- Home
- »
- Pharmaceuticals
- »
-
Respiratory Disease Vaccine Market, Industry Report, 2030GVR Report cover
![Respiratory Disease Vaccine Market Size, Share & Trends Report]()
Respiratory Disease Vaccine Market (2025 - 2030) Size, Share & Trends Analysis Report By Type (Viral, Bacterial, Combination), By Infection (COVID-19, Influenza), By Age Group, By Distribution Channel, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-093-9
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2018 - 2023
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
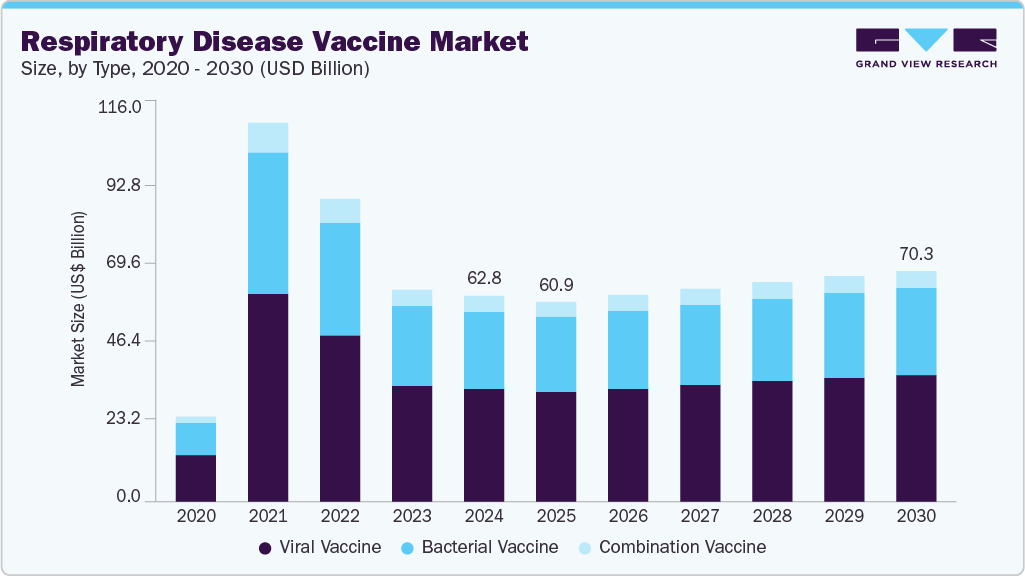
Respiratory Disease Vaccine Market Summary
The global respiratory disease vaccine market size was estimated at USD 62.76 billion in 2024 and is projected to reach USD 70.30 billion by 2030, growing at a CAGR of 2.9% from 2025 to 2030. The rising global burden of diseases such as influenza, pneumonia, pertussis, and respiratory syncytial virus (RSV) is a major reason for the market growth.
Key Market Trends & Insights
- The respiratory disease vaccine market in Asia Pacific held the largest market share of 30.55% in 2024.
- China respiratory disease vaccine marketis expected to witness robust growth over the forecast period.
- In terms of type segment, the viral vaccine segment held the largest market share of 54.84% in 2024.
- In terms of age group segment, the adult segment held the largest market share of 76.5% in 2024.
- In terms of infection segment, the COVID-19 segment held the largest market share of 40.03% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 62.76 Billion
- 2030 Projected Market Size: USD 70.30 Billion
- CAGR (2025-2030): 2.9%
- Asia Pacific: Largest market in 2024
- North America: Fastest growing market
The need to prevent and manage these illnesses increases the demand for vaccines. Advances in vaccine technology and government efforts to promote immunization programs also support market expansion. Supporting this trend, vaccine manufacturers are focusing on developing next-generation respiratory vaccines that incorporate the latest scientific advancements to address evolving pathogens and enhance immune response. Recent developments include improved vaccine formulations and delivery methods to increase effectiveness and patient compliance. For instance, in May 2024 , the U.S. Food and Drug Administration approved Moderna's mRESVIA (mRNA-1345), an mRNA-based vaccine for respiratory syncytial virus (RSV), marking the first mRNA vaccine approved for a disease other than COVID-19. This vaccine is designed to protect adults aged 60 years and older from lower respiratory tract disease caused by RSV infection. Such progress highlights the industry's dedication to leveraging technology to improve vaccine efficacy, streamline production, and ultimately reduce the impact of respiratory illnesses globally.
Moreover, technological progress in research and development has led to the creation of more effective and targeted respiratory disease vaccines. Enhanced formulations, novel adjuvants, and improved delivery systems are helping boost vaccine efficacy, immunogenicity, and safety, which supports wider adoption. These improvements help address existing challenges in vaccine performance, particularly in vulnerable groups such as infants, the elderly, and individuals with chronic illnesses. In addition, consistent investment in vaccine research by pharmaceutical companies and public health agencies plays a key role in strengthening the respiratory disease vaccines pipeline and expanding access across various regions.
Despite ongoing progress, the market faces several challenges that may hinder growth. The development, testing, and approval of vaccines require extensive time and resources, with manufacturers needing to meet strict regulatory and safety standards. These complexities often result in delays in product launches and broader market availability. In addition, ensuring a stable and equitable vaccine supply presents logistical difficulties, especially in regions with limited infrastructure. The need for reliable cold chain storage and transportation complicates vaccine distribution in remote or underserved areas. These factors collectively emphasize the importance of coordinated global efforts to overcome regulatory and logistical barriers and ensure consistent access to respiratory disease vaccines.
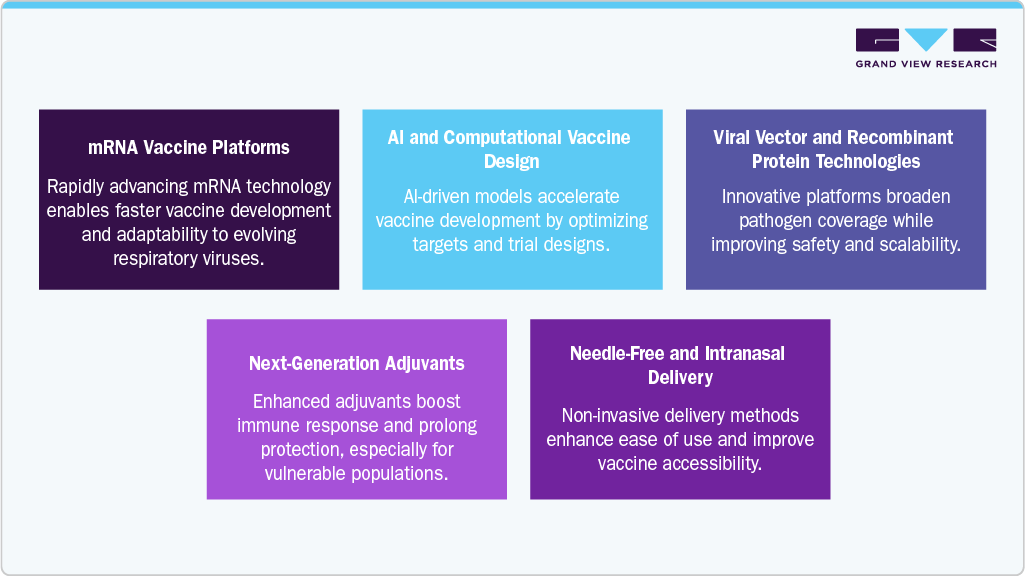
Key Technological Trends Shaping the Market
The respiratory disease vaccines industry is significantly transforming, shaped by advanced technological approaches to improve vaccine design, delivery, and responsiveness. These developments enhance protection against well-known respiratory pathogens and prepare the global healthcare infrastructure for future outbreaks. Below are some of the key trends influencing this market:
-
mRNA Vaccine Platforms: The success of mRNA vaccines during the COVID-19 pandemic has accelerated their development for other respiratory diseases such as influenza and RSV. These platforms allow for faster production and adaptability to mutating strains, offering a responsive approach to emerging respiratory threats.
-
Next-Generation Adjuvants: New adjuvant systems are being developed to improve immune responses and durability, particularly in older adults and immunocompromised individuals. These formulations aim to provide broader immunity while reducing the required doses.
-
Needle-Free and Intranasal Delivery: Intranasal sprays and microneedle patches are emerging as promising alternatives to traditional injections. These delivery methods improve ease of use, reduce the need for cold chain logistics, and increase patient compliance, especially in mass immunization programs.
-
Viral Vector and Recombinant Protein Technologies: Advances in vector-based and recombinant protein vaccine platforms are expanding the range of targetable respiratory pathogens. These methods provide enhanced safety and immunogenicity profiles and support rapid scale-up during public health emergencies.
-
AI and Computational Vaccine Design: Artificial intelligence is being applied to predict antigenic targets and optimize vaccine candidates. Machine learning models assist in analyzing pathogen evolution and improving trial design, which reduces development time and cost.
These emerging technologies are central to improving vaccine effectiveness, accessibility, and responsiveness. With the rising respiratory disease burden, these innovations can be essential in shaping global immunization strategies and public health preparedness.
Market Concentration & Characteristics
The respiratory disease vaccine industry is marked by rapid innovation in vaccine platforms such as mRNA, viral vectors, and recombinant proteins, which enable faster development and improved immune responses. Advances in adjuvants and delivery methods-including needle-free and intranasal options—enhance vaccine efficacy and patient compliance. Integrating artificial intelligence in vaccine design supports precision targeting of antigens and accelerates clinical development. These technological strides collectively improve vaccine adaptability to emerging respiratory pathogens, streamline production, and support broader immunization coverage.
The market has experienced consistent mergers and acquisitions focused on expanding vaccine platforms, strengthening research capabilities, and broadening global reach. Key deals target access to innovative technologies such as mRNA and viral vector platforms, advanced adjuvants, and novel delivery systems. Large pharmaceutical firms are acquiring smaller biotech companies with specialized vaccine candidates, while mid-sized players engage in partnerships and acquisitions to enhance their product pipelines and competitive positioning.
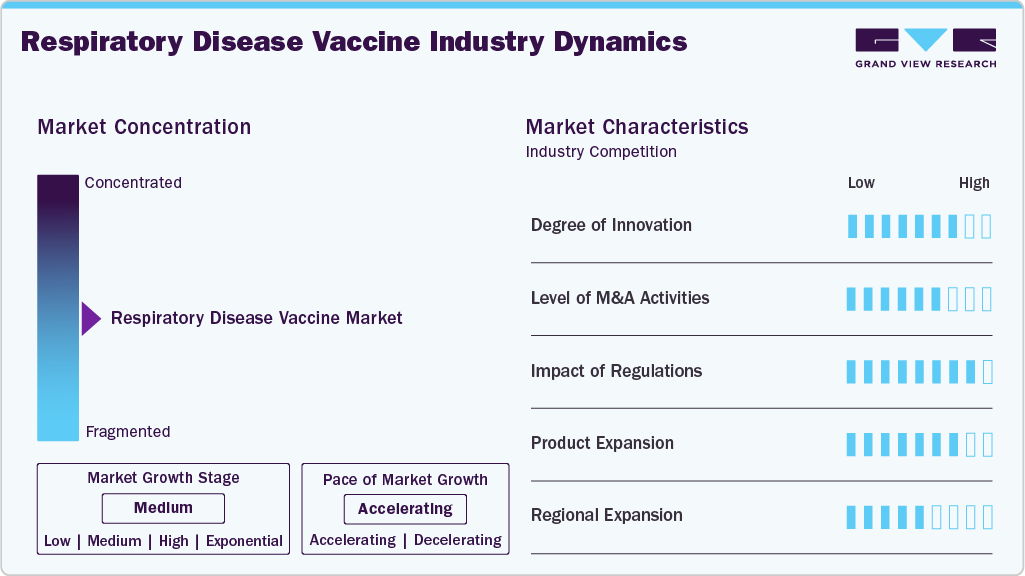
Regulation significantly influences the respiratory disease vaccine industry, with stringent approval processes in major regions like the U.S. and Europe. Compliance with regulatory standards, such as those set by the FDA and EMA, is critical for vaccine development, testing, and commercialization. Regulatory bodies enforce rigorous safety, efficacy, and quality requirements, which can extend development timelines. Additionally, evolving guidelines on novel vaccine technologies, including mRNA platforms and viral vectors, require manufacturers to adapt strategies to meet compliance, impacting market entry and availability.
The market is witnessing ongoing product expansion, with companies developing new vaccine candidates targeting a broader range of respiratory pathogens. A growing focus is improving vaccine formulations, adjuvants, and delivery systems to enhance efficacy and patient compliance. Efforts to expand product pipelines include next-generation mRNA vaccines, viral vector platforms, and needle-free delivery options to address unmet needs and emerging respiratory threats.
Regional expansion is prominent in the respiratory disease vaccine industry, with companies focusing on emerging markets in Asia Pacific, Latin America, and the Middle East due to increasing healthcare investments and rising incidence of respiratory illnesses. North America and Europe hold a significant market share because of advanced healthcare infrastructure and established vaccination programs. Growth strategies emphasize forming partnerships and expanding distribution channels in underserved regions to improve vaccine accessibility and meet growing demand.
Type Insights
The viral vaccine segment held the largest market share of 54.84% in 2024. This dominance is primarily driven by the widespread use of viral vaccines to prevent respiratory diseases such as influenza, COVID-19, and respiratory syncytial virus (RSV). These vaccines play a critical role in reducing disease burden and transmission, especially among vulnerable populations, including the elderly and immunocompromised individuals. Viral vaccines are favored for their ability to elicit strong and lasting immune responses, and advancements in vector-based and recombinant technologies have further supported their effectiveness and scalability. Continued development of targeted viral vaccines, particularly through platforms like mRNA and recombinant protein-based methods, is expected to sustain demand and support broader immunization programs across global markets.
The bacterial vaccine segment is expected to grow fastest during the forecast period. This growth is driven by rising concerns over antibiotic resistance and the need for effective prevention against bacterial respiratory infections such as pneumococcal and pertussis. These vaccines are increasingly integrated into national immunization schedules, particularly in developing countries. Ongoing research efforts and pipeline developments to expand bacterial vaccine coverage further support this segment’s accelerated growth.
Age Group Insights
The adult segment held the largest market share of 76.5% in 2024. This is mainly due to the rising prevalence of respiratory diseases among the adult population, particularly older adults who face a higher risk of infections and complications. With the global population aging, there is a growing focus on immunization strategies tailored for adults and efforts to promote vaccines that address their specific health needs. Increasing awareness among adults about the benefits of vaccination is also contributing to the segment’s continued growth.
Pediatric is expected to show the fastest growth rate over the forecast period. This growth is driven by the high number of vaccinations given to infants and young children to prevent respiratory diseases. For example, the pneumococcal conjugate vaccine (PCV) is administered in multiple doses starting at around 2 months of age to protect against pneumococcal infections, which can cause severe respiratory illnesses in children. The rising awareness about childhood immunization, expanded vaccination schedules, and strong government support continue to support this trend. In addition, the burden of respiratory infections and the lasting impact of the COVID-19 pandemic have further emphasized the importance of early immunization, boosting demand in this segment.
Infection Insights
The COVID-19 segment held the largest market share of 40.03% in 2024. This reflects extensive global vaccination efforts against SARS-CoV-2, with various vaccines, such as mRNA-based, viral vector, and inactivated types, rapidly developed and authorized for widespread use. Continued booster programs and immunization efforts help maintain immunity and support demand in this segment. Increased public awareness and government vaccination campaigns contribute to its sustained market leadership.
Respiratory Syncytial Virus (RSV) vaccine is anticipated to exhibit the fastest growth rate over the forecast period. The increasing prevalence of RSV-related infections and significant advancements in vaccine development drive this surge. GSK’s Arexvy became the first RSV vaccine approved by the U.S. FDA in May 2023, for adults aged 60 and older, demonstrating an overall efficacy of 82.6% in preventing lower respiratory tract disease caused by RSV, according to the pivotal AReSVi-006 Phase III trial. The rising awareness about RSV’s impact on vulnerable populations and growing government support for immunization programs further contribute to market expansion. In addition, ongoing research and development efforts continue to improve vaccine efficacy and safety, encouraging broader adoption. These factors collectively support strong growth prospects for the RSV vaccine segment.
Distribution Channel Insights
The hospital and retail pharmacies segment held the largest market share in 2024. This dominance is linked to the growing number of respiratory disease cases and the accessibility of vaccination services in these settings. Hospitals offer immunization through trained healthcare professionals, especially for high-risk groups such as older adults or individuals with chronic conditions. At the same time, retail pharmacies provide convenient, walk-in vaccination options without the need for appointments, improving access for the broader population. The combination of professional oversight in hospitals and flexible pharmacy services continues to support strong demand across these channels.
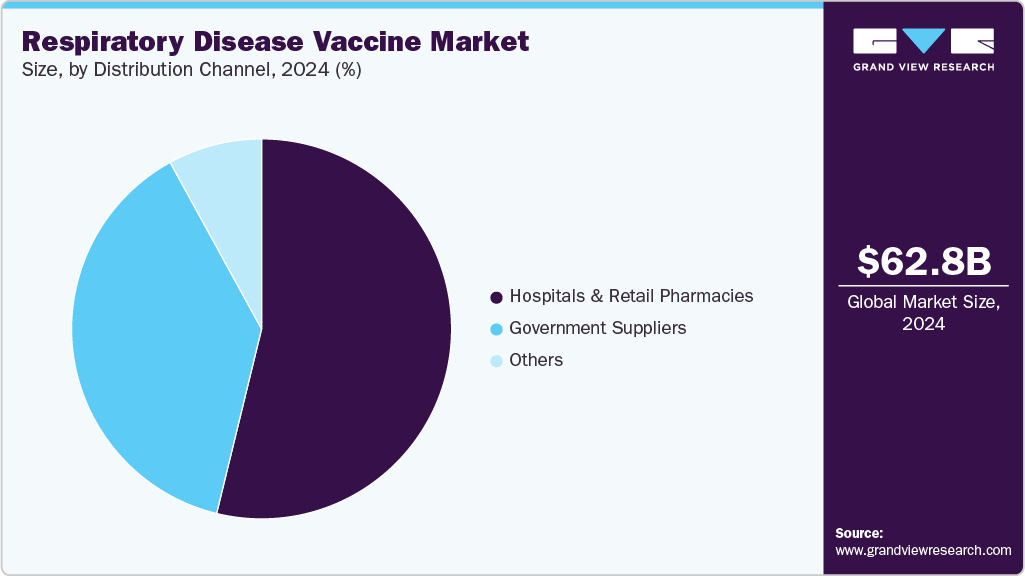
The government supplies segment is projected to grow fastest over the forecast period. This growth is driven by the expanding role of governments in supporting and executing vaccination programs across the globe. National initiatives, such as the National Vaccine Program, focus on research, safety testing, and efficient distribution of vaccines. In addition, partnerships with international organizations such as the World Health Organization (WHO) and the Pan American Health Organization (PAHO) have helped governments improve vaccine accessibility and affordability. Government-led vaccination drives, particularly those implemented during the pandemic, further highlight the critical role of public sector efforts in reaching broader populations.
Regional Insights
North America's respiratory disease vaccine market is expected to grow strongly over the forecast period, supported by rising public health awareness, a growing elderly population, and the early adoption of new vaccine technologies. The U.S. plays a significant role in this growth due to its strong immunization programs and widespread access to vaccines for illnesses such as influenza and pneumococcal infections. Vaccines are readily available through hospitals, clinics, pharmacies, and community health centers, helping to improve coverage across different age groups.
In addition, the region has seen progress in vaccine innovation and regulatory support. For example, in June 2024[3] , the U.S. FDA expanded the age indication for GSK's AREXVY vaccine, approving it for adults aged 50 to 59 at higher risk of RSV-related lower respiratory tract disease. This decision was supported by data showing that RSV leads to around 42,000 hospitalizations each year among U.S. adults aged 50 to 64. Such developments reflect North America's focus on improving protection against respiratory diseases through timely access to newer vaccines.
U.S. Respiratory Disease Vaccine Market Trends
The respiratory disease vaccine market in the U.S. is experiencing significant growth, supported by strong public health initiatives and increasing demand among the aging population. Established vaccination programs and a robust healthcare infrastructure ensure broad accessibility of vaccines for respiratory infections such as influenza, pneumococcal disease, and RSV. A favorable regulatory environment and substantial investments in research and development continue to drive innovation and the introduction of advanced vaccine technologies. In addition, growing awareness about the importance of vaccination for high-risk groups, including older adults and individuals with chronic conditions, has increased vaccine uptake. Collaborative efforts among government agencies, healthcare providers, and pharmaceutical companies are accelerating vaccine development and distribution. These factors solidify the U.S. as a key global market contributor, with ongoing advancements expected to improve prevention and reduce the burden of respiratory illnesses.
Europe Respiratory Disease Vaccine Market Trends
The respiratory disease vaccine market in Europe is experiencing steady growth, supported by strong public health policies, increased healthcare investment, and growing awareness about respiratory infections. Funding initiatives such as Horizon Europe promote research and development focused on innovative vaccine technologies. The European Medicines Agency provides clear regulatory pathways facilitating timely approval and market access. Countries like Germany, France, and the UK play a key role in vaccine innovation and manufacturing capacity, helping to meet the demand for effective respiratory vaccines. In addition, an aging population and rising prevalence of chronic respiratory conditions contribute to the growing need for vaccination programs across the region. These factors strengthen Europe’s position in the global market.
The UK respiratory disease vaccine market is growing significantly, driven by a substantial research and clinical infrastructure, government-supported vaccination programs, and a dynamic biotechnology sector. Leading academic institutions and NHS-affiliated health centers are actively involved in vaccine research and development, focusing on respiratory diseases that impact vulnerable populations. Funding from UK Research and Innovation (UKRI) and targeted public health initiatives support advancing and deploying innovative vaccine technologies. Regulatory guidance from the Medicines and Healthcare Products Regulatory Agency (MHRA) helps ensure the safe and timely introduction of new respiratory vaccines into clinical practice. In addition, the UK participates in collaborative projects across Europe, strengthening research networks and accelerating vaccine accessibility. These efforts position the UK as a key market player.
The respiratory disease vaccine market in Germanyplays a vital role in Europe’s market, supported by a strong research infrastructure, government-backed health programs, and active collaboration between academia and industry. The country’s well-established biotechnology and pharmaceutical sectors drive innovation in vaccine development, with leading research institutes such as the Robert Koch Institute contributing to epidemiological studies and vaccine evaluation. Germany’s regulatory authority, the Federal Institute for Vaccines and Biomedicines (PEI), ensures rigorous assessment and approval of vaccines, facilitating market access. Public-private partnerships and focused investments in precision medicine further accelerate progress in respiratory vaccine technologies. In addition, Germany’s aging population and rising respiratory disease burden increase demand for effective vaccines, reinforcing its position as a key market within the region.
Asia Pacific Respiratory Disease Vaccine Market Trends
The respiratory disease vaccine market in Asia Pacific held the largest market share of 30.55% in 2024 and is expected to maintain its dominant position throughout the forecast period. This leadership is driven by the high burden of respiratory diseases across several countries, combined with growing government initiatives to strengthen immunization programs. The region’s rapidly aging population, more susceptible to conditions such as pneumonia and influenza, contributes significantly to vaccine demand. Vaccination programs targeting older adults have been instrumental in reducing severe disease outcomes and alleviating strain on healthcare systems.
In addition, manufacturers are increasing their investments and footprint in Asia Pacific due to the considerable market potential. Efforts to improve vaccine access, supported by national and regional health agencies, reinforce this growth trend. Countries such as Japan, China, and India are scaling up procurement and distribution of respiratory vaccines as part of broader public health strategies. These factors, along with expanding healthcare infrastructure and heightened public awareness, are expected to support Asia Pacific's dominance in the market.
China respiratory disease vaccine market is expected to witness robust growth over the forecast period, supported by expanding immunization programs and a high burden of respiratory infections among both children and the elderly. Government initiatives to improve vaccination coverage, particularly for influenza, pneumonia, and emerging threats like RSV, have strengthened demand across healthcare settings. The country's aging population and urban pollution have increased susceptibility to respiratory illnesses, prompting greater public health focus on prevention. Domestic manufacturers are actively advancing vaccine development with support from national health authorities, while regulatory efforts by agencies such as the NMPA are streamlining product approvals. These factors collectively contribute to the rising adoption of respiratory vaccines in China.
The respiratory disease vaccine market in Japanholds a significant position in the Asia Pacific market, supported by strong government initiatives, a highly advanced healthcare system, and a well-established pharmaceutical industry. The country emphasizes preventive healthcare and vaccination programs, especially for vulnerable populations such as the elderly. Leading research institutions like the National Institute of Infectious Diseases actively contribute to vaccine development and surveillance of respiratory pathogens. Japan’s regulatory authority, the Pharmaceuticals and Medical Devices Agency (PMDA), ensures strict evaluation and approval processes, promoting the safety and efficacy of vaccines. Continued investment in innovative vaccine technologies and public health campaigns strengthens Japan’s role in advancing respiratory disease prevention across the region.
Latin America Respiratory Disease Vaccine Market Trends
The respiratory disease vaccine market in Latin America is showing steady growth, supported by expanding immunization programs, rising awareness of respiratory infections, and improvements in healthcare infrastructure. Brazil and Argentina are key contributors, with national vaccination campaigns targeting conditions such as influenza and pneumococcal disease. Government-backed efforts to increase vaccine accessibility enhance coverage rates, particularly in underserved communities. In addition, collaborations between public health institutions and international organizations facilitate the procurement and distribution of vaccines. These efforts, combined with a growing focus on preventive care and the modernization of cold chain logistics, are helping to strengthen the region’s position in the global market.
Middle East and Africa Respiratory Disease Vaccine Market Trends
The respiratory disease vaccine market in the Middle East and Africa is accelerating, driven by rising healthcare investment and a growing emphasis on preventive care. National strategies such as the UAE’s National Immunization Program and Saudi Arabia’s Vision 2030 prioritize expanded vaccine access for respiratory infections like influenza and RSV. Public-private partnerships are enabling wider distribution of vaccines, particularly in underserved regions. The region’s high burden of communicable respiratory diseases and emerging regulatory improvements encourage faster adoption of targeted vaccination programs. Collaborative efforts with global health organizations and local manufacturing initiatives strengthen the regional vaccine ecosystem.
Key Respiratory Disease Vaccine Company Insights
Key players in the market are concentrating on obtaining regulatory approvals for innovative vaccines, expanding their product portfolios, and forming strategic collaborations to strengthen their market position. For example, in June 2024, the U.S. FDA expanded the age indication for GSK’s RSV vaccine, Arexvy, to include adults aged 50 to 59 years who are at increased risk of RSV-related lower respiratory tract disease. This approval reflects the growing need for effective prevention among broader age groups and supports the rising demand for advanced respiratory vaccines.
Key Respiratory Disease Vaccine Companies:
The following are the leading companies in the respiratory disease vaccine market. These companies collectively hold the largest market share and dictate industry trends.
- GSK plc.
- Johnson & Johnson Services, Inc.
- Pfizer Inc.
- Sanofi
- Serum Institute of India Pvt. Ltd.
- SINOVAC
- Bavarian Nordic
- Merck & Co., Inc.
- AstraZeneca
- EMERGENT
- Novavax
- INOVIO Pharmaceuticals
- Moderna, Inc.
Recent Developments
-
In May 2025, the U.S. FDA approved Novavax’s Biologics License Application (BLA) for its COVID-19 vaccine, Nuvaxovid, for use in individuals 12 years and older. Nuvaxovid stands out as the only recombinant protein-based, non-mRNA COVID-19 vaccine currently authorized in the U.S. This approval supports broader immunization efforts and provides an alternative platform for individuals seeking a non-mRNA vaccine option within the market.
-
In March 2025, the World Health Organization (WHO) prequalified Pfizer's maternal respiratory syncytial virus (RSV) vaccine, Abrysvo, marking the first-ever prequalification of a maternal RSV vaccine. Designed to protect infants during their first six months of life through antibody transfer during pregnancy, Abrysvo addresses a significant global health concern, as RSV is a leading cause of acute lower respiratory infections in children under five, particularly in low- and middle-income countries. This prequalification enables procurement by UN agencies and paves the way for broader access in regions with the highest RSV burden.
-
In April 2025, Pfizer Inc. announced that the U.S. Centers for Disease Control and Prevention’s (CDC) Advisory Committee on Immunization Practices (ACIP) had voted to broaden its recommendation for RSV vaccines to include adults aged 50-59 who are at increased risk of RSV-related lower respiratory tract disease (LRTD). This updated guidance covers ABRYSVO (Respiratory Syncytial Virus Vaccine), which received FDA approval in October 2024 for preventing RSV-associated LRTD in at-risk adults aged 18 to 59.
Respiratory Disease Vaccine MarketReport Scope
Report Attribute
Details
Market size value in 2025
USD 60.95 billion
Revenue forecast in 2030
USD 70.30 billion
Growth rate
CAGR of 2.90% from 2025 to 2030
Base year for estimation
2024
Historical data
2018 - 2023
Forecast period
2025 - 2030
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, age group, infection, distribution channel, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
GSK plc; Johnson & Johnson Services, Inc.; Pfizer Inc.; Sanofi; Serum Institute of India Pvt. Ltd.; SINOVAC; Bavarian Nordic; Merck & Co., Inc.; AstraZeneca; EMERGENT; Novavax; INOVIO Pharmaceuticals; Moderna, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Respiratory Disease Vaccine Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global respiratory disease vaccine market report based on type, age group, infection, distribution channel and region.
-
Type Outlook (Revenue, USD Million, 2018 - 2030)
-
Viral Vaccine
-
Bacterial Vaccine
-
Combination Vaccine
-
-
Age Group Outlook (Revenue, USD Million, 2018 - 2030)
-
Pediatric
-
Adult
-
-
Infection Outlook (Revenue, USD Million, 2018 - 2030)
-
COVID-19
-
Influenza
-
Respiratory Syncytial Virus (RSV)
-
Pneumonia
-
Others
-
-
Distribution Channel Outlook (Revenue, USD Million, 2018 - 2030)
-
Hospital & Retail Pharmacies
-
Government Suppliers
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global respiratory disease vaccine market size was estimated at USD 62.76 billion in 2024 and is expected to reach USD 60.95 billion in 2025.
b. The global respiratory disease vaccine market is expected to grow at a compound annual growth rate of 2.90% from 2025 to 2030 to reach USD 70.30 billion by 2030.
b. North America dominated the respiratory disease vaccine market with a share of 28.48% in 2024. This is attributable to the presence of key players, rising healthcare infrastructure, growing cases of chronic disorders and availability of proper vaccination service
b. Some key players operating in the respiratory disease vaccine market include GSK plc., Serum Institute of India Pvt. Ltd., Johnson & Johnson Services, Inc., Pfizer Inc., Sanofi, Bavarian Nordic, Merck & Co., Inc., AstraZeneca, Novavax, EMERGENT, INOVIO Pharmaceuticals, SINOVAC, and Moderna Inc.
b. Key factors that are driving the market growth include the increasing awareness associated with the importance of vaccination, rising incidence of respiratory infections, and advancements in vaccine technology.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










