- Home
- »
- Medical Devices
- »
-
Peripheral Nerve Stimulators Market Size Report, 2033GVR Report cover
![Peripheral Nerve Stimulators Market Size, Share & Trends Report]()
Peripheral Nerve Stimulators Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Systems, Wearables/Accessories), By Modality (Rechargeable, Non-Rechargeable), By Application (Diabetic Peripheral Neuropathy Pain), By Connectivity, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-753-9
- Number of Report Pages: 300
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
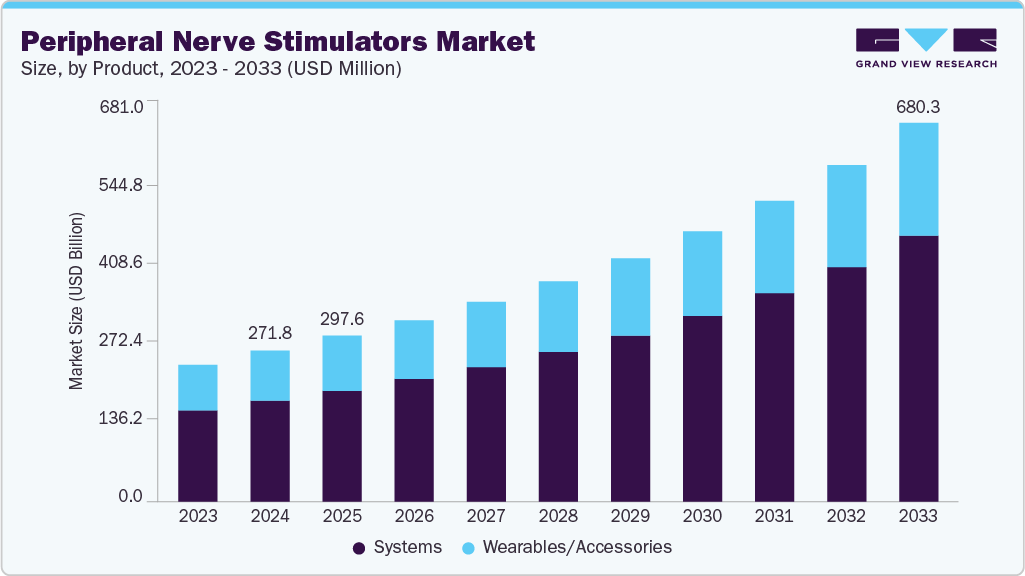
Peripheral Nerve Stimulators Market Summary
The global peripheral nerve stimulators market size was estimated at USD 271.79 million in 2024 and is projected to reach USD 680.30 million by 2033, growing at a CAGR of 10.89% from 2025 to 2033. The rising prevalence of chronic pain disorders, increasing preference for minimally invasive and non-opioid therapies, and continuous advancements in neuromodulation technologies are the primary driving factors contributing to the growth of the industry.
Key Market Trends & Insights
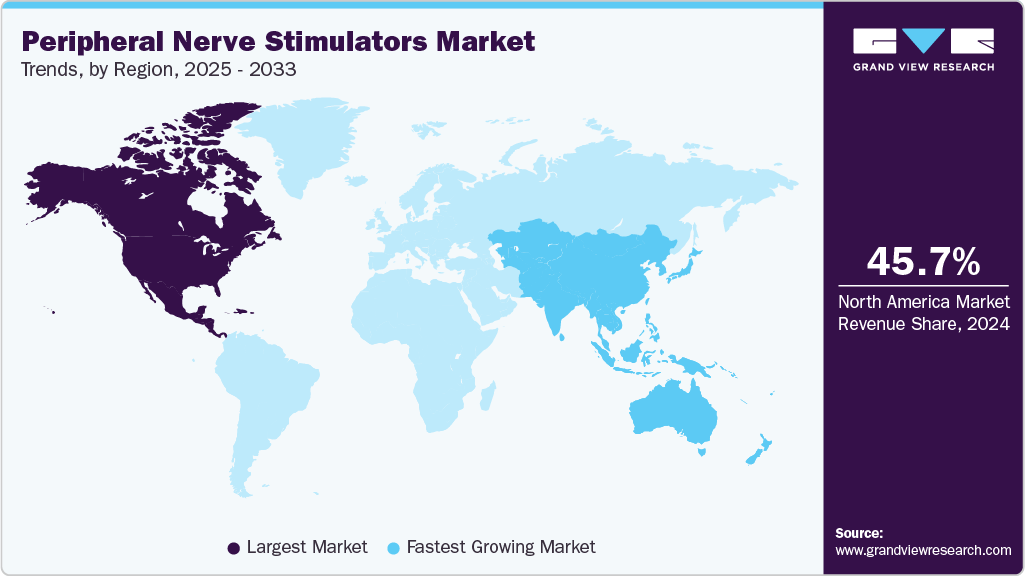
- North America dominated the peripheral nerve stimulators market with the largest share of 45.66% in 2024.
- The U.S. accounted for the largest revenue share of 84.72% in the peripheral nerve stimulators market in North America in 2024.
- By product, the systems segment led the market with the largest revenue share of 66.67%in 2024.
- By modality, the rechargeable segment led the market with the largest revenue share of 63.13% in 2024.
- By connectivity, the wireless segment led the market with the largest revenue share of 78.08% in 2024.
- By application, the chronic musculoskeletal pain segment led the market with the largest revenue share of 42.23% in 2024.
- By end use, the hospitals and clinics segment led the market with the largest revenue share of 52.12% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 271.79 Million
- 2033 Projected Market Size: USD 680.30 Million
- CAGR (2025-2033): 10.89%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
The rising prevalence of chronic pain disorders is one of the key driving factors of the peripheral nerve stimulators industry. Conditions like neuropathic pain, migraines, and post-surgical pain often cause long-term disability, while traditional treatments such as opioids carry risks of addiction and limited efficacy. For instance, according to recent data published by Brain Research UK, the following facts and statistics highlight the burden of neurological conditions in the UK:
Condition
Key Facts, UK
Brain Tumors
More under 40s die from brain tumors than any other cancer - 12,300 people diagnosed with a primary brain tumor each year - 5,500 lives lost annually
Brain & Spinal Cord Injury
Every 90 seconds, someone is admitted to a hospital with a brain injury - 2.6 million people live with the effects of traumatic brain injury or stroke - 105,000 people live with a spinal cord injury
Headache & Facial Pain
Headaches are the most common type of pain - 190,000 migraine attacks occur daily - 65,000 people suffer from cluster headache (the “suicide headache”)
Source: Brain Research UK
The growing shift toward non-opioid therapies is a significant driver of the peripheral nerve stimulators market. With the global opioid crisis highlighting the dangers of addiction, dependency, and adverse side effects, healthcare providers and policymakers are prioritizing safer alternatives for chronic pain management. For instance, according to the CDC, in May 2024, opioid overdose deaths remain high:
-
Opioid-involved overdose deaths: About 105,000 people died from drug overdoses in 2023, with nearly 80,000 deaths (~76%) involving opioids.
-
The number of opioid overdose deaths in 2023 was almost 10 times higher than in 1999.
Growing favorable reimbursement policies are one of the major contributors to market growth. The number of companies' peripheral nerve stimulation systems is reimbursed nationally by Medicare as a treatment for chronic pain of a peripheral origin. For instance, in January 2024, the Bioventus StimRouter Neuromodulation System Reimbursement Reference Guide was released to support healthcare providers and payers in navigating coverage and reimbursement pathways. This guide aimed to facilitate broader adoption of the StimRouter system by improving access for patients requiring peripheral nerve stimulation for chronic pain management. In addition, in January 2024, Bioventus’s StimRouter Neuromodulation System Reimbursement Reference Guide was released:
StimRouter Neuromodulation System Reimbursement Reference Guide January 2024, CPT Codes for StimRouter Device and Procedure
CPT Procedure Code & Description
Physician Fee
Schedule In-Office |
FacilityAmbulatory Surgery Center (ASC)Payment
RateHospital
Outpatient APCHospital
Outpatient
APC Payment
Rate64555: Percutaneous implantation of
neurostimulator electrode array; peripheral nerve (excludes sacral nerve)USD 2,084.50|
USD 316.96USD 5615.15
5462
USD 6516.28
64575: Open implantation of neurostimulator
electrode array; peripheral nerve (excludes sacral nerve)NA |USD 310.42
USD 11344.43
5463
USD 12978.95
64585: Revision or removal of peripheral
neurostimulator electrode arrayUSD 238.38 |
USD 141.78USD 1898.16
5461
USD 3241.90
64596: Insertion or replacement of a percutaneous electrode array, peripheral nerve, with an integrated neurostimulator, including imaging guidance, when
performed; initial electrode arrayTBD | TBD
USD 9224.45
5463
USD 12978.95
+64597: Insertion or replacement of percutaneous electrode array; peripheral nerve, with integrated neurostimulator, including imaging guidance, when performed; each additional electrode array (list separately in addition to the code for primary procedure)
TBD | TBD
SI = N1,
Packaged
service/item;
no separate
payment
made.NA
SI=N, Items
and Services
Packaged
into APC
Rates564598: Revision or removal of neurostimulator electrode array; peripheral nerve, with integrated neurostimulator
TBD | TBD
USD 1898.16
5461
USD 3241.9
Source: StimRouter (Bioventus)
Key Opinion Leaders
Company Name
KOLs
About KOL
Nalu Medical, Inc.
"Nalu Medical advanced wearable neurostimulation with the launch of its next-generation Therapy Disc, which was 39% smaller and significantly improved patient comfort and accessibility. This advancement expanded the reach of peripheral nerve stimulation therapy, allowing more chronic pain patients to be considered for a clinically proven treatment option."
Tom West, CEO and President of Nalu Medical.
Nalu Medical, Inc.
“Aetna’s recognition of Nalu as a medically necessary therapy is a powerful validation of our technology, clinical evidence, and commitment to patient outcomes,” said Tom West, Chief Executive Officer of Nalu Medical. “We’ve built a robust body of clinical data demonstrating that Nalu PNS provides safe, effective, and durable relief for patients living with chronic, intractable pain, as well as healthcare economic data that demonstrates use of Nalu PNS results in significant cost savings. We’re proud to see a leading payer like Aetna acknowledge that value, and we look forward to engaging with other health plans to expand access to this life-changing therapy.”
Tom West, CEO and President of Nalu Medical.
SPR
“The ASPN consensus guidelines were driven by the leading voices in the pain management space and will provide clinicians with a valuable tool to help assist them in optimal patient selection and the application of 60-day PNS throughout the body to deliver the best possible results for their patients.”
Dr. Timothy Deer, President & CEO, The Spine and Nerve Center of the Virginias, and Chairman and Founder of The American Society of Pain & Neuroscience.
Checkpoint Surgical
“This expansion of Checkpoint Surgical’s stimulator portfolio ensures that we provide the surgeon with the optimal solution for clinical nerve procedures.” “Checkpoint Gemini complements our existing stimulators with a device optimized for precise nerve surgeries.”
Derek Lewis, President, and CEO of Checkpoint Surgical
Source: Grand View Research Analysis
Market Concentration & Characteristics
The industry is characterized by growing demand for minimally invasive, non-opioid pain management solutions, driven by the rising prevalence of chronic and neuropathic pain conditions. Technological innovations such as wireless, miniaturized, and wearable devices are shaping market growth, expanding clinical applications, and increasing global adoption.
The industry demonstrates a high degree of innovation, with advancements in wireless technologies, miniaturized and wearable devices, rechargeable systems, and smart programming that enhance precision, patient comfort, and long-term outcomes. For instance, in August 2025, Nalu Medical advanced wearable neurostimulation technology by introducing a new therapy disc that is 39% smaller, offering patients a more comfortable and discreet option for peripheral nerve stimulation.
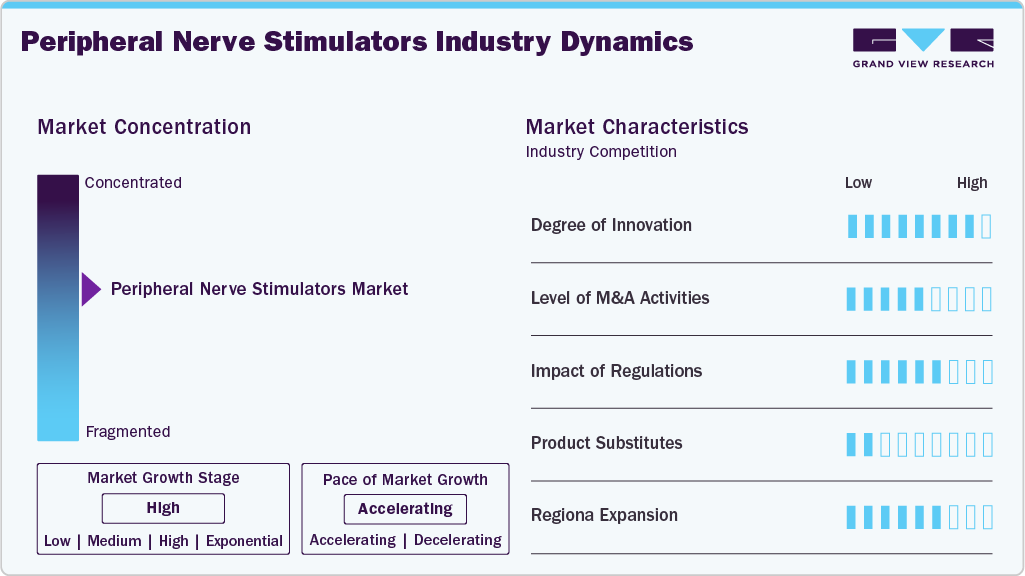
Regulations shape the peripheral nerve stimulators market by ensuring safety and efficacy, though stringent approvals may slow market entry, while supportive policies can accelerate innovation and adoption. For instance, in July 2025, Bioventus Inc., a global leader in innovations for active healing, announced it had received FDA 510(k) clearances for both TalisMann and StimTrial. These approvals mark a significant expansion of the company’s Peripheral Nerve Stimulation (PNS) portfolio, strengthening its position in delivering innovative, non-opioid solutions for chronic pain management.
The industry has witnessed moderate merger and acquisition activity, with companies pursuing strategic deals to expand product portfolios and strengthen technological capabilities. Such consolidations are enabling larger players to enhance market presence while supporting smaller innovators in scaling their advanced neuromodulation solutions. For instance, in March 2021, Bioventus Inc. acquired Bioness, Inc., a global leader in neuromodulation and rehabilitation devices, for USD 45 million upfront and up to USD 65 million in contingent milestone-based payments, strengthening its portfolio with innovative peripheral nerve stimulation (PNS) therapies and rehabilitation solutions.
Regional expansion activity in the market is growing as key players enter emerging markets in Asia-Pacific, Latin America, and the Middle East to tap into the rising demand for advanced pain therapies. Partnerships with local healthcare providers and investments in distribution networks are driving broader global adoption. For instance, in August 2025, Checkpoint Surgical hosted its 3rd Annual Nerve Summit in Cleveland, showcasing its commitment to advancing nerve care, supporting regional expansion, and fostering global collaborations in peripheral nerve treatment.
Product Insights
The systems segment dominated the market in 2024, driven by rising adoption of advanced technologies for chronic pain management and increasing preference for minimally invasive therapies. This segment includes implantable and non-implantable systems, with the latter comprising handheld devices and other external stimulators. For instance, in July 2025, Asuris Northwest Health published implantable peripheral nerve stimulation (PNS) and its modification, peripheral subcutaneous field stimulation (PSFS), relieving chronic pain by implanting electrodes near affected nerves or pain regions. Unlike other electrical therapies, these approaches target peripheral nerves or nerve fields rather than surrounding tissues or the spine.
The wearables/accessories segment is significantly growing due to its versatility, ease of use, and effectiveness in managing chronic pain conditions. Continuous technological advancements, including miniaturized wireless devices, smart controllers, and rising patient preference for non-invasive solutions, are expected to accelerate this segment’s growth further. For instance, in August 2025, ScienceDirect published a study showing that a wearable peripheral nerve stimulator can reduce essential tremor symptoms through targeted brain modulation, highlighting its potential as a non-invasive and effective therapy option.
Modality Insights
The rechargeable segment led the market in 2024, driven by demand for long-term, cost-effective pain management and advancements in miniaturized rechargeable systems. For instance, in January 2024, ScienceDirect published a study on peripheral nerve stimulation using a high-frequency electromagnetic coupled powered implanted receiver at the posterior tibial nerve for treating chronic foot pain, demonstrating its potential effectiveness as a targeted neuromodulation therapy.
The non-rechargeable segment is projected to grow significantly in the peripheral nerve stimulators market, driven by demand for simple, low-maintenance devices that offer ease of use and reliable pain relief. Their convenience, shorter treatment duration, suitability, and preference among certain patient groups are fueling adoption, aligning with the broader trend toward personalized and accessible pain management solutions.
Connectivity Insights
The wireless segment dominates the peripheral nerve stimulators market, driven by its ease of use, minimally invasive nature, and improved patient comfort compared to traditional wired systems. Growing adoption of compact, wearable, and remotely programmable devices further reinforces its strong market presence. For instance, in May 2025, Nature Communications published a study on a bioresorbable, wireless dual stimulator designed for peripheral nerve regeneration, highlighting its potential to enhance recovery while eliminating the need for device removal.
The wired segment is experiencing significant growth in the peripheral nerve stimulators market due to its reliability and consistent power delivery for chronic pain management. Advances in implantable wired systems enhance therapeutic outcomes, making them a trusted option for clinicians. Growing clinical adoption and proven long-term effectiveness further support the segment’s expansion.
Application Insights
The chronic musculoskeletal pain segment dominated the market in 2024, driven by its high prevalence and growing demand for non-opioid pain management solutions. Rising cases of arthritis, lower back pain, and sports-related injuries further supported segment growth. In February 2025, Elsevier published "Peripheral Nerve Stimulation for Pain Management: A Survey of Clinical Practice Patterns," providing valuable insights into how PNS is utilized across diverse clinical settings. The survey highlighted evolving physician preferences, adoption trends, and practical considerations shaping peripheral nerve stimulation's role in chronic pain management.
The Complex Regional Pain Syndrome (CRPS) segment is projected to be the fastest-growing area of the peripheral nerve stimulators market from 2025 to 2033. This growth is fueled by the increasing prevalence of trauma cases, burn injuries, and surgical interventions that often lead to chronic pain conditions. As awareness of non-opioid pain management rises, peripheral nerve stimulators are gaining prominence as a targeted therapy for CRPS, offering long-term relief and functional improvement. These factors position the CRPS segment as a key driver of market expansion over the forecast period.
End Use Insights
The hospitals and clinics segment dominated the market in 2024, capturing the largest share. This leadership was attributed to the high patient volume requiring advanced pain management and post-surgical care, where peripheral nerve stimulators play a critical role. Hospitals and clinics served as primary centers for procedures addressing neuropathic pain, complex surgical recoveries, and chronic pain conditions, driving strong adoption of these devices. The availability of skilled healthcare professionals, access to cutting-edge neuromodulation technologies, and a rising number of surgical interventions further reinforced hospitals’ position as the leading end-use setting for peripheral nerve stimulators globally.
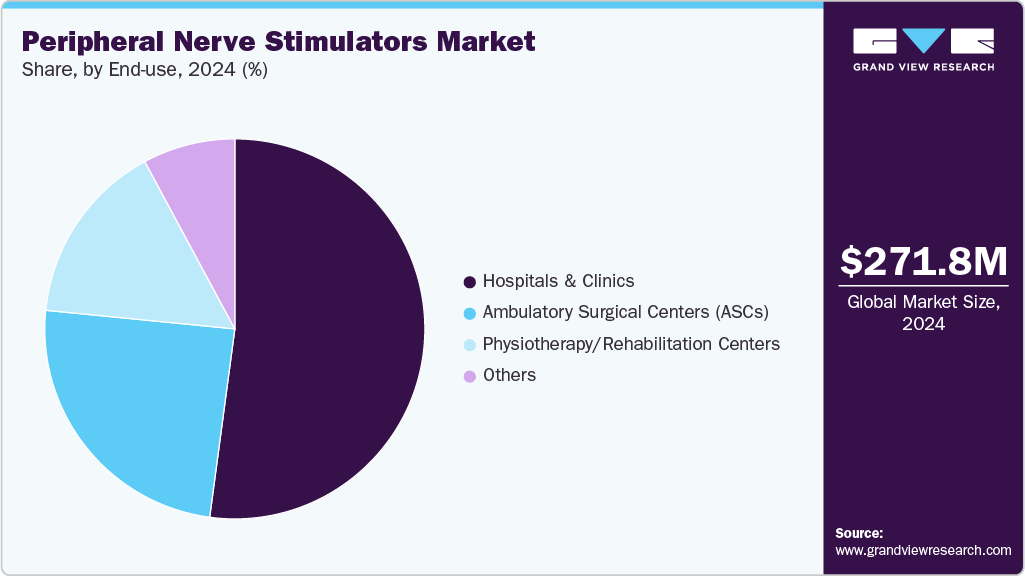
The ambulatory surgical centers (ASCs) segment is expected to be the fastest-growing segment during the forecast period. This growth is driven by the rising preference for minimally invasive procedures, cost-effective treatment settings, and shorter recovery times offered by ASCs. Increasing adoption of advanced neuromodulation therapies in outpatient environments positions ASCs as a key contributor to market expansion.
Regional Insights
North America peripheral nerve stimulators market held the largest revenue share in 2024. The region is also anticipated to grow fast over the forecast period, supported by the strong presence of leading players such as Medtronic, Abbott, and Boston Scientific, along with continuous innovation in minimally invasive neuromodulation devices. In addition, the rising prevalence of chronic pain conditions, coupled with an expanding aging population, is driving higher adoption of PNS therapies. For example, a report from the U.S. CDC highlighted that chronic pain affects more than 51.6 million adults in the country, underscoring the significant growth potential for advanced pain management solutions like peripheral nerve stimulators.
U.S. Peripheral Nerve Stimulators Market Trends
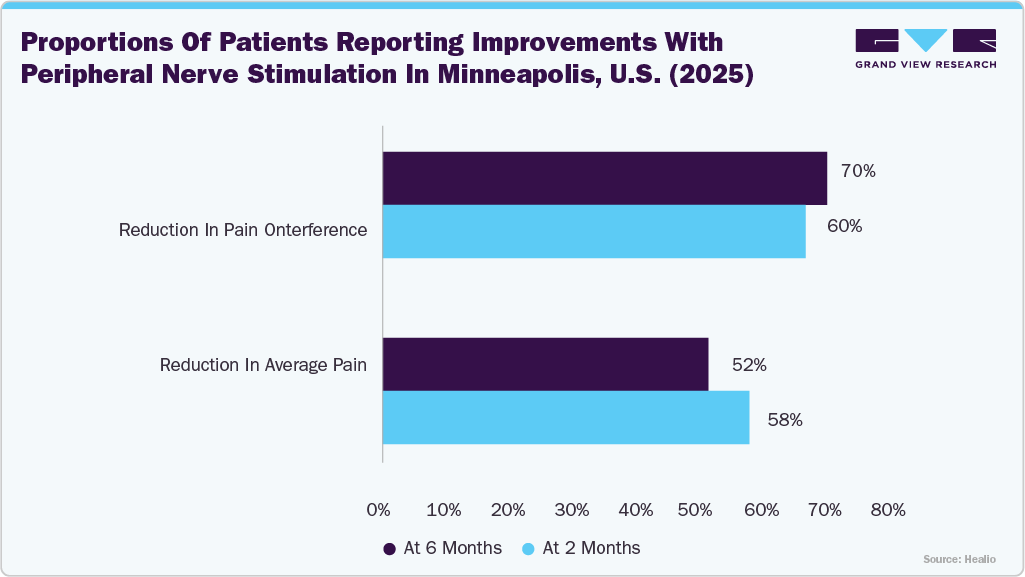
The peripheral nerve stimulators market in the U.S. held the largest share of 84.72% in 2024 in North America, and is gaining momentum as an effective non-opioid solution for managing chronic pain conditions. For instance, in June 2025, Healio highlighted that patients with occipital headache experienced significant relief after 60 days of peripheral nerve stimulation therapy. The graph below shows the proportions of patients reporting reductions in average pain and pain interference at 2 months and 6 months of PNS treatment.
Europe Peripheral Nerve Stimulators Market Trends
The peripheral nerve stimulators market in Europe is poised for notable growth in the coming years, fueled by increasing adoption of minimally invasive pain therapies, supportive reimbursement policies, and growing awareness of non-opioid alternatives for chronic pain management. In March 2025, the European Journal of Cardiovascular Medicine published a study on peripheral neuropathy's clinical, electrophysiological, and aetiological profile in a tertiary care center, highlighting patterns of diagnosis, underlying causes, and the importance of early intervention for effective management.
The UK peripheral nerve stimulators market is expected to see substantial growth in the coming years, supported by rising demand for non-opioid pain management, advancements in neuromodulation technologies, and increased adoption within NHS healthcare settings. In February 2022, the NHS UK reported on a PNS motor stimulation study that explored the effectiveness of peripheral nerve stimulators in improving pain management and functional outcomes. The study highlighted the potential of PNS therapy to reduce chronic pain while enhancing patient mobility and quality of life.
Asia Pacific Peripheral Nerve Stimulators Market Trends
The peripheral nerve stimulators market in Asia-Pacific is expanding rapidly, driven by a high prevalence of chronic pain and rising cases of diabetic neuropathy. Chronic pain, including cancer pain, neuropathic pain, and musculoskeletal pain, continues to increase in the region. For instance, in April 2023, The Lancet highlighted the burdens and progress of chronic pain management, noting that depending on the measurement methods used, the prevalence of chronic pain in Asian countries ranges from 7.1% to 90.8% among adults. Despite this high prevalence, awareness and effective management of pain have long been neglected, creating opportunities for broader adoption of advanced therapies like PNS.
India’s peripheral nerve stimulators market is growing due to a rising burden of chronic pain and diabetic neuropathy, coupled with increasing demand for non-opioid treatment options. Advancements in healthcare infrastructure and greater adoption of minimally invasive therapies further drive market expansion. For instance, in December 2023, the Indian Journal of Pain published Best Practice Recommendations for Implantable Neuromodulation Therapies for Pain II, covering intrathecal drug delivery systems, peripheral nerve stimulation (PNS), and sacral neuromodulation. The publication emphasized standardized clinical guidelines to improve patient selection, optimize outcomes, and enhance the safe and effective use of these advanced pain management therapies.
Latin America Peripheral Nerve Stimulators Market Trends
The peripheral nerve stimulators market in Latin America is showing significant growth, driven by the rising prevalence of chronic pain, neuropathic disorders, and an increasing diabetic population. Expanding access to advanced medical technologies through healthcare reforms and greater investments in pain management solutions supports adoption. In addition, growing awareness of non-opioid alternatives and minimally invasive procedures is creating opportunities for broader use of PNS devices across the region. International players are also entering the market through partnerships and product launches, further boosting availability and market penetration.
Middle East and Africa Peripheral Nerve Stimulators Market Trends
The peripheral nerve stimulators market in the Middle East and Africa is experiencing gradual growth, supported by a rising burden of chronic pain conditions, increasing cases of diabetes-related neuropathy, and a growing elderly population. Expanding healthcare infrastructure and ongoing investments in advanced medical technologies are boosting access to innovative pain management solutions. In addition, a shift toward non-opioid therapies and greater awareness of minimally invasive treatment options are encouraging the adoption of PNS devices across the region.
Saudi Arabia peripheral nerve stimulators market is witnessing the growing adoption of peripheral nerve stimulators (PNS) as demand for advanced pain management solutions increases. Rising cases of chronic pain and diabetic neuropathy, along with an expanding elderly population, are driving market growth. Healthcare modernization and greater awareness of non-opioid alternatives further support the country's uptake of innovative PNS technologies. For instance, in July 2023, the Saudi Press Agency reported that six peripheral nerve stimulator devices were successfully implanted at King Fahd Medical City, marking an unprecedented milestone in Saudi Arabia’s adoption of advanced pain management technologies.
Key Peripheral Nerve Stimulators Companies Insights
Key players operating in the peripheral nerve stimulators market are undertaking various initiatives to strengthen their presence and increase the reach of their products and services. Strategies such as expansion activities and partnerships are key in propelling the market growth.
Key Peripheral Nerve Stimulators Companies:
The following are the leading companies in the peripheral nerve stimulators market. These companies collectively hold the largest market share and dictate industry trends.
- B. Braun SE
- Nalu Medical, Inc.
- Curonix LLC.
- SPR
- Vygon
- AirLife
- Neuspera Medical
- AVNS
- Mercury Medical
- Medilogix
- Anesthesia Associates, Inc.
- Medtronic
- Boston Scientific Corporation
- Abbott
- Bioventus
- Checkpoint Surgical Inc.
- NEURIMPULSE s.r.l.
- Nervonik, Inc
- Epineuron Technologies Inc.
Recent Developments
-
In August 2025, Checkpoint Surgical marked a milestone with the first use of its investigational nerve regeneration system, Regen10, in the FASTR-TEN clinical trial. This advancement represented a significant step forward in exploring innovative approaches to promote peripheral nerve regeneration and improve outcomes for patients with nerve injuries.
-
In August 2025, Checkpoint Surgical hosted the 2025 Nerve Summit, bringing together leading international experts in peripheral nerve surgery. The event focused on advancing clinical knowledge, sharing best practices, and fostering collaboration to improve outcomes in nerve repair and regeneration.
-
In August 2025, Nalu Medical advanced wearable neurostimulation with the launch of its new Therapy Disc, which is 39% smaller than the previous version. This next-generation device enhanced patient comfort, expanded accessibility, and broadened eligibility for chronic pain patients to benefit from clinically proven peripheral nerve stimulation therapy.
-
In July 2025, SPR announced that it will feature five abstracts highlighting the SPRINT Peripheral Nerve Stimulation (PNS) System at the upcoming ASPN 2025 meeting. The presentations will showcase new clinical evidence on the role of SPRINT PNS in managing chronic pain, including data on patient outcomes, functional improvement, and real-world applications across multiple pain indications. This marks another step in expanding awareness and adoption of PNS as a minimally invasive, non-opioid treatment option for patients with unmet pain management needs.
-
In July 2025, Bioventus Inc., one of the global leaders in innovations for active healing, announced it had received FDA 510(k) clearances for both TalisMann and StimTrial. These approvals mark a significant expansion of the company’s Peripheral Nerve Stimulation (PNS) portfolio, strengthening its position in delivering innovative, non-opioid solutions for chronic pain management.
-
In May 2025, Imec and the Feinstein Institutes for Medical Research unveiled a novel concept for neuromodulation using intermittent interferential current stimulation (i²CS). The breakthrough, published in Nature Communications, demonstrates a more energy-efficient and precise method of activating neural tissue, reducing side effects while enhancing therapeutic outcomes. This innovation paves the way for new applications in treating chronic conditions such as depression and rheumatoid arthritis through advanced vagus nerve stimulation.
-
In March 2025, Nervonik, Inc., a medical device company developing an opioid-free peripheral nerve stimulation (PNS) system, announced the successful closing of a USD 13 million Series A funding round. The round was led by U.S. Venture Partners (USVP) with participation from Foothill Ventures, Correlation Ventures, and other investors. This financing builds on Nervonik’s earlier USD 4.4 million in SAFE and convertible note investments from Shangbay Capital, Camford Capital, Joyance Partners, Life Science Angel, Seraph Group, and others, further strengthening the company’s resources to advance development and clinical validation of its novel PNS platform.
-
In January 2025, Nalu Medical announced FDA clearance for expanded labeling of its Peripheral Nerve Stimulation (PNS) System, now approved for whole-body MRI-conditional use. This approval offers physicians increased flexibility in managing chronic pain by reducing MRI-related restrictions. With the updated MRI-conditional status of the Nalu Neurostimulation System, pain specialists can confidently deliver tailored treatments without concerns about future diagnostic imaging limitations.
-
In September 2024, Epineuron received Health Canada approval for its PeriPulse System, a novel technology designed to support nerve regeneration. This approval marked a significant milestone for the company and expanded access to innovative therapies aimed at improving recovery outcomes for patients with peripheral nerve injuries.
-
In February 2024, SPR announced USD 85 million in additional funding to support the rapid commercial expansion of the SPRINT PNS System. This investment aimed to accelerate market penetration, scale operations, and strengthen the company’s leadership in minimally invasive peripheral nerve stimulation therapies.
-
In January 2024, Nalu Medical, Inc. presented long-term and holistic outcomes from the COMFORT randomized controlled trial (RCT) for peripheral nerve stimulation (PNS) at the North American Neuromodulation Society (NANS) meeting. The results highlighted sustained pain relief, functional improvement, and quality-of-life benefits, reinforcing the clinical value of Nalu’s PNS therapy.
-
In January 2024, the Bioventus StimRouter Neuromodulation System Reimbursement Reference Guide was released to support healthcare providers and payers in navigating coverage and reimbursement pathways. This guide aimed to facilitate broader adoption of the StimRouter system by improving access for patients requiring peripheral nerve stimulation for chronic pain management.
-
In April 2023, Neuspera Medical announced FDA clearance of its system for Peripheral Nerve Stimulation (PNS). The minimally invasive technology is designed to provide adequate pain relief by targeting specific peripheral nerves with greater precision and flexibility. This milestone clearance supports broader clinical adoption of PNS as a non-opioid therapy option and strengthens Neuspera’s position in advancing next-generation neuromodulation solutions.
-
In October 2023, Boston Scientific received FDA approval for its spinal cord stimulation (SCS) therapy to treat people living with diabetic peripheral neuropathy (DPN). This approval expanded the company’s neuromodulation portfolio and provided a new treatment option for patients suffering from chronic pain associated with diabetes-related nerve damage.
-
In February 2023, SPR received 510(k) clearance for technological advancements and an expanded intended patient population for the SPRINT PNS System. This clearance enhanced the system’s clinical versatility and supported broader adoption of peripheral nerve stimulation as a minimally invasive therapy for pain management.
-
In September 2022, Checkpoint Surgical launched the CHECKPOINT GEMINI Bipolar Nerve Stimulator, designed to deliver precise stimulation for intraoperative nerve identification and protection. This innovation strengthened the company’s surgical solutions portfolio and supported safer, more effective peripheral nerve procedures.
-
In May 2022, SPR announced the completion of its 10,000th SPRINT PNS procedure for patients suffering from life-altering pain. This milestone highlighted the growing adoption of the SPRINT peripheral nerve stimulation system as a minimally invasive, non-opioid treatment option for chronic and acute pain management.
-
In March 2022, Bioventus received FDA 510(k) clearance for its StimRouter Pain Management Device. This milestone expanded the company’s neuromodulation portfolio and strengthened its position in the peripheral nerve stimulation market by offering patients a minimally invasive solution for chronic pain management.
-
In October 2021, SPR received FDA clearance for an expanded indication of the SPRINT PNS System, enabling its use for the treatment of pain in areas of the head, neck, and torso. This clearance significantly broadened the clinical utility of the system and reinforced its role as a versatile, minimally invasive solution for pain management.
Peripheral Nerve Stimulators Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 297.64 million
Revenue forecast in 2033
USD 680.30 million
Growth rate
CAGR of 10.89% from 2025 to 2033
Base year for estimation
2024
Actual data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, modality, connectivity, application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; and Middle East & Africa (MEA)
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; South Korea; Australia; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
B. Braun SE;Nalu Medical, Inc.; Curonix LLC.; SPR; Vygon; AirLife; Neuspera Medical; AVNS; Mercury Medical; Medilogix; Anesthesia Associates, Inc.; Medtronic; Boston Scientific Corporation; Abbott; Bioventus; Checkpoint Surgical Inc.; NEURIMPULSE s.r.l.; Nervonik, Inc; Epineuron Technologies Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Peripheral Nerve Stimulators Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global peripheral nerve stimulators market report based on product, modality, connectivity, application, end use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Systems
-
Implantable
-
Non-implantable
-
-
Handheld
-
Others
-
Wearables/Accessories
-
-
Modality Outlook (Revenue, USD Million, 2021 - 2033)
-
Rechargeable
-
Non-rechargeable
-
-
Connectivity Outlook (Revenue, USD Million, 2021 - 2033)
-
Wired
-
Wireless
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Diabetic Peripheral Neuropathy Pain
-
Chronic Musculoskeletal Pain
-
Complex Regional Pain Syndrome (CRPS)
-
Headache and migraine disorders
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals and Clinics
-
Ambulatory Surgical Centers (ASCs)
-
Physiotherapy/Rehabilitation Centers
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global peripheral nerve stimulators market size was estimated at USD 271.79 million in 2024 and is projected to reach USD 297.64 million in 2025.
b. The global peripheral nerve stimulators market is expected to grow at a CAGR of 10.89% from 2025 to 2033.
b. The wireless segment accounted for the largest revenue share in peripheral nerve stimualators market in 2024 driven by its ease of use, minimally invasive nature, and improved patient comfort compared to traditional wired systems.
b. The key companies include B. Braun SE, Nalu Medical, Inc., Curonix LLC., SPR, Vygon, AirLife, Neuspera Medical, AVNS, Mercury Medical, Medilogix, Anesthesia Associates, Inc., Medtronic, Boston Scientific Corporation, Abbott, Bioventus, Checkpoint Surgical Inc., NEURIMPULSE s.r.l., Nervonik, Inc, and Epineuron Technologies Inc.
b. The peripheral nerve stimulators market is primarily driven by growing demand for minimally invasive, non-opioid pain management solutions, and rising prevalence of chronic and neuropathic pain conditions.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










