- Home
- »
- Medical Devices
- »
-
U.S. Deep Brain Stimulation In Parkinson’s Disease Market Report, 2030GVR Report cover
![U.S. Deep Brain Stimulation In Parkinson’s Disease Market Size, Share & Trends Report]()
U.S. Deep Brain Stimulation In Parkinson’s Disease Market (2025 - 2030) Size, Share & Trends Analysis Report By Product (Single-channel, Dual-channel), Competitive Analysis, And Segment Forecasts
- Report ID: GVR-4-68039-951-4
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2018 - 2023
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
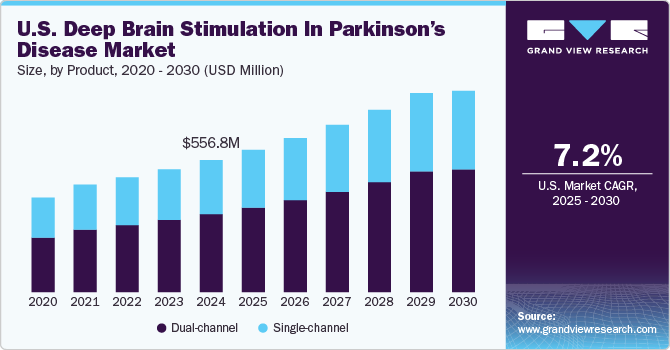
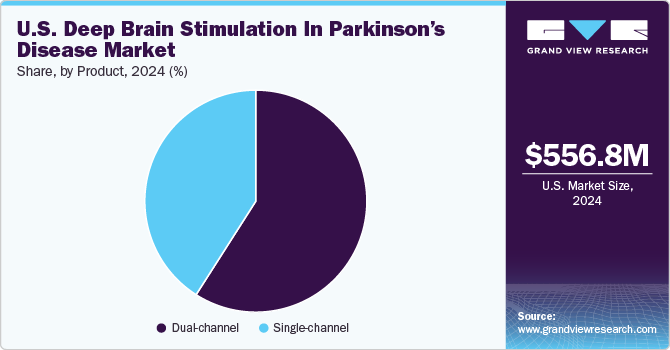
The U.S. deep brain stimulation in parkinson’s disease market size was estimated at USD 556.79 million in 2024 and is projected to grow at a CAGR of 7.19% from 2025 to 2030. The market is driven by numerous factors, such as technological advancements in deep brain stimulation (DBS) devices, increasing prevalence of Parkinson’s disease (PD), and government funding for research and development. Through the allocation of funds to research initiatives, the governments actively support groundbreaking discoveries, foster technological innovations, and promote the development of more effective DBS technologies.
For instance, In October 2024, the National Institute of Neurological Disorders and Stroke (NINDS) received funding through the National Advisory Neurological Disorders and Stroke (NANDS) Council. The NANDS Council comprises scientists, clinicians, and public members who advise the NINDS on research priorities, policies, and program planning. As per the data published in December 2023 by the National Institute of Neurological Disorders and Stroke (NINDS), PD research received support from NINDS with a contribution of approximately USD 125 million to the total funding of USD 259 million provided by the National Institutes of Health (NIH). This substantial financial support from NINDS highlighted the commitment to advancing research & understanding of PD, aiming to make strides in its diagnosis, treatment, and overall management.
Increasing government funding propels scientific advancements and encourages collaboration between researchers, medical professionals, and technology developers. The aim is to improve the lives of individuals suffering from PD by advancing the development of DBS therapies. In addition, government support can contribute to making DBS technology more accessible to a broader patient population. This may include initiatives to reduce costs, improve insurance coverage, or subsidize expenses associated with DBS procedures. It plays a pivotal role in catalyzing this progress, propelling market growth, and creating a vibrant landscape for research & development endeavors.
Technological advancements in DBS, which acts as abrainpacemaker for the treatment of PD, are anticipated to propel the market growth in the forecast period. Innovations, such as robot-assisted implantation, enhanced microelectrode designs, multi-target stimulation, rechargeable Implantable Pulse Generators (IPGs), personalized directed programming, and integration of Magnetic Resonance Imaging (MRI), are opening new possibilities. These breakthroughs not only enhance the precision and efficiency of DBS procedures but also pave the way for personalized & more effective treatments. Ongoing advancements in technology enhance DBS treatments, offering enhanced prospects for patients in search of neurostimulation therapies.
For instance, in January 2024, The FDA approved new deep brain stimulation (DBS) systems and software designed to treat movement disorders, including Parkinson's disease. Abbott's Liberta RC and Medtronic's Percept RC are among these systems, both of which offer advanced options for managing these conditions. Additionally, innovative software and algorithms are being developed to enhance the effectiveness and personalization of Parkinson's disease treatments, further advancing the field of neurostimulation.
These new DBS systems provide more precise stimulation and advanced algorithms, improving motor control and reducing symptoms such as tremors, rigidity, and bradykinesia. Advanced software integration enables real-time monitoring and adaptive programming, tailoring the treatment to individual patient needs and response patterns. Systems such as Liberta RC and Percept RC are designed with rechargeable capabilities, reducing the frequency of surgeries required for battery replacement and enhancing patients' convenience. Advanced algorithms and software simplify programming and maintenance, improving the overall experience for patients and healthcare providers.
Moreover, the growing incidence of Parkinson’s disease plays a significant role in driving the market for DBS devices. This condition results from a combination of genetic factors, such as mutations in the alpha-synuclein gene, and environmental influences, including repeated head injuries. Age remains a primary risk factor for the development of PD, highlighting the importance of addressing this health concern. For instance, As of May 2024, Parkinson's Europe estimated that approximately 10 million people worldwide have Parkinson's disease. The number of cases is increasing due to longer life expectancy. According to Parkinson's Foundation's 2022 estimates, number of individuals living with PD was projected to reach 1.2 million by 2030. In the U.S., 90,000 people received PD diagnosis, surpassing numbers for muscular dystrophy, multiple sclerosis, and amyotrophic lateral sclerosis combined. Furthermore, approximately 60,000 new cases of PD are identified each year in the U.S. Rising incidence of Parkinson's Disease (PD) has led to an increased need for DBS as a viable treatment option. DBS has proven essential in providing effective solutions for managing and relieving PD symptoms.
Market Concentration & Characteristics
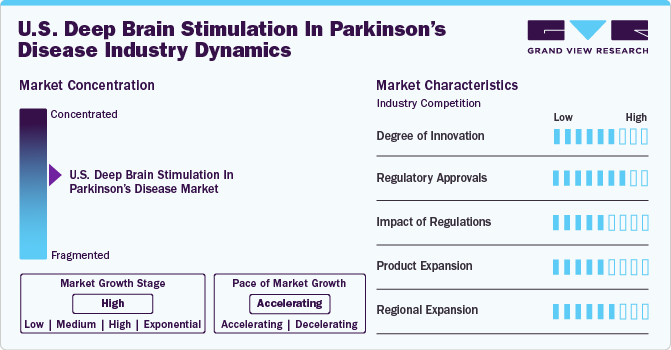
The market growth stage is high, and the pace is accelerating. The market is characterized by increasing cases of PD, technological advancements, and increasing regulatory approval for novel product launches.
Key strategies implemented by major players and government entities are increasing clinical studies for assessments for PD, acquisitions, partnerships, collaborations, etc. In November 2023, Surgical Information Systems (SIS) revealed a novel dubbed VISION (NCT05774041), a post-marketed clinical study focusing on exploring the potential of SIS visualization technology to enhance the precision of DBS for people dealing with PD. The main goal was to evaluate how well this technology can improve the accuracy of DBS procedures, by offering notable advancements in the treatment of PD. This research aimed to contribute valuable insights into refining methods and effectiveness of DBS, ultimately benefiting individuals affected by PD
Innovation in the precision of electrode placement through advanced targeting techniques and imaging technologies helps optimize therapeutic outcomes and minimize side effects, enhancing the degree of innovation
The major regulatory authority overseeing medical devices in the U.S. is the FDA. In the case of DB devices, which act as abrainpacemaker intended for treating PD, FDA approval is a prerequisite before these devices can be sold and employed for clinical applications
Continuous regulatory oversight involves ongoing surveillance of the safety and performance of DBS devices post-market release. This monitoring is essential for ensuring the long-term safety of patients. Regulatory bodies set forth guidelines for the proper use and labeling of DBS devices, aiming to provide healthcare professionals and patients with precise information
Manufacturers may develop electrodes with enhanced designs to enhance accuracy and effectiveness in targeting specific regions of the brain. Upgrades to programming software can provide healthcare providers with additional customization options, allowing them to optimize therapy more precisely based on individual patient responses
The U.S. deep brain stimulation in Parkinson’s disease industry is characterized by high concentration, with a few key players accounting for the majority of market share. These players typically hold strong positions due to factors, such as established brand recognition, extensive distribution networks, and significant investments in R&D. Market concentration has led to intense competition among these companies, driving innovation and the development of new technologies. It also influences pricing strategies, with companies often leveraging their market dominance to maintain premium pricing for their products. However, regulatory scrutiny and the potential for disruptive innovations could impact market concentration in the future
The U.S. has a significant population of target patients, contributing to a substantial market for DBS as a treatment option. Advances in DBS technology, including improvements in electrode design, programming algorithms, and surgical techniques, can impact market growth
Product Insights
The dual-channel segment led the market with the largest revenue share of 59.04% in 2024 and is expected to register at the fastest CAGR from 2025 to 2030. Dual-channel systems can target multiple brain regions simultaneously. This can be beneficial for patients with complex movement disorders or those experiencing a range of symptoms that may require separate stimulation parameters. Dual-channel DBS offers greater flexibility in adjusting and optimizing therapy for patients with diverse & evolving symptoms. As compared to single-channel systems, dual-channel DBS allows for personalized treatment approaches. Flexibility to stimulate different brain regions independently can be valuable in tailoring therapy to the specific needs of each patient. Moreover, the growing adoption of double-channel DBS for treating Parkinson’s disease is one of the key factors driving segment growth.
The new product launches and technological advancements are facilitating segment growth. For instance, in January 2024, Medtronic Plc announced the U.S. FDA confirmation for its Percept RC DBS system. As the latest addition to Medtronic Percept portfolio, comprising BrainSense technology, Percept PC neurostimulator, and SenSight directional leads, this rechargeable neurostimulator sets a new standard in sensing-enabled DBS systems. Uniquely personalized for patients with movement disorders like PD, essential tremor, dystonia, and epilepsy, the Percept portfolio empowers physicians to tailor treatments. The Percept RC is the most compact and delicate dual-channel neurostimulator in this market. The product is readily available across the U.S., and it is further supported by obtaining CE Mark approval for distribution in Europe. In addition, it is accessible in the Japanese market.
Key U.S. Deep Brain Stimulation In Parkinson’s Disease Company Insights:
LivaNova PLC and Functional Neuromodulation, Ltd. are some of the emerging players in the U.S. deep brain stimulation in Parkinson’s disease industry. The market has experienced a notable expansion in recent years, propelled by technological advancements, rising PD prevalence, and a growing elderly demographic. DBS, a surgical intervention, entails the implantation of a device similar to a pacemaker within the brain, administering electrical impulses to mitigate Parkinson's symptoms, including tremors and rigidity. New entrants are leveraging technological advancements to introduce innovative solutions and gain a competitive edge.
Start-ups and smaller companies are actively engaged in R&D to bring forth novel therapies and enhance existing treatment options. Moreover, the growing patient pool and rising awareness about the benefits of DBS have created opportunities for market entry. Companies are focusing on strategic collaborations, partnerships, and mergers to strengthen their industry position and expand their product portfolios. In addition, increasing investments in clinical trials and regulatory approvals are helping emerging players navigate the complex landscape of the market.
Key U.S. Deep Brain Stimulation In Parkinson’s Disease Companies:
- ABBOTT
- Medtronic
- Boston Scientific Corporation
Recent Developments
-
In January 2025, Medtronic achieved CE Mark approval for its BrainSense Adaptive Deep Brain Stimulation and Electrode Identifier, a significant advancement in personalized care for individuals with Parkinson's disease. This breakthrough leverages innovative brain-computer interface technology, offering new possibilities in real-time patient monitoring and therapy adjustment.
-
In September 2024, Abbott announced the initiation of a clinical trial, named the TRANSCEND study, to evaluate the effectiveness of its deep brain stimulation system in managing treatment-resistant depression (TRD), a severe form of major depressive disorder (MDD). This trial marks a significant step in exploring DBS as a potential therapeutic option for patients with TRD who do not respond to conventional treatments such as medication and psychotherapy. The TRANSCEND study and its use of DBS for treatment-resistant depression share a deep connection with the use of DBS in Parkinson’s disease.
-
In August 2024, Medtronic received FDA approval for Asleep Deep Brain Stimulation (DBS) surgery for Parkinson's disease and essential tremor. This is the first time a company has been approved to offer DBS surgery while a patient is asleep or awake.
-
In March 2023, Michigan Tech researchers successfully advanced the field of DBS systems for Parkinson's patients by integrating neuromorphic computing. This innovative approach aimed to enhance the efficiency and energy utilization of DBS systems, crucial for managing Parkinson's disease. Neuromorphic computing, often referred to as brain-inspired or neuroscience-powered artificial intelligence (AI), employs microchips and algorithms to replicate the functionality of a nervous system. The application of this technology marks a significant evolution in optimizing the therapeutic capabilities of DBS systems while concurrently addressing energy
U.S. Deep Brain Stimulation In Parkinson’s Disease Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 600.22 million
Revenue forecast in 2030
USD 849.50 million
Growth rate
CAGR of 7.19% from 2025 to 2030
Base year for estimation
2024
Historical data
2018 - 2023
Forecast period
2025 - 2030
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends, and volume analysis
Segments covered
Product
Key companies profiled
ABBOTT; Medtronic; Boston Scientific Corporation
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Deep Brain Stimulation In Parkinson’s Disease Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the U.S. deep brain stimulation in Parkinson’s disease market report based on product:
-
Product Outlook (Revenue, USD Million, 2018 - 2030)
-
Single-channel
-
Dual-channel
-
Frequently Asked Questions About This Report
b. The U.S deep brain stimulation in Parkinson's disease market size was estimated at USD 556.79 million in 2024 and is expected to reach USD 600.22 million in 2025.
b. The U.S deep brain stimulation in Parkinson's disease market is expected to grow at a compound annual growth rate of 7.19% from 2025 to 2030 to reach USD 849.50 million by 2030.
b. Dual-channel segment dominated the U.S deep brain stimulation in Parkinson's disease market with a share of 59.04% in 2024. Dual channel DBS offers the best overall support, exhibiting greater effectiveness in reducing patient tremors and enhancing the quality of life.
b. There are three major players in the market—Medtronic, Abbott, and Boston Scientific—with FDA-approved DBS systems for Parkinson’s disease. Among these, Medtronic has been in the market for a longer duration compared to Abbott and Boston Scientific. All three players give unique service and product offerings such as greater stimulation, increased contact points, greater battery life, and ease of implantation, catering to a broad range of patients affected with PD.
b. The major factors impacting the growth of the U.S deep brain stimulation devices in Parkinson's disease market are introduction of technologically advanced products, increasing incidence of chronic diseases such as PD and supportive government initiatives.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










