- Home
- »
- Healthcare IT
- »
-
North America Real World Evidence Solutions Market Report, 2033GVR Report cover
![North America Real World Evidence Solutions Market Size, Share & Trends Report]()
North America Real World Evidence Solutions Market (2026 - 2033) Size, Share & Trends Analysis Report By Component (Services, Data Sets), By Application (Drug Development & Approvals), By End Use, By Therapeutic Area (Oncology, Cardiology), By Country, And Segment Forecasts
- Report ID: GVR-4-68040-878-1
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
North America Real World Evidence Solutions Market Summary
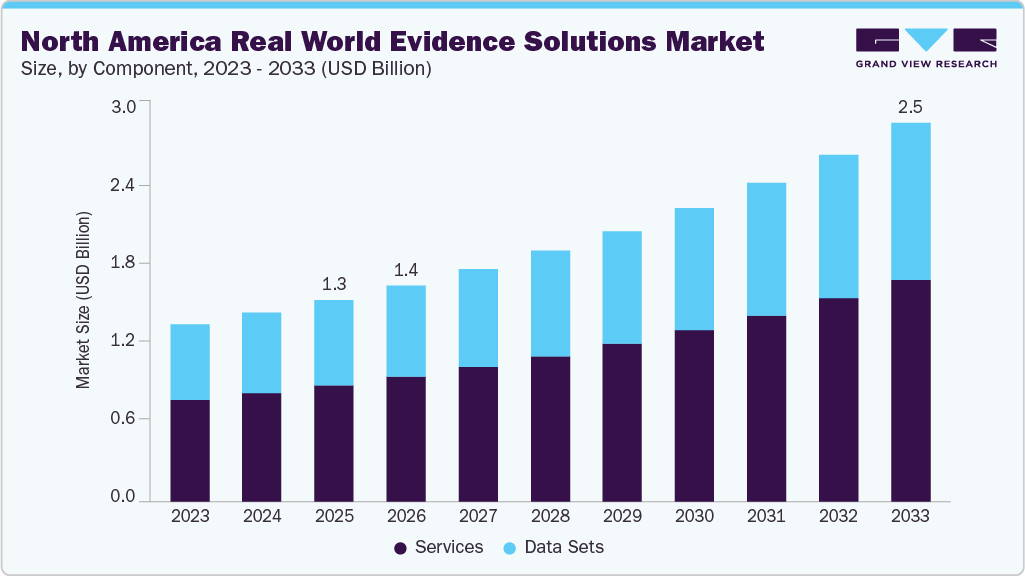
The North America real world evidence solutions market size was estimated at USD 1.34 billion in 2025 and is projected to reach USD 2.52 billion by 2033, growing at a CAGR of 8.29% from 2026 to 2033. Market growth is driven by the increasing use of real-world data in regulatory decision-making, the rising demand for value-based healthcare, and the need among pharmaceutical and biotechnology companies to generate post-market evidence on drug safety and effectiveness.
Key Market Trends & Insights
- The U.S. dominated the North America real world evidence solutions market with a share of 79.7% in 2025.
- Based on component, the services segment held the largest market share of 57.9% in 2025.
- Based on application, drug development & approvals held the dominant market share of 28.6% in 2025.
- Based on end use, the healthcare companies segment held the largest revenue share in 2025.
- Based on therapeutic area, oncology held the dominant market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1.34 Billion
- 2033 Projected Market Size: USD 2.52 Billion
- CAGR (2026-2033): 8.29%
- U.S.: Largest market in 2025
- Canada: Fastest growing market
The North America Real-World Evidence (RWE) solutions industry is growing, driven by strong regulatory support for the adoption of RWE solutions and expanding investments in biomedical and translational research. For instance, according to the National Center for Science and Engineering Statistics, in 2024, R&D expenditures in health sciences reached USD 38.5 billion, up USD 2.8 billion from 2023, while biological and biomedical sciences accounted for USD 20.8 billion, up USD 1.3 billion from 2023. Together, these fields represented 50% of total higher education R&D spending. The growth in academic medical center research and life sciences funding supports a greater generation of real-world data, observational studies, and clinical research outputs, thereby increasing demand for advanced RWE analytics and evidence-generation platforms.
Moreover, the shift from volume-based to value-based care is anticipated to fuel market growth. The rising geriatric population and the increasing prevalence of chronic diseases further drive market expansion. In addition, the region’s strong market position is supported by the presence of key industry players across the U.S. and Canada. Favorable government regulations and the growing number of RWE service providers in the region are expected to further accelerate market growth.
Key Market Trend: Increasing Integration of Real-World Evidence in Regulatory and Drug Development Decisions
The integration of real-world evidence into regulatory decision-making and drug development has emerged as a key trend in the market. The increasing use of real-world data from electronic health records, insurance claims, and patient registries is enabling pharmaceutical, biotechnology, and medical device companies to generate clinical insights beyond traditional clinical trials. This approach supports faster regulatory approvals, improved post-market surveillance, and stronger evidence for reimbursement and market access decisions. Some of the recent developments include:
-
In February 2026, the U.S. Food and Drug Administration, United States, held a town hall to discuss its final guidance on the use of real-world evidence (RWE) for medical device regulatory decision-making. The guidance clarified evaluation standards for real-world data quality and updated recommendations for sponsors submitting RWE to support device regulatory submissions.
-
In December 2025, the U.S. Food and Drug Administration removed a key limitation on the use of RWE in regulatory submissions by announcing that certain real-world evidence could be accepted without requiring identifiable individual patient data in medical device applications. This decision allows broader use of large de-identified healthcare databases, helping accelerate the evaluation of drugs, biologics, and medical devices.
“We’re removing unnecessary barriers that have prevented us from using powerful real-world evidence to get life-changing treatments to patients faster. This common-sense reform will unlock access to vast databases like cancer and cystic fibrosis registries that contain critical insights about how treatments work in the real world.”
-FDA Commissioner Marty Makary, MD, MPH
As a result, pharmaceutical and biotechnology companies are increasingly adopting RWE solutions to optimize clinical development, generate evidence for reimbursement, and support faster regulatory decision-making across North America.
Market Characteristics
The chart below illustrates the relationship between market concentration, industry characteristics, and industry participants. The x-axis represents the level of industry concentration, ranging from low to high. The y-axis represents various industry characteristics, including industry competition, impact of regulations, level of partnerships & collaborations activities, degree of innovation, and regional expansion. For instance, the real world evidence solutions market is slightly fragmented, with many product & service providers entering the market. The degree of innovation, the level of partnerships & collaboration, and the impact of regulations on the industry are high. However, the regional expansion observes moderate growth.
North America leads the Real-World Evidence (RWE) solutions industry, driven by advanced digital health infrastructure, widespread EHR adoption, and growing use of AI-based analytics. Regulatory support for RWE in clinical and post-market decision-making further accelerates innovation in the region.

The North America real world evidence solutions market shows a high level of partnerships and collaborations, driven by alliances among pharmaceutical companies, healthcare providers, and technology firms to integrate real-world data and strengthen analytics capabilities.
North America experiences a strong regulatory impact in the Real-World Evidence (RWE) solutions market, as supportive frameworks and guidance from regulatory authorities encourage the use of real-world data in drug approvals, post-market surveillance, and healthcare decision-making, thereby promoting the adoption of RWE solutions.
The demand in the North America real world evidence solutions industry is driven by increasing adoption across pharmaceutical companies, healthcare providers, and research organizations, along with growing investments in real-world data infrastructure and analytics platforms. For instance, in August 2025, SOPHiA GENETICS expanded its collaboration with AstraZeneca across North America and Europe to leverage AI-driven multimodal data analytics to generate real-world evidence and improve breast cancer treatment outcomes.
Component Insights
The service segment held the largest market share of 57.9% in 2025 and is expected to grow at the fastest CAGR throughout the forecast period, driven by the growing demand for data integration, analytics, consulting, and regulatory support services required to generate and interpret real-world evidence. Pharmaceutical and biotechnology companies increasingly rely on specialized service providers to manage large healthcare datasets, conduct real-world studies, and support regulatory submissions and market access strategies.
The data set segment is expected to grow at a significant CAGR during the forecast period, driven by the rapid expansion of therapy-specific and indication-specific curated datasets, particularly in oncology, rare diseases, and immunology. The increasing need for high-quality real-world data to support clinical research, regulatory submissions, and healthcare decision-making is further accelerating demand.
Application Insights
The drug development & approvals segment led the North America real world evidence solutions market, accounting for the largest revenue share of 28.6% in 2025. Regulators are increasingly accepting RWE in approval processes, particularly for rare diseases, oncology, and accelerated approval pathways, where traditional randomized controlled trials (RCTs) may be limited. By integrating RWE into drug development, companies can enhance regulatory confidence, streamline approvals, and generate evidence for market access, ultimately reducing time-to-market and supporting more informed, patient-centric therapeutic decisions.
The reimbursement/coverage and regulatory decision-making segment of the North America real world evidence solutions industry is expected to grow at the fastest CAGR during the forecast period, driven by the increasing use of real-world evidence to support payer decisions, demonstrate treatment value, and meet evolving regulatory requirements for drug and medical device approvals.
End Use Insights
Healthcare companies held the largest share of the North America real world evidence solutions market in 2025. These organizations leverage RWE to support clinical development, regulatory submissions, market access, safety monitoring, and value-based decision-making across the product lifecycle. For instance, in January 2026, Atropos Health launched its AI-driven Commercial Analytics solution in the U.S., enabling life science companies to rapidly generate actionable real-world evidence for market, brand, and patient insights.
The healthcare payers segment of the North America real world evidence solutions industry is expected to register the fastest CAGR during the forecast period, supported by the growing need to evaluate treatment effectiveness, optimize reimbursement decisions, and manage rising healthcare costs through data-driven insights derived from real-world patient outcomes.
Therapeutic Area Insights
Oncology held the dominant market share in 2025, driven by the high burden of cancer, increased oncology drug development, and the growing use of real-world evidence to assess treatment effectiveness, safety, and long-term patient outcomes. For instance, in January 2026, U.S.-based Verana Health merged with COTA, significantly expanding its real-world data capabilities across oncology, ophthalmology, urology, and neurology. The combined company now serves 17 of the top 20 global biopharma firms, accessing over 95 million patients and 20,000 clinicians, delivering high-fidelity, research-ready datasets and analytics to accelerate clinical trials, regulatory submissions, and evidence generation.
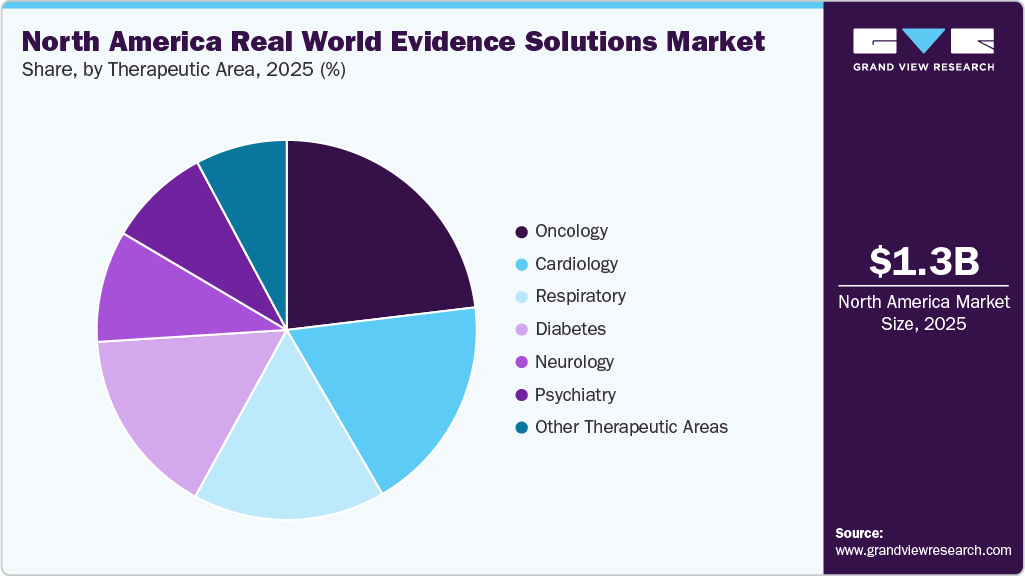
The cardiology segment in the North America Real-World Evidence (RWE) Solutions market is expected to witness significant growth over the forecast period, driven by the rising prevalence of cardiovascular diseases, increasing clinical research in cardiology, and growing use of real-world data to evaluate treatment effectiveness, patient outcomes, and long-term therapy performance.
Country Insights
U.S. Real World Evidence Solutions Market Trends
The real-world evidence solutions market in the U.S. is growing rapidly as healthcare and life sciences organizations adopt advanced digital platforms and AI-driven tools to generate insights from clinical and real-world data. For instance, in April 2025, Scientist.com launched Clinical Labs Navigator to streamline clinical trial service management and improve research efficiency. Market growth is further supported by increasing use of real-world data by healthcare payers and supportive initiatives from the U.S. Food and Drug Administration to expand the use of real-world evidence in regulatory decision-making.
Canada Real World Evidence Solutions Market Trends
The real-world evidence (RWE) solutions market in Canada is driven by rising demand for real-world data in drug development, increasing regulatory acceptance of RWE, and growing R&D investments by biopharmaceutical companies. Strategic collaborations also support market growth. For instance, in June 2022, Dacima Software partnered with EVIDENTIQ to deliver an end-to-end eClinical platform for clinical trials and patient registries.
Key North America Real World Evidence Solutions Company Insights
The North America real-world evidence (RWE) solutions market is characterized by the presence of several established healthcare analytics, technology, and life sciences service providers competing through advanced data platforms, strategic partnerships, and acquisitions. Key companies are expanding their RWE capabilities by integrating artificial intelligence, real-world data sources, and analytics tools to support clinical research, regulatory submissions, and value-based healthcare decision-making, thereby strengthening their market positions.
Key North America Real World Evidence Solutions Companies:
- Oracle
- Merative (formerly IBM Watson Health, acquired by Francisco Partners in June 2022)
- IQVIA
- ICON, plc
- Syneos Health
- Cegedim Health Data is (a division of Cegedim Group)
- Medpace
- Optum Inc. (UnitedHealth Group)
- SAS Institute Inc.
- Aetion, Inc. (acquired by Datavant in May 2025)
- Flatiron Health
- Cytel Inc.
- Trinity
- Thermo Fisher Scientific Inc.
- Parexel International Corporation
- NTT DATA, Inc.(a part of the NTT Group)
- Cognizant
Recent Developments
- In January 2026, Thermo Fisher Scientific announced the launch of the CoreVitas Obesity Registry, a real‑world evidence initiative designed to collect and analyze comprehensive longitudinal data on patients living with obesity to better understand treatment patterns and outcomes. The registry aims to support healthcare decision‑making, clinical research, and improved patient care through high‑quality real‑world insights.
“Insights generated from this robust, real-world evidence base will inform the healthcare community on treatment effectiveness, safety and the patient experience associated with obesity care, which will advance care for millions living with the condition.”
- Leslie Harrold, M.D., vice president and global head of real-world science and strategy for PPD CorEvitas Clinical Registries, Thermo Fisher Scientific
- In January 2026, Oracle announced the Life Sciences AI Data Platform, a generative AI‑enabled solution that unifies diverse data sources, including de‑identified longitudinal real‑world health records, with advanced AI and agentic reasoning to accelerate research, clinical development, post‑market safety monitoring, and commercialization for pharmaceutical, medical device, and life sciences organizations.
“Fragmented, inconsistent data is a major barrier to progress, holding back life sciences organizations from delivering the medical breakthroughs that could transform and even save lives.”
-Executive vice president and general manager, Oracle Health and Life Sciences
- In December 2025, Thermo Fisher Scientific announced the launch of the PPD CorEvitas Alzheimer’s Disease Registry, enrolling the first patient in an international, multi‑country initiative designed to generate harmonized real‑world data on Alzheimer’s therapy safety, effectiveness, and treatment patterns to support regulators, clinicians, and researchers. The longitudinal registry aims to improve understanding of long‑term outcomes and unmet needs in Alzheimer’s care and expand regulatory‑grade evidence generation.
“Together, we are building a regulatory-grade, real-world evidence foundation that will help shape the understanding of current and next generation of therapies.”
-Peter Wahl, Sc.D, vice president and global head of scientific affairs for PPD CorEvitas Clinical Registries, Thermo Fisher Scientific
North America Real World Evidence Solutions Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.44 billion
Revenue forecast in 2033
USD 2.52 billion
Growth rate
CAGR of 8.29% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Market Value in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Component, application, end use, therapeutic area, country
Regional scope
North America
Country scope
U.S.; Canada; Mexico
Key companies profiled
Oracle; Merative (formerly IBM Watson Health, acquired by Francisco Partners in June 2022); IQVIA; ICON, plc; Syneos Health; Cegedim Health Data is (a division of Cegedim Group); Medpace; Optum Inc. (UnitedHealth Group); SAS Institute Inc.; Aetion, Inc. (acquired by Datavant in May 2025); Flatiron Health; Cytel Inc.; Trinity; Thermo Fisher Scientific Inc.; Parexel International Corporation; NTT DATA, Inc.(a part of the NTT Group); Cognizant
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
North America Real World Evidence Solutions Market Report Segmentation
This report forecasts revenue growth at the regional and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the North America real world evidence solutionsmarket report based on service, component, application, end use, therapeutic area, and region:
-
Component Outlook (Revenue, USD Million, 2021 - 2033)
-
Services
-
Data Sets
-
Clinical Settings Data
-
Claims Data
-
Pharmacy Data
-
Patient-Powered Data
-
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Drug Development & Approvals
-
Medical Device Development & Approvals
-
Reimbursement/Coverage and Regulatory Decision Making
-
Post Market Safety & Adverse Events Monitoring
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Healthcare Companies (Pharmaceutical, Biopharmaceutical, Medical Device)
-
Clinical research
-
Commercial (inclusive of marketing, etc.)
-
HEOR
-
Others (market access, etc.)
-
-
Healthcare Payers
-
Healthcare Providers
-
Others
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
Cardiology
-
Neurology
-
Diabetes
-
Psychiatry
-
Respiratory
-
Other Therapeutic Areas (Immunology, Gastroenterology, etc.)
-
-
Country Outlook (Revenue, USD Million, 2021 - 2033)
-
U.S.
-
Canada
-
Mexico
-
Frequently Asked Questions About This Report
b. The North America real world evidence solutions market size was estimated at USD 1.34 billion in 2025 and is expected to reach USD 1.44 billion in 2026.
b. The North America real world evidence solutions market is expected to grow at a compound annual growth rate of 8.29% from 2026 to 2033 to reach USD 2.52 billion by 2033.
b. Based on components, the service segment held the largest market share of 57.9% in 2025 and expected to grow with the fastest rate throughout the forecast period, driven by the growing demand for data integration, analytics, consulting, and regulatory support services required to generate and interpret real-world evidence.
b. Some key players operating in the North America real world evidence solutions market include Oracle; Merative (formerly IBM Watson Health, acquired by Francisco Partners in June 2022); IQVIA; ICON, plc; Syneos Health; Cegedim Health Data is (a division of Cegedim Group); Medpace; Optum Inc. (UnitedHealth Group); SAS Institute Inc.; Aetion, Inc. (acquired by Datavant in May 2025); Flatiron Health; Cytel Inc.; Trinity; Thermo Fisher Scientific Inc.; Parexel International Corporation; NTT DATA, Inc.(a part of the NTT Group); Cognizant
b. Key factors that are driving the market growth include the increasing use of real-world data in regulatory decisions, rising demand for value-based healthcare, and the need among pharmaceutical and biotechnology companies to generate post-market evidence for drug safety and effectiveness.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










