Industry Insights
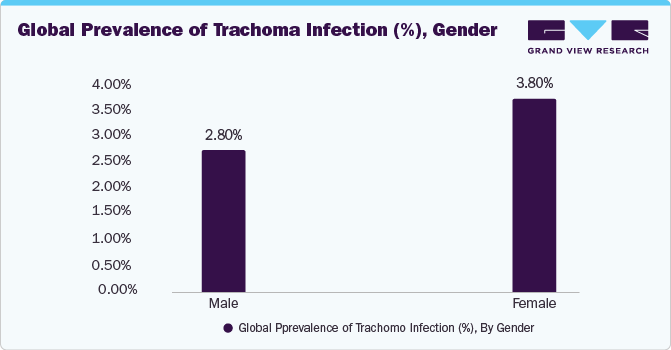
The global trachoma treatment market size was valued at USD 514.6 million in 2023 and is expected to grow at a Compound Annual Growth Rate (CAGR) of 7.09% from 2024 to 2030. The market is experiencing growth due to the increasing incidence and prevalence of trachoma infectious disease. For instance, there are 125 million individuals residing in regions where trachoma is endemic, placing them at risk of experiencing blindness due to the disease.
Moreover, government initiatives play a pivotal role in bolstering the market by implementing comprehensive programs and policies designed to control and ultimately eliminate this preventable eye disease. These initiatives encompass vital aspects such as allocating financial resources to support treatment distribution and surgeries, ensuring access to antibiotics in endemic regions, and promoting improved hygiene practices. For instance, as per the Australian Trachoma Surveillance Report published in 2021, trachoma continues to persist in certain Aboriginal and Torres Strait Islander communities in the country. The Australian government actively reports its efforts to combat trachoma to the World Health Organization's (WHO) global program aimed at eradicating the disease worldwide. Additionally, in June 2022, the Ministry of Public Health and Social Assistance of Guatemala (MPHSA) collaborated with the Pan American Health Organization (PAHO) on a healthcare campaign in Sololá. The objective was to perform surgeries on individuals affected by trachoma, aiming to prevent the disease from progressing and causing vision loss.
The trachoma treatment market was significantly influenced by the COVID-19 pandemic, which led to interruptions in standard healthcare services, particularly in trachoma treatment programs.
Route of Administration Insights
On the basis of the route of administration, the market is segmented into oral and topical. The oral segment held the largest market share in 2023 due to its effectiveness in treating the disease caused by Chlamydia trachomatis bacteria. Furthermore, they are easy to administer, improve patient compliance, provide systemic coverage, reduce contamination risk, and have a proven track record in large-scale trachoma control programs, making them a reliable route of administration.
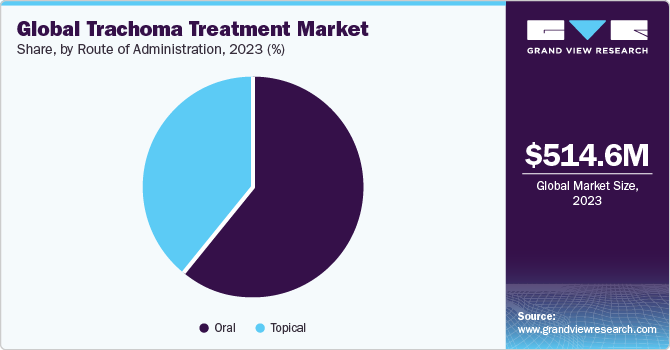
On the other hand, the topical segment is expected to witness lucrative growth over the forecast period.
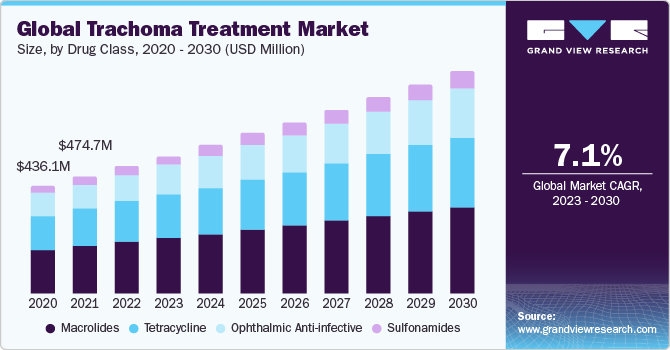
Drug Class Insights
Based on drug class, the market is segmented into macrolides, tetracycline, ophthalmic anti-infective, and sulfonamides. The macrolides segment dominated the market in 2023 due to its high effectiveness against bacteria-causing trachoma and simplified treatment regimens. Additionally, their reduced risk of antibiotic resistance and favorable safety profile further solidify their prominence in treating trachoma. On the other hand, tetracycline is expected to witness lucrative growth over the forecast period.
Regional Insights
North America dominated the market in 2023. The region benefits from robust healthcare infrastructure and access to medical resources, enabling efficient diagnosis and treatment of trachoma cases. Government initiatives and support for healthcare, along with well-established healthcare systems, play a pivotal role in the success of trachoma control programs.
On the other hand, Asia Pacific is expected to grow at a lucrative growth over the forecast period. The regional growth is mainly attributed to the increasing patient population, the high geriatric population, and an increasing number of regional companies developing novel treatment options.
Competitive Insights
Key players operating in the market are Allergan (AbbVie Inc.), Apotex, Pfizer Inc., Teva Pharmaceutical Industries Ltd, Fresenius Kabi AG, Sandoz Inc. (Novartis AG), Merck & Co. Inc., and Arbor Pharmaceuticals Inc. (Azurity Pharmaceuticals, Inc.) The market participants are working towards new product development, M&A activities, and other strategic alliances to gain new market avenues. The following are some instances of such initiatives.
-
In June 2022, Pfizer declared the extension of its antibiotic donation initiative until 2030, ensuring sustained support for trachoma elimination efforts across 18 nations. This long-standing program, conducted in collaboration with the International Trachoma Initiative (ITI) since 1998, has successfully provided 1 billion doses of donated antibiotics, aligning with the WHO-endorsed SAFE strategy.
-
In February 2023, AbbVie broadened its partnership with Capsida Biotherapeutics to create precision genetic therapies for eye conditions characterized by significant unmet medical needs. This initiative will make use of Capsida's advanced AAV engineering platform and manufacturing capacities.














