD 93.15 million in 2025.
b. The Middle East thyroid cancer diagnostics market is expected to grow at a compound annual growth rate of 5.4% from 2025 to 2033 to reach USD 142.27 million by 2033.
b. In the Middle East, papillary carcinoma dominates the thyroid cancer diagnostics market, contributing the largest revenue share at around 80.01% in 2024, driven by its high prevalence across GCC and neighboring countries. Increased awareness, government-led screening initiatives, and the growing availability of advanced imaging and molecular testing have improved early detection rates. Additionally, environmental exposures, lifestyle transitions, and the relatively younger average age at diagnosis in the region continue to elevate case numbers, supporting strong demand for accurate diagnostic solutions.
b. Abbott; F.Hoffmann-La Roche Ltd.; Thermo Fisher Scientific, Inc.; Siemens Healthcare GmbH; Bio-Rad Laboratories, Inc; GE HealthCare; Hologic, Inc.; Koninklijke Philips N.V.; Toshiba Corporation; Agilent Technologies, Inc.; Illumina, Inc
b. The Middle East thyroid cancer diagnostics market shows rising demand, driven by gender-skewed incidence, with female rates notably higher, especially in the UAE. Current practice relies on ultrasound, FNA, and cytology, while molecular testing adoption remains low (~20%), leaving significant growth potential for NGS panels and mutation/fusion assays. Regulatory oversight varies across countries, and reimbursement challenges slow uptake. However, opportunities exist in precision oncology, Bethesda III/IV nodule classifiers, and partnerships with local hospitals and labs to expand access and reduce overtreatment.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










