- Home
- »
- Medical Devices
- »
-
Optical Preclinical Imaging Market Size Report, 2020-2027GVR Report cover
![Optical Preclinical Imaging Market Size, Share & Trends Report]()
Optical Preclinical Imaging Market Size, Share & Trends Analysis Report By Product Type (Device, Consumables, Software), By End Use, By Region, And Segment Forecasts, 2020 - 2027
- Report ID: GVR-4-68038-458-1
- Number of Report Pages: 36
- Format: PDF, Horizon Databook
- Historical Range: 2016 - 2018
- Forecast Period: 2020 - 2027
- Industry: Healthcare
Report Overview
The global optical preclinical imaging market size was valued at USD 419.1 million in 2019 and is projected to expand at a CAGR of 3.3% during the forecast period. Increasing focus of market players on research and development, strategic collaborations, novel drug development to cater to unmet medical needs, and non-invasive diagnostic techniques is a major factor fueling the market growth.
Technological advancements favoring animal safety and the use of alternative models that could mimic the functions for preclinical evaluation are driving the market for optical preclinical imaging. For instance, in June 2019, researchers from Hesperos, a biotech firm collaborated with Roche and the University of Central Florida (UCF) for the development of a multi-organ in vitro model that could effectively measure compound efficacy and toxicity, thus transforming drug development safety studies.
Moreover, a surge in the number of preclinical studies aimed at developing various effective therapeutics for various diseases is increasing the adoption of optical preclinical imaging systems. Rising focus of pharmaceutical companies on R&D activities for the discovery and development of novel drugs is expected to raise demand for optical preclinical imaging.
According to the Pharmaceutical Research and Manufacturers of America (PhRMA), in 2018, PhRMA member biopharmaceutical research companies invested around USD 79.6 billion for discovery and development of novel medicines. Furthermore, in June 2019, BioCelerate collaborated with the Food and Drug Administration (FDA) to develop data-driven methods, thereby enabling effective preclinical analysis for the researchers.
Moreover, optical imaging systems offer a rapid, easy use, cost-effective technique to help researchers and drug developers in the non-invasive study of biological and molecular disease processes. For instance, in October 2019, the National Institute of Health (NIH) granted US$ 2.8 million grant to the Sanders-Brown Center on Aging (SBCoA) for preclinical study of new dementia treatment.
Product Type Insights
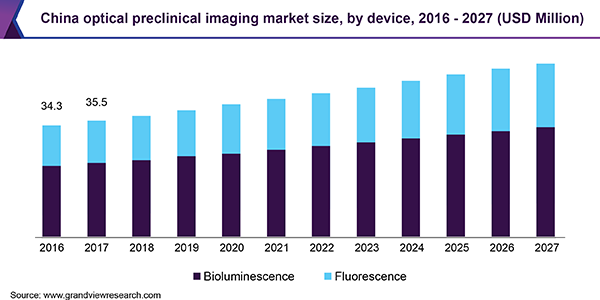
Based on product type, the market for optical preclinical imaging is segmented into devices, consumables, and software. The device segment is further segmented into fluorescence and bioluminescence. The bioluminescence segment is anticipated to witness the highest growth in the optical preclinical imaging market.
High sensitivity, ease of use, and short acquisition time facilitate the usage of bioluminescence imaging in the research and pharma industries. In addition, a detailed analysis of disease mechanisms offered by these optical imaging systems helps the researchers accelerate drug discovery development and in vivo imaging research.
End-use Insights
Based on end use, the market for optical preclinical imaging is segmented into pharma and biotech companies, research institutes, and others. The pharma and biotech companies accounted for a major share in the market for optical preclinical imaging in 2019. The companies adopt effective imaging modalities to gather drug safety information in preclinical trials to ensure safe use in patients during clinical trials. According to a 2016 survey conducted by the Physicians Committee for Responsible Medicine, around 95% of the drug fails in Phase 1 clinical trials as they differ in therapeutic efficacy and safety in animals.
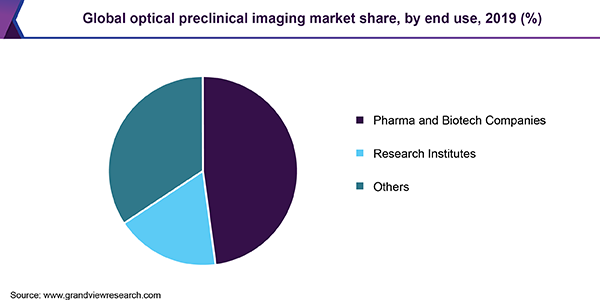
According to the U.S. Food and Drug Administration (FDA), more than 100,000 human deaths are caused annually due to adverse drug reactions. Adverse drug reactions are also the fourth major cause of death in the U.S. Therefore, ensuring an effective approach in drug development studies to facilitate the development of novel drug candidates and limit the occurrence of drug failure in clinical trials compels the biotech and pharma companies to adopt the modern optical preclinical imaging technologies.
Regional Insights
North America dominated the market for optical preclinical imaging in 2019 owing to factors such as growing use of preclinical optical imaging for cancer research and overall growth in cancer drug development pipeline in U.S. Moreover, product launches by the market players in the region focused on developing non-invasive imaging systems are propelling the market growth. For instance, in September 2017, PerkinElmer, Inc. launched IVIS Lumina S5 and Lumina X5 systems for high throughput in vivo imaging. These optical imaging systems will further help researchers to understand disease progression and develop effective therapies targeting infectious diseases and cancer.
Asia Pacific is expected to witness lucrative growth over the forecast period. Rising government funding to facilitate research and development and increasing preclinical trials by research institutes to cater to unmet medical needs for rare diseases are some of the key factors expected to drive the market for optical preclinical imaging in this region.
Optical Preclinical Imaging Market Share Insights
Leading players operating in the market for optical preclinical imaging include Bruker Corporation; Perkin Elmer, Inc.; MR Solutions; BioTek Instruments, Inc.; Milabs B.V.; Fujifilm Holdings Corporation; Magnetic Insight, Inc.; MBF Bioscience; and Mediso Ltd. Product launches, distribution agreements, and product innovation are the major strategies adopted by the players to retain their share in the optical preclinical imaging industry. For instance, in 2014, PerkinElmer Inc. launched an optical molecular imaging system – Solaris. This optical imaging system is designed for use in animal studies.
Report Scope
Attribute
Details
Base year for estimation
2019
Actual estimates/Historical data
2016 - 2018
Forecast period
2020 - 2027
Market representation
Revenue in USD Million and CAGR from 2020 to 2027
Regional scope
North America, Europe, Asia Pacific, Latin America, and Middle East & Africa
Country scope
U.S., Canada, U.K., Germany, France, Italy, Spain, Russia, Japan, China, Malaysia, India, South Korea, Thailand, Brazil, Mexico, Argentina, Columbia, South Africa, Saudi Arabia, UAE, and Qatar
Report coverage
Revenue forecast, company share, competitive landscape, growth factors, and trends
15% free customization scope (equivalent to 5 analysts working days)
If you need specific information, which is not currently within the scope of the report, we will provide it to you as a part of customization
Segments Covered in the ReportThis report forecasts revenue growth at the global, regional, and country levels as well as provides an analysis of the latest industry trends and opportunities in each of the sub-segments from 2016 to 2027. For the purpose of this study, Grand View Research has segmented the global optical preclinical imaging market report on the basis of product type, end use, and region:
-
Product Type Outlook (Revenue, USD Million, 2016 - 2027)
-
Device
-
Fluorescence
-
Bioluminescence
-
-
Consumables
-
Fluorescence
-
Green fluorescent proteins
-
Red fluorescent proteins
-
Infrared dyes
-
Others
-
-
Bioluminescence
-
Luciferins
-
Proluciferins
-
Coelenterazine
-
Others
-
-
-
Software
-
-
End-use Outlook (Revenue, USD Million, 2016 - 2027)
-
Pharma and Biotech Companies
-
Research Institutes
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2016 - 2027)
-
North America
-
The U.S.
-
Canada
-
-
Europe
-
The U.K.
-
Germany
-
France
-
Italy
-
Spain
-
Russia
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Thailand
-
Malaysia
-
South Korea
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
Columbia
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Qatar
-
-
Frequently Asked Questions About This Report
b. The global optical preclinical imaging market size was estimated at USD 419.1 million in 2019 and is expected to reach USD 434.0 million in 2020.
b. The global optical preclinical imaging market is expected to grow at a compound annual growth rate of 3.3% from 2020 to 2027 to reach USD 543.0 million by 2027.
b. North America dominated the optical preclinical imaging market with a share of 37.7% in 2019. This is attributable to the growing use of preclinical optical imaging for cancer research and overall growth in cancer drug development pipeline in the U.S.
b. Some key players operating in the optical preclinical imaging market include Bruker Corporation; Perkin Elmer, Inc.; MR Solutions; BioTek Instruments, Inc.; Milabs B.V.; Fujifilm Holdings Corporation; Magnetic Insight, Inc.; MBF Bioscience; and Mediso Ltd.
b. Key factors that are driving the market growth include increasing focus of market players on research and development, strategic collaborations, novel drug development to cater to unmet medical needs, and non-invasive diagnostic techniques.
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





