- Home
- »
- Healthcare IT
- »
-
Southeast Asia Digital Pharmaceutical Supply Chain Management Market, 2033GVR Report cover
![Southeast Asia Digital Pharmaceutical Supply Chain Management Market Size, Share & Trends Report]()
Southeast Asia Digital Pharmaceutical Supply Chain Management Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Software, Hardware, Services), By Mode Of Delivery, By Country (Thailand, Singapore, Indonesia, Philippines, Vietnam, Malaysia), And Segment Forecasts
- Report ID: GVR-4-68040-880-2
- Number of Report Pages: 90
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Southeast Asia Digital Pharmaceutical Supply Chain Management Market Summary
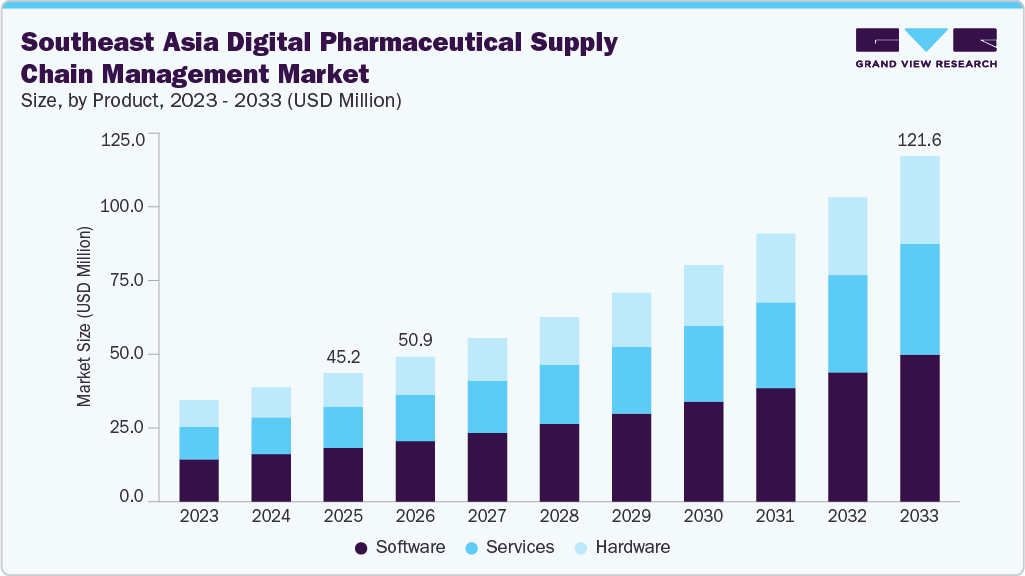
The Southeast Asia digital pharmaceutical supply chain management market size was estimated at USD 45.23 million in 2025 and is projected to reach USD 121.63 million by 2033, growing at a CAGR of 13.23% from 2026 to 2033. This growth is attributed to rising pharmaceutical demand and expanding healthcare infrastructure, increasing adoption of AI, IoT, and advanced analytics in supply chain operations, growing focus on drug traceability and anti-counterfeit regulations, expansion of digital procurement and B2B pharmaceutical distribution platforms, and government initiatives promoting digital transformation and supply chain modernization.
Key Market Trends & Insights
- Singapore dominated the Southeast Asia digital pharmaceutical supply chain management market with a revenue share of 26.9% in 2025.
- The Indonesia digital pharmaceutical supply chain management market is expected to grow at the fastest pace from 2026 to 2033.
- Based on product, the software segment held the largest market share of 41.8% in 2025.
- Based on mode of delivery, the on-premise segment held the dominant market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 45.2 Million
- 2033 Projected Market Size: USD 121.6 Million
- CAGR (2026 - 2033): 13.2%
- Singapore: Largest market in 2025
- Indonesia: Fastest growing market
The expansion of pharmaceutical manufacturing and contract manufacturing facilities across Southeast Asia is contributing to the growth of the digital pharmaceutical supply chain management market by increasing the scale and complexity of regional drug distribution networks. Rising production volumes, growing exports, and expanding cross-border pharmaceutical trade are driving the adoption of digital platforms for inventory management, track-and-trace compliance, procurement, and logistics coordination. For instance:-
In September 2025, a leading biotech firm announced a USD 150 million expansion of its manufacturing facilities in Singapore to significantly boost production capacity and create over 200 new jobs, supporting advanced therapeutics and biologics production.
-
In September 2025, a major generics manufacturer announced plans to open a state‑of‑the‑art generics manufacturing facility in Southeast Asia with over USD 50 million investment, aimed at boosting local supply chains and meeting rising demand for affordable medicines across the region. The facility, set to begin operations next quarter, will deploy advanced digital technology to enable efficient production and create around 300 jobs, supporting broad access to high-quality generics.
-
In October 2024, the Global Fund to Fight AIDS, Tuberculosis, and Malaria supported Indonesia in launching the SMILE e-LMIS, a digital logistics management system under the SATUSEHAT Logistics framework to improve tracking, distribution, and availability of essential health products across the country’s healthcare supply chain.
Such initiatives highlight the rapid growth and regional integration of pharmaceutical manufacturing in Southeast Asia, which is driving the demand for digital supply chain management solutions to efficiently manage procurement, distribution, regulatory compliance, and real-time inventory tracking.
In addition, the growing focus on drug traceability and anti-counterfeit regulations is driving the adoption of digital pharmaceutical supply chain management solutions in Southeast Asia. Regulatory authorities across the region are increasingly implementing track-and-trace mandates, serialization requirements, and anti-counterfeit measures to ensure drug safety, prevent the sale of falsified medicines, and improve patient outcomes. This is promoting pharmaceutical companies, distributors, and contract manufacturers to deploy digital platforms capable of real-time product tracking, automated compliance reporting, and secure data sharing, thereby strengthening supply chain transparency, minimizing losses, and maintaining regulatory compliance across complex regional distribution networks.
Moreover, AI integration is increasingly shaping the market, enabling smarter forecasting, real-time inventory tracking, and predictive demand planning. Healthcare organizations and distributors are adopting AI-powered analytics and automation to optimize procurement, reduce stockouts, prevent wastage, and enhance overall operational efficiency, supporting a more resilient and responsive supply chain across the region.
Market Concentration & Characteristics
The chart below illustrates the relationship between market concentration, industry characteristics, and industry participants. The x-axis represents the level of industry concentration, ranging from low to high. The y-axis represents various industry characteristics, including industry competition, impact of regulations, level of partnerships & collaborations activities, degree of innovation, and regional expansion. For instance, the Southeast Asia digital pharmaceutical supply chain management market is slightly fragmented, with many providers entering the market. The degree of innovation, the level of partnerships & collaboration, and the impact of regulations on the industry are high. However, the regional expansion observes moderate growth.
Innovation in the Southeast Asia digital pharmaceutical supply chain management industry is high, driven by the need for smarter, real-time, and efficient supply chain operations. Advances in AI-enabled analytics, digital integration, and connected platforms are helping healthcare organizations streamline procurement, inventory management, and distribution, enhance visibility of medical supplies, reduce costs, and support the timely delivery of medicines across the region. For instance, in September 2025, Oracle announced new AI-powered capabilities in its Oracle Fusion Cloud Applications to help healthcare organizations improve real-time visibility, boost efficiency, and enhance procurement workflows, further supporting better patient care.
Partnerships in the Southeast Asia digital pharmaceutical supply chain market remain limited, but collaboration is growing as companies expand their digital platforms to integrate required stakeholders. This trend strengthens digital infrastructure, interoperability, and real-time supply visibility, enabling more efficient procurement, inventory management, and distribution across the region.

Regulations play a critical role in the Southeast Asia digital pharmaceutical supply chain management industry by ensuring drug safety, authenticity, and compliance across complex supply networks. Regional differences in enforcement and traceability requirements across countries such as Indonesia, Malaysia, Vietnam, Thailand, and the Philippines create operational challenges that require manufacturers, distributors, and logistics providers to adopt customized compliance strategies. At the same time, these regulations are driving investments in digital track-and-trace systems, serialization technologies, inventory visibility platforms, and secure data management, enabling pharmaceutical supply chains to operate efficiently while maintaining product integrity, regulatory compliance, and patient safety.
The Southeast Asia digital pharmaceutical supply chain management market is growing due to rising drug demand, complex distribution networks, and expanding healthcare infrastructure, with digital platforms and collaborations between pharma companies and logistics providers enhancing supply chain visibility, efficiency, and compliance across the region.
Product Insights
The software segment led the Southeast Asia digital pharmaceutical supply chain management market, accounting for the largest revenue share of 41.7% in 2025, owing to its applications in the digital pharmaceutical supply chain management market. Software is also expected to be the fastest-growing segment over the forecast period. The increasing adoption of software, such as SaaS, which can offer next-generation cloud computing applications, including supply chain management & enterprise resource planning, is among the key factors boosting the market. In addition, the software product segment of the market is further sub-segmented into planning & analytics, procurement, manufacturing, logistics, and inventory management. The manufacturing software module segment dominated the market.
The services segment is expected to grow at the second-fastest CAGR over the forecast period, driven by advancements and rising demand for support & maintenance, consulting, and education & training. The development of a range of platforms for monitoring, diagnosis, and disease prevention and wellness is another key factor contributing to the segment's growth. The growing trend of implementing software upgrades to incorporate a more diverse array of healthcare applications is further contributing to segment growth.
Mode of Delivery Insights
The cloud-based segment dominated the Southeast Asia digital pharmaceutical supply chain management industry, accounting for a revenue share of 41.78% in 2025. Cloud-based deployment is also expected to be the fastest-growing segment over the forecast period. Its growth can be attributed to rising demand for user-friendly technology, cost-effectiveness, and increasing adoption. The growing adoption of cloud computing among healthcare providers to maintain inventory and procurement information is expected to further drive the segment.
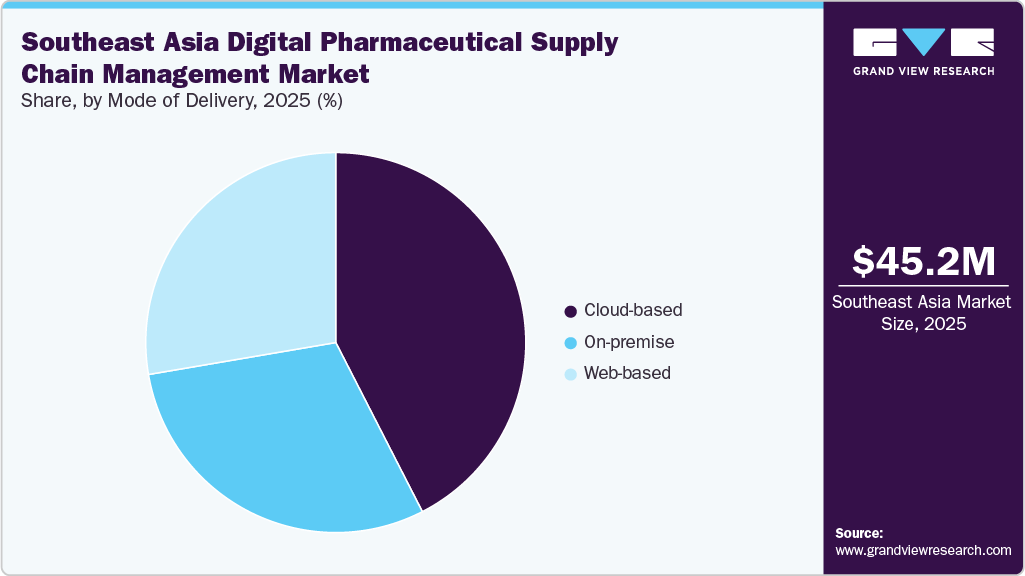
Moreover, the on-premises segment held a substantial share in the market owing to the preference of pharmaceutical manufacturers and distributors for greater control over sensitive drug manufacturing, regulatory, and serialization data, along with concerns related to data security, compliance requirements, and integration with legacy enterprise systems.
Country Insights
Thailand Digital Pharmaceutical Supply Chain Management Market
The digital pharmaceutical supply chain management market in Thailand is advancing as healthcare logistics adopt digital solutions to improve inventory visibility, real‑time tracking, and distribution efficiency. Government initiatives to modernize industrial and digital capabilities are driving the adoption of digital supply chain platforms and analytics tools. For instance, in December 2025, DKSH Business Unit Healthcare rolled out its “A‑Frame” automation system at its main healthcare logistics hub in Thailand, boosting fulfilment productivity by about 40% and enhancing the efficiency and digitalization of pharmaceutical supply chain operations.
Indonesia Digital Pharmaceutical Supply Chain Management Market
Indonesia is expected to grow at the fastest CAGR during the forecast period in the Southeast Asia digital pharmaceutical supply chain management market, driven by rapidly expanding pharmaceutical manufacturing, rising exports, and the increasing adoption of digital supply chain technologies. Investments in automation, warehouse management systems, and track-and-trace solutions are enhancing operational efficiency and regulatory compliance across the country’s pharmaceutical supply chains. For instance, in October 2024, the Ministry of Health of Indonesia, in collaboration with the United Nations Development Programme, introduced SATUSEHAT Logistics, a digital platform integrating pharmaceutical and medical device supply chain processes from procurement to distribution, enabling real-time tracking and improving the efficiency of national health logistics management.
Key Southeast Asia Digital Pharmaceutical Supply Chain Management Company Share Insights
Key players in the Southeast Asia digital pharmaceutical supply chain management market are adopting innovative strategies, including AI-enabled analytics, digital procurement platforms, and real-time inventory monitoring to enhance operational efficiency and supply visibility. Integration of platforms and collaborations with logistics providers, distributors, and healthcare organizations are further driving adoption, improving access to medicine, and expanding the reach of efficient supply chain solutions across the region.
Key Southeast Asia Digital Pharmaceutical Supply Chain Management Companies:
- SAP
- Oracle
- Infor
- TraceLink
- InterSystems
- Tecsys
- Jump Technologies
- Palantir Technologies
- Terso Solutions
- CenTrak (Halma plc)
- Biolog-id
- ANTARES VISION S.p.A
Recent Developments
-
In December 2025, Fujitsu announced the development of a multi‑AI agent collaboration technology that enables secure cooperation among AI agents from different companies to optimize supply chain operations. The technology will begin field trials in January 2026 with Rohto Pharmaceutical and Science Tokyo to streamline logistics, improve responsiveness to demand changes, and enhance resilience across supply networks.
-
In October 2025, Malaysia announced it will launch a supply chain intelligent management system (SCIMS) in December 2025, a government initiative covering eight key sectors, including pharmaceuticals and medical devices, to enhance supply chain resilience and better prioritise critical industries.
-
In April 2025, DHL Group launched a USD 11.5 million (€10 million) pharmaceutical logistics hub in Singapore as part of its USD 576.6 million (€500 million) Asia-Pacific healthcare investment to strengthen life sciences supply chain infrastructure and support rising regional demand for pharmaceutical and medical products
Southeast Asia Digital Pharmaceutical Supply Chain Management Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 50.97 million
Revenue forecast in 2033
USD 121.63 million
Growth rate
CAGR of 13.23% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Market Value in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, mode of delivery, country
Country scope
Thailand; Singapore; Indonesia; Philippines; Vietnam; Malaysia
SAP; Oracle; Infor; TraceLink; InterSystems; Tecsys; Jump Technologies; Palantir Technologies; Terso Solutions; CenTrak (Halma plc); Biolog-id; Antares Vision Group
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Southeast Asia Digital Pharmaceutical Supply Chain Management Market Report Segmentation
This report forecasts revenue growth at the regional and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Southeast Asia digital pharmaceutical supply chain management market report based on product, mode of delivery, and country:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Software
-
Planning & Analytics
-
Procurement
-
Manufacturing
-
Logistics
-
Inventory Management
-
Others
-
-
Hardware
-
Services
-
-
Mode of Delivery Outlook (Revenue, USD Million, 2021 - 2033)
-
On-premise
-
Cloud-based
-
Web-based
-
-
Country Outlook (Revenue, USD Million, 2021 - 2033)
-
Thailand
-
Singapore
-
Indonesia
-
Philippines
-
Vietnam
-
Malaysia
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










