- Home
- »
- Medical Devices
- »
-
Orthopedic Digit Implants Market Size, Industry Report, 2033GVR Report cover
![Orthopedic Digit Implants Market Size, Share & Trends Report]()
Orthopedic Digit Implants Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Metatarsal Joint Implants, Metacarpal Joint Implants, Hemi Phalangeal Implants), By Material (Titanium, Nitinol), By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-265-3
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Orthopedic Digit Implants Market Summary
The global orthopedic digit implants market size was estimated at USD 283.44 million in 2024 and is projected to reach USD 488.89 million by 2033, growing at a CAGR of 6.17% from 2025 to 2033. The rising prevalence of orthopedic conditions such as rheumatoid arthritis and osteoarthritis is expected to drive market growth.
Key Market Trends & Insights
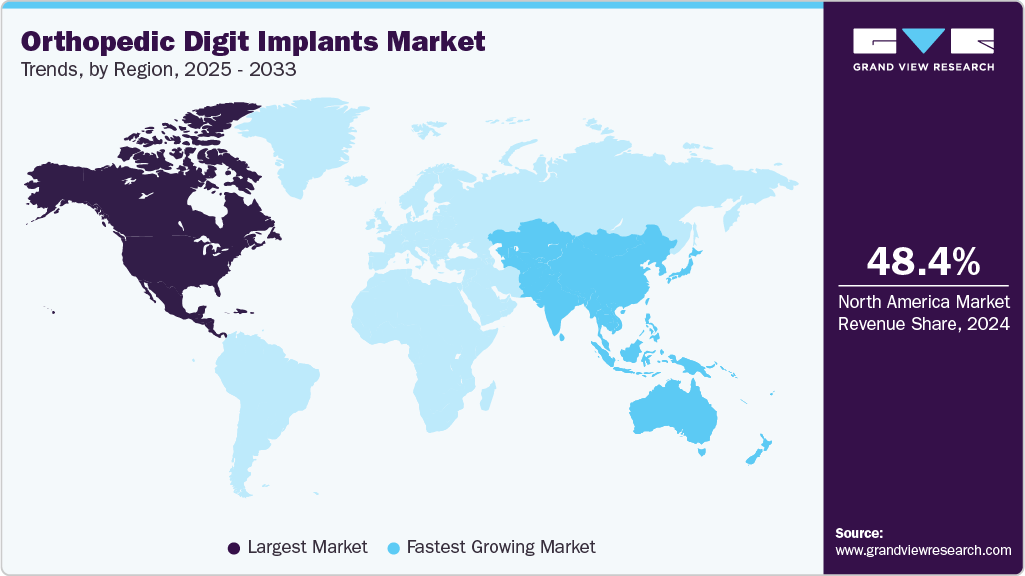
- North America orthopedic digit implants market held the largest share of 48.37% of the global market in 2024.
- The orthopedic digit implants industry in the U.S. is expected to grow significantly over the forecast period.
- By product type, the metatarsal joint implants segment held the largest market share of 47.73% in 2024.
- Based on material, the titanium segment held the largest market share in 2024.
- Based on end use, the outpatient facilities segment held the largest market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 283.44 Million
- 2033 Projected Market Size: USD 488.89 Million
- CAGR (2025-2033): 6.17%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
This trend, coupled with an aging population and increasing demand for joint preservation solutions, is fueling the adoption of advanced orthopedic implants. In February 2025, SingleCare reported that nearly 1 in 4 American adults has arthritis, with osteoarthritis being the most common form. The article highlights that more than 33 million U.S. adults are affected by osteoarthritis, and the economic burden exceeds USD 300 billion annually. Arthritis is also a leading cause of work limitations in the U.S., with 25.7 million adults experiencing physical restrictions. Increasing technological advancements in orthopedic implants including orthopedic digit implants is anticipated to accelerate market growth. Advanced biomaterials with better biocompatibility and durability have improved the quality of implants, leading to an increase in their demand. These biomaterials consist of ceramics and alloys that imitate the properties of natural bone and joints, which has further contributed to market growth. The use of computer-aided design and biomechanics studies has also played a significant role in the development of precise implant designs, further boosting the market growth. In March 2024, OrthoVentions LLC made an investment in MotioOV, which is an early-stage company that was created in partnership with the Hospital for Special Surgery (HSS). MotioOV's focus is to develop a unique 3D printed implant as well as custom instruments that can help address issues related to stiffness and pain caused by arthritis of the first metatarsophalangeal (MTP) joint.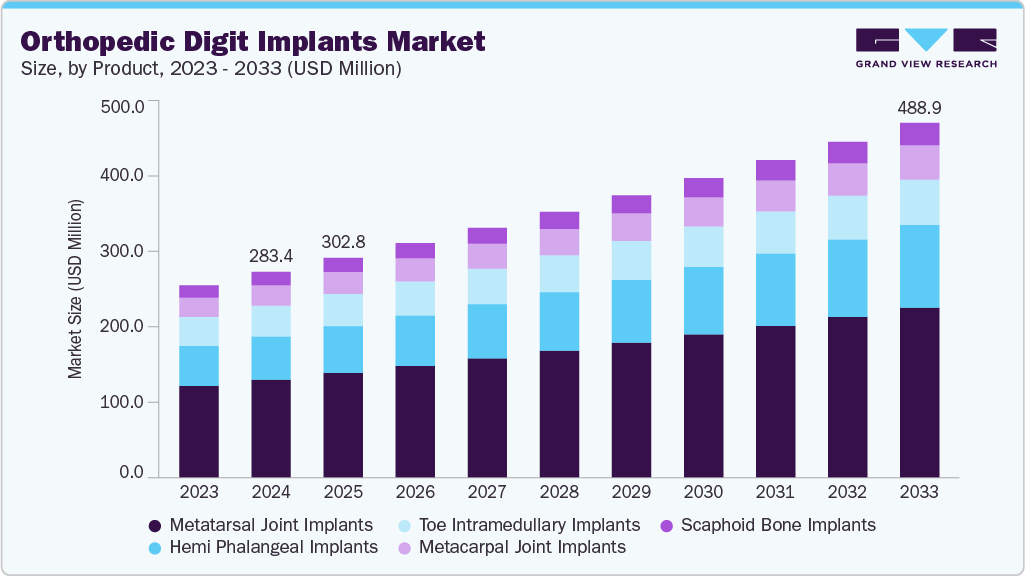
Increasing demand for minimally invasive procedures drives the demand for orthopedic digit implants. The increasing adoption of organic and inorganic strategies by the key market players is expected to spur market growth. For instance, in January 2024, Extremity Medical LLC, a medical engineering firm, has announced a strategic relationship with Henry Schein, Inc., a global healthcare solutions company. This partnership will help Extremity Medical expand its innovative orthopedic extremity implant systems. In addition, the company completed growth debt financing from June through November, which will help fuel its growth. Such initiatives undertaken by market players will foster market growth.
The orthopedic industry is expected to experience rapid growth due to the increasing number of new players entering the market. In April 2024, Henry Schein, Inc., the largest provider of healthcare solutions for dental and medical practitioners, announced the completion of its acquisition of TriMed, Inc. TriMed is a global developer of orthopedic solutions for the treatment of upper extremities like hands and wrists, and lower extremities like feet and ankles. Henry Schein had announced its intention to acquire a majority interest in TriMed on December 20, 2023. With this acquisition, Henry Schein entered the market for orthopedic extremities.
Increasing government and non-government initiatives escalate the market growth. For instance, in June 2022, Loci Orthopaedics, a Medtech start-up, received USD 8 million in funding and financial support from the European Commission's European Innovation Council (EIC) Accelerator Programme. The grant and equity investment will be utilized to conduct a clinical trial of the company's implant treatment for thumb arthritis, before proceeding to full-scale commercialization.
Market Concentration & Characteristics
The orthopedic digit implants industry has witnessed varying degrees of innovation over the years, driven by advancements in materials science, biomechanics, and surgical techniques. Innovations such as custom-made implants and minimally invasive surgical approaches have contributed to better outcomes and faster recovery times for patients. In June 2025 , OSSIO Inc. launched the OSSIOfiber 2.5mm Suture Anchors to expand its metal-free, bio-integrative implant line. The device offers 55% greater pull-out strength and is designed for soft tissue fixation across multiple joints. It aims to improve procedures like Brostrom repair while maintaining a familiar surgical workflow.
The orthopedic digit implants industry is characterized by a high level of merger and acquisition (M&A) activity by the leading players. These mergers and acquisitions facilitate access to complementary technologies, expertise, and distribution channels, enabling companies to accelerate product development, improve operational efficiency, and capture a larger share of the market. In March 2024, Stryker acquired SERF SAS, a joint replacement company based in France, from Menix. This acquisition enhanced the company’s product portfolio.
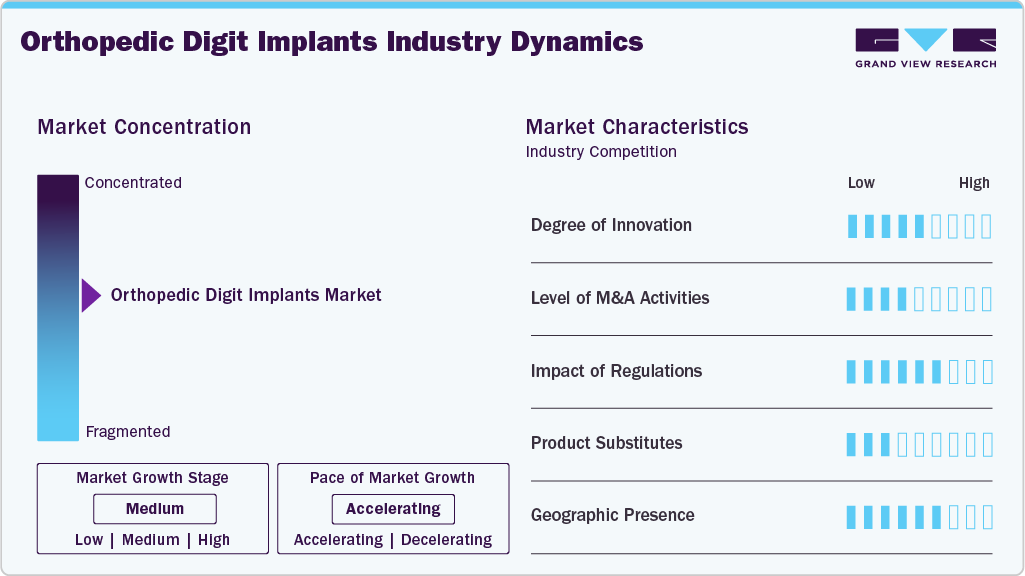
Regulations play a crucial role in shaping the orthopedic digit implants industry, ensuring the safety, efficacy, and quality of implants available to patients. Regulatory requirements govern various aspects of implant development, manufacturing, and distribution, imposing standards for materials, design, sterilization, and labeling. While these regulations are essential for safeguarding patient health and promoting innovation, they can also pose challenges for manufacturers, increasing time and cost barriers to market entry.
Product substitutes are alternative methods or technologies that healthcare facilities can use instead of performing orthopedic digit implants surgery. One notable substitute in the industry is minimally invasive procedures, along with regenerative medicine. These substitutes provide patients and physicians with options that may moderate certain risks associated with traditional implants, offer tailored solutions for complex cases, and potentially speed up the healing process. Another emerging substitute is regenerative medicine techniques, such as tissue engineering and stem cell therapy, which hold the promise of regrowing or repairing damaged digits without the need for implants.
Several industry players are expanding their business by entering new geographical regions to strengthen their market position and expand their product portfolio. Rising product launches create more opportunities for market players to enter new regions. In February 2023, Tyber Medical LLC acquired ADSM-Synchro Medical, a French company that specializes in developing and distributing implants for surgical forefoot pathologies. The acquisition was a part of Tyber Medical LLC's strategy to strengthen its global presence.
Product Insights
By product, the metatarsal joint implants segment dominated the market in 2024 and accounted for the largest revenue share of 47.73%. Metatarsal joints are more prone to fractures, thereby fostering segmental growth. Increasing demand for advanced reconstructive solutions and the growing preference for patient-specific implants are further accelerating innovation in this space. Technological advancements in 3D printing have enabled the creation of anatomically accurate, durable implants that support bone ingrowth and long-term functionality. This has opened new possibilities for treating joint degeneration and osteoarthritis more effectively. For instance, in February 2024 , Anatomic Implants unveiled the first 3D printed metatarsophalangeal (MTP) toe joint replacement at AAOS 2024. Developed in partnership with AddUp Inc., the implant mimics natural anatomy using titanium lattice structures. A 510(k) submission is underway, with FDA clearance expected in Q3 2024.
The hemi phalangeal implants segment in the market is anticipated to witness the fastest CAGR over the forecast period. Increasing incidence of fractures among the growing population is expected to boost market growth. Advances in implant design, including hemi phalangeal implants, are driving improved surgical outcomes and faster patient recovery. In July 2025 , a study published in the Journal of Hand Surgery Global Online highlighted the consistent use of hemi phalangeal implants in phalangeal fracture surgeries. These implants were favored for preserving joint motion in complex cases. Their usage remained stable across socioeconomic groups, underscoring clinical preference.
Material Insights
By material, the titanium segment dominated the market in 2024 and accounted for the largest revenue share of 59.41% and is anticipated to grow at the fastest CAGR over the forecast period. Titanium orthopedic digit implants are expected to experience significant growth due to their superior biocompatibility and strength-to-weight ratio. This makes them perfect for implants requiring durability and compatibility with the human body. For instance , according to an article published in ScienceDirect in 2023, Ti and its alloys are the preferred materials for orthopedic implants. This is due to their lower elastic moduli, which range from 48 to 112 GPa, in comparison to 316L stainless steel, which has a moduli of 210 GPa, and cobalt-chromium alloys, which have a moduli of 240 GPa. In addition, advancements in titanium manufacturing techniques have enabled the production of complex geometries, allowing for a better anatomical fit and long-term performance.
The stainless steel segment accounted for the second-largest share of the market by material in 2024. Its continued use is driven by key advantages, including high tensile strength, corrosion resistance, and cost efficiency. Stainless steel is particularly well-suited for digit implants requiring reliable structural support and durability under load. While it does not possess the shape-memory properties of advanced alloys like nitinol, its proven clinical performance, manufacturing versatility, and regulatory familiarity make it a widely adopted material in orthopedic procedures. In August 2024, a critical review published in The International Journal of Advanced Manufacturing Technology explored the role of stainless steel and other metals in 3D-printed orthopedic implants. While stainless steel offers high strength, the study flagged its mechanical mismatch with bone, leading to stress shielding and long-term implant failure.
End Use Insights
The outpatient facilities segment dominated the market in 2024 with a revenue share of 63.11% and is anticipated to grow at the fastest CAGR over the forecast period. Outpatient facilities provide a convenient and easily accessible option for patients who require minor orthopedic procedures such as digit implants, without the need for prolonged hospital stays. As the trend toward ambulatory care continues to grow, outpatient facilities are becoming increasingly popular for patients seeking quicker recovery times and reduced healthcare costs associated with inpatient hospital stays. For instance, in October 2023, a report by MJH Life Sciences highlighted a sharp rise in demand for outpatient orthopedic procedures. Outpatient knee replacements surged by 293% since 2019, reaching over five times the inpatient volume, with monthly procedures rising from 373,266 in January 2022 to 484,857 in May 2023.
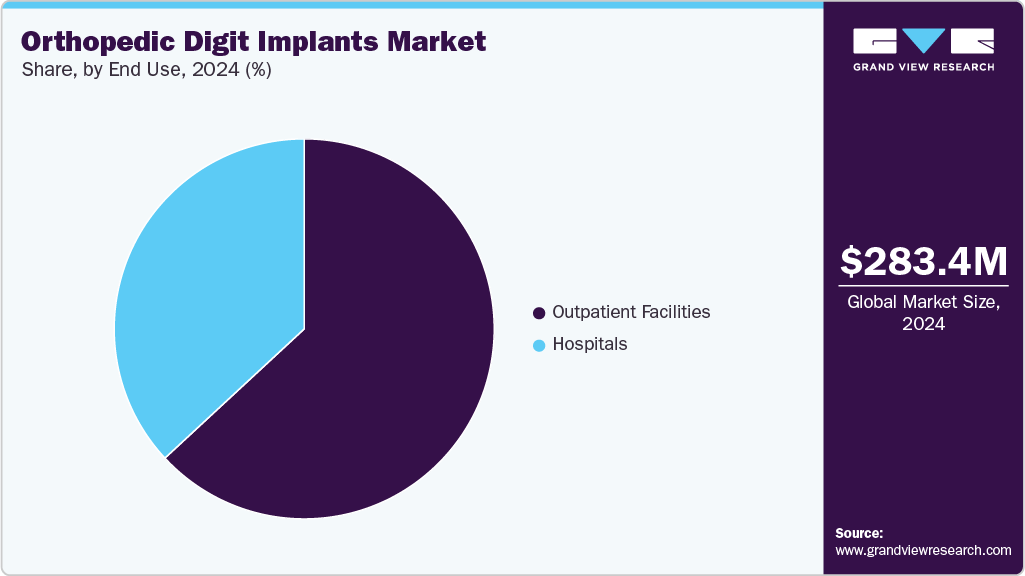
The hospitals segment in the market is anticipated to register a significant CAGR over the forecast period. Hospitals are primary centers for orthopedic surgeries, including digit implants, where patients receive comprehensive care from diagnosis to post-operative rehabilitation. Advancements in minimally invasive techniques for digit implant surgeries have made orthopedic procedures more feasible, further driving demand for these facilities. In June 2025 , a review published in MedComm emphasized the growing role of AI in orthopedic surgery, from preoperative planning to post-op rehab. It highlighted integration with robotics and 3D modeling, enhancing surgical precision and reducing complications. Hospitals are increasingly adopting AI-driven digital implants and surgical planning tools, signaling a shift toward data-informed, personalized orthopedic care.
Regional Insights
North America orthopedic digit implants industry dominated globally in 2024 and accounted for the largest revenue share of 48.37% owing to the rise in musculoskeletal disorders, such as osteoarthritis and rheumatoid arthritis. The presence of well-established healthcare infrastructure and reimbursement policies further stimulate market growth. In March 2025 , a study published in Medicine analyzed US arthritis prevalence from 2019 to 2022, revealing a slight overall increase from 21.4% to 21.6%. Females showed a higher and rising prevalence compared to males, with rates peaking in adults aged 75 and older.
U.S. Orthopedic Digit Implants Market Trends
The orthopedic digit implants industry in the U.S. held the largest share of 92.2% in 2024. Growing government initiatives to accelerate the research for arthritis are boosting market growth. For instance, in March 2024, Congress approved a legislative funding package for federal fiscal year 2024 expenditure. President Biden signed the package into law, which includes a provision to allocate USD 685 million dollars for the National Institute of Arthritis, Musculoskeletal and Skin Diseases (NIAMS). This funding is intended to support research aimed at preventing, treating, and understanding the causes of arthritis and other related conditions that affect many patients.
Canada orthopedic digit implants industry is anticipated to register the fastest growth rate during the forecast period. The country's aging population and increasing prevalence of musculoskeletal disorders contribute to rising demand for orthopedic digit implants. For instance, according to a Springer Nature article, the proportion of seniors (aged 65+) in Canada was projected to increase from 16.3% in 2016 to 25.4% by 2060.
Europe Orthopedic Digit Implants Market Trends
Europe orthopedic digit implants industry is anticipated to register the fastest CAGR during the forecast period. The region's strong focus on research and development fosters continuous innovation in implant technologies, enhancing product efficacy and patient outcomes. Favorable healthcare policies and robust regulatory frameworks ensure the safety and quality of orthopedic digit implants, thereby increasing their demands among the population.
Germany orthopedic digit implants industry is anticipated to register a considerable growth rate during the forecast period. The rising incidence of road accidents in the country contributes to increased demand for orthopedic digit implants. According to the Statistisches Bundesamt (Destatis) report, in February 2023, around 21,600 people were injured in road accidents in Germany.
The UK orthopedic digit implants industry is anticipated to register a considerable CAGR during the forecast period. The UK's National Health Service (NHS) provides universal healthcare coverage, ensuring access to orthopedic procedures for all residents, which stimulates market growth. In addition, increasing aging population will escalate market growth. According to the Office for National Statistics, the number of people aged 75 and over is expected to double from 5 million to nearly 10 million by 2039 in UK.
Italy orthopedic digit implants industry is anticipated to register the fastest CAGR during the forecast period. Increasing prevalence of osteoarthritis among geriatric population is fostering market growth. For instance , according to a survey published in NIH in 2022, 32% of people aged 65 and over, and 48% of those aged over 85, have serious chronic diseases and multimorbidity. The most common illnesses are osteoarthritis (48%) and hypertension (47%), followed by heart disease (19%) and diabetes (17%). The percentage of people with at least one serious chronic disease is 43% for those over 65 years old, and 48% for those between 75 and 84 years old.
Asia Pacific Orthopedic Digit Implants Market Trends
The Asia Pacific orthopedic digit implants industry is experiencing significant growth owing to the rising prevalence of orthopedic conditions such as osteoarthritis and rheumatoid arthritis due to an aging population and lifestyle changes. Advancements in healthcare infrastructure and increasing healthcare expenditure in countries such as China, India, and Japan are fostering the adoption of orthopedic digit implants. In February 2025 , an article published in Medical Sciences discussed the prevalence and risk factors of rheumatoid arthritis (RA) among Korean adults, focusing on age and sex differences. The study found that RA affected 1.1% of the population, with women showing nearly three times higher prevalence than men, especially in older age groups.
China orthopedic digit implants industry is anticipated to register the fastest CAGR during the forecast period. Rapidly expanding healthcare infrastructure and a growing elderly population, presents a significant growth driver for the orthopedic digit implants industry. In October 2024 , an official report by China’s Ministry of Civil Affairs and the China National Committee on Ageing revealed that 21.1% of China’s population was aged 60 and above in 2023, underscoring the nation’s growing aging population.
Japan orthopedic digit implants industry is anticipated to register the fastest CAGR during the forecast period. With an aging population, there is a growing need for orthopedic treatments, including digit implants, to address age-related conditions, thereby escalating market growth. In January 2025, an article published in Modern Rheumatology reported that approximately 822,000 individuals in Japan are affected by rheumatoid arthritis (RA), with the highest prevalence among those aged 65 to 85 years.
Latin America Orthopedic Digit Implants Market Trends
Latin America orthopedic digit implants industry is witnessing a steady growth due to the increasing awareness regarding the availability and benefits of orthopedic implants among both patients and healthcare professionals. The region's growing geriatric population and a rise in chronic diseases such as diabetes, which can lead to complications requiring orthopedic interventions, are further driving market growth. In June 2025, a blog article published on Number Analytics reported that the proportion of individuals aged 60 and over in Latin America and the Caribbean is projected to rise from 12.4% in 2015 to 25.3% by 2050, according to United Nations estimates.
Brazil orthopedic digit implants industry is anticipated to register the fastest CAGR during the forecast period. Increasing emphasis on sports and physical activity in the country is driving market growth. According to a study published in NIH titled "Injuries and complaints in the Brazilian national men's volleyball team: a case study" in 2023, out of 41 athletes who played for the team during the analyzed period, 12 athletes suffered from 28 injuries, and 38 athletes reported a total of 402 complaints. Such increasing cases of injuries escalate market growth.
Middle East and Africa Orthopedic Digit Implants Market Trends
The Middle East and Africa region are experiencing notable growth in the orthopedic digit implants industry. Increasing incidence of orthopedic conditions such as osteoarthritis and osteoporosis due to factors such as sedentary lifestyles, obesity, and an aging population are accelerating market growth. In March 2023 , an article in Open Access Rheumatology assessed osteoarthritis burden in Africa and the Middle East. While most studies focused on knee and hip OA, the 2019 ACR Guidelines cited in the review recommended hand orthoses for first carpometacarpal (CMC) joint OA.
South Africa orthopedic digit implants industry is driven by the improving healthcare system, along with increasing investments in medical technology that is enabling better access to orthopedic treatments, thereby supplementing market growth. In June 2025 , an article in Osteoarthritis and Cartilage Open reported that hand osteoarthritis prevalence in South Africa was 2,999.6 per 100,000, higher than the global average. It highlighted that best evidence rehabilitation practices, such as orthoses for thumb base (CMC) OA, strengthening exercises, and ergonomic training, are underutilized in Africa.
Key Orthopedic Digit Implants Company Insights
Key participants in the orthopedic digit implants industry are focusing on devising innovative business growth strategies in the form of product portfolio expansions, partnerships & collaborations, mergers & acquisitions, and business footprint expansions.
Key Orthopedic Digit Implants Companies:
The following are the leading companies in the orthopedic digit implants market. These companies collectively hold the largest market share and dictate industry trends.
- Extremity Medical LLC
- Stryker
- Zimmer
- Smith & Nephew
- Depuy
- Arthrex Inc
- Acumed LLC
- Anika Therapeutics, Inc.
- VILEX, LLC
- Paragon 28
Recent Developments
-
In September 2024 , Tyber Medical’s ToeGrip Hammer Toe implant family earned MDR certification, becoming one of the first hammer toe implants to hold both FDA 510(k) clearance and MDR approval. The lineup-comprising the Classic, Evo, and Retrograde versions (the latter offering added K-Wire fixation)-has been used in over 55,000 patients. These implants, made from biocompatible PEEK, are designed for toe joint arthrodesis.
-
In August 2024, Anatomic Implants partnered with AddUp to develop the 3D-printed Anatomic Great Toe Joint (1st MTP joint), aiming for FDA 510(k) clearance by end-2024. Manufactured using AddUp’s FormUp 350 in Ti64 per ASTM F3001 standards, the implant features lattice structures to enable osseointegration.
-
In January 2024, Extremity Medical announced a strategic partnership with Henry Schein to expand its orthopedic extremity implant systems. The collaboration aims to improve access and distribution of devices for fusion, fixation, and motion preservation in the digits and extremities. The company also secured growth financings to support this expansion.
Orthopedic Digit Implants Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 302.78 million
Revenue forecast in 2033
USD 488.89 million
Growth rate
CAGR of 6.17% from 2025 to 2033
Actual data
2021 - 2024
Forecast data
2025 - 2033
Quantitative units
Revenue in USD million and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, material, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; Spain; Italy; France; Norway; Denmark; Sweden; Japan; China; India; Australia; Thailand; South Korea; Brazil; Mexico; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Stryker; Zimmer Biomet; Smith+Nephew; DePuy Synthes; Arthrex Inc.; Acumed LLC; Anika Therapeutics, Inc.; VILEX, LLC; Paragon 28; Extremity Medical LLC
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Orthopedic Digit Implants Market Report Segmentation
This report forecasts revenue and volume growth at global, regional, and country level and provides an analysis on industry trends in each of the sub segments from 2021 to 2033. For this study, Grand View Research, Inc. has segmented the global orthopedic digit implants market report based on product, material, end use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Metatarsal joint implants
-
Metacarpal joint implants
-
Hemi phalangeal implants
-
Scaphoid bone implants
-
Toe intramedullary implants
-
-
Material Outlook (Revenue, USD Million, 2021 - 2033)
-
Titanium
-
Nitinol
-
Silicon pyrocarbon
-
Stainless steel
-
Other materials
-
-
End Use Outlook (Revenue USD Million, 2021 - 2033)
-
Hospitals
-
Outpatient Facilities
-
-
Regional Outlook Revenue USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
Spain
-
Italy
-
France
-
Denmark
-
Norway
-
Sweden
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
MEA
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global orthopedic digit implants market size was estimated at USD 283.44 million in 2024 and is expected to reach USD 302.78 million in 2025.
b. The global orthopedic digit implants market is expected to grow at a compound annual growth rate of 6.17% from 2025 to 2033 to reach USD 488.89 million by 2033.
b. North America dominated the orthopedic digit implants market with the highest share of 48.37% in 2024. This is attributable to growing preference of minimally invasive surgeries and increased number of patient pool suffering from orthopedic diseases.
b. Some key players operating in the orthopedic digit implants market include Stryker, Zimmer Biomet, Smith+Nephew, DePuy Synthes, Arthrex Inc., Acumed LLC, Anika Therapeutics, Inc., VILEX, LLC, Paragon 28, and Extremity Medical LLC.
b. The rising prevalence of orthopedic conditions such as rheumatoid arthritis and osteoarthritis is expected to drive market growth. This trend, coupled with an aging population and increasing demand for joint preservation solutions, is fueling the adoption of advanced orthopedic implants.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










