- Home
- »
- Medical Devices
- »
-
U.S. Cell & Gene Therapy Contract Research Organizations Market Report, 2033GVR Report cover
![U.S. Cell & Gene Therapy Contract Research Organizations Market Size, Share & Trends Report]()
U.S. Cell & Gene Therapy Contract Research Organizations Market (2025 - 2033) Size, Share & Trends Analysis Report By Type (Drug Discovery, Preclinical, Clinical), By Service, By Indication, By Modality, And Segment Forecasts
- Report ID: GVR-4-68040-688-5
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
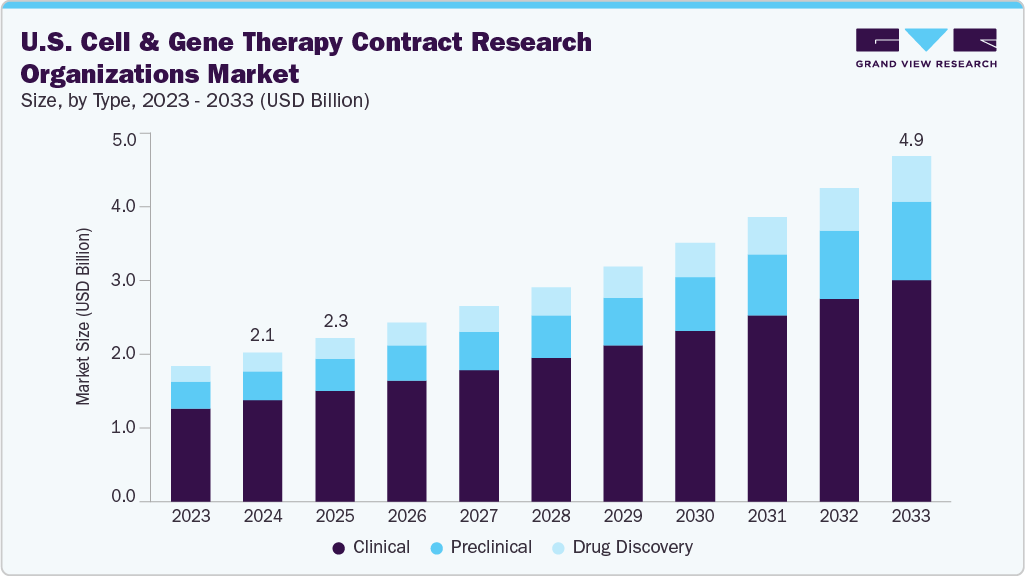
The U.S. cell & gene therapy contract research organizations market size was estimated at USD 2.09 billion in 2024 and is projected to reach USD 4.86 billion by 2033, growing at a CAGR of 9.91% from 2025 to 2033. The growth of the market is due to growing complexity of cell- and gene-based therapeutics, rising clinical trial volume in CGT, increased outsourcing by biotech firms, regulatory complexity, among others.
The market is being driven by growing collaboration among scientific innovation, regulatory changes, and evolving sponsor needs. A growing number of advanced therapies-including autologous cell therapies, gene editing interventions, and in vivo viral vector-based treatments-are reaching investigational and clinical stages, often requiring highly tailored preclinical and clinical development programs. Most small-to-midsize biotech firms lack the internal infrastructure to navigate the complex clinical, regulatory, and bioanalytical demands of such therapies, especially those targeting rare or ultra-rare conditions. This gap fuels demand for CROs with niche expertise in translational biology, GMP-compliant sample management, and immunogenicity testing.
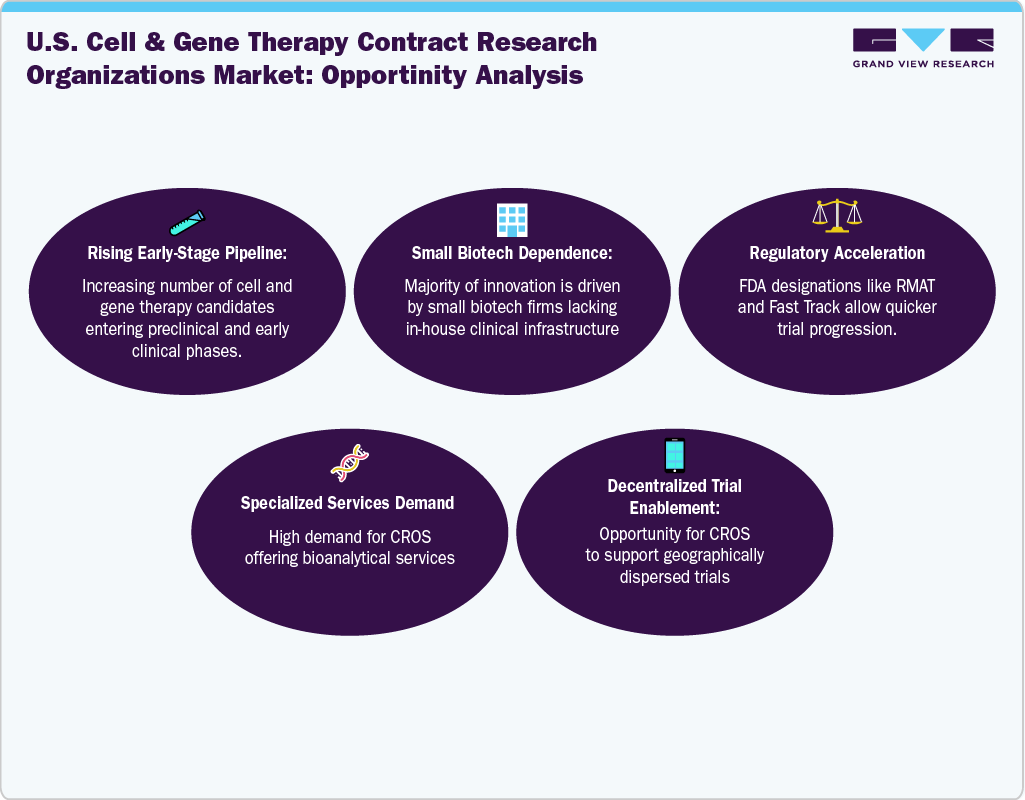
Furthermore, an increase in strategic outsourcing models, where sponsors form integrated partnerships with CROs to reduce fixed operational costs and increase scalability, is also one of the factors driving the market growth. The high logistical and patient-specific complexity of therapies such as CAR-T or AAV-based interventions necessitates deep coordination between trial sponsors, clinical sites, and regulatory bodies, prompting sponsors to engage CROs that bring both scientific depth and operational agility. The expansion of academic-industry partnerships, government initiatives supporting rare disease trials, and growing venture capital inflows into cell and gene therapy startups further amplify CRO demand.
Technological Advancements
The technological landscape of the U.S. cell and gene therapy CRO market is evolving rapidly, driven by the need for greater precision, efficiency, and regulatory compliance in clinical development. CROs are integrating advanced tools such as artificial intelligence for patient stratification and protocol optimization, cloud-based platforms for real-time trial monitoring, and high-throughput genomic and proteomic analytics for deeper biological insights. Digital twin modeling and in-silico simulations are also being explored to predict patient responses and streamline trial design.

Pricing Analysis
The pricing model in the U.S. cell and gene therapy CRO market is shaped by the high complexity, customization, and regulatory sensitivity of these therapies. Unlike traditional clinical trials, pricing here is often based on a hybrid model that blends fixed-fee and milestone-based components. Fixed pricing is typically used for well-defined preclinical services (e.g., assay development, toxicology), while milestone-based pricing is applied across clinical phases tied to deliverables such as IND submission, patient recruitment targets, or data lock. Due to the variability in protocol design, patient population (often rare), and the need for specialized bioanalytical assays, time-and-materials (T&M) pricing is frequently used for exploratory studies or evolving trial scopes.

Type Insights
On the basis of type segment, the market is classified into drug discovery, preclinical, and clinical. The clinical segment accounted for the largest revenue share in the cell and gene therapy CRO industry of 68.43% in 2024. The growth of the segment is due to the sharp increase in investigational new drug (IND) applications and first-in-human studies involving complex genetic and cellular constructs.
Preclinical segment is anticipated to grow at a fastest CAGR during the forecast period. The segment growth is driven due to the rising volume of early-stage cell and gene therapy candidates entering the pipeline, particularly from small biotech firms and academic spin-offs. These therapies require highly specialized preclinical testing services such as vector characterization, biodistribution studies, in vivo toxicology, immunogenicity assessments, and mechanism-of-action analyses-all of which must be conducted under strict regulatory frameworks. The need for sophisticated animal models and GLP-compliant laboratories has driven sponsors to partner with CROs offering integrated preclinical platforms tailored to cell and gene therapies.
Service Insights
On the basis of service segment, the market is segregated into project & clinical trial management, regulatory strategy, data management & medical writing, clinical monitoring, quality management / GMP compliance, biostatistics & safety monitoring, patient & site recruitment, technology transfer, and others. The clinical monitoring segment held the largest market share in 2024 due the highly specialized nature of cell and gene therapy trials, which require intensive oversight to ensure patient safety, protocol adherence, and data integrity. These therapies often involve complex procedures such as leukapheresis, autologous cell reinfusion, or direct in vivo vector delivery, which demands close supervision at each clinical site.
The regulatory strategy segment is anticipated to grow at the fastest CAGR over the forecast period. The growth of the segment is due to the increasing complexity of navigating the evolving regulatory landscape for cell and gene therapies. These therapies often require customized regulatory pathways, including accelerated approval programs such as RMAT and orphan drug designations, which demand specialized expertise to efficiently manage submissions and agency interactions. Sponsors, especially smaller biotech firms, depend heavily on CROs to develop robust regulatory strategies that ensure compliance, facilitate faster approvals, and reduce time to market.
Indication Insights
On the basis of indication segment, the market is segregated into oncology, CNS Disorders, infectious diseases, immunological disorders, cardiovascular diseases, respiratory diseases, diabetes, ophthalmology, pain management, and others. The oncology segment held the largest market share in 2024 due to the rapid advancements and widespread adoption of cell and gene therapies targeting various cancers, including hematologic malignancies and solid tumors. The success of CAR-T therapies and other gene-modified immune cell treatments has significantly expanded the pipeline of oncology-focused clinical trials.
The CNS disorders segment is anticipated to grow at the fastest CAGR over the forecast period. The growth of the segment is due to the rising focus on gene and cell-based therapies for previously untreatable or poorly managed neurological conditions such as Parkinson’s disease, Alzheimer’s disease, amyotrophic lateral sclerosis (ALS), and rare inherited neurodegenerative disorders. Traditional treatment options for these disorders often offer limited efficacy and fail to address the underlying causes, creating a strong incentive for the development of regenerative and gene-modifying therapies.
Modality Insights
On the basis of modality segment, the market is segregated into cell-based therapies, gene therapies, and gene-modified cell therapies. The cell-based therapies segment held the largest market share in 2024 due to the widespread adoption of autologous and allogeneic cell therapies across multiple therapeutic areas, especially oncology and regenerative medicine. These therapies, such as CAR-T and stem cell treatments, have demonstrated significant clinical success, driving increased clinical trial activity and commercialization efforts.
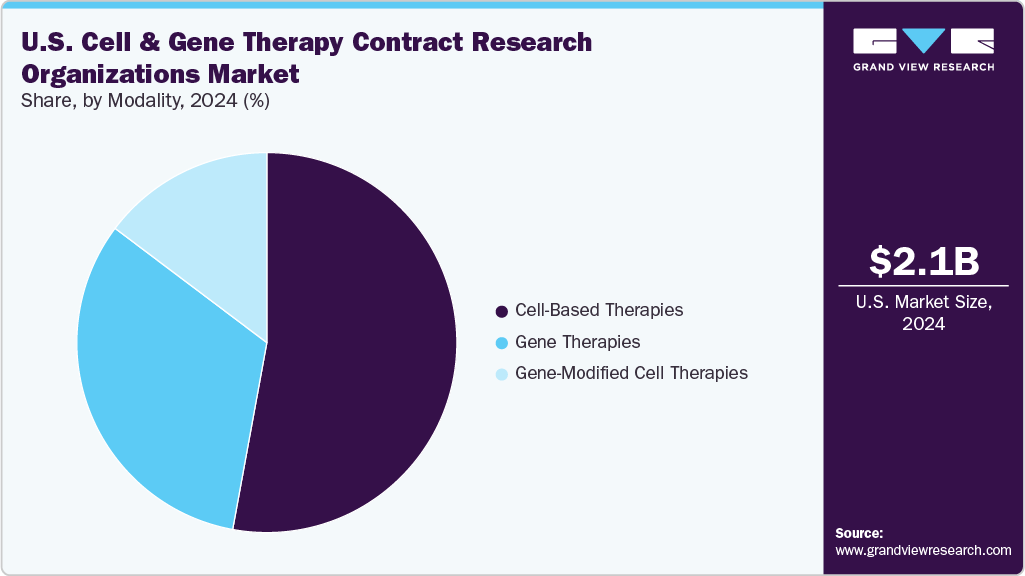
In addition, the gene therapies segment is anticipated to witness the fastest CAGR growth during the forecast period. This growth can be attributed to the increasing number of gene therapy candidates entering clinical pipelines, driven by advancements in gene editing tools such as CRISPR, AAV-based vectors, and lipid nanoparticle delivery systems. Gene therapies offer the potential for curative treatment of a wide range of genetic, metabolic, and rare disorders by directly addressing the root cause at the molecular level.
Key U.S. Cell and Gene Therapy Contract Research Organizations Company Insights
Several key players are acquiring various strategic initiatives to strengthen their market position offering diverse services to customers. The prominent strategies adopted by companies are service launches, mergers & acquisitions/joint ventures merger, partnership & agreements, expansions, and others to increase market presence & revenue and gain a competitive edge drives the market growth.
Key U.S. Cell and Gene Therapy Contract Research Organizations Companies:
- Altasciences
- Allucent
- Labcorp
- Linical
- Medpace
- Thermo Fisher Scientific Inc.
- Precision Medicine Group, LLC.
- QPS Holdings
- Syneos Health
- ICON plc
Recent Developments
-
In April 2024, Labcorp announced the expansion of its precision oncology portfolio to support pharmaceutical, biopharma, and clinical research in advancing drug development programs. The expansion solidifies the company's commitment to advancing cancer research and patient care by investing in scientific, diagnostic, and laboratory innovations.
-
In June 2025, Thermo Fisher Scientific, Inc. entered into a collaboration agreement with Cellular Origins, with an aim to transform production output and labor requirements for cell and gene therapy (CGT) manufacturing using automated robotics capabilities.
U.S. Cell & Gene Therapy Contract Research Organizations Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 2.28 billion
Revenue forecast in 2033
USD 4.86 billion
Growth rate
CAGR of 9.91% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, service, indication, modality
Key companies profiled
Altasciences; Allucent; Labcorp; Linical; Medpace; Thermo Fisher Scientific Inc.; Precision Medicine Group, LLC.; QPS Holdings; Syneos Health; ICON plc
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Cell & Gene Therapy Contract Research Organizations Market Report Segmentation
This report forecasts revenue growth at country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. cell & gene therapy contract research organizations market report based on type, phase, service, indication, and modality:
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Drug Discovery
-
Target Validation
-
Lead Identification
-
Lead Optimization
-
-
Preclinical
-
Clinical
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Project & Clinical Trial Management
-
Regulatory Strategy
-
Data Management & Medical Writing
-
Clinical Monitoring
-
Quality Management / GMP Compliance
-
Biostatistics & Safety Monitoring
-
Patient & Site Recruitment
-
Technology Transfer
-
Others
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
CNS Disorders
-
Infectious Diseases
-
Immunological Disorders
-
Cardiovascular Diseases
-
Respiratory Diseases
-
Diabetes
-
Ophthalmology
-
Pain Management
-
Others
-
-
Modality Outlook (Revenue, USD Million, 2021 - 2033)
-
Cell-Based Therapies
-
Gene Therapies
-
Gene-Modified Cell Therapies
-
Frequently Asked Questions About This Report
b. The U.S. cell & gene therapy contract research organizations market size was estimated at USD 2.09 billion in 2024 and is expected to reach USD 2.28 billion in 2025.
b. The U.S. cell & gene therapy contract research organizations market is expected to grow at a compound annual growth rate of 9.91% from 2025 to 2033 to reach USD 4.86 billion by 2033.
b. Clinical segment dominated the U.S. cell & gene therapy contract research organizations market with a share of 68.43% in 2024. The growth of the segment is due to the sharp increase in investigational new drug (IND) applications and first-in-human studies involving complex genetic and cellular constructs.
b. Some key players operating in the U.S. cell & gene therapy contract research organizations market include Altasciences, Allucent, Labcorp, Linical, Medpace, Thermo Fisher Scientific Inc., Precision Medicine Group, LLC., QPS Holdings, Syneos Health, ICON plc
b. Key factors that are driving the market growth include growing complexity of cell- and gene-based therapeutics, rising clinical trial volume in CGT, and increased outsourcing by biotech firms, regulatory complexity, among others.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










