- Home
- »
- Biotechnology
- »
-
North America, Europe & Asia Pacific Gene Therapy Cell Culture Media Market, 2033GVR Report cover
![North America, Europe & Asia Pacific Gene Therapy Cell Culture Media Market Size, Share & Trends Report]()
North America, Europe & Asia Pacific Gene Therapy Cell Culture Media Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Specialty Media, Serum-free Media), By Vector Type (AAV, LV), By End User, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-762-6
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
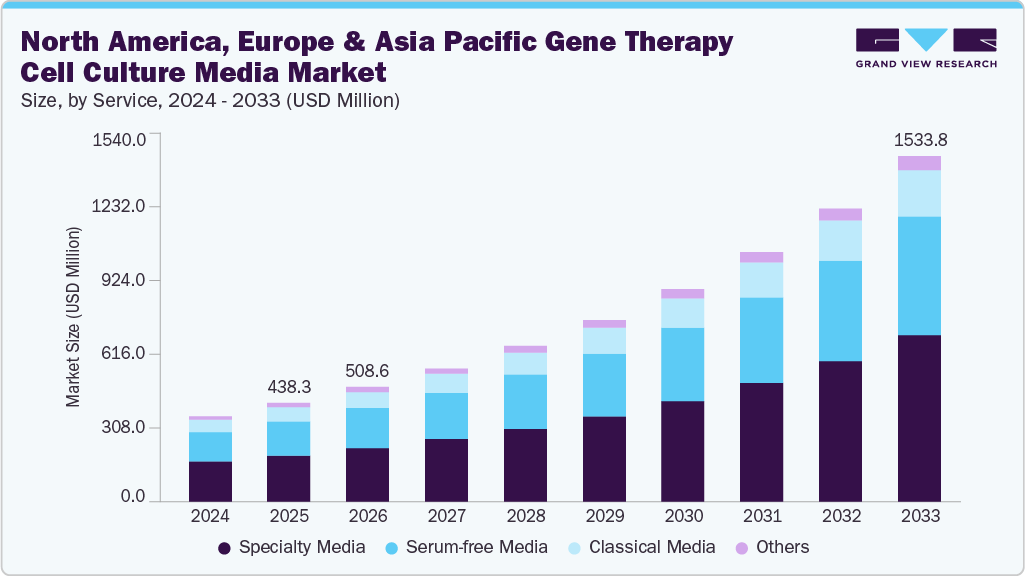
North America, Europe & Asia Pacific Gene Therapy Cell Culture Media Market Summary
The North America, Europe & Asia Pacific gene therapy cell culture media market size was estimated at USD 438.3 million in 2025 and is expected to reach a value of USD 1,533.8 million by 2033, growing at a CAGR of 17.08% from 2026 to 2033. The gene therapy cell culture media market in North America, Europe, and the Asia Pacific is driven by rising clinical trial activity, expanding biomanufacturing capacity, and the growing need for GMP-compliant, chemically defined media. Together, these regions are accelerating the adoption of high-performance media to boost vector yields and streamline production.
Key Market Trends & Insights
- The North America in North America, Europe & Asia Pacific gene therapy cell culture media market held the largest share of 40.48% of the market in 2025.
- The North America, Europe & Asia Pacific gene therapy cell culture media industry in the U.S. is expected to grow significantly over the forecast period.
- By product, the specialty media segment held the highest market share of 46.75% in 2025.
- Based on vector type, the AAV production segment held the highest market share in 2025.
- By end user, the pharmaceutical and biotechnology companies segment held the highest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 438.3 Million
- 2033 Projected Market Size: USD 1,533.8 Million
- CAGR (2026-2033): 17.08%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Growth In Rare Genetic Disease Therapies And Strong Regulatory SupportThe push to develop therapies for rare genetic disorders has become a major driver in the gene therapy cell culture media market. Although each rare disease affects fewer than 200,000 people in the U.S., there are nearly 7,000 such conditions worldwide, collectively impacting more than 25 million Americans. Many of these disorders stem from single-gene mutations, which makes them especially suitable for gene therapy approaches that correct or replace faulty genes. Unlike treatments that only manage symptoms, gene therapy has the potential to offer long-term, sometimes even curative outcomes. Achieving this, however, depends on specialized culture media. Researchers and manufacturers need serum-free, chemically defined formulations that can nurture fragile cells across every stage of development from early lab work to preclinical studies and full-scale clinical production. Standard, one-size-fits-all media often falls short, creating demand for tailored solutions for sensitive or patient-derived cell types.
Key Statistics Of Rare Disease
Particulars
Value
People are Affected by Rare Disease
1 in 10
Million People Suffer From a Rare Disease Globally
400 million
% of Rare Diseases Lack an FDA Approved Treatment
95%
Rare Diseases Start in Childhood
7 in 10
Rare Diseases are Genetic
8 in 10
Distinct types of Rare and Genetic Diseases
10,000+
Source: Global Genes, Secondary Research, Grand View Research
At the same time, regulatory policies are making this field more attractive for investment. Many countries now offer programs that fast-track approvals, grant tax breaks, extend market exclusivity, or provide direct funding, all aimed at reducing the risk of working on therapies for small patient populations. While these incentives vary by region, they all support the same idea of making treatments for rare diseases financially viable and available to the patients who need them. This blend of scientific opportunity and regulatory support is evolving the landscape to make it attractive for biopharma companies and contract manufacturers to invest talent and resources into developing scalable and high-performing media solutions. As rare disease programs expand, the need for reliable, customized culture media will only grow, reinforcing its central role in the future of gene and cell therapies.
Increasing Capacity For Viral Vector Production
There is a growing need for viral vectors, most often AAVs, lentiviruses, and adenoviruses, that act as vehicles for carrying therapeutic genes. As more gene therapy candidates move through the pipeline, what used to be manageable with small, academic-scale vector production is not enough. Early-stage lab work can rely on limited batches. Still, the gap between demand and capacity becomes obvious once therapies advance into larger trials with multiple study sites, or even toward full commercial release. This has created urgency around finding more efficient ways to manufacture these vectors at scale, and media formulations are at the heart of that challenge.
The industry trend toward bioreactors and single-use systems has only amplified the need for dedicated media. These systems require consistency in product performance, maintaining healthy cells while pushing viral yields higher and ensuring every batch behaves the same way. Contract development and manufacturing organizations often shoulder the responsibility of simultaneously scaling production for many different sponsors, especially those that depend on high-quality, GMP-grade media. Traditional small-scale production methods are insufficient to meet clinical and market demand, especially as therapies transition from early research stages to large, multi-center clinical trials and eventual commercial launch.
Market Concentration & Characteristics
Innovation in North America, Europe, and Asia Pacific gene therapy cell culture media industry varies by region but is accelerating overall. North America focuses on cutting-edge, chemically defined, serum-free media driven by biotech growth. Europe emphasizes safety, consistency, and regulatory compliance, while Asia Pacific combines rapid facility expansion and government support to experiment with scalable, cost-effective solutions. Across all regions, the goal is to make media reliably support the journey from lab bench to patient-ready therapies.
M&A activity in the North America, Europe & Asia Pacific gene therapy cell culture media industry has steadily risen as companies aim to strengthen their capabilities and expand their portfolios. Firms are acquiring or merging with specialized media developers to gain access to innovative formulations, proprietary technologies, and scalable production solutions. These deals facilitate research and development activities and enhance manufacturing efficiency and commercialization. All in all, mergers and acquisitions are a cornerstone of growth, innovation, and fulfilling rising consumer demand for innovative, quality, reliable cell culture media.
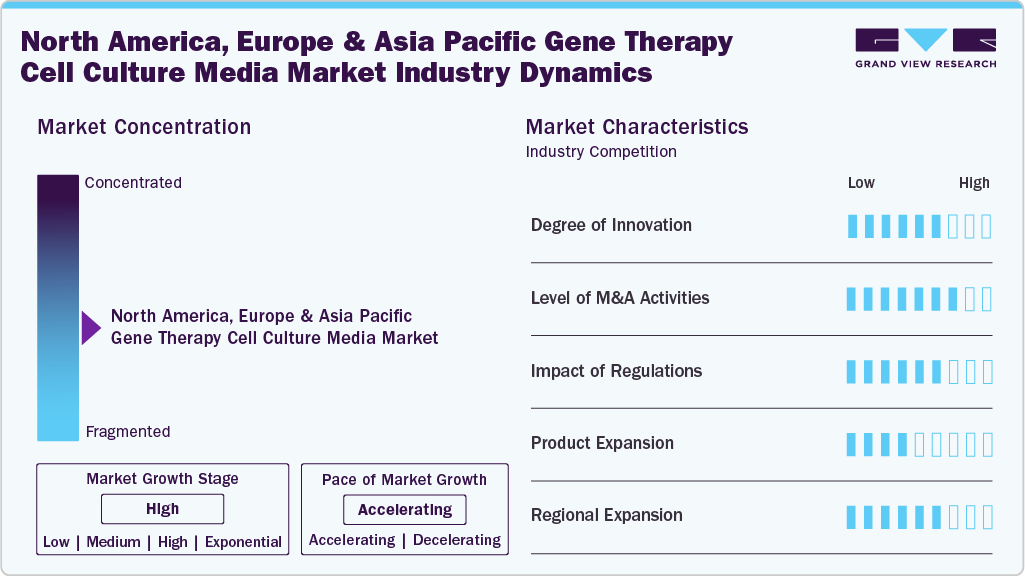
Regulations profoundly impact the North America, Europe & Asia Pacific gene therapy cell culture media industry. Strict guidelines around quality, safety, and compliance ensure that media used in research and manufacturing meet high standards, particularly for clinical and commercial applications. These rules push companies to develop serum-free, chemically defined, and GMP-compliant formulations, which can support sensitive cells while minimizing variability. At the same time, regulatory frameworks provide clarity and structure, giving manufacturers confidence to scale production and invest in innovative solutions. While regulations can be demanding, they drive higher-quality products and foster trust in gene and cell therapy processes.
In North America, Europe & Asia Pacific gene therapy cell culture media industry, product offerings are expanded due to the demand for the increasing complexity of therapies and cell types. Companies are developing new formulations, including sterile-free and chemically defined specialty media tailored to certain cell types to enhance cell growth, viability, and reproducibility. Expanding media product offerings can be a way to solve existing customer needs, as well as to keep up with the changing landscape of the industry and stay competitive and responsive to the changing demand for scalable, high-performance media.
Regional expansion in North America, Europe & Asia Pacific gene therapy cell culture media industry is focused on reaching emerging biotech hubs and expanding manufacturing and distribution networks. Companies are establishing new facilities, partnerships, and collaborations to serve growing markets and improve accessibility to high-quality media. By entering new regions, manufacturers can better meet local demand, reduce supply chain bottlenecks, and support the increasing number of gene and cell therapy programs worldwide. This expansion helps drive market growth while strengthening regional presence and competitiveness.
Product Insights
The specialty media segment accounted for the largest market share of 46.75% in 2025 due to its critical role in supporting the growth and productivity of sensitive cell types used in gene and cell therapies. These media are specifically designed to improve cell viability and increase production of viral vectors while allowing for consistency across batches in regulatory-approved settings, which is crucial for research and commercial-scale manufacturing. As a custom product, many of these serum-free and chemically defined media provide significant value for heightened performance and have seen compelling adoption rates across biopharmaceutical companies and contract manufacturing organizations.
The serum-free media segment is expected to grow significantly over the forecast period, driven primarily by the demand for safer, clinically compliant cell culture solutions with greater consistency. Serum-free media can be used with sensitive cell types and in therapeutic applications. In addition, preferences for media without animal components from regulatory bodies and growth in manufacturing gene and cell therapies are also driving serum-free media adoption, as companies seek media that will reliably and sustainably deliver the level of performance required to support the commercial production of their products.
Vector Type Insights
The AAV production segment dominated the market with a share of 51.49% in 2025, driven by the broad use of AAV in in vivo gene therapy applications for rare genetic disorders, ocular disorders, and neuromuscular conditions. In addition, the transition from adherent to suspension-based AAV production platforms that need optimized media that can support high-density cell culturing and robust triple-transfection workflows in bioreactors is expected to drive the growth of this segment.
The LV production segment is expected to grow at a significant CAGR during the forecast period, fueled by the shift from small academic batches to large-scale GMP manufacturing. Chemically defined, serum-free, and animal-origin-free media are preferred to minimize variability and streamline regulatory approvals.
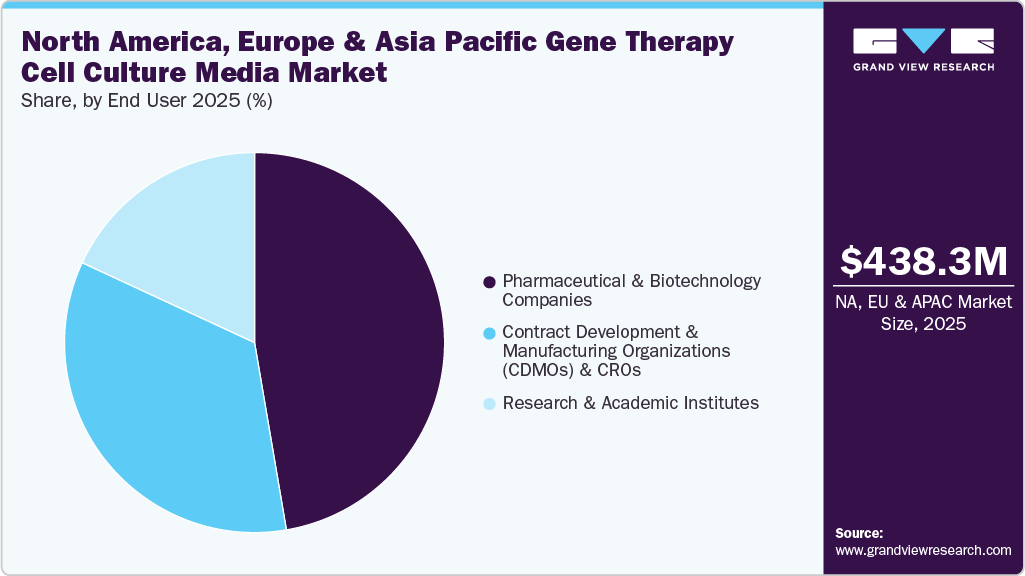
End User Insights
The pharmaceutical and biotechnology companies segment captured the largest market share, 47.31%, in 2025. Pharmaceutical companies are increasing their investment in advanced therapy manufacturing infrastructure, which directly stimulates demand for high-quality cell culture media. Companies such as Pfizer, Novartis, and Roche have expanded their in-house vector production capabilities, requiring scalable media solutions for suspension cell cultures and fed-batch processes. Thus, the growth of the biopharmaceutical industry, the rise in the number of clinical trials, and initiatives for developing novel drugs are factors expected to boost market growth.
The hospital and diagnostic laboratory segment is projected to grow significantly during the forecast period. This growth is driven by the rising use of North America, Europe & Asia Pacific gene therapy cell culture media in diagnostic testing, the increasing prevalence of chronic diseases, and the growing adoption of advanced diagnostic technologies. The need for hospitals with advanced facilities has increased with ongoing changes in the healthcare industry. The rise in hospitals and diagnostic laboratories has also led to high segment growth. With the increase in the prevalence of chronic diseases, such as cancer and infectious and autoimmune diseases, the need for hospitals and diagnostic centers to detect antigens in cancer cells is increasing, thus driving the North America, Europe & Asia Pacific gene therapy cell culture media market.
Regional Insights
North America dominated the gene therapy cell culture media industry in 2025, holding a 40.48% share. This dominance is attributed to supportive policies, significant investment in research and development, and a large concentration of biopharma and biotech hubs. North America's favorable policies, research and development infrastructure, and expanding therapy pipeline will see it emerge as the main growth driver of the gene therapy cell culture market.
U.S. Gene Therapy Cell Culture Media Market Trends
The U.S. gene therapy cell culture media industry is highly competitive because of a well-established R&D infrastructure and a favorable regulatory landscape, which is rapidly evolving to adapt to the ongoing research progress in this sector. Investments by the government and leading players in the pharmaceutical and biotechnology industries are growing. In April 2023, the California Institute for Regenerative Medicine (CIRM) Board invested USD 89 million in stem cell and gene therapy research, which leads to market expansion in the country.
Europe Gene Therapy Cell Culture Media Market Trends
The Europe gene therapy cell culture media market is witnessing steady growth, fueled by strong regulatory support, public funding, and others. Programs such as Horizon Europe are funding research initiatives in regenerative medicine and personalized therapies, which inherently rely on high-performance, clinical-grade media. Regulatory authorities such as the European Medicines Agency (EMA) are also raising quality expectations, emphasizing GMP compliance, traceability, and a shift toward serum-free and animal-component-free media to reduce contamination risks. This regulatory and funding push is driving demand for highly consistent, reproducible media formulations supporting early-stage research and commercial manufacturing.
The UK gene therapy cell culture media market is experiencing significant growth, driven by regulatory advancements, increased clinical adoption, and substantial infrastructure investments. Moreover, in collaboration with Cellular Origins and Resolution Therapeutics, the Cell and Gene Therapy Catapult has formed a consortium to deliver robotics-based manufacturing solutions for cell and gene therapies. These initiatives are expected to bolster UK's position in the gene therapy landscape, driving further demand for high-quality, scalable cell culture media solutions.
Germany’s strong biotechnology ecosystem, supported by significant public sector funding, advanced regulatory infrastructure, and well-established manufacturing capabilities, is a major driver of demand for high-quality, GMP-grade cell culture media. Companies developing gene therapies, viral vectors, CAR-T, and similar cell therapies need media and supplements that are consistent, free from animal components, and capable of supporting scale-up and further boost the demand in the country.
Asia Pacific Gene Therapy Cell Culture Media Market Trends
Asia Pacific is expected to grow the fastest in the gene therapy cell culture media market, with a CAGR of 19.32% during the forecast period. This rapid growth is fueled by increasing investment in biotechnology infrastructure, rising clinical trial activity, supportive government policies, and expanding manufacturing capabilities. The region’s focus on cost-effective production, growing awareness, and adoption of advanced therapies are driving strong demand for high-quality, scalable cell culture media solutions.
The China gene therapy cell culture media industry is growing rapidly, driven by robust policy support, substantial investments in biotechnology infrastructure, and an expanding clinical trial landscape. Furthermore, the shift in focus of the industry in China from generics to innovative biopharmaceuticals is creating growth opportunities. In addition, the availability of funding, enhanced focus on research, and cross-border deals facilitate biopharmaceutical industry growth in the region. For instance, over 60 biopharmaceutical licensing deals were signed between Chinese companies and international players in 2020. These factors are expected to create a strong demand for cell culture media for the mass production of biopharmaceuticals.
The Japan gene therapy cell culture media market is growing as it has one of the region's most developed pharmaceutical and biotechnology sectors. Increasing investments in the cell culture domain also drive the market. For instance, Japan-based Fujifilm announced an acquisition of IS Japan (ISJ) and Irvine Scientific Sales Company (ISUS) for USD 800 million, expanding into the cell culture media space. Such investments and expansion activities can generate substantial revenue for cell culture media.
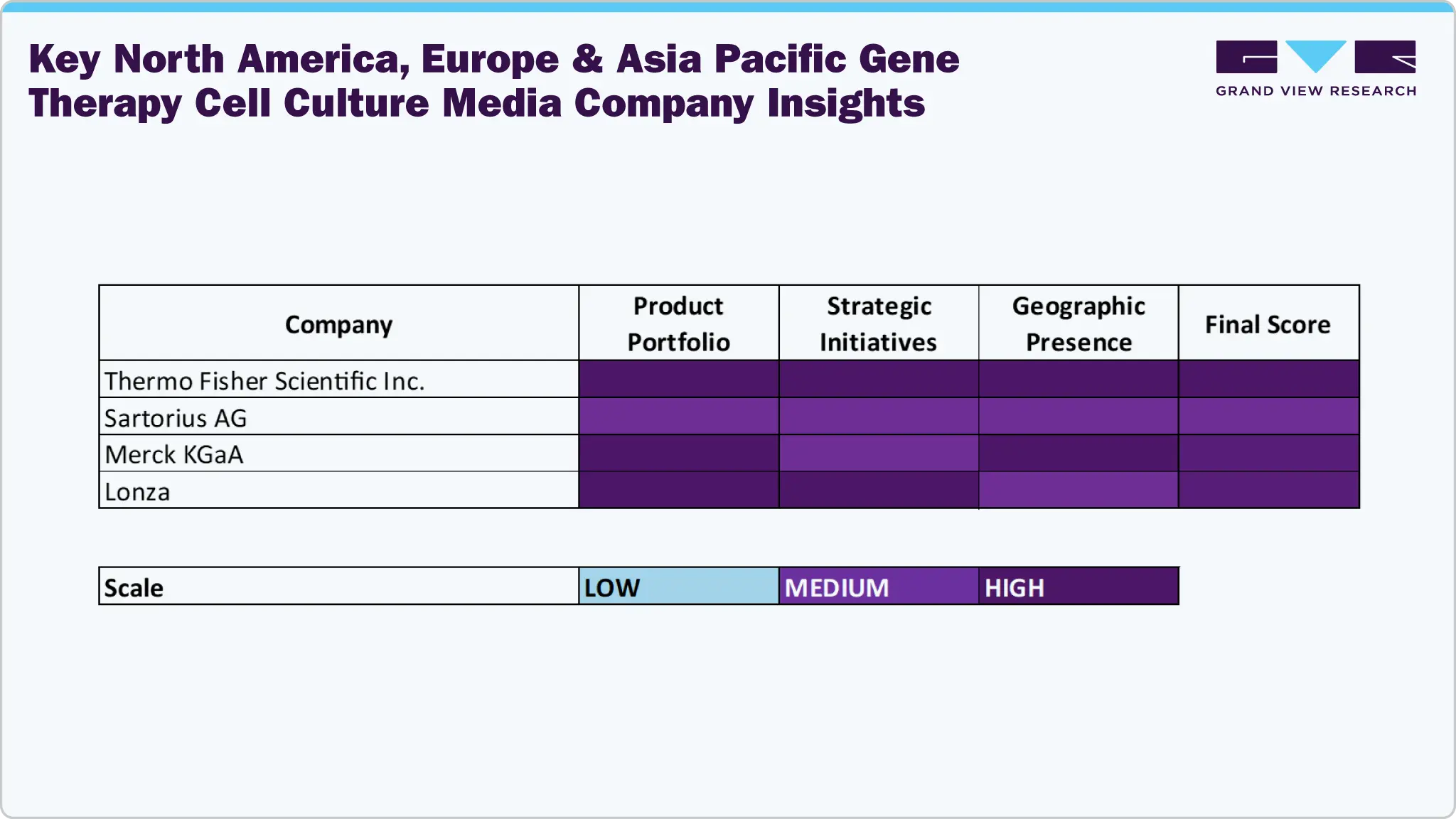
Key North America, Europe & Asia Pacific Gene Therapy Cell Culture Media Company Insights
The North America, Europe & Asia Pacific gene therapy cell culture media industry features several very notable companies, exhibiting leadership through extensive product portfolios, strategic partnerships and continued investment in R&D. Prominent players such as Thermo Fisher Scientific Inc., Sartorius AG, Merck KGaA, Lonza, Danaher Corporation (Cytiva), and FUJIFILM Holdings Corporation have cultivated meaningful market share by producing advanced, high performance media solutions, with broad application support and large distribution networks.
Organizations such as MP Biomedicals, Corning Incorporated, PAN-Biotech, Bio-Techne (R&D Systems, Inc.), InVitria Inc., Florabio AS, Revvity, and Virica are growing their reach through the supply of specialized and customizable media that meet the unique needs of gene and cell therapy applications. These products serve a range of end-users, including biopharmaceutical firms, contract manufacturing companies, and research organizations, to meet the need for media that provides cell viability, potency, and reproducibility.
The development of this market shows a coming together of science and solutions directed at the customer. These leading players are in a solid position to shape the future of gene and cell therapy manufacturing while adhering to broader healthcare and quality standards.
Key North America, Europe & Asia Pacific Gene Therapy Cell Culture Media Companies:
- Thermo Fisher Scientific Inc.
- Sartorius AG
- Merck KGaA
- Lonza
- Danaher Corporation (Cytiva)
- FUJIFILM Holdings Corporation
- MP Biomedicals
- Corning Incorporated
- PAN-Biotech
- Bio-Techne (R&D Systems, Inc.)
- InVitria Inc.
- Florabio AS
- Revvity
- Virica
Recent Developments
-
In September 2025, Fujifilm Biosciences launched BalanCD HEK293 Perfusion A medium. This medium enables scalable, high-density viral vector production for gene therapies through suspension HEK293 cells, perfusion technology, and broad workflow compatibility.
-
In August 2024, Nucleus Biologics launched QuickStart Media in the U.S., offering customizable, high-performance cell culture media to accelerate therapy development and streamline cell and gene therapy research.
North America, Europe & Asia Pacific Gene Therapy Cell Culture Media Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 508.6 million
Revenue forecast in 2033
USD 1,533.8 million
Growth rate
CAGR of 17.08% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 – 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, vector type, end user, region
Regional scope
North America; Europe; Asia Pacific
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; India; China; Japan; Australia; South Korea
Key companies profiled
Thermo Fisher Scientific Inc.; Sartorius AG; Merck KGaA; Lonza; Danaher Corporation (Cytiva); FUJIFILM Holdings Corporation; MP Biomedicals; Corning Incorporated; PAN-Biotech; Bio-Techne (R&D Systems, Inc.); InVitria Inc.; Florabio AS; Revvity; Virica
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
North America, Europe & Asia Pacific Gene Therapy Cell Culture Media Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For the purpose of this report, Grand View Research has segmented the North America, Europe & Asia Pacific gene therapy cell culture media market on the basis of product, vector type, end user, and region.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Specialty Media
-
Serum-free Media
-
Classical Media
-
Others
-
-
Vector Type Outlook (Revenue, USD Million, 2021 - 2033)
-
AAV Production
-
LV Production
-
Other Vectors
-
-
End User Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical and Biotechnology Companies
-
Contract Development & Manufacturing Organizations (CDMOs) & CROs
-
Research and Academic Institutes
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
India
-
Japan
-
South Korea
-
Australia
-
Thailand
-
-
Frequently Asked Questions About This Report
b. The North America, Europe & Asia Pacific gene therapy cell culture media market size was estimated at USD 438.3 million in 2025 and is projected to reach USD 508.6 million in 2026.
b. The North America, Europe & Asia Pacific gene therapy cell culture media market is expected to reach a value of USD 1,533.8 million by 2033, growing at a CAGR of 17.08% from 2026 to 2033.
b. The AAV production segment dominated the market with a share of 51.49% in 2025, driven by the broad use of AAV in in vivo gene therapy applications for rare genetic disorders, ocular disorders, and neuromuscular conditions.
b. Key players of the market include Thermo Fisher Scientific Inc.; Sartorius AG; Merck KGaA; Lonza; Danaher Corporation (Cytiva); FUJIFILM Holdings Corporation; MP Biomedicals; Corning Incorporated; PAN-Biotech; Bio-Techne (R&D Systems, Inc.); InVitria Inc.; Florabio AS; Revvity; Virica
b. The gene therapy cell culture media market in North America, Europe, and the Asia Pacific is driven by rising clinical trial activity, expanding biomanufacturing capacity, and the growing need for GMP-compliant, chemically defined media. Together, these regions are accelerating the adoption of high-performance media to boost vector yields and streamline production.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










