- Home
- »
- Pharmaceuticals
- »
-
Preclinical CRO Market Size & Share, Industry Report, 2033GVR Report cover
![Preclinical CRO Market Size, Share & Trends Report]()
Preclinical CRO Market (2026 - 2033) Size, Share & Trends Analysis Report By Service (Bioanalysis And DMPK studies, Toxicology Testing, Compound Management), By Development Stage, By Indication, By Model, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-2-68038-035-4
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
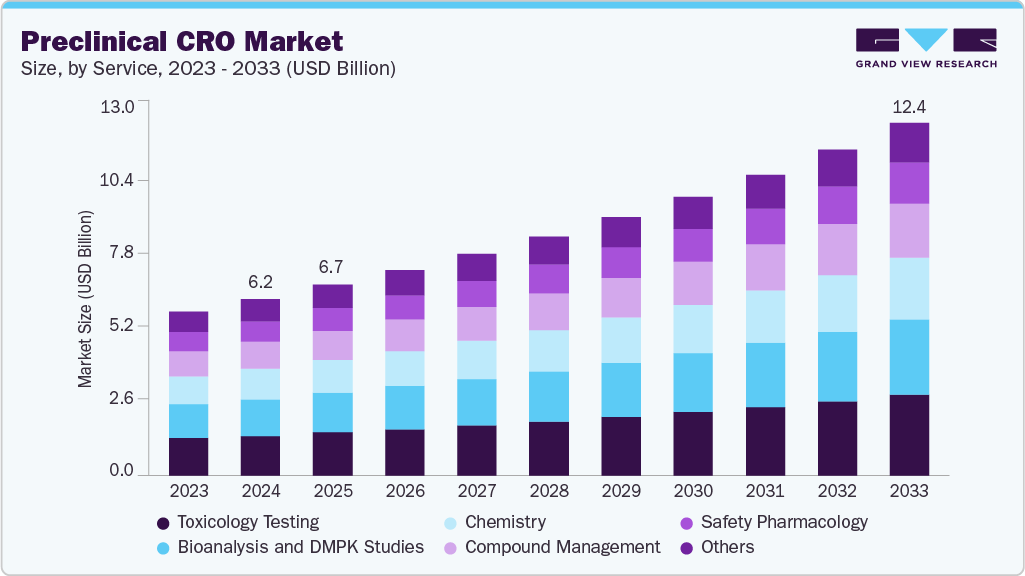
Market Size, 2025$6.7BMarket Estimate, 2026$7.3BMarket Forecast, 2033$12.8BCAGR, 2026 - 20338.4%Preclinical CRO Market Summary
The global preclinical CRO market size was estimated at USD 6.7 billion in 2025 and is projected to reach USD 12.8 billion by 2033, growing at a CAGR of 8.4% from 2026 to 2033. The market growth is driven by economic outsourcing, regulatory mandates, and scientific complexities, which, in turn, make preclinical CROs a critical component of the early-stage drug development process.

Key Market Trends & Insights
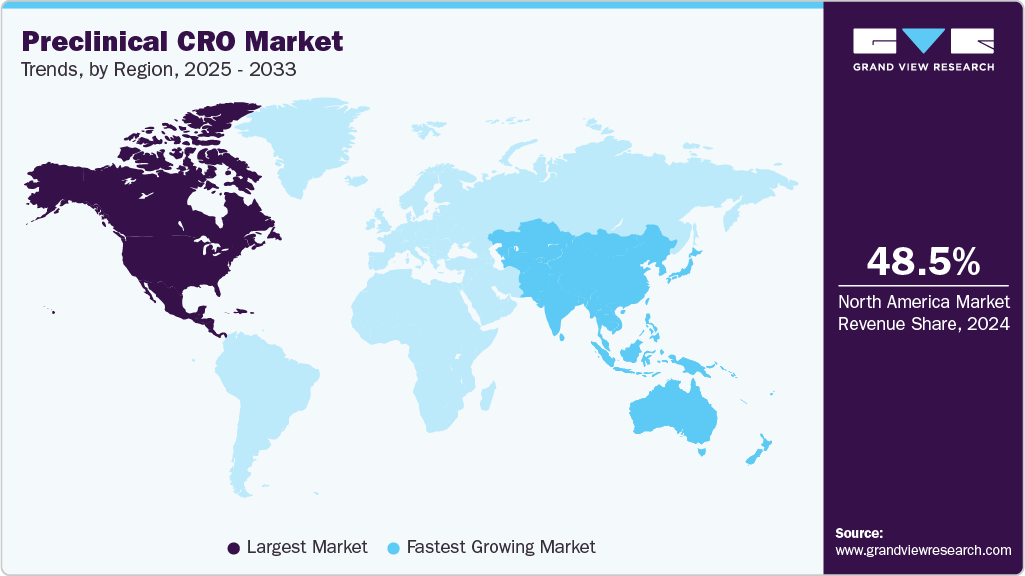
- The North America preclinical CRO market held the largest share of 48.1% of the global market in 2025.
- The preclinical CRO market in the U.S. is expected to grow significantly over the forecast period.
- Based on the development stage, the preclinical optimization, efficacy & PK/PD segment held the largest market share in 2025.
- Based on service, the toxicology testing segment is anticipated to dominate the preclinical CRO market.
- Based on the indication, the oncology segment held the largest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 6.7 Billion
- 2033 Projected Market Size: USD 12.8 Billion
- CAGR (2026-2033): 8.4%
- North America: Largest market in 2025
- Asia Pacific: Fastest-growing region
Besides, the rising demand for toxicology, pharmacology, and efficacy studies is expected to drive market growth. The outsourcing trends of preclinical research, particularly among biotech and biopharma companies seeking to minimize fixed infrastructure costs, shorten development timelines, and access specialized expertise, are expected to significantly contribute to future growth. Moreover, increasing complexity in drug development, higher biopharma R&D investments, and stringent regulations requiring Good Laboratory Practice (GLP)-compliant studies are influencing the market growth. Thus, these factors are driving manufacturers to rely more on CROs to optimize costs and reduce operational burdens, thereby fueling market growth.The market is anticipated to grow owing to growing R&D offshoring and the rising outsourcing trend, increasing demand for personalized medicine and advanced therapeutics, increasing pharmaceutical R&D investments, and the integration of translational biomarkers into preclinical studies, among others.
The trend of R&D offshoring is attributed to structural shifts in the pharmaceutical and biotechnology industries. Since the R&D costs for developing new drugs are high, coupled with longer discovery timelines and increasing complexity, the manufacturers are relying on outsourcing to CROs at cost-effective locations for conducting preclinical studies. This R&D offshoring allows companies to leverage the availability of highly skilled resources, advanced infrastructure, and laboratory capacity at significantly lower cost than in the U.S. and Europe, especially for smaller and mid-sized biopharma companies without internal capabilities, and for large pharma to improve capital efficiency.
The developing markets, including India and China, have emerged as major centers with GLP-compliant facilities, integrated service offerings, and cost advantages of 30-60% over other locations. The CROs in these countries offer comprehensive capabilities, including discovery, toxicology, DMPK, and in vivo studies, enabling faster, more flexible studies. Moreover, the development of hybrid R&D operations, including both onshore strategy and offshore execution, has further strengthened this trend.
In addition, strategic collaborations and initiatives undertaken by the companies are further bolstering the outsourcing growth. For instance, in February 2026, Syngene International signed a partnership agreement with Johns Hopkins University, leveraging discovery and preclinical capabilities to advance early-stage research into clinical candidates. Similarly, in January 2026, Syngene International extended its existing collaboration with Bristol Myers Squibb through 2035, providing a dedicated R&D extension including discovery, preclinical, and manufacturing capabilities. Such initiatives are likely to propel the market growth over the forecast period.
Furthermore, the demand for personalized medicine and advanced therapeutics is driving the growth of preclinical CROs, driven by factors such as the rising need for new drug discoveries to combat the increasing incidence of chronic illnesses worldwide, as well as companies' strategic initiatives and technology integration. For instance, in March 2026, NJ Bio and Ajinomoto Bio-Pharma Services partnered to enhance NJ Bio's capabilities in antibody-drug conjugate development by integrating the AJICAP technology into its workflow. This will allow modification of the antibody at a site-specific level, thereby improving the stability, efficacy, and pharmacokinetics of the antibody-drug conjugate.
Similarly, in March 2026, Eli Lilly and Insilico Medicine partnered by signing an agreement valued at up to USD 2.75 billion to enhance drug discovery using artificial intelligence technology. The partnership agreement grants Eli Lilly exclusive global rights to the development and commercialization of selected oral drug candidates currently in preclinical development. In return, Insilico Medicine will receive royalty payments and leverage its AI technology to discover new disease targets and speed up its pipeline. Thus, an increase in the demand for advanced therapeutics and personalized medicines is likely to impel the market’s growth over the coming years.
Opportunity Analysis
The market offers significant growth opportunities within the CROs market for preclinical services, driven by the increasing complexity of drug development pipelines, which necessitate outsourcing toxicology, bioanalysis, and pharmacokinetics, among other capabilities. The pharmaceutical and biotechnology companies are actively working to reduce their fixed costs to allocate resources more efficiently, thereby expanding the target market for CRO providers.
In addition, the expanding role of biological agents and the emergence of new therapeutic areas, such as cell and gene therapy, require increasingly complex in vivo testing and analysis, necessitating specialized niche capabilities from CROs. Moreover, the growing importance of regulatory agencies in validating the quality and safety of data is a driving factor compelling companies to rely on CRO partners that consistently comply with regulatory guidelines. In addition, small and medium biotechnology organizations have become the industry's major customers due to their need to simultaneously develop multiple compounds and to rely on CRO networks for support.
Furthermore, geographic expansion into cost-effective regions like the Asia Pacific ensures scalability without compromising quality standards and supports CROs in a larger pool of clients. Moreover, increased investments in rare disease and oncology research have led to more specialized preclinical studies that may require customized procedures and greater technical involvement, thereby improving revenues. In addition, growing demand for end-to-end service offerings from target discovery through IND-enabling studies enables CROs to act as partners, helping them secure bigger contracts and ensure better client retention rates. Besides, developments in biomarker and imaging technologies offer CROs opportunities to deliver premium, high-value services, helping them improve their margins. Finally, regulatory convergence across geographies makes it easier to conduct multicenter trials, thereby favoring greater outsourcing and ensuring greater engagement for CROs.
Impact of U.S. Tariffs on the Global Preclinical CRO Market
The impact of U.S. tariffs on laboratory instruments, reagents, and biological materials raises input costs for pre-clinical CROs, thereby increasing outsourcing costs. Thus, rising costs prompt pharmaceutical and biotechnological firms to reconsider their strategies, shifting their share of demand to U.S. CROs despite higher base prices. In addition, international CROs face increased pressure due to higher import costs, necessitating cost-cutting measures through procurement from multiple suppliers and local sources. Moreover, China-based imports are subject to additional tariffs, which are affecting the timeline for the launch of the toxicology and bioanalysis study. In addition, CROs may opt to develop their infrastructure in the Asia Pacific region, helping decentralize the process of conducting tests.
Moreover, sponsors prefer CROs that can handle studies across multiple regions, enabling them to assign studies to regions with minimal tariff effects and robust logistics. Furthermore, tariffs indirectly affect capital investment, as high equipment costs delay investments in more sophisticated testing models, such as patient-derived xenograft (PDX) and organoid platforms.
The technological advancements within the preclinical market are changing the research process from study design to advanced human-relevant in vitro testing systems, which enhance disease modeling capabilities and facilitate the early identification of safety and efficacy indicators. The improved data quality from such models will positively impact the data in computational and AI-enabled discovery programs used to identify targets and prioritize compounds. In addition, AI-driven tools can reduce reliance on sequential experimentation, thereby shortening discovery periods. Moreover, automation and high-throughput experimentation enhance processing capabilities, allowing CROs to handle higher volumes of studies while maintaining consistent output quality.

Besides, automation eliminates variations in experiment execution, enhancing replicability and facilitating the regulatory approval of generated data. Moreover, high-throughput systems generate vast amounts of data that can be analyzed using multi-omics and imaging tools, enabling better biological understanding and enhanced decision-making. Furthermore, multi-omics methodologies will correlate genomic, proteomic, and phenotypic information, leading to efficient biomarker identification and aligning well with clinical trials. In addition, high-content imaging tools add a quantitative component to cell analysis, thereby enhancing drug response assessment within biological systems.
Furthermore, there is an ever-increasing need for analytical systems that enable efficient integration of information throughout the discovery and pre-clinical testing processes. In addition, integrating AI, automation, and data platform systems helps eliminate operational fragmentation, thereby facilitating better collaboration among CRO service units. Moreover, sponsors benefit from consolidated workflow processes, which can help eliminate delays when working with multiple vendors. Thus, through the implementation of integrated technology platforms, CROs can increase their scalability, which would enable them to cover a range of different pipelines, including biologic and innovative therapy options.
Pricing Model Analysis
The pricing models for preclinical CRO services differ depending on project scope and the sponsors' and CROs' risk-handling capacity, with fee-for-service arrangements charging per assay or other outputs delivered. This provides transparent cost information and facilitates swift negotiations in routine projects. The fee-for-service model suits standard activities like in vitro testing and ADME. However, changes in scope can raise overall costs and cause costs to vary over time. The time-and-material arrangements and full-time equivalents provide pricing flexibility based on scientific effort, making them suitable for iterative activities such as medicinal chemistry and computation.
PRICING MODEL
DEFINITION
HOW IT WORKS
WHERE IT IS USED
COMMERCIAL ADVANTAGES
COMMERCIAL RISKS
Fee-for-Service (FFS) / Per-Deliverable
Transactional pricing where sponsors pay per study, assay, or deliverable.
CRO provides a rate card; cost is defined per assay/sample/animal. Scope changes add cost.
Standardized in vitro assays, ADME, routine in vivo studies, and formulation screens.
Predictable costs; faster contracting; ideal for routine work.
Scope creep increases costs; CRO absorbs some rework pressure if the requirements are poorly defined.
Time & Materials (T&M) / Full-Time Equivalent (FTE)
Billing based on scientist hours (T&M) or fully dedicated staff (FTE).
Monthly billing for time or dedicated teams; flexible project execution.
Medicinal chemistry, computational chemistry, mechanistic studies, and platform development.
High flexibility; stable utilization for CROs; adequate for iterative discovery.
Budget unpredictability necessitates robust project management to prevent inefficiencies.
Fixed-Price / Milestone-Based
Pre-agreed price for a defined study or phase with milestone payments.
Price set upfront; payments triggered by milestones (initiation, necropsy, report).
GLP toxicology, safety pharmacology, and IND-enabling study packages.
High cost predictability; strong efficiency incentives for CROs.
CRO bears the cost of overruns; a rigid scope requires strict change-order controls.
Bundled / Integrated Program Pricing
Consolidated pricing across multiple sequential services under one program.
One contract covers multi-stage work (chemistry → DMPK → in vivo → toxicity).
End-to-end discovery; IND-enabling programs; multi-species PK/PD studies.
Cost efficiencies, reduced turnaround times, and single-vendor accountability.
Lower flexibility: performance risk concentrated with one CRO.
Capacity Reservation / Retainer
Upfront/periodic payment to reserve lab, animal, or platform capacity.
Sponsors pay a monthly/quarterly fee, guaranteeing access to facilities and staff.
High-demand animal models, GLP tox slots, imaging platforms, specialized vivarium.
Guaranteed scheduling; stable CRO revenue; reduced waiting times.
Inefficient if underutilized; may require CRO to maintain standby capacity.
Outcome-Linked / Risk-Sharing (Emerging)
Payments are tied partly to scientific outcomes or performance metrics.
Hybrid: base fee + success milestone based on predefined criteria.
Early discovery platforms, strategic collaborations, and target validation.
Aligns incentives and lowers upfront costs for biotechs.
Hard to define objective success; high CRO risk if milestones are misaligned.
In addition, fixed-price and milestone models offer predetermined prices tied to specific phases within studies; this is an advantage for better budget planning and aligns perfectly with GLP toxicology and IND enablement services. Similarly, bundled pricing is advantageous in terms of offering an incentive to reduce the costs associated with conducting work by CROs, although cost overruns may affect margins in the case of complex projects. Similarly, pricing that is linked to capacity ensures that the capacity of CROs is utilized effectively, since they charge clients based on periodic payment plans; this allows sponsors to get their work done without delay. Finally, pricing linked to outcomes is beneficial because it encourages performance-based work, aligns the interests of both parties, and minimizes financial burden.
Market Concentration & Characteristics
The preclinical CRO market is at a medium growth stage, with growth accelerating. The market is characterized by the degree of innovation, level of M&A activities, regulatory impact, service expansion, and regional expansion.
Innovation is likely to shape the competitive landscape, as preclinical CROs increasingly use advanced in vitro and in vivo technologies such as 3D cell cultures, organoids, patient-derived xenograft models, and AI-based toxicology assays. They enable CROs to predict human drug reactions more accurately, reduce failures in later stages of development, and accelerate the time to market for new products. In addition, automation and digitization increase the efficiency of testing.
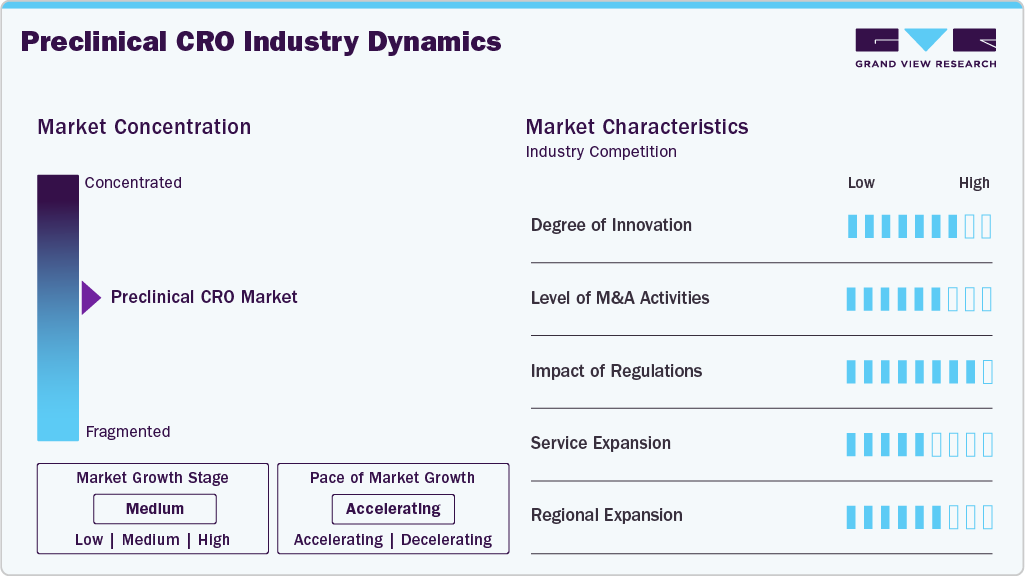
Mergers and acquisitions will contribute to the enhancement of preclinical CROs' service portfolio and geographical coverage. The firms will merge with small companies specializing in toxicology, bioanalysis, and the development of animal models for studying various diseases. Besides, M&A enables firms to gain access to modern research facilities and qualified specialists who can work with complex biologics and gene therapies.
The regulations play an important role in the way preclinical CROs operate. For instance, GLP regulations, animal welfare standards, and data integrity are key regulatory considerations for preclinical CRO service offerings. Furthermore, the application of other international regulations, such as FDA and OECD principles, also plays a critical role in study design, approval process, and data interpretation. The experienced preclinical CROs with regulatory know-how help ensure fast and efficient submission and approval of IND applications.
The preclinical CROs continue to expand their service offerings, ranging from target discovery to IND-enabling activities, including safety, toxicology, DMPK, and bioanalysis. Additionally, increased use of integrated service providers helps address the complexity of managing multiple vendors, thereby offering greater value to clients seeking comprehensive outsourcing solutions for drug development.
The expansion on the regional level helps CROs achieve benefits such as access to lower costs and availability of diverse patient-derived sample material, in addition to improved ability to deliver services globally. Furthermore, rising investment levels in the Asia Pacific region, especially in India and China, help expand capacity and deliver studies more rapidly, as facilities located across several regions enhance CROs' logistics capabilities.
Service Insights
On the basis of service, the toxicology testing segment accounted for the largest revenue share of 25.5% in the preclinical CRO industry in 2025. The segment's growth is driven by increasing demand for comprehensive safety evaluations of drug candidates before entering clinical trials. Moreover, rising regulatory scrutiny from agencies such as the U.S. FDA, EMA, and PMDA has made toxicology studies a mandatory, resource-intensive step in the development pipeline, encompassing acute, sub-chronic, chronic, reproductive, developmental, and genotoxicity assessments.
The bioanalysis and DMPK studies segment is anticipated to grow at the fastest CAGR during the forecast period. The segment growth is driven by the increasing complexity of drug candidates, which require detailed pharmacokinetic, pharmacodynamic, and bioanalytical profiling to meet stringent regulatory requirements. As therapeutic modalities expand into biologics, biosimilars, nucleic acid-based drugs, and antibody-drug conjugates, the need for highly sensitive and specific assays, such as LC-MS/MS, ligand-binding assays, and hybrid approaches, has increased. These studies are critical for determining absorption, distribution, metabolism, and excretion characteristics, as well as understanding potential drug-drug interactions and bioavailability. Outsourcing to preclinical CROs provides access to specialized instrumentation, validated methods, and experienced scientific teams capable of handling complex matrices and low-concentration analytes.
Development Stage Insights
On the basis of the development stage, the preclinical optimization, efficacy & PK/PD segment dominated the market in 2025. The segment growth is driven by the growing demand for translational data to reduce the risks associated with clinical trials. The complexity of drug modalities, including biologics and gene therapies, is increasing, while regulatory bodies are placing greater emphasis on robust dose-selection data.
The bioanalysis & analytics segment is expected to grow significantly over the forecast period, attributed to the growing complexity of drug pipelines, including biologics, biosimilars, and innovative therapies such as gene and cell therapies. Bioanalysis and analytics focus on the quantitative and qualitative evaluation of drugs, metabolites, and biomarkers in biological matrices to facilitate early-stage drug development. This segment encompasses a variety of studies, including LC-MS/MS bioanalysis, ligand-binding assays (LBA), immunogenicity testing, biomarker quantification, product analysis in accordance with ICH guidelines, stability testing, and specialized studies such as metabolite identification and drug-drug interaction (DDI) assessments which play a major role for assessing drug behavior, safety, and efficacy before clinical trials begin. These segments play a major role in generating high-quality, regulatory-compliant data necessary for investigational new drug (IND) submissions.
Indication Insights
On the basis of indication, the oncology segment held the largest share of the preclinical CRO industry in 2025. The growth is driven by rising global cancer incidence, strong investment in targeted therapies and immuno-oncology, and the need for predictive tumor models. The increasing use of patient-derived xenografts, biomarker-driven studies, and combination therapies is accelerating demand for specialized CRO capabilities to improve translational success and reduce clinical trial failure rates.
The rare diseases segment is expected to grow significantly during the forecast period. The growth is driven by rising regulatory incentives, the availability of orphan drug designations, and a surge in investments in gene and cell therapies. Due to the small patient populations, there is a need for effective preclinical models and biomarker strategies. CROs play a crucial role in accelerating development by providing specialized expertise, implementing innovative study designs, and utilizing translational strategies to enhance success rates and reduce time-to-market.
Model Insights
On the basis of the model, the in vivo segment is anticipated to hold the largest market share in 2025, driven by the need for these services to conduct safety, toxicology, and pharmacokinetics research, which is expected to drive higher revenue for this segment. Moreover, due to the high price involvement and longer duration of these studies, usually taking weeks and months, the in vivo segment holds a dominant market share despite having relatively low volumes. This is further supported by the need for an in vivo model in IND-enabling research by the biopharmaceutical industry to develop drugs for clinical trials.
The ex vivo segment is projected to grow rapidly due to its role as a link between in vitro and in vivo testing. This is attributed to their greater relevance to human tissues. Furthermore, the growing emphasis on minimizing the use of animal tests and improving predictability is further contributing to increased usage of ex vivo models in preclinical stages. Furthermore, advancements in technologies such as tissue engineering and organ storage have enabled wider utilization.
End Use Insights
On the basis of end use, the pharmaceutical and biopharmaceutical companies segment dominated the preclinical CRO market in 2025. Pharmaceutical and biopharmaceutical companies are driven by expanding therapeutics requirements and increasing pressure to accelerate drug development while managing costs and complexity. These companies rely heavily on preclinical CROs to conduct essential early-stage research activities, including target validation, lead optimization, pharmacokinetics, toxicology, and safety assessment. As drug pipelines become more diverse and technologically advanced, small molecules, biologics, gene therapies, and cell-based treatments are expected to drive demand for specialized expertise and infrastructure, further encouraging outsourcing to CROs.
Some other factors contributing to market growth include rising R&D expenditure across the pharmaceutical and biotech sectors, coupled with enhanced productivity and reduced drug development costs.
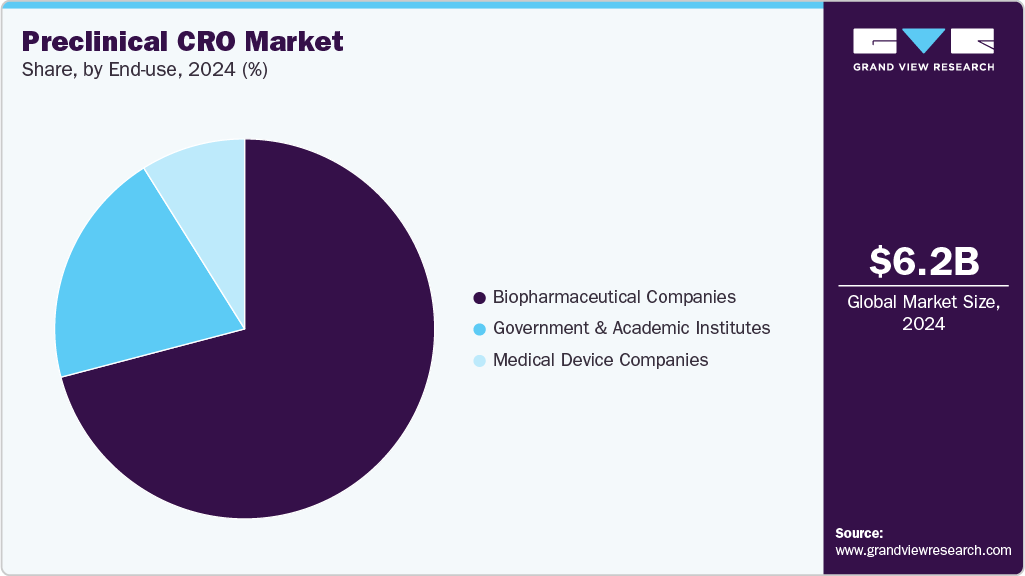
The medical device companies segment is expected to grow significantly during the forecast period. The medical device companies segment is driven by the increasing complexity of device innovation and stringent regulatory requirements. These companies depend on preclinical CROs to conduct essential early-stage evaluations, which include biocompatibility testing, safety assessments, performance validation, and toxicological studies. This ensures that medical devices meet regulatory standards, a crucial factor supporting the progress of clinical trials.
In addition, rapid advances in technologies, such as implantable devices, wearable health monitors, and minimally invasive surgical tools, are expected to drive demand for specialized preclinical expertise. Some other factors contributing to market growth include rigorous global Regulatory Frameworks that require extensive preclinical data on safety, functionality, and risk assessment, which is expected to drive the market. As CROs play a vital role in generating data and supporting companies in streamlining their approval processes and reducing time-to-market.
Regional Insights
The North America preclinical CRO industry dominated the global market in 2025, accounting for 48.1% of revenue. The market growth is driven by established contract research organizations (CROs) and a growing demand for pharmaceutical, biotech, and medical device products. Other factors contributing to segment growth include outsourcing early development phases to lower costs, speeding up timelines, and gaining access to specialized expertise. In addition, the region benefits from a stringent Regulatory Framework that meets FDA standards, advanced infrastructure, and the rising adoption of innovative technologies, including organoid models, humanized mice, and automation-driven laboratories. Besides, the expansion of requirements for biologics and the development of cell & gene therapy pipelines are anticipated to drive the demand for GLP toxicology, bioanalysis, and in vivo pharmacology services. Moreover, established CROs specializing in early drug discovery, such as Charles River Laboratories and LabCorp, are further contributing to regional growth.
U.S. Preclinical CRO Market Trends
The preclinical CRO industry in the U.S. accounted for the largest share in the North America market, due to the expansion of contract research organizations, increased investment in biopharmaceutical R&D, a rising number of clinical trials, coupled with growing requirements for personalized medicine, and strong regulatory oversight by the FDA, which further promotes the high-quality analytical standards. In addition, most CROs in the country offer a range of capabilities, including genotoxicity & immunotoxicity testing services, which contribute to market growth. In addition, rapid growth in emerging areas of medicine, such as personalized medicines, orphan drugs, and biosimilars, is expected to drive the U.S. preclinical outsourcing market.
The Canada preclinical CRO industry is expected to grow at a significant CAGR during the forecast period. The market is driven by strong government support for biomedical research, rising R&D spending by pharmaceutical and biotech companies, and an expanding startup ecosystem focused on novel therapies. The country's advanced research infrastructure, favorable regulations, and increasing demand for specialized services such as toxicology, PK/PD studies, and humanized models further accelerate growth. Collaboration between academia and industry enhances innovation, while the trend toward outsourcing to reduce costs and shorten development timelines boosts CRO engagement. In addition, the country's presence of a scientific workforce and focus on precision medicine contribute to a consistent demand for high-quality preclinical research services.
Europe Preclinical CRO Market Trends
The preclinical CRO industry in Europe benefits from technological advancements, an increasing number of clinical trials, and a rising number of key players offering preclinical CRO services. Besides, the market is driven by major biopharmaceutical companies, such as F. Hoffmann-La Roche Ltd. and Novartis, which are expected to drive market growth over the forecast period. Furthermore, preclinical testing is generally conducted at the central drug development center; outsourcing preclinical services to a local CRO in the region is expected to drive the market over the estimated period.
The UK preclinical CRO industry held the largest share in 2025. The UK market is driven by strong government funding, rising pharmaceutical manufacturing capabilities, stringent regulatory requirements, extensive R&D initiatives, and robust clinical research activities. Further, the growing demand for outsourcing discovery, toxicology, ADME, and PK/PD studies is fueling market growth. Moreover, the country's advanced regulatory environment, emphasis on precision medicine, and innovation in therapeutic areas such as oncology, gene therapy, and AI-driven drug discovery are further contributing to market growth. Moreover, the presence of skilled scientific talent, high-quality laboratory infrastructure, and strong industry-academia collaborations enhances the country's position as a preferred hub for preclinical research services.
The preclinical CRO industry in Germany is expected to grow significantly over the forecast period. The growth is driven by factors such as strong pharmaceutical and biotechnology sectors, advanced R&D infrastructure, highly skilled workforce, and stringent regulatory standards. Besides, the increasing prevalence of chronic diseases has driven a rising demand for biologics, biosimilars, and specialty drugs, further underscoring the need for reliable, compliant bioanalytical testing services. Some other factors contributing to market growth are the increasing number of CROs offering comprehensive services such as LC-MS/MS, immunoassays, pharmacokinetics, and biomarker analysis, further supporting the market growth.
Asia Pacific Preclinical CRO Market Trends
The preclinical CRO industry in the Asia Pacificis expected to grow at a significant CAGR over the forecast period. The region has seen high growth in recent years, primarily driven by cost advantages, which in turn have helped global pharmaceutical and biotechnology companies outsource early-stage drug discovery in regions like China and India. This cost advantage, coupled with the availability of a large scientific workforce, helps in accelerating toxicology, DMPK, and bioanalytical studies, thus reducing the overall development time for drugs. This has made the Asia Pacific region a hub for biologics and biosimilars development, driven by the increasing prevalence of chronic diseases, thereby boosting the number of preclinical studies in the region.
The China preclinical CRO industry isone of the emerging markets driven by rising investments, increasing acquisition activities, low operational costs, adoption of R&D standards, and robust demand for preclinical CRO. Other factors contributing to market growth include skilled professionals, cost efficiency, and the growing pharmaceutical industry. These factors are expected to contribute to the growth of preclinical trials outsourcing in the country. Moreover, numerous market players are focused on technology-based value-added services due to the demand for drug discovery and pre-clinical studies for various diseases. In addition, increased funding in pharmaceutical companies, R&D institutes, and hospitals drives the market growth. For instance, in July 2025, WuXi Biologics launched WuXiHigh 2.0, a next-generation development platform for high-concentration biologics capable of achieving protein concentrations up to 230 mg/mL, exceeding current FDA-approved limits. The proprietary platform uses advanced excipient blends and predictive instruments to reduce viscosity by up to 90%, maintain stability and injectability, and streamline manufacturing from early formulation through commercial scale.
The preclinical CRO industry in Japan is driven by the growing pharmaceutical industry in the country, contributing to the increasing demand for preclinical trial outsourcing. There has been a significant increase in the number of preclinical trials conducted in the country, as the Japanese government is taking steps to provide regulatory support and promote preclinical research. This is expected to stimulate the market growth for preclinical CRO services as it is to be conducted in proximity to central development centers.
The India preclinical CRO industry is experiencingsteady growth, driven by strong advancements in the pharmaceutical and biotech industries, along with cost benefits and a skilled scientific workforce. The country offers a wide range of preclinical services, including toxicology, pharmacokinetics, bioanalysis, and in vivo and in vitro studies, further strengthening its role as a preferred outsourcing destination for international drug developers. Other factors, such as rising research and development investments, increasing demand for biosimilars and biologics, and supportive government initiatives, further boost the market.
Latin America Preclinical CRO Market Trends
The preclinical CRO industry in Latin America is driven by increasing pharmaceutical R&D activity, which is gradually expanding beyond clinical trials into preclinical activities and, in turn, into preclinical CRO services. This is evident in countries such as Brazil and Mexico, where improving transparency within regulatory agencies, such as COFEPRIS and ANVISA, is helping global pharmaceutical companies navigate these processes and, in turn, evaluate local outsourcing opportunities.
The Brazil preclinical CRO industry held a significant market share in Latin America and is expected to maintain its position over the forecast period. Among Latin American countries, Brazil conducts the majority of preclinical trials due to its cost-effectiveness and diverse population. The country has witnessed substantial demand from the biotechnology and healthcare industries worldwide. Moreover, the increasing R&D capabilities in Brazil and government initiatives to collaborate with global companies are likely to attract investments from pharmaceutical and biotech companies. This is expected to boost market growth during the forecast period.
Middle East & Africa Preclinical CRO Market Trends
The preclinical industry in the Middle East and Africa is in the early stages of development and is gradually gaining traction due to the governments in the region, especially in the Gulf Cooperation Council (GCC) countries, that are recognizing the importance of diversification in the healthcare sector and investing in the development of the region’s life sciences industry. This is evident in the investments made by the governments in the region, such as in the United Arab Emirates and Saudi Arabia, in the development of biotech clusters and research infrastructure, which has been translating into opportunities for the preclinical market.
The UAE preclinical CRO industry is expected to grow steadilydue to the growing investments in complex biologic molecules development, a supportive regulatory environment, the entry of new players, and thegrowing year-on-year investments of foreign pharmaceutical companies & CROs. Moreover, the market is driven by domestic players expanding their R&D capabilities and geographical presence through various strategies. Such factors are expected to support the overall market growth in the country.
Key Preclinical CRO Company Insights
The key players operating across the market are adopting strategic initiatives such as service launches, mergers & acquisitions, partnerships & agreements, and expansions to gain a competitive edge in the market. For instance, in February 2026, Scantox acquired DuplexSeq genomic safety business from TwinStrand Biosciences. Such initiatives expand advanced mutagenesis testing, strengthen regulatory-ready data generation, and position Scantox as a leader in genomic safety assessment services.
Key Preclinical CRO Companies:
The following key companies have been profiled for this study on the preclinical CRO market.
- Eurofins Scientific
- ICON plc
- WuXi AppTec
- Medpace Holdings, Inc.
- Charles River Laboratories
- PPD (Thermo Fisher Scientific, Inc.)
- SGS SA (SGS Société Générale de Surveillance SA.)
- Intertek Group plc (IGP)
- Labcorp
- Crown Bioscience
- Scantox
Recent Developments
-
In March 2026, Scantox integrated Gentronix under its brand as Scantox Manchester. This strengthens genetic toxicology capabilities, enhances genomic safety services, and supports end-to-end preclinical offerings, improving competitiveness in regulatory toxicology services.
-
In February 2026, Synthace collaborated with Charles River Laboratories International, Inc. to improve assay development using automation. These kind of initiatives enhances reproducibility, reduce delays, and accelerate drug discovery timelines, strengthening efficiency and technological advancement in the market.
-
In September 2025, Crown Bioscience opened a North Carolina model development center to expand PDX, organoid, and translational oncology model capabilities. Such initiatives propel the demand for preclinical services in the region.
Preclinical CRO Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 7.3 billion
Revenue forecast in 2033
USD 12.8 billion
Growth rate
CAGR of 8.4% from 2026 to 2033
Actual Data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Service, development stage, indication, model, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East and Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Thailand; South Korea; Australia; Brazil; Argentina; South Africa; UAE; Saudi Arabia; Kuwait; Qatar; Oman
Key companies profiled
Eurofins Scientific; ICON plc; WuXi AppTec; Medpace Holdings, Inc.; Charles River Laboratories; PPD (Thermo Fisher Scientific, Inc.); SGS SA (SGS Société Générale de Surveillance SA.); Intertek Group plc (IGP); Labcorp, Crown Bioscience; Scantox
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Preclinical CRO Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global preclinical CRO market report based on service, development stage, indication, model, end use, and region.
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Bioanalysis and DMPK studies
-
In vitro ADME
-
In-vivo PK
-
-
Toxicology Testing
-
GLP
-
Non-GLP
-
-
Compound Management
-
Process R&D
-
Custom Synthesis
-
Others
-
-
Chemistry
-
Medicinal Chemistry
-
Computation Chemistry
-
-
Safety Pharmacology
-
Others
-
-
Development Stage Outlook (Revenue, USD Million, 2021 - 2033)
-
Target Discovery
-
Preclinical Optimization, Efficacy & PK/PD
-
IND-Enabling Safety (GLP)
-
Bioanalysis & Analytics
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
CNS
-
Infectious Diseases
-
Cardiovascular Diseases
-
Metabolic Disorders
-
Rare Diseases
-
Others
-
-
Model Outlook (Revenue, USD Million, 2021 - 2033)
-
In Vitro
-
In Vivo
-
Ex Vivo
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical and Biopharmaceutical Companies
-
Medical Device Companies
-
Academic and Research Institutes
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
UAE
-
Saudi Arabia
-
Kuwait
-
Qatar
-
Oman
-
-
Frequently Asked Questions About This Report
The global preclinical CRO market size was estimated at USD 6.70 billion in 2025 and is expected to reach USD 7.3 billion in 2026.
The global preclinical CRO market is expected to grow at a compound annual growth rate of 8.4% from 2026 to 2033 to reach USD 12.8 billion by 2033.
North America dominated the preclinical CRO market with a share of 48.07% in 2025. The regional growth is driven by the presence of established contract research organizations (CROs) and a growing demand for pharmaceutical, biotech, and medical device products. Some other factors contributing to segment growth include outsourcing early development phases to lower costs, speeding up timelines, and gaining access to specialized expertise.
Some key players operating in the preclinical CRO market include Eurofins Scientific, ICON plc, WuXi AppTec, Medpace Holdings, Inc., Charles River Laboratories, PPD (Thermo Fisher Scientific, Inc.), SGS SA (SGS Société Générale de Surveillance SA.), Intertek Group plc (IGP), Labcorp, Crown Bioscience, and Scantox among others.
Key factors that are driving the preclinical CRO market growth include outsourcing trends of preclinical research, particularly among biotech and biopharma companies seeking to minimize fixed infrastructure costs, shorten development timelines, and access specialized expertise, is expected to significantly contribute to future growth. Moreover, increasing complexity in drug development, higher biopharma R&D investments, and stringent regulations requiring Good Laboratory Practice (GLP)-compliant studies are influencing the market growth
About the authors:
Author: GVR Pharmaceuticals Research Team | Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









