- Home
- »
- Pharmaceuticals
- »
-
U.S. Antifungal Drugs Market Size, Industry Report, 2030GVR Report cover
![U.S. Antifungal Drugs Market Size, Share & Trends Report]()
U.S. Antifungal Drugs Market (2025 - 2030) Size, Share & Trends Analysis Report By Drug Class (Azoles, Echinocandins, Polyenes, Allylamines), By Indication, By Dosage Form, By Distribution Channel, And Segment Forecasts
- Report ID: GVR-4-68040-668-1
- Number of Report Pages: 80
- Format: PDF
- Historical Range: 2018 - 2023
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Antifungal Drugs Market Summary
The U.S. antifungal drugs market was estimated at USD 6.05 billion in 2024 and is projected to reach USD 7.20 billion by 2030, growing at a CAGR of 3.0% from 2025 to 2030. The growing incidence of fungal infections drives market expansion.
Key Market Trends & Insights
- By drug class, the azoles segment held the highest market share of 47.4% in 2024.
- Based on the indication, the candidiasis segment held the highest market share in 2024.
- By dosage form, the oral drugs segment held the highest market share in 2024.
- By distribution channel, retail pharmacies are expected to grow rapidly over the forecast period.
Market Size & Forecast
- 2024 Market Size: USD 6.05 Billion
- 2030 Projected Market Size: USD 7.20 Billion
- CAGR (2025-2030): 3.0%
Fungal infections cover a broad range of conditions, from athlete’s foot, ringworm, and vaginal yeast infections to severe systemic diseases such as candidemia, aspergillosis, and mucormycosis. The CDC identifies Candida auris and resistant Aspergillus species as serious threats, necessitating therapeutic advances and enhanced infection control. The U.S. antifungal drugs industry is mainly driven by a consistent increase in fungal infections among immunocompromised groups, such as cancer patients, transplant recipients, and individuals with HIV; a rise in ICU admissions; and the increased use of medical devices such as catheters and ventilators. This population is particularly vulnerable to invasive infections such as candidemia and aspergillosis, which require systemic antifungal treatments. Furthermore, superficial fungal infections such as onychomycosis and dermatophytosis remain widespread and frequently recur, maintaining steady demand for oral and topical antifungal medications. Surveillance data show that non-albicans Candida species cause approximately two-thirds of bloodstream infections, with 6% of isolates resistant to fluconazole and 2% of C. glabrata isolates resistant to echinocandins, highlighting the need for newer and more effective antifungal therapies.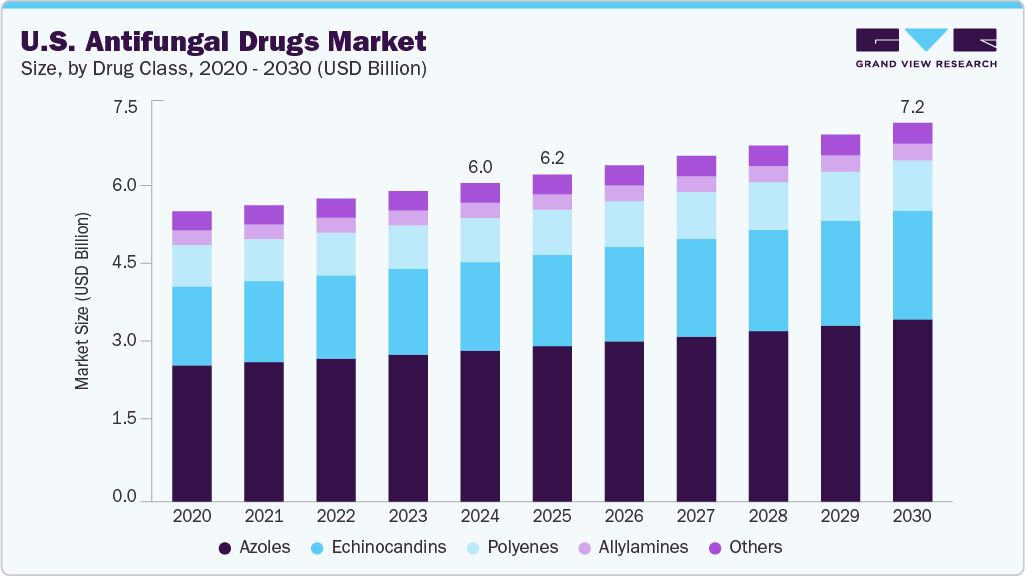
Systemic fungal infections continue to pose a significant healthcare challenge in the U.S. Each year, more than 75,000 hospitalizations and nearly 9 million outpatient visits are attributed to fungal diseases, as per the CDC in May 2024. As per April 2024 data, ab out 25,000 cases of candidemia occur annually, with roughly one-third of patients dying during their hospital stay. Public health initiatives are steadily raising awareness of fungal diseases. The CDC’s Fungal Disease Awareness Week, scheduled for September 2025, aims to promote early diagnosis and enhance resistance surveillance.
Drug Class Insights
The azole drug class segment dominated the market and accounted for a revenue share of 47.4% in 2024. This dominance is supported by the clinical success of widely used antifungal agents such as Vfend, Cresemba, Noxafil and Diflucan. Known for their broad-spectrum activity, azoles are effective against various infections, including ocular fungal diseases, systemic candidiasis, and blastomycosis. In December 2023, Astellas Pharma US, Inc. announced that the U.S.FDA approved CRESEMBA (isavuconazonium sulfate) for treating invasive mucormycosis and invasive aspergillosis in children as young as 1 year and over, making it the only azole antifungal authorized for pediatric patients of this age.
The echinocandins segment is expected to grow at the fastest CAGR of 3.5%, due to their targeted action against fungal cell wall synthesis and strong safety profile. Administered intravenously, these drugs are a cornerstone of hospital treatment protocols for candidemia and invasive candidiasis, where rapid fungal clearance is crucial. In March 2023, the FDA approved Rezzayo (rezafungin), an echinocandin antifungal, for adults aged 18 and older with candidemia and invasive candidiasis, particularly when other treatment options are limited or unavailable.
Indication Insights
The candidiasis segment held the largest market share in 2024. In the U.S., candidiasis, including candidemia and invasive candidiasis, remains the most common systemic fungal infection, driven by factors such as the widespread use of central venous catheters, broad-spectrum antibiotics, and an increasing population of immunocompromised patients, such as those undergoing cancer treatment or transplants and patients in ICU settings. These risk factors lead to high morbidity and mortality, making early and effective systemic treatment crucial. In March 2023, Scynexis signed an exclusive license agreement with GSK to commercialize ibrexafungerp (Brexafemme) for treating vulvovaginal candidiasis (VVC) and to reduce the incidence of recurrent VVC and to continue to develop ibrexafungerp , also currently in a phase III trial for potential treatment of a life-threatening fungal infection. invasive candidiasis (IC).
The aspergillosis segment is expected to register a significant CAGR during the forecast period. The incidence of aspergillosis is rising in the U.S. antifungal drugs industry, especially among patients with hematologic cancers, transplant recipients, and those recovering from severe viral pneumonia. In September 2024, F2G secured USD 100 million in funding from investors, including the AMR Action Fund, supported by major pharmaceutical companies such as Johnson & Johnson and Pfizer, to advance the development of treatments targeting drug-resistant “superbug” infections.
Dosage Form Insights
Oral drugs dominated the market in 2024 due to their convenience, effective systemic absorption, and suitability for inpatient step-down and outpatient treatment. Physicians commonly prescribe oral medications such as fluconazole, voriconazole, and posaconazole for step-down therapy in candidemia and long-term prophylaxis in high-risk patients such as hematopoietic stem cell transplant recipients. Their ease of use improves patient adherence, while well-established pharmacokinetic and safety profiles facilitate monitoring and dose adjustments. The rising number of immunocompromised patients and a growing focus on outpatient care for milder infections continue to boost the demand for oral antifungals. In September 2024, Basilea launched the Phase 3 FAST-IC study to evaluate fosmanogepix, a first-in-class broad-spectrum antifungal treatment cadidare with a novel mechanism, available in both Intravenous (IV) and oral forms, for treating Candidemia
The ointments segment is expected to grow at the fastest CAGR over the forecast period. This can be attributed to its convenience in managing mild fungal infections, including dermatophytosis, onychomycosis, and other superficial fungal infections. Advances in lipid-based and nano-emulsion technologies improve skin absorption and patient comfort, while new excipients help reduce application frequency and irritation. Dermatologists and primary care providers increasingly recommend ointments for mild to moderate infections when systemic treatments are unsuitable, boosting retail pharmacy sales and OTC use. In January 2024, Vanda Pharmaceuticals Inc. (VNDA) received FDA approval for its Investigational New Drug application for VTR-297, intended to treat onychomycosis.
Distribution Channel Insights
The hospital pharmacies segment held the largest share of the U.S. antifungal drugs industry in 2024 due to their key role in treating severe and systemic fungal infections, particularly in immunocompromised patients in ICU and transplant units. Hospitals also serve as a primary hub for early diagnosis and prompt treatment, which is vital for managing high-mortality fungal diseases. In August 2023, Sandoz acquired Mycamine (micafungin sodium) from Astellas Pharma. This acquisition strengthened Sandoz’s hospital antifungal portfolio, especially for treating invasive and esophageal candidiasis.
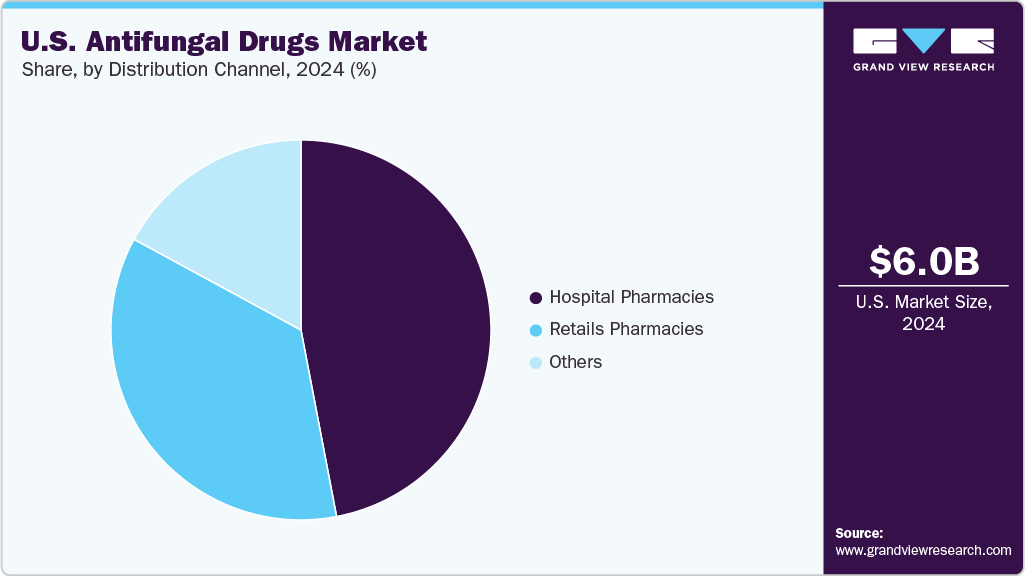
The retail pharmacies segment is expected to grow at a significant CAGR over the forecast period. This is due to the rising cases of superficial fungal infections such as athlete’s foot, vaginal candidiasis, and ringworm. These conditions are often self-diagnosed and treated with OTC antifungal creams, sprays, and oral medications. The ease of walk-in access, growing consumer awareness, and e-commerce integration with major pharmacy chains such as CVS and Walgreens have boosted market adoption. Furthermore, the wider availability of branded generics and pharmacist-led consultations for recurring infections has enhanced access and treatment adherence.
Key U.S. Antifungal Drugs Company Insights
Some key players operating in the market include Astellas Pharma US, Inc.;Pfizer Inc.; Merck & Co., Inc.; and Abbott.
- Astellas Pharma US, Inc. is a well-known provider of systemic antifungal therapies. It leverages its broad-spectrum azole portfolio to treat invasive and preventive needs in immunocompromised patients. Its client base includes transplant centers, oncology centers, and infectious disease specialists.
Key U.S. Antifungal Drugs Companies:
- Novartis Pharmaceuticals Corporation
- Pfizer Inc.
- Bayer AG
- Sanofi.
- Merck & Co., Inc.
- GSK plc.
- Abbott.
- GLENMARK PHARMACEUTICALS INC., USA
- Enzon Pharmaceuticals, Inc.
- Astellas Pharma US, Inc.
U.S. Antifungal Drugs Market Report Scope
Report Attribute
Details
Revenue forecast in 2030
USD 7.20 billion
Growth rate
CAGR of 3.0% from 2025 to 2030
Base year for estimation
2024
Historical data
2018 - 2023
Forecast period
2025 - 2030
Quantitative units
Revenue in USD Billion and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Drug class, indication, dosage form, distribution channel
Key companies profiled
Novartis Pharmaceuticals Corporation; Pfizer Inc.; Bayer AG; Sanofi.; Merck & Co., Inc.; GSK plc.; Abbott.; GLENMARK PHARMACEUTICALS INC., USA; Enzon Pharmaceuticals, Inc.; Astellas Pharma US, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Antifungal Drugs Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2018 to 2030. For the purpose of this report, Grand View Research has segmented the U.S. antifungal drugs market on the basis of drug class, indication, dosage form, and distribution channel:
-
Drug Class Outlook (Revenue, USD Billion, 2018 - 2030)
-
Azoles
-
Echinocandins
-
Polyenes
-
Allylamines
-
Others
-
-
Indication Outlook (Revenue, USD Billion, 2018 - 2030)
-
Dermatophytosis
-
Aspergillosis
-
Candidiasis
-
Others
-
-
Dosage Form Outlook (Revenue, USD Billion, 2018 - 2030)
-
Oral Drugs
-
Ointments
-
Powders
-
Others
-
-
Distribution Channel Outlook (Revenue, USD Billion, 2018 - 2030)
-
Hospital Pharmacies
-
Retails Pharmacies
- Others
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










