- Home
- »
- Clinical Diagnostics
- »
-
Polymerase Chain Reaction Market, Industry Report, 2033GVR Report cover
![Polymerase Chain Reaction Market Size, Share & Trends Report]()
Polymerase Chain Reaction Market (2026 - 2033) Size, Share & Trends Analysis Report By Type (Conventional PCR, Others), By Product (Consumables & Reagents, Instruments), By Application (Medical & Non-Medical), By Region, And Segment Forecasts
- Report ID: GVR-4-68039-188-0
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Polymerase Chain Reaction Market Summary
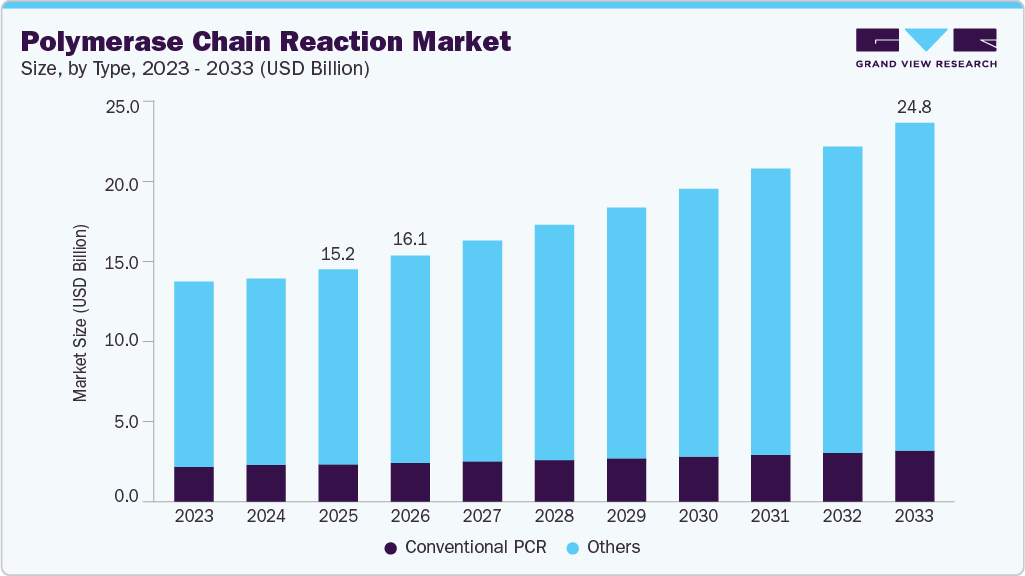
The global polymerase chain reaction market size was estimated at USD 15.19 billion in 2025 and is projected to reach USD 24.78 billion by 2033, growing at a CAGR of 6.36% from 2026 to 2033. The market growth is driven by the increasing demand for rapid and accurate molecular diagnostics across clinical, research, and forensic applications.
Key Market Trends & Insights
- North America dominated the global polymerase chain reaction market with the largest revenue share of 44.06% in 2025.
- The polymerase chain reaction industry in the U.S. accounted for the largest market revenue share in North America in 2025.
- By type, the others segment, including dPCR, qPCR, and other novel technologies, led the market with the largest revenue share of 83.92% in 2025.
- By application, the non-medical application segment is anticipated to expand at the fastest CAGR of 8.85% over the forecast period.
- By product, the consumables and reagents segment led the market with the largest revenue share of 74.07% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 15.19 Billion
- 2033 Projected Market Size: USD 24.78 Billion
- CAGR (2026-2033): 6.36%
- North America: Largest market in 2025
PCR technology enables the amplification of small segments of DNA or RNA, allowing for precise detection of pathogens, genetic mutations, and biomarkers.The growing prevalence of infectious diseases, genetic disorders, and cancer has accelerated the adoption of PCR-based testing in hospitals, diagnostic laboratories, and research institutes. In addition, the expansion of molecular diagnostics and personalized medicine has strengthened the role of PCR technologies in modern healthcare systems.However, some of the key factors, such as the rising application of polymerase chain reaction (PCR) in research and forensic laboratories and increasing demand for advanced diagnostics, are anticipated to increase the demand for polymerase chain reaction tests. In addition, the launch of novel technologies and the increasing number of contract research organizations are likely to drive demand for PCR tests forward during the forecast period.
The increase in the prevalence of chronic and infectious diseases and genetic disorders is expected to boost the demand during the forecast period. According to the CDC, chronic diseases such as cancer and diabetes, and chronic kidney and respiratory diseases such as asthma, are responsible for 7 in 10 deaths in the U.S. each year. Furthermore, the number of patients suffering from congenital heart disease is increasing, requiring medical professionals to look for molecular diagnostic tools for accurate diagnoses. According to the CDC, congenital heart defects are the most common birth defects in the U.S., affecting approximately 1% of births every year. Thus, the increasing prevalence of diseases is increasing the demand for novel molecular diagnostic tests during the forecast period.
The rising demand for prenatal genetic testing procedures owing to growing awareness regarding genetic disorders, such as cystic fibrosis, hemophilia, and thalassemia, is expected to propel the industry growth. In the early stages of pregnancy, fetal DNA present in the mother’s blood or the amniotic fluid can be analyzed to detect genetic defects before birth using qPCR and dPCR techniques. Possible genetic aberrations and fetuses with defective genes can then be treated during early pregnancy.
Technological advancements such as real-time PCR (qPCR), digital PCR (dPCR), and multiplex PCR have improved sensitivity, specificity, and throughput in diagnostic testing. The COVID-19 pandemic significantly boosted global PCR testing infrastructure, leading to increased investments in automated systems and high-throughput platforms. Major companies such as Thermo Fisher Scientific, Bio-Rad Laboratories, QIAGEN, F. Hoffmann-La Roche, and Agilent Technologies continue to develop advanced PCR instruments, reagents, and consumables to support clinical diagnostics, pharmaceutical research, and biotechnology applications. The growing focus on early disease detection, expanding genomic research, and increasing government funding for molecular biology research are expected to further drive market growth.
Market Concentration & Characteristics
Innovation in the polymerase chain reaction industry remains high, driven by continuous advancements in molecular diagnostics, automation, and high-throughput testing technologies. Newer technologies such as real-time PCR (qPCR), digital PCR (dPCR), and multiplex PCR have made detection more sensitive, improved accuracy, and made workflows more efficient compared to older PCR methods. Major companies such as Thermo Fisher Scientific, Bio-Rad Laboratories, QIAGEN, F. Hoffmann-La Roche, and Agilent Technologies are investing in better reagents, integrated instruments, and automated sample preparation systems. The use of artificial intelligence, microfluidics, and portable PCR devices is helping deliver faster diagnostic results and making point-of-care molecular testing more common in clinics, labs, and fieldwork.
Currently, mergers and acquisitions in the polymerase chain reaction industry are at moderate levels. Most companies are focusing on partnerships, licensing deals, and joint research to build up their molecular diagnostics offerings. Instead of big mergers, the market often sees PCR technology developers, biotech companies, and diagnostic labs working together to bring new testing solutions to market faster. For instance, manufacturers often team up with pharmaceutical companies and research institutions to help develop companion diagnostics, support genomic research, and run infectious disease surveillance programs. These partnerships help companies reach more regions and make PCR-based testing more accessible, all without incurring large, expensive acquisitions.
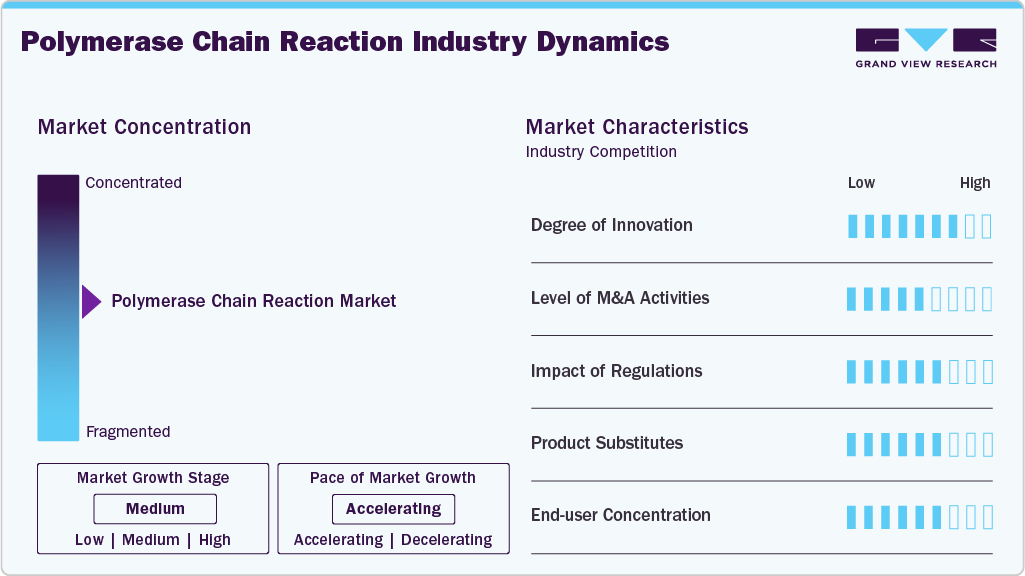
Regulations have a big impact on the polymerase chain reaction industry, especially for clinical diagnostic uses. PCR diagnostic kits and instruments must meet strict regulations set by agencies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other national regulators. Getting approval usually means conducting extensive clinical testing to demonstrate that the products are accurate, reliable, and consistent. These rules help keep products safe and high-quality, but they can also make development take longer and raise costs for manufacturers. In emerging economies, regulations for molecular diagnostics are still developing. This can slow market entry or limit the widespread adoption of advanced PCR systems.
There is only moderate substitution in the polymerase chain reaction industry, as PCR remains one of the most reliable and widely used methods for amplifying nucleic acids and detecting pathogens. Although new molecular technologies such as next-generation sequencing (NGS), loop-mediated isothermal amplification (LAMP), and CRISPR-based diagnostics are emerging, PCR remains the standard because it is accurate, cost-effective, and widely supported by labs. Within PCR, labs are moving from traditional systems to real-time and digital PCR platforms, which offer faster processing, greater sensitivity, and better quantification.
Type Insights
The other segment, including dPCR, qPCR, and other novel technologies, led the market with the largest revenue share of 83.92% in 2025 and is projected to grow at the fastest CAGR during the forecast period. The introduction of technologically advanced PCR tests, increasing prevalence of targeted diseases, and the expansion of existing technologies are expected to drive the segment over the forecast period. In October 2024, Luminex Molecular Diagnostics offered its NxTAG Respiratory Pathogen Panel + SARS-CoV-2, a multi-analyte RT-PCR-based diagnostic test, reinforcing its presence in the PCR diagnostics market. Moreover, novel PCR has wide applications in the quantification of gene expression, pathogen detection, copy number variation, microarray verification, viral quantification, SNP genotyping, and microRNA analysis.
The conventional PCR segment is anticipated to witness at a significant CAGR of 4.02% during the forecast period. The growth of conventional PCR is augmented by the rising application of conventional PCR in food testing and safety, and a surge in product launches. For instance, in December 2025, Roche received CE Mark approval for its cobas BV/CV PCR assay, enabling accurate, single-sample detection of bacterial vaginosis and candida vaginitis, thereby improving diagnostic accuracy and streamlining sexual health testing workflows.Roche also rolled out the next‑generation cobas 6800/8800 systems and software in the U.S., expanding menu capacity and lab efficiency for high‑volume PCR‑based infectious‑disease testing.
Product Insights
The consumables and reagents segment led the market with the largest revenue share of 74.07% in 2025. The growth of the segment is attributed to the increased burden of infectious and chronic diseases, rising demand for consumables and reagents in various types of diagnostic assays, and a rise in product launches. For instance, In March 2025, Visby Medical, Inc. secured authorization for its Visby Medical Respiratory Health Test, a rapid PCR-based multi-analyte diagnostic designed for use by healthcare professionals and patients.
The instruments segment is anticipated to grow at a signficant CAGR during the forecast period, owing to the high penetration of PCR instruments in central laboratories and the increasing introduction of technologically advanced instruments in recent years. In February 2025, Bio-Rad Laboratories announced a binding offer to acquire Stilla Technologies, aiming to strengthen its digital PCR portfolio and accelerate the development of next-generation high-throughput molecular diagnostic solutions.The software and services segment is anticipated to register the fastest growth rate during the forecast period owing to the rising strategic collaborations for the development and advancement of software.
Application Insights
The medical segment led the market with the largest revenue share of 72.78% in 2025, owing to the high adoption rate of PCR tests for clinical diagnostics and research activities to study diseases and drug development. Moreover, the growing application of PCR in the detection of SARS-CoV-2 infections and drug development and research activities is increasing the demand for tests. Furthermore, high precision of testing methods, increasing commercialization of qPCR and dPCR reagents for diagnostics, and increasing applications for disease diagnosis, monitoring, and treatment are fueling the market growth. For instance, in September 2024, Roche launched its TAGS-based cobas Respiratory flex test, a novel PCR-based technology enabling high-throughput, multi-target detection of up to 15 respiratory pathogens from a single sample, significantly improving diagnostic efficiency in clinical laboratories.
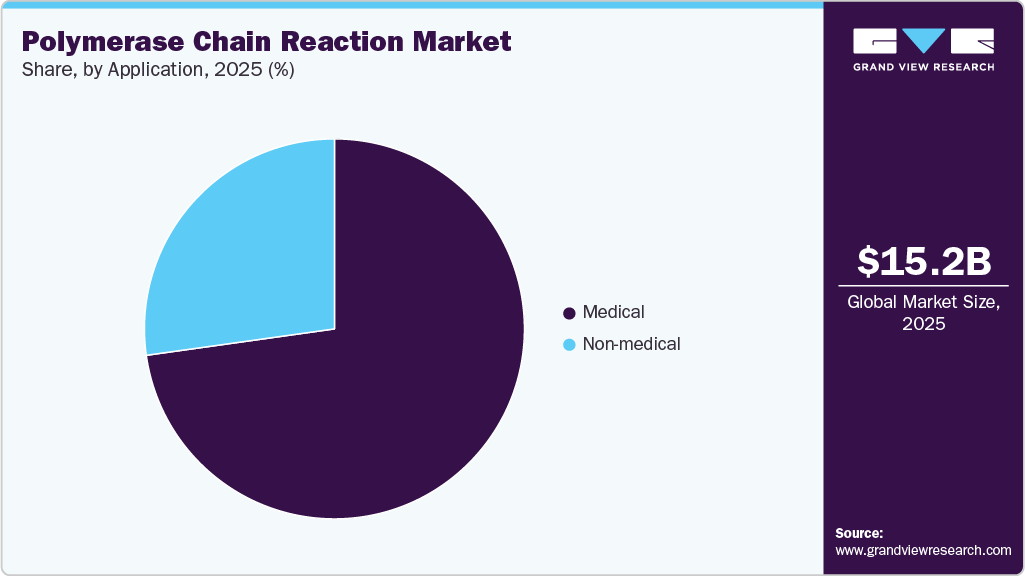
The non-medical application segment is anticipated to expand at the fastest CAGR of 8.85% over the forecast period. Increasing adoption of PCR techniques to test food and other consumables is expected to drive the segment during the forecast period. The food industry is adopting novel and reliable tests to reduce the risk of contamination and enhance food safety. The companies usually offer their product through different distributers in different countries to reach a wider customer base. For instance,Labindia Instruments has partnered with Bio-Rad Laboratories to distribute and promote their food-safety testing products in India, including the iQ-Check PCR Automation system and various PCR kits for pathogen detection. This partnership leverages Bio-Rad’s advanced real-time PCR technology for food safety applications.
Regional Insights
North America dominated the global polymerase chain reaction market with the largest revenue share of 44.06% in 2025. North America leads the polymerase chain reaction (PCR) market owing to its strong healthcare system, many top biotechnology companies, and widespread use of advanced molecular diagnostic tools. PCR is widely used in the U.S. for clinical diagnostics, infectious disease testing, cancer research, and genetic screening. Major companies offer a wide range of PCR instruments, reagents, and automated systems. For instance, many hospitals and labs use Cepheid’s GeneXpert systems for rapid, on-site PCR tests for diseases such as COVID-19, tuberculosis, and influenza. Advanced reference labs such as Quest Diagnostics and LabCorp also boost North America’s testing capacity and support the use of high-throughput PCR methods.
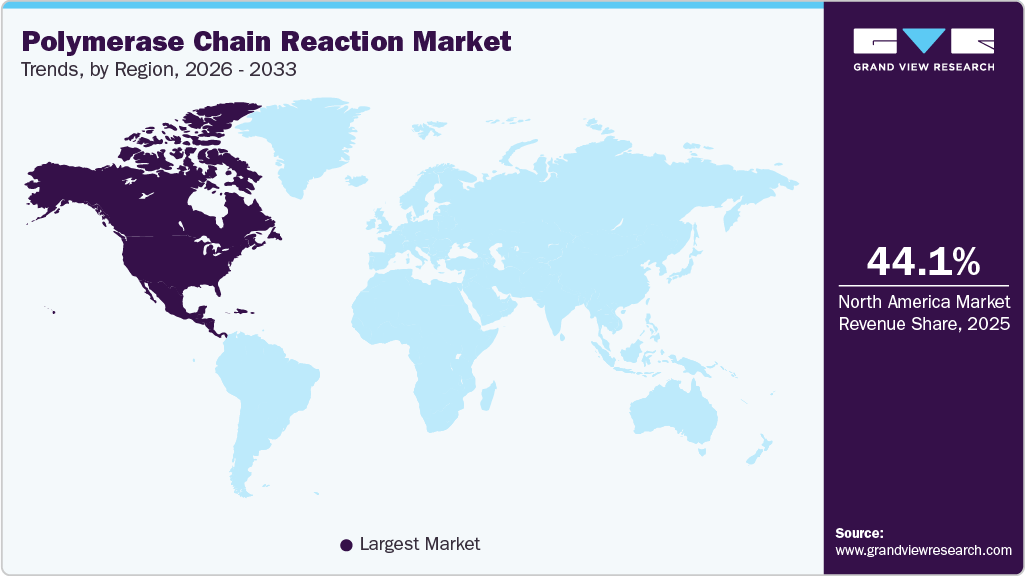
The region benefits from strong government support, research funding, and updated regulations that help bring PCR technologies into healthcare and life sciences. Organizations such as the National Institutes of Health (NIH) and the Centers for Disease Control and Prevention (CDC) fund genomic research and disease-monitoring programs, where PCR is key for early detection and tracking. The COVID-19 pandemic led to major investments in PCR infrastructure, expanding automated and high-throughput testing systems throughout North America. Digital PCR (dPCR) is increasingly common in cancer research and precision medicine. Companies now offer advanced tools for detecting rare mutations and performing quantitative analysis.
U.S. Polymerase Chain Reaction Market Trends
The polymerase chain reaction market in the U.S. accounted for the largest market revenue share in North America in 2025. This is mainly because PCR is widely used in clinical diagnostics, life sciences research, and biopharmaceutical development. The U.S. has a highly advanced healthcare system and is home to major industry leaders, including Thermo Fisher Scientific, Bio-Rad Laboratories, Danaher Corporation (Cepheid), Agilent Technologies, and F. Hoffmann-La Roche. Hospitals and diagnostic labs in the U.S. use PCR to detect infectious diseases, perform cancer testing, screen for genetic disorders, and analyze blood. Platforms such as Cepheid’s GeneXpert and Roche’s cobas PCR systems enable fast, high-volume testing. Large reference labs have greatly increased their PCR testing, especially for respiratory infections, sexually transmitted diseases, and molecular screening panels.
Strong government support, clear regulations, and ongoing investment in research and innovation make the U.S. market even stronger. Groups like the National Institutes of Health (NIH), Centers for Disease Control and Prevention (CDC), and Biomedical Advanced Research and Development Authority (BARDA) are key funders of molecular diagnostics and public health programs that depend on PCR technologies. There is also a growing focus on precision medicine and cancer research, leading to greater use of advanced methods such as digital PCR (dPCR) to detect rare mutations and analyze biomarkers. Outside of healthcare, PCR is widely used in forensic science, food safety, agricultural biotechnology, and environmental monitoring. This helps keep demand strong and supports ongoing advances in the U.S. market.
Europe Polymerase Chain Reaction Market Trends
The polymerase chain reaction market in Europe has a technologically advanced and highly regulated market for Polymerase Chain Reaction (PCR). Molecular diagnostics are widely used in everyday clinical practice and public health systems across the region. Germany, the UK, France, and Italy lead the way, owing to strong laboratory networks and universal healthcare systems that make PCR testing widely accessible. Major companies such as QIAGEN (Germany), F. Hoffmann-La Roche (Switzerland), and bioMérieux (France) are based in Europe. They develop and sell PCR kits, reagents, and automated platforms for infectious disease diagnostics and genetic analysis. PCR is widely used in European hospitals and central laboratories for respiratory disease panels, antimicrobial resistance detection, and prenatal genetic screening. Multiplex assays are increasingly used to improve diagnostic efficiency.
Strict regulations and quality standards, especially the In Vitro Diagnostic Regulation (IVDR), shape the European market. These rules focus on clinical evidence, traceability, and the reliability of products. These regulations improve test accuracy and patient safety, but they also make compliance more demanding for manufacturers. This affects how companies develop products and enter the market. Europe is also seeing greater use of decentralized, near-patient PCR solutions, especially in outpatient clinics and smaller labs seeking faster results. Public health agencies and private companies often work together, especially in disease surveillance and pandemic preparedness. More investment in genomics and cancer research, backed by regional funding, is helping advanced PCR techniques like real-time and digital PCR become more common.
Asia-Pacific Polymerase Chain Reaction Market Trends
The polymerase chain reaction market in the Asia Pacific is anticipated to grow at the fastest CAGR of 7.15% over the forecast period. This growth is fueled by improved healthcare infrastructure, greater investment in molecular diagnostics, and a higher burden of infectious and chronic diseases. China, India, Japan, and South Korea are major contributors to this growth. These countries have large patient populations and better access to diagnostic services. For instance, China performs millions of PCR tests each year. This number grew during and after the COVID-19 pandemic, as the country expanded its network of molecular testing labs. India also has more than 3,000 PCR labs in both public and private sectors. These labs support large-scale testing for diseases like tuberculosis, COVID-19, and viral hepatitis. Companies such as BGI Genomics, Sansure Biotech, Thermo Fisher Scientific, and QIAGEN are expanding their presence in the region by establishing local manufacturing facilities, forming partnerships, and building distribution networks.
More people in the region are using PCR technologies for various purposes, including clinical diagnostics, agricultural biotechnology, food safety testing, and academic research. Government programs are important for market growth. For instance, national disease control efforts in India and China depend on PCR for early detection and tracking of infectious diseases. Japan and South Korea have advanced healthcare systems that are helping to increase the use of real-time and digital PCR platforms, especially in cancer care and precision medicine. There is also a growing effort to improve diagnostic capabilities in Southeast Asia. This is increasing demand for portable, affordable PCR solutions that can be used outside major hospitals. Regulatory rules vary by country, but there are ongoing efforts to standardize diagnostics and increase healthcare spending. In several emerging economies, healthcare spending in Asia-Pacific is expected to grow by more than 6 to 8 percent each year. These trends are likely to help PCR technologies become more widely used in the region.
Latin America Polymerase Chain Reaction Market Trends
The polymerase chain reaction market in Latin America is experiencing steady growth, supported by improving healthcare infrastructure, rising awareness of molecular diagnostics, and increasing government focus on infectious disease control. Public‑health programs in Brazil, Mexico, and Argentina have scaled real‑time PCR systems in public hospitals and reference labs for HIV, TB, influenza, and other infectious diseases. Companies such as F. Hoffmann-La Roche, QIAGEN, Thermo Fisher Scientific, and bioMérieux are actively operating in the region through distribution partnerships and localized support.
Middle East and Africa Polymerase Chain Reaction Market Trends
The polymerase chain reaction market in the Middle East & Africa (MEA) presents emerging opportunities in the PCR industry, driven by increasing investments in healthcare infrastructure and growing demand for advanced diagnostic technologies. Countries such as the UAE, Saudi Arabia, and South Africa are leading adopters, with PCR widely utilized in infectious disease diagnostics, including HIV, tuberculosis, and respiratory infections, as well as in hospital and research laboratory settings. Government initiatives aimed at strengthening disease surveillance systems and pandemic preparedness are encouraging the adoption of PCR technologies, often through partnerships with international organizations and diagnostic companies.
Key Polymerase Chain Reaction Company Insights
Some of the leading players operating in the polymerase chain reaction industry include QIAGEN, Thermo Fisher Scientific Inc., Danaher, and Abbott. Companies focus on capturing the market by increasing their presence using various business initiatives, such as partnerships & collaborations with government. Moreover, these companies have well-established product portfolios, which help them capture major market share.
Stilla, Standard BioTools, and Microsynth AG are some of the emerging market participants in the polymerase chain reaction industry. Developing & launching new and improved diagnostic tools that offer faster results, higher accuracy, and easier usability are prevalent operating strategies for these companies. The players may face challenges in penetrating the market due to competition and regulatory hurdles.
Key Polymerase Chain Reaction Companies:
The following key companies have been profiled for this study on the polymerase chain reaction market.
- Thermo Fisher Scientific Inc.
- Danaher
- Abbott
- Bio-Rad Laboratories, Inc.
- Agilent Technologies, Inc.
- QIAGEN
- F. Hoffmann-La Roche Ltd.
- Stilla
- Standard BioTools
- Microsynth AG
Recent Development
-
In December 2025, Roche received CE Mark approval for its cobas BV/CV PCR assay, enabling accurate, single-sample detection of bacterial vaginosis and candida vaginitis, thereby improving diagnostic accuracy and streamlining sexual health testing workflows.
-
In March 2025, Visby Medical, Inc. secured authorization for its Visby Medical Respiratory Health Test, a rapid PCR-based multi-analyte diagnostic designed for use by healthcare professionals and patients.
-
In February 2025, Bio-Rad Laboratories announced a binding offer to acquire Stilla Technologies, aiming to strengthen its digital PCR portfolio and accelerate the development of next-generation high-throughput molecular diagnostic solutions.
Polymerase Chain Reaction Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 16.10 billion
Revenue forecast in 2033
USD 24.78 billion
Growth rate
CAGR 6.36% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, Volume in thousands and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, product, application, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Italy; Spain; China; Japan; India; Australia; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE
Key companies profiled
Thermo Fisher Scientific Inc.; Danaher; Abbott; Bio-Rad Laboratories, Inc.; Agilent Technologies, Inc.; QIAGEN; F. Hoffmann-La Roche Ltd.; Stilla; Standard BioTools; Microsynth AG
Customization scope
Free report customization (equivalent to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Global Polymerase Chain Reaction Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the global polymerase chain reaction market report based on the type, product, application, and region:
-
Type Method Outlook (Volume, Thousands; Revenue, USD Million, 2021 - 2033)
-
Conventional PCR
-
Others
-
-
Product Outlook (Volume, Thousands; Revenue, USD Million, 2021 - 2033)
-
Consumables & Reagents
-
Instruments
-
Software & Services
-
-
Application Outlook (Volume, Thousands; Revenue, USD Million, 2021 - 2033)
-
Medical
-
Clinical
-
Pathology Testing
-
Oncology Testing
-
Blood Screening
-
Others
-
-
Research
-
Forensics and Others
-
-
Non-medical
-
Food
-
Others
-
-
-
Regional Outlook (Volume, Thousands; Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
-
Frequently Asked Questions About This Report
The global polymerase chain reaction market is expected to witness a compound annual growth rate of 6.36% from 2026 to 2033 to reach USD 24.78 billion by 2033.
Increasing demand for advanced diagnostic techniques; rising number of CROs, forensic & research laboratories, prenatal genetic testing procedures, and the launch of novel technologies are expected to increase the demand for PCR tests over the forecast period.
Consumables & reagents held the largest polymerase chain reaction market share of 74.07% in 2025 due to increased burden of infectious and chronic diseases, rising demand for consumables and reagents in various types of diagnostic assays, and a rise in product launches.
Some key players operating in the polymerase chain reaction market include Thermo Fisher Scientific Inc., Danaher, Abbott, Bio-Rad Laboratories, Inc., Agilent Technologies, Inc., QIAGEN, F. Hoffmann-La Roche Ltd., Stilla, Standard BioTools, and Microsynth AG
The global polymerase chain reaction market size was estimated at USD 15.19 billion in 2025 and is expected to reach USD 16.10 billion in 2026.
About the authors:
Author: GVR Clinical Diagnostics Research Team | Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









