- Home
- »
- Biotechnology
- »
-
Tissue Engineering Market Size And Share Report, 2030GVR Report cover
![Tissue Engineering Market Size, Share & Trends Report]()
Tissue Engineering Market (2025 - 2030) Size, Share & Trends Analysis Report By Application (Cord Blood & Cell Banking, Cancer, GI & Gynecology, Dental, Orthopedics, Musculoskeletal, & Spine, Urology), By Region And Segment Forecasts
- Report ID: 978-1-68038-768-1
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2018 - 2024
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Tissue Engineering Market Summary
The global tissue engineering market size was estimated at USD 19.36 billion in 2024 and is anticipated to reach USD 43.13 billion by 2030, growing at a CAGR of 14.3% from 2025 to 2030. The increasing prevalence of chronic diseases, including cardiovascular conditions, diabetes, and orthopedic issues, has heightened the need for innovative tissue engineering solutions that can effectively repair or replace damaged tissues.
Key Market Trends & Insights
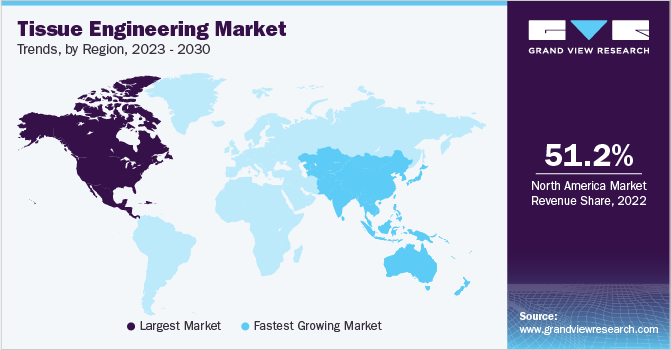
- North America dominated the tissue engineering market and accounted for the largest revenue share of 51.30% in 2024.
- The tissue engineering market in the U.S. dominated the North American market in 2024.
- Based on application, the orthopedics, musculoskeletal, & spine segment held the largest revenue of 59.21% market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 19.36 Billion
- 2030 Projected Market Size: USD 43.13 Billion
- CAGR (2025-2030): 14.3%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
Improvements in biomaterials, 3D bioprinting, stem cell research, and gene editing techniques have propelled recent advancements in this field. These developments have facilitated the creation of complex and functional tissues, enhancing the potential for successful therapeutic interventions. Technological advancements in tissue engineering are transforming the field with innovations. 3D Bioprinting, for instance, enables the precise layering of cells and biomaterials to create complex tissues and organ structures. In addition, increasing government funding for medical and academic research activities would enhance the market growth for tissue engineering throughout the forecast period. For instance, in August 2024, Dr. Josephine Wu from Trinity College Dublin received USD 8,82,604 (€800,000) in Wellcome Trust funding to develop a novel platform called OPTO-BIOPRINTING. This project aims to create a method for tissue engineering using light to control protein production within cells, offering promising advances in regenerative. Such funding efforts highlight the increasing global support for tissue engineering innovations.
The increasing number of clinical studies in Tissue Engineering and tissue engineering is driven by the growing interest in developing new therapies for a range of conditions, such as tissue repair, wound healing, and organ regeneration. Clinical trials investigating tissue-engineered patches for heart tissue regeneration have gained momentum. For example, in January 2023, a research team led by Dr. Hasan Erbil Abaci at Columbia University has been developing techniques to create 3D-engineered skin tailored to the shapes of complex body parts. These custom grafts can be transplanted with minimal suturing, enhancing surgical efficiency. In their recent study, the team evaluated their skin-culture system using models of human hands and mouse hindlimbs. In addition, several studies are exploring 3D bioprinting in orthopedic applications. In 2024, researchers launched clinical trials to assess the effectiveness of bioprinted scaffolds for bone regeneration in patients suffering from bone defects due to trauma or disease.
Furthermore, stem cell technology enhances regenerative potential by using induced pluripotent stem cells (iPSCs) and adult stem cells for tissue growth. Stem cell therapies are promising treatments for various clinical conditions, leading to significant global research and clinical translation investments. The rapid progress in stem cell research has contributed to enhanced disease management. Consequently, as the prevalence of cancer, diabetes, and other chronic disorders rises, the focus on stem cell research has intensified.
Application Insights
The orthopedics, musculoskeletal, & spine segment held the largest revenue of 59.21% market share in 2024, owing to the increasing prevalence of musculoskeletal disorders. According to the Global RA Network, arthritis affects over 350 million people worldwide, and projections suggest that by 2040, approximately 78 million U.S. adults will suffer from the condition. This increase underscores the growing burden of arthritis globally and within the U.S. Tissue engineering is gaining prominence as a therapeutic approach for orthopedic surgeons in treating a variety of musculoskeletal conditions. These include meniscal deficits in young athletes and osteochondral defects in joints like the glenohumeral joint (shoulder). By offering regenerative solutions, tissue engineering is emerging as a vital option in managing conditions that impact joint function and overall mobility.
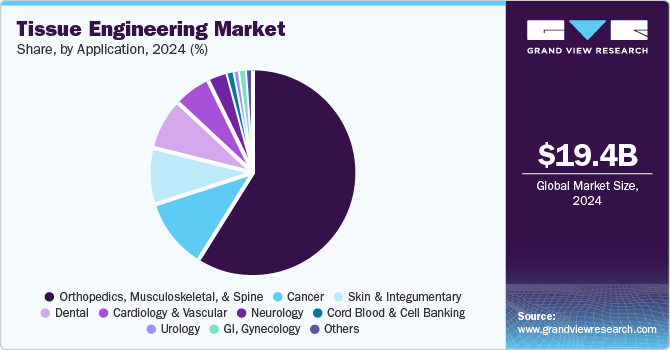
The cardiology & vascular segment is expected to register the highest CAGR over the forecast period owing to the rising prevalence of cardiovascular disorders. Notably, key players in the industry are actively engaged in developing stem cell therapies aimed at repairing, restoring, and re-vascularizing damaged heart tissues. In addition, research is being conducted on gene therapy, advanced biologics, and small molecules to stimulate the regeneration of damaged heart cells.
Regional Insights
North America dominated the tissue engineering market and accounted for the largest revenue share of 51.30% in 2024, owing to rising awareness of stem cell therapy, the growing geriatric population, and the rising incidence of chronic diseases. Moreover, advanced technology for diagnosis and treatment of chronic disorders, private and government funding availability, and high healthcare spending are some factors responsible for its high share.
U.S. Tissue Engineering Market Trends
The tissue engineering market in the U.S. dominated the North American market in 2024. Advancements in tissue engineering technology and the presence of prominent market players are significantly driving growth through continuous product launches in the U.S. For instance, Organovo, a pioneer in 3D bioprinting, has been working on commercializing 3D-printed human tissues for drug testing and therapeutic applications. Their liver and kidney tissue models are being advanced for therapeutic applications in the market. Similarly, Advanced Cell Technology (ACT) has been actively developing stem-cell-based tissue regeneration technologies, targeting therapies for conditions such as macular degeneration.
Europe Tissue Engineering Market Trends
The tissue engineering market in Europe is growing significantly due to the aging population, particularly in countries like Germany, Italy, and France. This population is increasing the prevalence of chronic conditions such as osteoarthritis, cardiovascular diseases, and bone degeneration. The elderly population's growing demand for regenerative therapies, such as those provided by tissue engineering, is driving innovation in this sector.
The UK tissue engineering market is anticipated to witness significant growth over the forecast period due to increased investments by various big companies and governments, including the NHS and research laboratories.
The tissue engineering market in Germany is expected to witness increasing competition over the forecast period due to the growing demand for gene and cell therapy in the country. Companies are focusing on expanding their markets.
Asia Pacific Tissue Engineering Market Trends
The market for tissue engineering in Asia Pacific is estimated to witness the fastest CAGR of 19.04% during the forecast period. Japan is one of the leading countries that have fostered technological advances in the tissue engineering field. In addition, an increase in the prevalence of clinical disorders, including cancer, in Asian countries, coupled with strong government support and robust research ecosystems, are expected to propel the growth of the market for tissue engineering in this region is considered one of the emerging markets in the Asia Pacific owing Japan tissue engineering market to the rising demand for Tissue Engineering & gene therapy products and the presence of enthusiastic investors.
The tissue engineering market in China is anticipated to witness significant growth over the forecast period due to increasing demand for cell and gene therapy. The companies and authorities operating in the country focus on cell and gene therapy.
MEA Tissue Engineering Market Trends
The tissue engineering market in MEA is expected to grow significantly over the forecast period. Middle Eastern countries in the MEA face several challenges, such as complexity and a lack of standardization in licensing and recruitment regulation. Investment by the private sector is low; more than 75% of healthcare spending in these countries is by the government. However, the rising incidence of lifestyle-related diseases and increasing demand for standard healthcare provisions are expected to create demand for better healthcare solutions.
South Africa tissue engineering market is expected to grow over the forecast period. The South African government is undertaking major initiatives to encourage the development of the country's biotechnology sector.
The tissue engineering market in the UAE is supported by several initiatives undertaken by various organizations to boost the country's biotechnology and biopharmaceutical sectors.
Key Tissue Engineering Company Insights
The market is highly competitive, with many manufacturers accounting for most of the market share. Market participants employ essential business strategies such as product launches, approvals, strategic acquisitions, and continuous innovations to sustain and expand their global presence.
Key Tissue Engineering Companies:
The following are the leading companies in the tissue engineering market. These companies collectively hold the largest market share and dictate industry trends.
- Zimmer Biomet Holdings, Inc.
- AbbVie (Allergan)
- Becton Dickinson and Company
- B. Braun
- Integra LifeSciences Corporation
- Organogenesis Holdings Inc.
- Medtronic
- ACell, Inc.
- Athersys, Inc.
- Tissue Regenix Group plc
- Stryker Corporation
- RTI Surgical, Inc.
- ReproCell, Inc.
- Baxter International, Inc.
Recent Developments
-
In March 2024, HVD Life Science partnered with Poietis to distribute Poietis’ NGB-R platform in Australia and Germany. This collaboration brings cutting-edge bioprinting technology to the forefront of R&D in tissue engineering and regenerative medicine.
-
In July 2024, PacBio partnered with Novogene to drive genomic research, including applications that benefit the tissue engineering sector, such as rare diseases and oncology.
-
In January 2023, BioMed X and AbbVie extended their research partnership, as the independent German biomedical research organization reported. The new U.S.-based research partnership will focus on immunology and tissue engineering after a first joint project on Alzheimer's disease at the BioMed X Institute in Heidelberg, Germany.
-
In May 2023, Canon entered the Tissue Engineering space by acquiring cell culture manufacturer Kyoto Seisakusho.
Tissue Engineering Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 22.13 billion
Revenue forecast in 2030
USD 43.13 billion
Growth rate
CAGR of 14.3% from 2025 to 2030
Actual data
2018 - 2024
Forecast period
2025 - 2030
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Application, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; Saudi Arabia; South Africa; UAE; Kuwait
Key companies profiled
Zimmer Biomet Holdings, Inc.; AbbVie (Allergan); Becton Dickinson and Company; B. Braun; Integra LifeSciences Corporation; Organogenesis Holdings Inc.; Medtronic; ACell, Inc.; Athersys, Inc.; Tissue Regenix Group plc; Stryker Corporation; RTI Surgical, Inc.; ReproCell, Inc.; Baxter International, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Tissue Engineering Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global tissue engineering market based on application and region:
-
Application Outlook (Revenue, USD Million, 2018 - 2030)
-
Cord blood & Cell Banking
-
Cancer
-
GI, Gynecology
-
Dental
-
Skin & Integumentary
-
Urology
-
Orthopedics, Musculoskeletal, & Spine
-
Neurology
-
Cardiology & Vascular
-
Others
-
-
Regional Outlook (Revenue in USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Sweden
-
Norway
-
Denmark
-
-
Asia Pacific
-
China
-
Japan
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa
-
Saudi Arabia
-
South Africa
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
The global tissue engineering market size was estimated at USD 19.36 billion in 2024 and is expected to reach USD 22.13 billion in 2025.
The global tissue engineering market is expected to grow at a compound annual growth rate of 14.3% from 2025 to 2030 to reach USD 43.13 billion by 2030.
North America dominated the tissue engineering market with a share of 51.30% in 2024. This is attributable to rising awareness of stem cell therapy as well as the growing geriatric population and rising incidence of chronic diseases.
Some key players operating in the tissue engineering market include Medtronic plc; Zimmer Biomet Holdings, Inc; Allergan plc; Athersys, Inc; ACell, Inc.; Organogenesis Holdings Inc; Tissue Regenix Group plc; Stryker Corporation; RTI Surgical, Inc.; Integra LifeSciences Corporation; ReproCell, Inc.; and Baxter International, Inc.
Key factors driving the market growth include advancements in stem cell technology & tissue engineering, a rise in the number of clinical studies for regenerative medicine and tissue engineering, and increasing tissue engineering research funding.
About the authors:
Author: GVR Biotechnology Research Team | Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









