- Home
- »
- Medical Devices
- »
-
North America Clinical Trials Market, Industry Report, 2033GVR Report cover
![North America Clinical Trials Market Size, Share & Trends Report]()
North America Clinical Trials Market (2025 - 2033) Size, Share & Trends Analysis Report By Phase (Phase I, Phase II, Phase III, Phase IV), By Study Design, By Indication, By Indication by Study Design, By Service, By Sponsor, By Country, And Segment Forecasts
- Report ID: GVR-4-68040-202-2
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
North America Clinical Trials Market Summary
The North America clinical trials market size was estimated at USD 43.03 billion in 2024 and is projected to reach USD 77.32 billion by 2033, growing at a CAGR of 6.98% from 2025 to 2033. The market growth is driven by a robust ecosystem of academic research institutions, growing research and development funding by pharmaceutical companies, and specialized contract research organizations (CROs) that support a high volume of early-to late-phase studies.
Key Market Trends & Insights
- The North America clinical trials industry in the U.S. is expected to grow significantly over the forecast period.
- By phase, the phase III segment led the market with the largest revenue share of 53.68% in 2024.
- Based on study design, the interventional trials segment led the market with the largest revenue share in 2024.
- By indication, the oncology segment held the highest market share in 2024.
- By sponsor, the pharmaceutical & biopharmaceutical companies segment held the largest market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 43.03 Billion
- 2033 Projected Market Size: USD 77.32 Billion
- CAGR (2025-2033): 6.98%
- U.S.: Largest market in 2024
- Mexico: Fastest growing market
The North America clinical trials market is propelled by a highly developed research infrastructure and a concentration of global pharmaceutical, biotechnology, and medical device companies that consistently prioritize clinical development. The presence of several academic research centers and hospitals such as the Mayo Clinic, MD Anderson Cancer Center, and Harvard Medical School is driving the innovation and partnerships in early-stage trials. Moreover, the regulatory agencies such as the U.S. FDA are offering well-established frameworks for Investigational New Drug (IND) applications, Orphan Drug Designations, and adaptive trial designs which is lowering the entry barriers and accelerating the clinical timeline.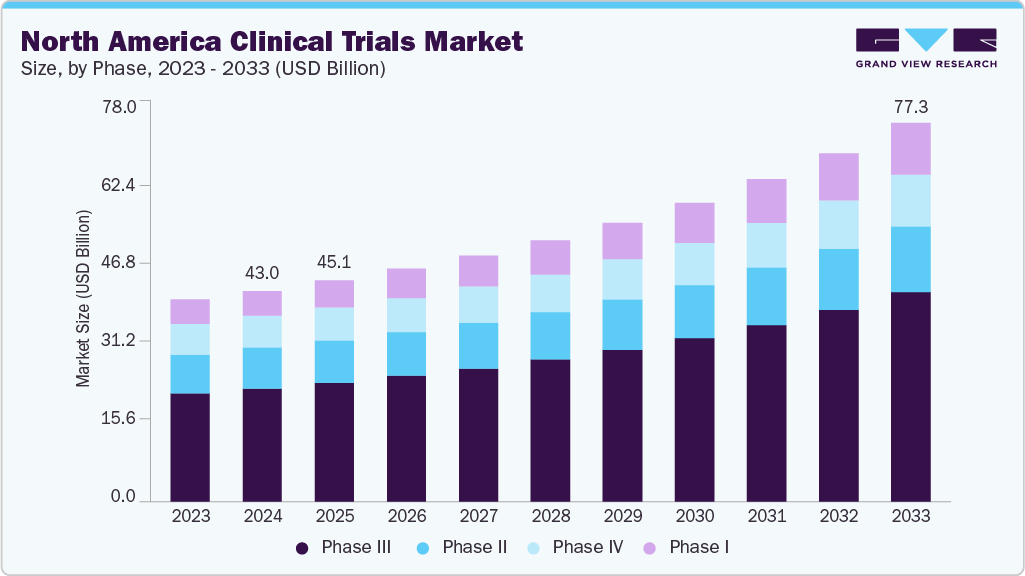
Furthermore, increasing collaboration between industry stakeholders, digital health innovators, and patient advocacy groups. Pharmaceutical companies are not only expanding their in-house R&D pipelines but are also actively partnering with CROs to streamline study design, site management, and regulatory submissions. This trend allows sponsors to accelerate timelines without compromising on data integrity or patient safety. Similarly, rising patient awareness and education, often supported by advocacy foundations and public health campaigns has resulted in greater trial participation, particularly in complex areas such as oncology and rare diseases. The shift toward more inclusive enrollment strategies, supported by federal guidelines and diversity mandates, ensures that clinical outcomes better reflect real-world populations.
Opportunity Analysis
The North America clinical trials market presents strong opportunities driven by evolving trial models, increasing demand for personalized medicine, and advancements in data-driven research methodologies. The shift toward decentralized clinical trials (DCTs), enabled by wearable technologies, remote monitoring tools, and telemedicine platforms, opens avenues to reach wider and more diverse patient populations, especially in underserved and rural regions. Opportunities also lie in the growing focus on precision therapies, where trials require smaller cohorts but deeper molecular profiling, creating demand for specialized CROs and integrated diagnostic services.

Technological Advancements
Technological advancements are playing a transformative role in reshaping the clinical trials landscape across North America. The widespread integration of electronic data capture (EDC) systems, cloud-based platforms, and real-time monitoring tools has significantly improved the efficiency and transparency of trial operations. Sponsors and CROs are increasingly adopting artificial intelligence (AI) and machine learning (ML) algorithms to optimize protocol design, predict patient dropout rates, and identify ideal trial sites based on historical and demographic data. Wearable devices and mobile health applications have enabled remote patient monitoring, allowing for decentralized and hybrid trial models that reduce the dependency on physical sites and improve participant convenience. Blockchain technologies are also gaining traction for secure and tamper-proof data management, especially in multi-site and global trials. These innovations not only accelerate the speed of drug development but also enhance regulatory compliance and patient engagement, and positioning.

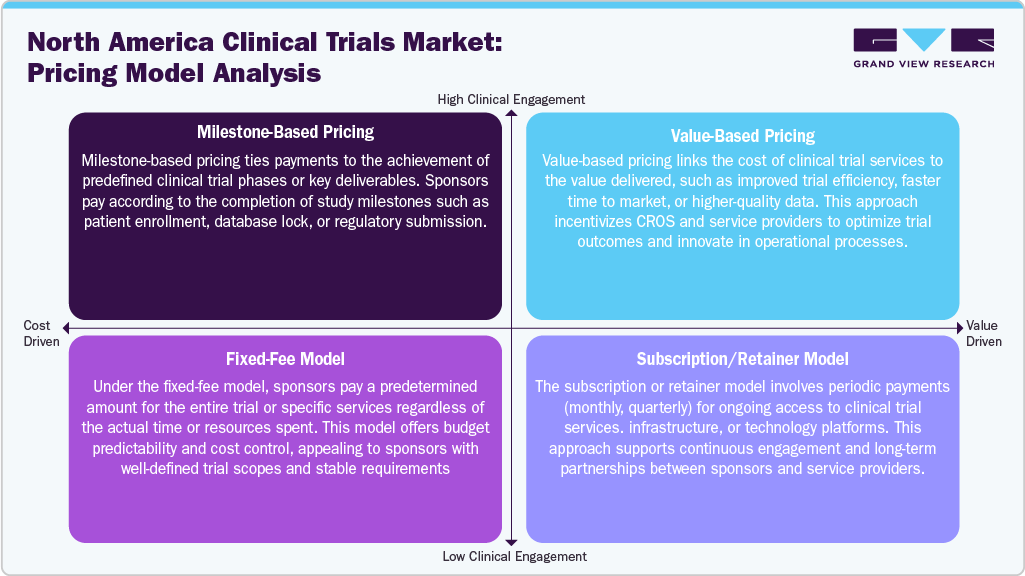
Pricing Analysis
The pricing structure in the North America clinical trials market is shaped by the complexity of study design, therapeutic area, trial phase, and level of technological integration. Typically, service providers including CROs adopt three primary pricing models: fixed-price, time-and-materials (T&M), and hybrid models. In early-phase studies, especially Phase I trials, T&M pricing is common due to the unpredictability of patient response and protocol modifications. Sponsors are billed based on actual hours worked and resources utilized, offering flexibility but requiring close budget tracking. In addition, fixed-price models are favoured for later-stage trials (Phase III), where study parameters are well-defined. This model allows sponsors to manage costs upfront but shifts the risk of overruns to the CRO. Hybrid pricing, which combines both approaches, is increasingly used for complex, multi-country trials where some elements are predictable and others variable.
Phase Insights
The phase III segment accounted for the largest revenue share of the North America clinical trials industry, with 53.68% in 2024. The growth is due to the high complexity, extended duration, and large patient populations involved in Phase III studies, which significantly increase operational costs. The segment’s growth is driven by rising investments from pharmaceutical and biotechnology companies to validate drug efficacy and safety at scale before regulatory approval.
The phase I segment is anticipated to grow at the fastest CAGR during the forecast period. The segment growth is driven by the rising number of early-stage biotech and pharma startups, increased venture capital funding, and growing focus on first-in-human studies for novel therapeutics. Expansion in areas such as gene therapies, cell therapies, and precision oncology is contributing to higher demand for Phase I trials that assess safety, tolerability, and optimal dosing.
Study Design Insights
The interventional trials segment held the largest share of the North America clinical trials market in 2024, due to the high volume of pharmaceutical and biotechnology companies conducting Phase I–IV clinical trials to evaluate the safety and efficacy of novel therapeutics and medical devices. Interventional studies are the cornerstone of drug development and are essential for regulatory approvals, particularly in areas like oncology, neurology, and infectious diseases.
The observational trials segment is anticipated to grow at the fastest CAGR over the forecast period. The growth of the segment is due to the rising emphasis on real-world evidence (RWE) to support regulatory decision-making, post-marketing surveillance, and health economics outcomes research. Pharmaceutical and medical device companies are increasingly leveraging observational studies to understand long-term safety, treatment patterns, and patient behavior outside the controlled environment of interventional trials.
Indication Insights
The oncology segment held the largest share of the North America clinical trials industry in 2024, due to the increasing incidence of various cancers across the region, particularly breast, lung, prostate, and colorectal cancers, which has fueled the demand for innovative cancer therapeutics and diagnostics. A significant portion of clinical research funding and pipeline development is concentrated in oncology, leading to a higher volume of Phase I–IV cancer trials.
The diabetes segment is anticipated to grow at the second-fastest CAGR over the forecast period. The growth is driven by the rising prevalence of both type 1 and type 2 diabetes, fueled by aging populations, sedentary lifestyles, and increasing obesity rates. The demand for novel therapies such as GLP-1 receptor agonists, SGLT2 inhibitors, and insulin delivery technologies is spurring clinical research activity in this area.
Service Insights
The laboratory services segment held the largest share of the North America clinical trials market in 2024, due to its critical role in generating high-quality, regulatory-compliant data required for drug and device approvals. Increasing demand for biomarker validation, pharmacokinetics/pharmacodynamics (PK/PD) analysis, genetic testing, and central lab services has significantly expanded the scope of laboratory functions in clinical trials.
The patient recruitment segment is anticipated to grow at the fastest CAGR over the forecast period. The growth is due to increasing challenges in enrolling suitable participants for clinical studies, which has prompted sponsors and CROs to invest heavily in innovative recruitment strategies. The adoption of digital recruitment platforms, social media outreach, and patient-centric engagement approaches is improving enrollment efficiency and retention rates.
Sponsor Insights
The pharmaceutical & biopharmaceutical companies segment held the largest market share in 2024. due to their substantial investments in drug development pipelines, including innovative therapies for oncology, autoimmune diseases, and rare disorders. These companies lead the majority of clinical research initiatives, driven by the high demand for novel and improved therapeutics as well as regulatory requirements for robust clinical evidence.
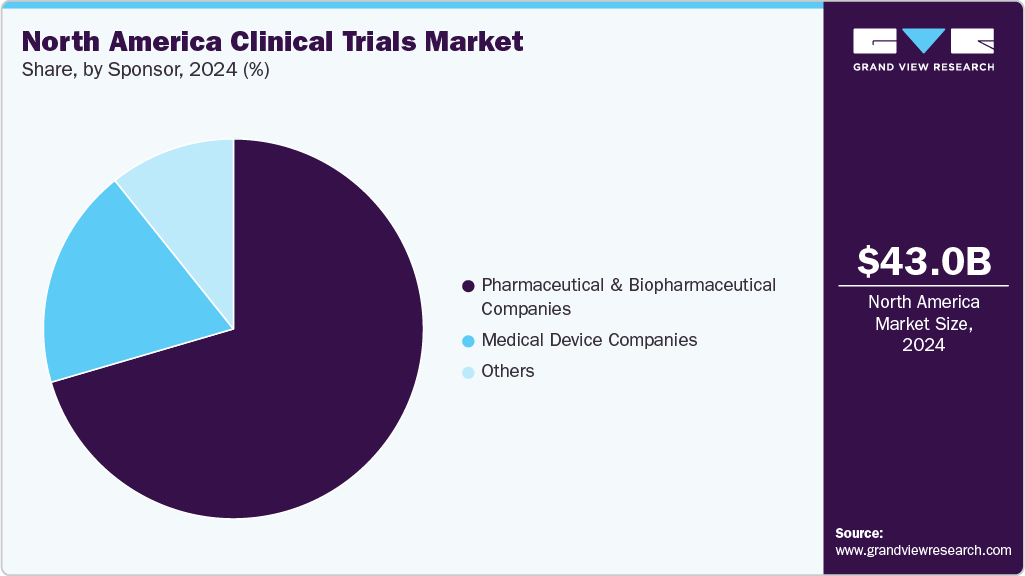
The medical device companies segment is anticipated to grow at the second-fastest CAGR over the forecast period. The segment growth is due to the increasing demand for innovative diagnostic and therapeutic devices, including minimally invasive tools, wearable health monitors, and AI-integrated technologies. Regulatory bodies such as the FDA have introduced streamlined approval pathways like the Breakthrough Devices Program, encouraging rapid clinical validation and market entry of high-impact devices.
Country Insights
U.S. Clinical Trials Market Trends
The clinical trials market in the U.S. held the largest share in 2024. The country’s growth is due to a robust regulatory framework, strong presence of leading pharmaceutical and biotech companies, and a well-established network of clinical research organizations (CROs). The country’s growth is also supported by substantial R&D investments, government funding through agencies like NIH, and a high volume of ongoing clinical trials across therapeutic areas such as oncology, neurology, and rare diseases.
Mexico Clinical Trials Market Trends
The clinical trials market in Mexico is anticipated to grow at the fastest CAGR over the forecast period. The high growth is driven by several key factorssuch as lower operational costs compared to the U.S. and Canada, a large and diverse patient population enabling faster recruitment, and growing regulatory alignment with international standards such as ICH-GCP. Moreover, Mexico’s expanding network of certified clinical research centers, increasing public and private investment in healthcare R&D, and rising participation in multinational trials are further propelling market growth. Government support and proximity to the U.S. also make Mexico a strategic hub for regional clinical trial expansion.
Key North America Clinical Trials Company Insights
Several key players are acquiring various strategic initiatives to strengthen their market position, offering diverse services to customers. The prominent strategies adopted by companies are service launches, mergers & acquisitions/joint ventures, mergers, partnerships & agreements, expansions, and others to increase market presence & revenue, and gain a competitive edge drives the market growth.
Key North America Clinical Trials Companies:
- Pfizer Inc.
- Merck & Co., Inc.
- AbbVie Inc.
- Amgen Inc.
- Bristol Myers Squibb
- Gilead Sciences, Inc.
- Eli Lilly and Company
- Biogen Inc.
- Moderna, Inc.
- Labcorp
- IQVIA Inc.
- Syneos Health
- Paraxel International
- ICON plc
- Thermo Fisher Scientific, Inc.
- Charles River Laboratories International, Inc.
- Medpace Holdings, Inc.
- Fortrea
- Worldwide Clinical Trials
- Clinipace Inc.
Recent Developments
-
In May 2025, IQVIA announced to introduce a site lab navigator to optimize clinical trial workflows. This innovative suite of solutions is designed to automate and optimize laboratory workflows for both clinical trial sponsors and investigator sites.
-
In April 2025, Thermo Fisher Scientific Inc. announced to use advanced platform technology along with a new CHO K-1 cell line, which can shorten the timeline for Investigational New Drug (IND) filings from 13 months to nine months. This innovation assists biotech and pharmaceutical companies in navigating the logistical challenges associated with pre-clinical biologic drug development.
-
In January 2025, ICON plc announced the enhancement of its range of artificial intelligence (AI) tools, which aim to improve efficiency throughout the clinical trial process. This includes areas such as study initiation, document handling, resource forecasting, and reporting of metrics.
North America Clinical Trials Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 45.08 billion
Revenue forecast in 2033
USD 77.32 billion
Growth rate
CAGR of 6.98% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Phase, study design, indication, indication by study design, service, sponsor
Country scope
U.S.; Canada; Mexico
Key companies profiled
Pfizer Inc.; Merck & Co., Inc.; AbbVie Inc.; Amgen Inc.; Bristol Myers Squibb; Gilead Sciences, Inc.; Eli Lilly and Company; Biogen Inc.; Moderna, Inc.; Labcorp; IQVIA Inc.; Syneos Health; Paraxel International; ICON plc; Thermo Fisher Scientific, Inc.; Charles River Laboratories International, Inc.; Medpace Holdings, Inc.; Fortrea; Worldwide Clinical Trials; Clinipace Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
North America Clinical Trials Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the North America clinical trials market report based on phase, study design, indication, indication by study design, service, sponsor, and country:
-
Phase Outlook (Revenue, USD Million, 2021 - 2033)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Study Design Outlook (Revenue, USD Million, 2021 - 2033)
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Autoimmune/Inflammation
-
Rheumatoid Arthritis
-
Multiple Sclerosis
-
Osteoarthritis
-
Irritable Bowel Syndrome (IBS)
-
Others
-
-
Pain Management
-
Chronic Pain
-
Acute Pain
-
-
Oncology
-
Blood Cancer
-
Solid Tumors
-
Others
-
-
CNS Conditions
-
Epilepsy
-
Parkinson's Disease (PD)
-
Huntington's Disease
-
Stroke
-
Traumatic Brain Injury (TBI)
-
Amyotrophic Lateral Sclerosis (ALS)
-
Muscle Regeneration
-
Others
-
-
Diabetes
-
Obesity
-
Cardiovascular Diseases
-
Others
-
-
Indication by Study Design Outlook (Revenue, USD Million, 2021 - 2033)
-
Autoimmune/Inflammation
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Pain Management
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Oncology
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
CNS Conditions
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Diabetes
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Obesity
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Cardiovascular Diseases
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Others
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Protocol Designing
-
Site Identification
-
Patient Recruitment
-
Laboratory Services
-
Analytical Testing Services
-
Clinical Trial Data Management Services
-
Others
-
-
Sponsor Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical & Biopharmaceutical Companies
-
Medical Device Companies
-
Others
-
-
Country Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Frequently Asked Questions About This Report
b. The North America clinical trials market is estimated at USD 43.0 billion in 2024 and is expected to reach USD 45.1 billion in 2025.
b. The North America clinical trials market is expected to grow at a CAGR of 6.98% from 2025 to 2030 to reach USD 77.32 billion in 2033.
b. Phase III dominated the market with a revenue share of 53.68% in 2024. The growth of the segment is due to the high complexity, extended duration, and large patient populations involved in Phase III studies, which significantly increase operational costs.
b. Some of the prominent players operating in the market include Labcorp, IQVIA, Syenos Health, Paraxel International, ICON plc, Thermo Fisher Scientific, Inc., Charles River Laboratories International, Inc., WuXi AppTec, Eli Lilly and Company, Holdings, SGS SA
b. Key factors driving the market growth include robust ecosystem of academic research institutions, growing research and development funding by pharmaceutical companies, and specialized contract research organizations (CROs) that support a high volume of early- to late-phase studies
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










