- Home
- »
- Pharmaceuticals
- »
-
Osteoarthritis Injectable Market Size, Industry Report, 2033GVR Report cover
![Osteoarthritis Injectable Market Size, Share & Trends Report]()
Osteoarthritis Injectable Market (2026 - 2033) Size, Share & Trends Analysis Report By Injection Type (Hyaluronic Acid Injections, Corticosteroids Injections), By Anatomy (Knee Osteoarthritis, Hip Osteoarthritis), By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-291-5
- Number of Report Pages: 137
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Osteoarthritis Injectable Market Summary
The global osteoarthritis injectable market size was valued at USD 8.18 billion in 2025 and is projected to reach USD 14.16 billion by 2033, growing at a CAGR of 7.17% from 2026 to 2033. This growth is primarily driven by the increasing prevalence of osteoarthritis (OA), particularly among the aging population, as well as rising awareness of minimally invasive treatment options.
Key Market Trends & Insights
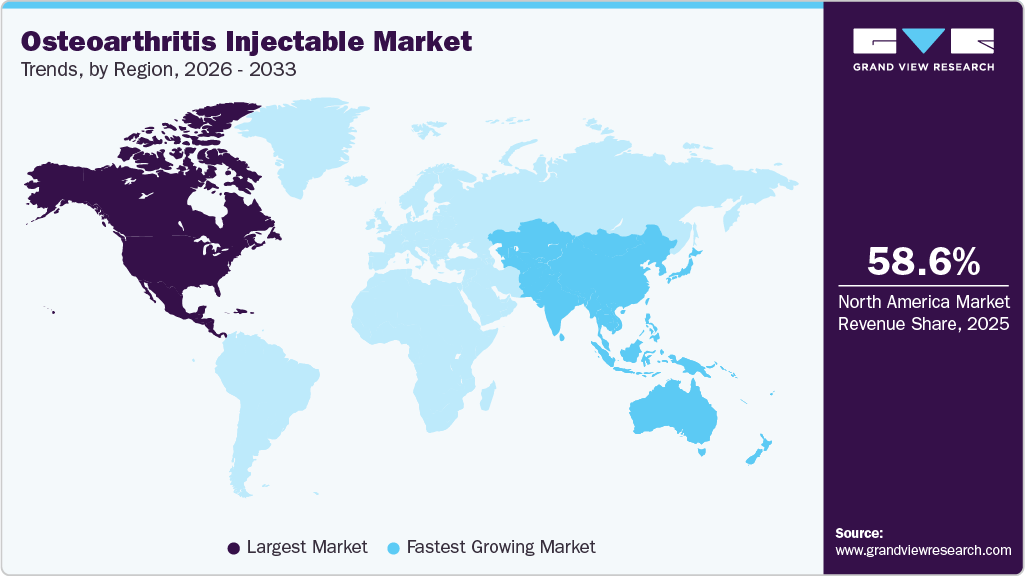
- North America osteoarthritis injectable market held the largest share of 58.58% of the global market in 2025.
- The industry in the U.S. is expected to grow significantly over the forecast period.
- By injection type, the hyaluronic acid injections segment held the highest market share of 39.23% in 2025.
- By anatomy, the knee osteoarthritis segment held the highest market share in 2025.
- By end use, the hospital pharmacies segment held the highest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 8.18 Billion
- 2033 Projected Market Size: USD 14.16 Billion
- CAGR (2026-2033): 7.17%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Additionally, advancements in biologic therapies and next-generation injectable, along with supportive reimbursement policies in key regions, are expected to further propel market expansion over the forecast period. R&D activities in injectable osteoarthritis therapies are accelerating due to challenges in delivering drugs into dense joint tissues. Companies are focusing on intra-articular biologics and disease-modifying agents. Candidates like lorecivivint from Biosplice Therapeutics and TPX-100 from OrthoTrophix highlight ongoing innovation. For instance, in January 2026, Biosplice Therapeutics submitted a New Drug Application (NDA) to the U.S. FDA for lorecivivint, an intra-articular injection for knee osteoarthritis, which showed promising Phase III results for pain reduction and functional improvement.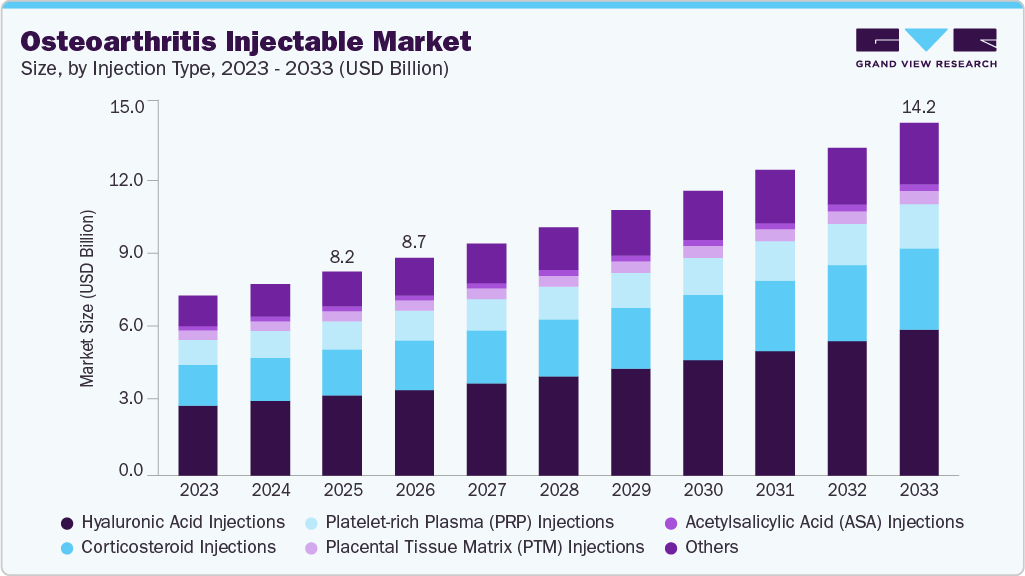
Similarly, in March 2026, OrthoTrophix advanced its Phase 2b clinical trial of TPX-100 by completing patient enrollment, reinforcing its position in developing disease-modifying intra-articular therapies targeting cartilage regeneration in osteoarthritis. The growing emphasis on personalized medicine is reshaping the OA injectable market, with a greater focus on patient-specific biologics and regenerative therapies. Research from Columbia University on cartilage-regenerating stem cells highlights the potential for precision-based injectable treatments targeting early-stage disease progression.
Market Concentration & Characteristics
The market is witnessing strong innovation, driven by a shift toward disease-modifying and regenerative therapies. Advanced intra-articular delivery systems, including sustained-release formulations, nanoparticles, and hydrogel-based carriers, are enhancing drug retention and targeted action within joints. Biologic injectable such as platelet-rich plasma (PRP), stem cell therapies, and gene-based approaches are gaining traction, moving beyond symptomatic relief toward cartilage regeneration. The pipeline remains robust with numerous novel therapies under development, while innovations in minimally invasive, long-acting injectable and precision medicine are improving patient outcomes and addressing unmet clinical needs.
The level of M&A activity in the osteoarthritis (OA) injectable market remains moderate but strategically significant, with key players such as Anika Therapeutics, Bioventus, Ferring Pharmaceuticals, and Sanofi actively engaging in collaborations, licensing agreements, and selective acquisitions to strengthen their orthobiologics and viscosupplementation portfolios. Rather than large-scale mergers, companies are increasingly focusing on strategic partnerships and pipeline expansion to enhance innovation, expand geographic presence, and accelerate the development of next-generation injectable therapies.
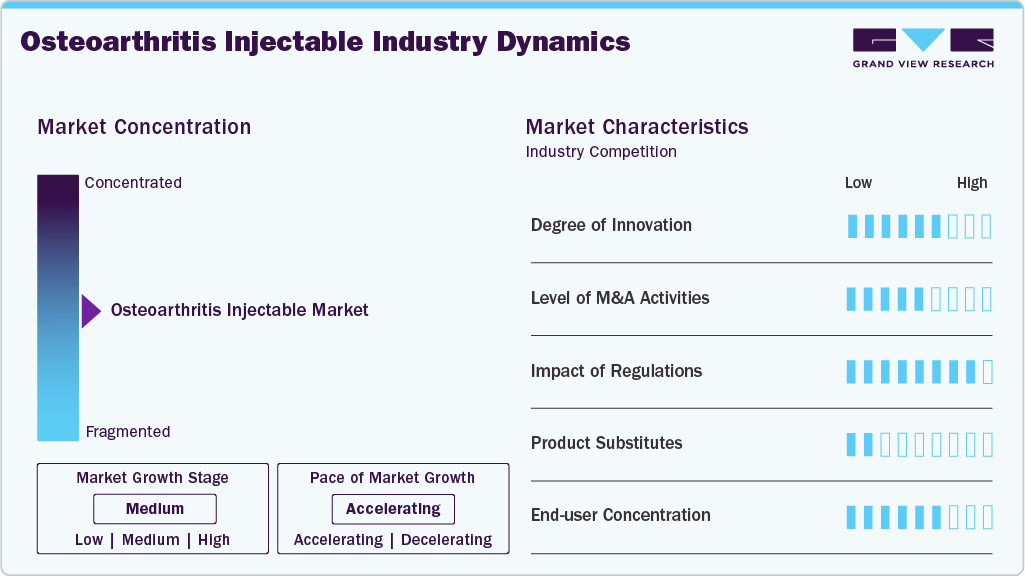
The regulatory landscape for osteoarthritis (OA) injectable remains complex, particularly for biologics and regenerative therapies, due to stringent safety and efficacy requirements and the lack of approved disease-modifying OA drugs (DMOADs). However, evolving regulatory support, including accelerated pathways and incentives for advanced therapies, is encouraging innovation in intra-articular treatments. At the same time, the widespread use of alternative options such as corticosteroids, hyaluronic acid injections, and over-the-counter drugs like acetaminophen and NSAIDs continues to limit market growth despite their primarily symptomatic benefits.
The osteoarthritis injectables market faces competition from several non-injectable and alternative therapeutic approaches. Key substitutes include oral medications such as NSAIDs and analgesics, which provide symptomatic pain relief and are widely used as first-line treatments. Physical therapy and lifestyle interventions, including weight management and exercise, are also common alternatives that improve joint function and delay disease progression. Additionally, surgical options such as joint replacement are considered in advanced cases. Emerging biologic and regenerative therapies, including platelet-rich plasma (PRP) and stem cell treatments, further act as substitutes to conventional injectable. These alternatives, along with topical therapies and assistive devices, can limit the demand for injectable osteoarthritis treatments.
The osteoarthritis (OA) injectable market is experiencing steady geographical expansion, driven by increasing adoption across hospitals, specialty orthopedic clinics, and ambulatory care centers in both developed and emerging regions. Companies are expanding their presence through distribution partnerships and regulatory approvals, improving access to advanced injectable therapies such as biologics, corticosteroids, and viscosupplements. This growing accessibility, particularly in Asia-Pacific and Latin America, is enhancing patient reach, supporting early intervention, and improving overall outcomes in OA management.
Injection Type Insights
The Hyaluronic Acid (HA) injections segment dominated the market, accounting for a share of 39.23% in 2025 and is expected to grow at the fastest CAGR during the forecast period. Hyaluronic acid continues to maintain high prescription rates in osteoarthritis (OA) due to its ability to improve joint lubrication and pain management. Recent advancements in HA-based hydrogel therapeutics are further driving market growth by enabling prolonged intra-articular residence and supporting cartilage function. For instance, in January 2025, Contura Orthopaedics partnered with Pure Sports Medicine to expand access to Arthrosamid, a long-acting hydrogel injectable for knee OA, enhancing the availability of advanced viscosupplementation treatments. In parallel, the broader orthobiologics space is evolving with the emergence of cell-based therapies, indicating future competition beyond traditional HA injections.
The corticosteroid injections segment is anticipated to witness steady growth over the forecast period, driven by its widespread clinical use for short-term pain relief in osteoarthritis (OA). Corticosteroids reduce inflammation by inhibiting pro-inflammatory mediators such as interleukins and prostaglandins, thereby improving joint function. Clinical guidelines, including those from the Osteoarthritis Research Society International, support their use for short-term symptom management, particularly in patients with inflammatory OA phenotypes. However, their limited duration of action and lack of disease-modifying effects have shifted market focus toward longer-acting and regenerative injectable therapies. This trend is reflected in recent developments beyond corticosteroids. For instance, in January 2025, Contura Orthopaedics partnered with Pure Sports Medicine to expand access to Arthrosamid, improving the availability of long-acting intra-articular treatment options in the osteoarthritis injectables market.
Anatomy Insights
The knee osteoarthritis segment dominated the market, accounting for a share of 42.18% in 2025. Knee osteoarthritis is a prevalent condition, particularly affecting older individuals, and is a leading cause of pain and disability among adults aged 50 years and above in the U.S. This condition significantly impacts the quality of life of patients, necessitating effective prevention and treatment strategies to alleviate pain and improve functionality. While pharmacological therapies such as Nonsteroidal Anti-inflammatory Drugs (NSAIDs) offer relief from pain and inflammation, they often only provide symptomatic relief without addressing disease progression. As the aging population continues to grow, the prevalence of knee osteoarthritis is expected to rise, emphasizing the urgent need for comprehensive management approaches to mitigate disability rates and enhance quality of life.
The hip osteoarthritis segment is poised to grow at the highest CAGR of 7.63% over the forecast period. Hip osteoarthritis poses significant challenges to mobility and functionality, often leading to joint replacement in advanced stages. The condition represents a growing public health concern, particularly as the population ages. As life expectancy increases, the burden of hip osteoarthritis is expected to escalate, driving demand for effective treatment solutions. For instance, in March 2025, Stryker Corporation expanded its hip portfolio through advancements in robotic-assisted joint replacement technologies, including the introduction of enhanced Mako Total Hip capabilities for primary and revision procedures, strengthening its enabling technologies platform and supporting improved surgical precision.
End Use Insights
The hospital pharmacies segment dominated the market with a share of 47.36% in 2025. Hospital pharmacies play a pivotal role in the management of osteoarthritis (OA), particularly for patients requiring advanced therapies administered in inpatient settings. The complexity of treatment regimens necessitates continuous monitoring and optimization of therapy. Supporting this, a study published in Cureus in March 2026 demonstrated that clinical pharmacist-led medication reviews improved medication safety and facilitated identification of therapy-related issues in hospitalized patients. Additionally, evidence from 2025 studies highlights the role of clinical pharmacists in improving clinical outcomes and medication management in hospital settings, reinforcing their importance in multidisciplinary care. Furthermore, hospital pharmacies enable timely access to specialized treatments and ensure appropriate dosing and administration, particularly for patients with comorbid conditions. Their integration within clinical teams also supports coordinated care delivery and enhances overall treatment efficiency in managing chronic conditions such as OA.
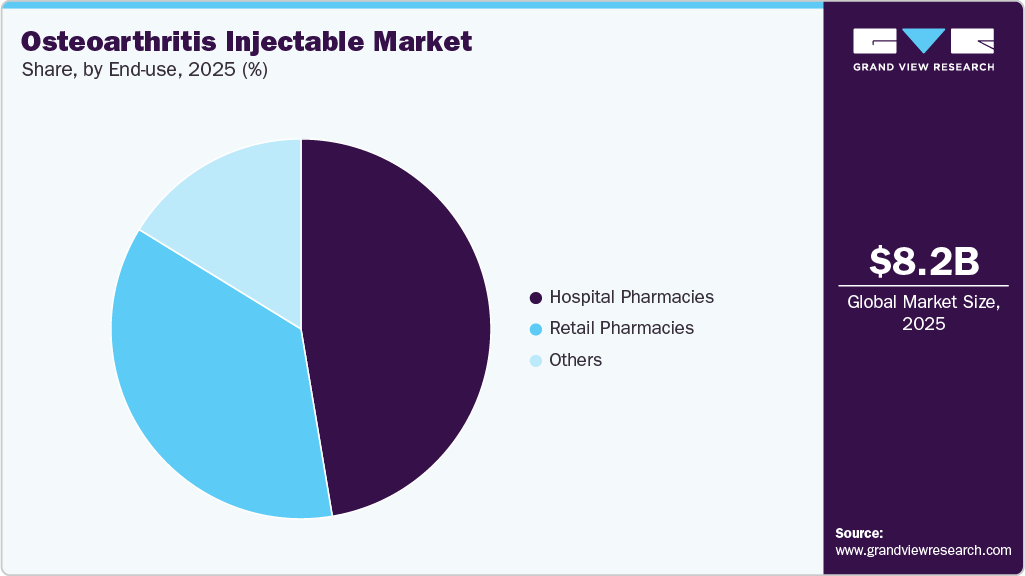
The retail pharmacies segment is anticipated to witness the fastest CAGR of 7.88% during the forecast period. Retail pharmacies serve as important access points within the osteoarthritis (OA) treatment landscape, primarily by ensuring the availability and distribution of prescribed therapies to patients in outpatient settings. While hospital pharmacies dominate the administration of injectable treatments, retail pharmacies play a complementary role by improving accessibility, offering extended hours, and supporting patient engagement and adherence through personalized services. For instance, in 2026, CVS Health introduced new pharmacy-only store formats focused exclusively on prescription services, immunizations, and pharmacist-led care, aimed at improving healthcare accessibility and delivering faster, patient-centric services in community settings. Additionally, evolving models of retail pharmacy emphasize their transition into community-based healthcare hubs, reinforcing their role in enhancing convenience, continuity of care, and medication access for chronic disease management, including osteoarthritis.
Regional Insights
North America osteoarthritis injectable market dominated the market and accounted for a 58.58% share in 2025. The North American market's dominance is evident due to factors such as the region's high obesity prevalence and supportive government policies. Key market players such as Teva Pharmaceuticals, Zimmer Biomet, and Ferring Pharmaceuticals Inc. further solidify its position. Strategic initiatives such as therapeutic study trials, mergers, acquisitions, and partnerships among pharmaceutical giants are actively shaping the competitive landscape. For instance, in March 2026, Enlivex Therapeutics received U.S. FDA Investigational New Drug (IND) clearance to initiate a global Phase 2b clinical trial of Allocetra, an intra-articular injectable therapy for moderate-to-severe knee osteoarthritis, highlighting continued innovation and advancement of injectable treatments in the region.
U.S. Osteoarthritis Injectable Market Trends
The U.S. osteoarthritis injectable market is expected to grow over the forecast period, driven by a strong pharmaceutical industry and the presence of leading innovators such as Zimmer Biomet, Bioventus, and Remedium Bio, which are advancing the development of novel therapeutics. For instance, in December 2025, Zimmer Biomet partnered with OneStep to integrate mobility data solutions aimed at improving joint procedure outcomes and recovery monitoring, highlighting the growing convergence of digital health and orthopedic therapeutics. This collaboration reflects the increasing emphasis on data-driven and patient-centric approaches in musculoskeletal care. Furthermore, ongoing strategic initiatives and technological advancements are expected to strengthen the development and adoption of next-generation injectable therapies in the U.S. market.
Europe Osteoarthritis Injectable Market Trends
The Europe osteoarthritis injectables market continues to present a lucrative opportunity for pharmaceutical companies, driven by the rising prevalence of OA, particularly among aging populations. Ongoing pharmaceutical advancements and increasing focus on disease-modifying therapies are supporting market expansion. Regulatory authorities such as European Medicines Agency continue to emphasize stringent evaluation criteria for drug efficacy, safety, and clinical trial design, shaping product approvals. Viscosupplementation agents remain widely used for symptom management, although their adoption varies across countries due to evolving clinical guidelines and reimbursement policies. Additionally, increasing research activity and strategic collaborations among regional and global players are further accelerating innovation in the current market scenario.
The UK osteoarthritis injectable market is witnessing steady growth, driven by an aging population and increasing awareness of osteoarthritis and its early management. Corticosteroid injections remain widely used for short-term symptom relief, particularly during acute flare-ups, while the use of hyaluronic acid injections is more selective due to evolving clinical guidelines. Recommendations from the National Institute for Health and Care Excellence emphasize evidence-based and cost-effective treatment approaches, influencing adoption patterns across the country. Additionally, increasing focus on early diagnosis, personalized care, and ongoing research into advanced therapies is shaping the current market landscape and supporting continued growth.
The osteoarthritis injectable market in France is experiencing steady growth, driven by an aging population and the increasing prevalence of osteoarthritis. Pharmaceutical companies are actively focusing on innovation to develop more effective and safer injectable therapies, particularly in viscosupplementation and next-generation formulations. For instance, in August 2025, Synartro initiated a Phase 1/2a clinical trial for SYN321, a novel intra-articular injection combining hyaluronic acid with diclofenac for osteoarthritis treatment, highlighting ongoing advancements in combination injectable therapies in Europe. Additionally, continued investments in research and strategic collaborations are further strengthening the competitive landscape in France.
The osteoarthritis injectable market in Germany is expected to grow over the forecast period, driven by an aging population, increasing obesity rates, and a rising incidence of musculoskeletal injuries. Strong emphasis on research and development by pharmaceutical and biotechnology companies continues to support innovation in advanced injectable therapies. Supporting this trend, in October 2025, clinical research highlighted the continued use and evaluation of low-dose radiation therapy for osteoarthritis in European countries, including Germany, demonstrating significant pain reduction and improved mobility in patients, reflecting ongoing efforts to expand and optimize treatment approaches for OA. Additionally, advancements in intra-articular drug delivery systems and biologic therapies are further strengthening Germany’s position as a key innovation hub in the European OA treatment landscape.
Asia Pacific Osteoarthritis Injectable Market Trends
The Asia Pacific osteoarthritis injectable market is expected to grow at highest CAGR due to the high prevalence of osteoarthritis, increasing healthcare spending, population growth, and ongoing medical advancements across the region. Countries such as China, Japan, and India are key contributors, driven by aging populations and rising demand for advanced treatment options. Pharmaceutical companies are actively investing in research and development to introduce innovative therapies. Supporting this trend, in March 2025, Belief BioMed received Investigational New Drug (IND) clearance from China’s regulatory authority for its gene therapy candidate BBM-A101 for knee osteoarthritis, highlighting the region’s growing focus on next-generation injectable and regenerative treatments.
The osteoarthritis injectable market in China is expected to grow over the forecast period, driven by increasing disease prevalence, rising adoption of pain management therapies, and a growing susceptible population. Favorable regulatory policies and expanding healthcare access are further supporting demand for both symptomatic and disease-modifying treatments. Pharmaceutical companies are actively engaging in research collaborations and product development to introduce innovative therapies. Supporting this trend, in March 2026, Sino Biopharmaceutical Limited received clinical trial approval from China’s National Medical Products Administration for TRD221, a novel therapy targeting osteoarthritis, highlighting the country’s accelerating focus on advanced injectable and disease-modifying treatments.
The osteoarthritis injectable market in Japan is expected to grow over the forecast period, driven primarily by the country’s rapidly aging population and increasing demand for effective pain management and mobility-enhancing therapies. Japanese pharmaceutical companies are actively expanding their presence through research and development of advanced injectable treatments. For instance, in April 2025, Ono Pharmaceutical entered into a basic agreement with Seikagaku Corporation for the co-development and commercialization of Gel-One, a hyaluronic acid-based intra-articular injectable currently undergoing Phase III clinical trials in Japan, highlighting strong domestic innovation and pipeline expansion.
Latin America Osteoarthritis Injectable Market Trends
The osteoarthritis injectable market in Latin America is expected to grow over the forecast period, driven by an increasing disease burden, rising awareness, and gradual improvements in healthcare access across the region. While osteoarthritis continues to significantly impact health-related quality of life and poses economic challenges, gaps in treatment access and clinical data persist in several countries. However, growing adoption of injectable therapies and expanding clinical research activity are supporting market development. For instance, in February 2025, a global Phase III clinical trial evaluating pentosan polysulfate sodium for knee osteoarthritis included sites across North and South America, highlighting increasing participation of Latin American regions in advanced osteoarthritis research and therapeutic development.
The osteoarthritis injectable market in Brazil is expected to grow over the forecast period, driven by a high disease burden, increasing healthcare expenditure, and rising demand for effective pain management therapies. Osteoarthritis continues to significantly impact disability and quality of life, placing a considerable burden on the healthcare system. Recent developments also indicate growing participation of Brazil in global clinical research activities, supporting the adoption of advanced injectable therapies and strengthening the country’s position in the Latin American market.
Middle East Osteoarthritis Injectable Market Trends
The osteoarthritis injectable market in the Middle East and Africa is expected to grow over the forecast period, driven by an aging population, improving healthcare infrastructure, and increasing adoption of minimally invasive treatment options such as viscosupplementation. Expanding access to healthcare services and rising awareness of osteoarthritis management are further supporting market growth. Additionally, increasing involvement of regional healthcare providers in international research initiatives and the gradual introduction of advanced therapies are contributing to the evolving treatment landscape across the region.
The osteoarthritis injectable market in Saudi Arabia is expected to grow over the forecast period, driven by an aging population, increasing prevalence of osteoarthritis, and expanding healthcare infrastructure under national initiatives. Market players in the region are actively undertaking strategic collaborations to strengthen their presence and expand access to advanced therapies. Supporting this trend, in May 2025, Tabuk Pharmaceutical Manufacturing Company announced an exclusive licensing partnership with Hanmi Pharmaceutical to introduce innovative therapies across Saudi Arabia and the MENA region, highlighting growing investment and collaboration in advanced pharmaceutical products.
Key Osteoarthritis Injectable Company Insights
Key players such as Anika Therapeutics, Inc., Bioventus, Ferring Pharmaceuticals Inc., and Sanofi S.A. implement a variety of strategies to sustain their competitive advantage. These approaches include ongoing investment in new product development, securing patent protection for innovations, obtaining necessary regulatory approvals, and establishing collaborations and partnerships to broaden market reach and enhance their product portfolios. In addition, there are notable competitors in the market such as Flexion Therapeutics, Inc., Zimmer Biomet, Arthrex, Inc., and Royal Biologics. These emerging players employ diverse strategies to bolster their presence in the market. Strategies such as a relentless focus on innovation to differentiate their products, expanding their geographic footprint to tap into new markets, and forging strategic partnerships to leverage complementary strengths help these companies in achieving their growth objectives. As competition intensifies, the ability to innovate, adapt to evolving regulatory landscapes, and forge strategic alliances will be pivotal for both established and emerging players to maintain and expand their market share in the dynamic landscape of OA injectable.
Key Osteoarthritis Injectable Companies:
The following key companies have been profiled for this study on the osteoarthritis injectable market.
- Anika Therapeutics, Inc.
- Bioventus.
- Ferring Pharmaceuticals Inc.
- Sanofi S.A.
- Flexion Therapeutics, Inc.
- Zimmer Biomet
- Arthrex, Inc.
- Royal Biologics
- Teva Pharmaceutical Industries Ltd.
Recent Developments
-
In October 2025, Ferring Pharmaceuticals announced optimisation of its operations under a new Enterprise Model to accelerate innovation, improve flexibility, and enhance investment capacity in advanced therapeutic areas, positioning the company to support future developments in injectable and biologic treatments.
-
In February 2026, Zimmer Biomet Holdings, Inc. announced that it will present new clinical and operational data and showcase its latest orthopaedic innovations at the American Academy of Orthopaedic Surgeons (AAOS) 2026 Annual Meeting, underscoring its ongoing commitment to advancing treatments for musculoskeletal conditions, including osteoarthritis, through enhanced joint solutions and technologies that support improved patient outcomes.
-
In January 2025, Bioventus completed the divestiture of its Advanced Rehabilitation business to Accelmed Partners, allowing the company to sharpen its focus on core biologics and next‑generation osteoarthritis injectable like DUROLANE, supporting broader distribution and treatment optimization.
Osteoarthritis Injectable Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 8.72 billion
Revenue forecast in 2033
USD 14.16 billion
Growth rate
CAGR of 7.17% from 2026 to 2033
Actual years
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Injection Type, Anatomy, End Use, and Region
Regions covered
North America; Europe; Asia Pacific; Latin America; MEA
Countries covered
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Anika Therapeutics, Inc.; Bioventus.; Ferring Pharmaceuticals Inc.; Sanofi S.A.; Flexion Therapeutics, Inc.; Zimmer Biomet; Arthrex, Inc.; Royal Biologics; Teva Pharmaceutical Industries Ltd.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Osteoarthritis Injectable Market Repoprt Segmentation
This report forecasts revenue growth at, regional & country levels and provides an analysis on the industry trends in each of the sub-segments from 2021 to 2033. For the purpose of this study, Grand View Research has segmented the global osteoarthritis injectable market report on the basis of injection type, anatomy, end use, and region:
-
Injection Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Hyaluronic Acid Injections
-
Corticosteroid Injections
-
Platelet-rich Plasma (PRP) Injections
-
Placental Tissue Matrix (PTM) Injections
-
Acetylsalicylic Acid (ASA) Injections
-
Others
-
-
Anatomy Outlook (Revenue, USD Million, 2021 - 2033)
-
Knee Osteoarthritis
-
Hip Osteoarthritis
-
Hand Osteoarthritis
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospital Pharmacies
-
Retail Pharmacies
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global osteoarthritis injectables market size was estimated at USD 7.23 billion in 2023 and is expected to reach USD 7.68 billion in 2024.
b. The global osteoarthritis injectables market is expected to grow at a compound annual growth rate of 6.82% from 2024 to 2030 to reach USD 11.42 billion by 2030.
b. Hyaluronic Acid (HA) injections segment dominated the market, accounting for a share of 38.73% in 2023. HA has high prescription rate in OA. Recent advancements in HA injection therapy have propelled space growth trajectory.
b. Market players operating in market include Anika Therapeutics, Inc., Bioventus., Ferring Pharmaceuticals Inc., Sanofi S.A., Flexion Therapeutics, Inc., and Zimmer Biomet.
b. The increasing prevalence of osteoarthritis, growing risk of developing osteoarthritis in the aging population, and rising Research & Development (R&D) activities to develop novel therapies are some key factors expected to drive market growth over the forecast period.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










