- Home
- »
- Medical Devices
- »
-
U.S. Dural Sealants And Fibrin Glues Market Report, 2035GVR Report cover
![U.S. Dural Sealants And Fibrin Glues Market Size, Share & Trends Report]()
U.S. Dural Sealants And Fibrin Glues Market (2025 - 2035) Size, Share & Trends Analysis Report By CPT Procedure Code (61510 / 61512, 61320 - 61323, 61518, 62140 / 62141, 63707, 63709, 63710), By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-810-6
- Number of Report Pages: 125
- Format: PDF
- Historical Range: 2025 - 2035
- Forecast Period: 2025 - 2035
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
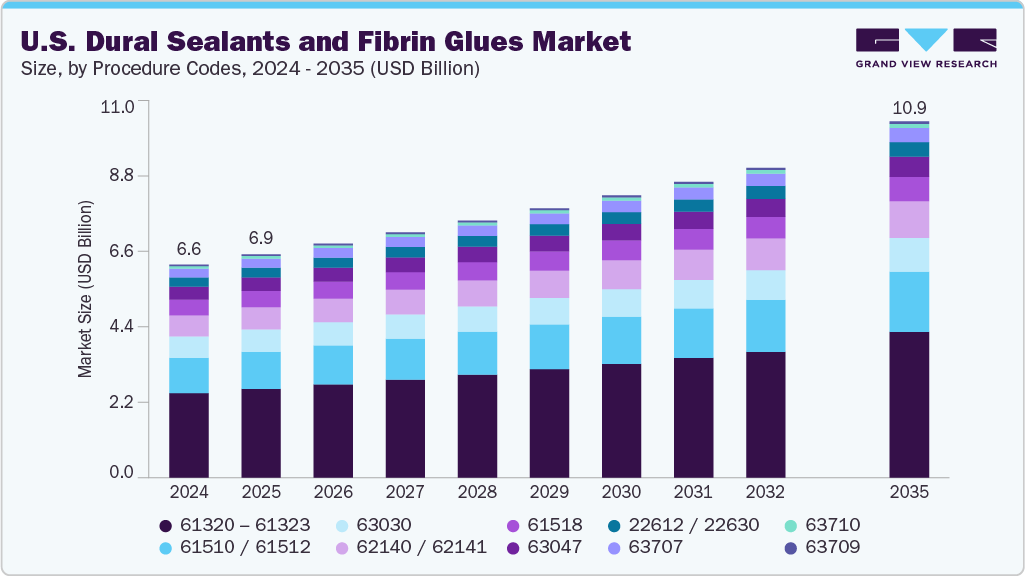
The U.S. dural sealants and fibrin glues market size was valued at USD 6.57 billion in 2024 and is expected to reach USD 10.99 billion by 2035, growing at a CAGR of 4.78% from 2025 to 2035. Growth is driven by increasing neurosurgical and spinal procedure volumes, particularly complex cranial, posterior fossa, and spinal duraplasty cases where cerebrospinal fluid (CSF) leak prevention is critical.
According to a 2024 study published in the Journal of Neurosurgery: Spine by Dykhouse et al., spine procedure volumes in the U.S. increased from approximately 800,000 in 2013 to over 1.1 million in 2022, representing a steady and sustained growth. This expansion is closely linked with a surge in degenerative spine disorders, aging demographics, and improved surgical safety, particularly for older patients.
Hospitals and ambulatory surgical centers prioritize sealants and glues that ensure watertight closure, reduce postoperative complications, and support value-based outcomes. The rising adoption of advanced products, such as PEG-based hydrogels, collagen grafts, and synthetic sealants, along with favorable reimbursement and increasing outpatient surgical volumes, further fuels market expansion. Hospitals and surgical centers are prioritizing products that minimize cerebrospinal fluid (CSF) leaks, shorten recovery times, and lower readmission rates. According to the Agency for Healthcare Research and Quality (AHRQ), postoperative CSF leaks account for up to 3-8% of neurosurgical complications, often requiring revision surgery. These reoperations not only elevate costs by nearly USD 25,000-USD 40,000 per case, but also significantly extend hospital stays and increase infection risk.
Moreover, the accelerating adoption of robotic-assisted and image-guided surgery (IGS) presents a clear growth pathway for dural sealants and fibrin glues in the U.S. As neurosurgeons and spine teams migrate toward precision platforms, demand rises for adjunctive products that are reliable, compatible with minimally invasive workflows, and deliver predictable watertight closure in constrained operative fields. A 2025 scientometric analysis published in Cureus reviewed the publication trends for dural sealants in spine surgery. It found that nearly 40% of research originates in the U.S., and recent research topics have shifted from “types of sealants” to “clinical outcomes, safety, efficacy, and workflow integration”.
Furthermore, an industry review of surgical robotics published by MPO Magazine in October 2025 described the field entering a “new phase” where platforms emphasize software-led differentiation, outpatient redesign and compatibility with adjunct devices. These data points demonstrate a growing intersection between advanced surgical platforms (robotics/IGS) and dural adjunct technologies, creating a substantive momentum for sealants and glues optimized for these environments. These factors are collectively contributing to the growth of market.
Market Concentration & Characteristics
TheU.S. dural sealants and fibrin glues industry is a moderately concentrated market where Integra LifeSciences, Stryker, Baxter, Ethicon (J&J) have the most significant market share. These companies provide a comprehensive range of advanced solutions across orthopedics, spine, and neurosurgery. The dural sealants and fibrin glues market is continually evolving with innovations, particularly through biocompatible materials and next-generation sealant delivery systems. Additionally, the rising prevalence of neurosurgical and spinal disorders with increasing treatment procedure, is fueling market growth and encouraging the entry of new players offering innovative surgical solutions.
The dural sealants and fibrin glues industry in the U.S. is undergoing major innovations and advancements. In March 2025, the company Pramand Inc. announced the launch of “SpineSeal Spine Sealant”, specifically marketed for spinal dural repair adjunctive use, signaling active commercial innovation in the dural graft (spinal) segment. Similarly, emerging innovations such as PEG-based hydrogel sealants and next-generation fibrin matrices are reducing CSF leak recurrence rates by up to 50% compared to traditional suturing or patch-only methods. These materials are being integrated into image-guided and robotic-assisted workflows, where surgical precision demands flexible, biocompatible, and rapidly polymerizing biomaterials.

Partnerships and collaborations are significant in improving access to emerging advanced and innovative solutions in dural sealants and fibrin glues industry. For instance, in November 2022 Ethicon’s collaborated with CMR Surgical which supports the U.S. dural sealants and fibrin glues market by expanding minimally invasive and robotic surgical access. As robotic neurosurgery adoption grows, demand for adjunctive biomaterials like dural sealants and fibrin glues will rise, enhancing precision and outcomes in cranial and spinal procedures.
The dural sealants and fibrin glues industry is regulated strictly to ensure safety, efficacy, and reliability. In the U.S., dural sealants and fibrin glues are regulated as biologics (BLAs) under FDA’s vaccines/blood & biologics center. FDA regulates pharmacovigilance activities for dural sealants and fibrin glues for performing clinical procedures. Manufacturers must comply with the labeling requirements of CPT procedure to ensure these solutions continue to perform safely and effectively.
Availability and improved efficacy of alternative dural closure techniques, which reduce reliance on sealant-based adjuncts works as an alternative solution for the dural sealants and fibrin glues. For example, a 2025 technical note published in Acta Neurochirurgica described a uni-portal full-endoscopic dural repair technique using standard instruments plus a custom knot-pusher derived from a drain catheter no specialized sealant or glue system required. Similarly, a recent retrospective systematic review found that in spinal surgery specifically, primary closure combined with a patch or graft achieved a CSF-leak rate of 5.5%, whereas adding sealant alone gave 13.7% leakage rate suggesting that sealants may not always provide incremental benefit over established mechanical methods.
CPT Procedure Codes Insights
The 61320 - 61323 - craniectomy or craniotomy, decompressive (with or without duraplasty) segment had the largest market share of 39.65% in 2024. This dominance is attributed to the high clinical importance of this segment. A decompressive craniectomy or craniotomy (CPT 61320-61323) involves removing a section of the skull to relieve elevated intracranial pressure resulting from trauma, stroke, infection, or swelling. The procedure may include duraplasty, where the dura mater is expanded using grafts to accommodate brain swelling. Dural sealants and fibrin glues play a vital role in reinforcing dural closure, maintaining watertight integrity, and reducing the risk of cerebrospinal fluid (CSF) leakage during reconstruction or subsequent cranioplasty. In addition, innovations in synthetic and biologic dural substitutes combined with advanced fibrin sealants have enhanced outcomes in decompressive neurosurgery. Modern sealants offer faster polymerization, improved tissue adherence, and superior handling in edematous or bleeding fields, leading to fewer postoperative CSF leaks and better healing profiles. These factors collectively driving the growth of segment.
The 62140 / 62141- cranioplasty or dural reconstruction segment is growing at a fastest CAGR of 5.17% during the forecast period. A cranioplasty or dural reconstruction is performed when a skull defect (from prior craniectomy, trauma or tumor resection) is repaired, or when the dura mater (the brain’s protective covering) needs reconstruction and sealing. The procedure often involves replacing a bone flap or synthetic implant, reconstructing the dura (with graft, patch or primary suture), and applying adjunct materials such as dural sealants and fibrin glues to achieve watertight closure and reduce cerebrospinal fluid (CSF) leakage. Moreover, education on clinical evidence, hospital formulary integration, surgeon preference, and coding clarity (especially bundling/billing rules) will influence uptake and growth.
End Use Insights
The hospitals segment held the largest revenue share in 2024. Hospitals serve as the key channel for deployment of dural sealants and fibrin glues in neurosurgery and spinal surgery, because they perform the majority of complex dural closure procedures where cerebrospinal fluid (CSF) leak risk is high. For example, the DuraSeal Dural Sealant System was approved by the U.S. Food & Drug Administration in 2005 for preventing CSF leaks in dural surgery; the pivotal trials, which took place across 10 U.S. hospitals, recorded zero intra-operative CSF leaks in 111 patients treated with DuraSeal. Similarly, a 2024 retrospective neurosurgical study reported that among 119 craniotomy patients treated with another sealant (Hemopatch) as the primary dural sealant at a tertiary center, the postoperative CSF-leak rate was just 1.68% (2 out of 119), with a surgical site infection rate of 0.84%. These figures are meaningful for hospitals as CSF leaks are associated with extended lengths of stay, increased readmissions and higher costs. In sum, hospitals remain the pivotal end use setting where dural sealants and fibrin glues impact both clinical outcomes and institutional economics.
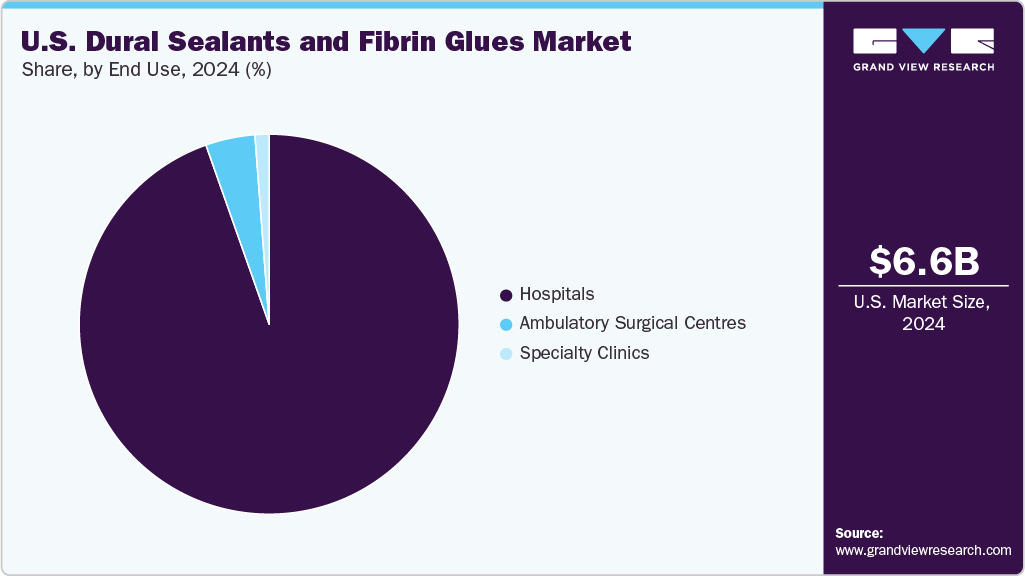
The ambulatory surgical centers (ASCs) segment is expected to grow significantly during the forecast period. ASCs are an increasingly important end use channel for dural sealants and fibrin glues as more spine and selected neurosurgical procedures migrate to outpatient settings. ASCs now account for a substantial share of elective surgical volume in the U.S.: industry trackers report roughly 12,007 ASCs nationally (Q4 2024), with about 6,300-6,500 Medicare-certified facilities, and the ASC market valued at approximately USD 45.6 billion figures that underscore the scale and purchasing power of this channel. These settings increasingly perform minimally invasive spinal surgeries, lumbar decompressions, and spinal cord stimulation procedures operations where CSF leak prevention is critical. Fibrin glues and dural sealants ensure watertight closure and minimize postoperative complications that could otherwise require hospital transfer. A 2025 clinical review reported successful prophylactic use of fibrin glue in outpatient spinal procedures, emphasizing its role in preventing cerebrospinal fluid leaks during same-day surgeries.
Key U.S. Dural Sealants And Fibrin Glues Company Insights
The U.S. dural sealants and fibrin glues market is highly competitive and has several key players. The major market players are focused on expanding their geographical presence, forming partnerships to enhance patient care, taking advantage of important cooperation activities, and exploring mergers and acquisitions.
Key U.S. Dural Sealants And Fibrin Glues Companies:
- Integra LifeSciences
- Stryker
- Baxter
- Ethicon (J&J)
- Takeda Pharmaceutical
- B. Braun Medical
- Cook Medical
- Polyganics
Recent Developments
-
In October 2025, Takeda completed a USD 170 million expansion at its Thousand Oaks campus, adding a 15,000-square-foot facility for rare disease therapies, including Hunter syndrome. The project enhances manufacturing capacity, sustainability with solar arrays, and local employment to 500. It underscores Takeda’s commitment to innovation and advancing biologic therapies for rare conditions.
-
In September 2024, Stryker completed its acquisition of Nico Corporation, expanding its neurotechnology portfolio in minimally invasive tumor and intracerebral hemorrhage (ICH) surgeries. Nico’s BrainPath and Myriad systems enhance outcomes for ICH treatment, reinforcing Stryker’s leadership in neurosurgical access, tumor resection, and advanced minimally invasive brain surgery solutions.
-
In March 2023, Collagen Matrix, Inc. rebranded as Regenity Biosciences to reflect its leadership in regenerative implant science. With over 25 years of experience and 71 global product lines, Regenity aims to advance bioresorbable technologies for tissue and bone repair, expanding innovation through its Polyganics acquisition.
U.S. Dural Sealants And Fibrin Glues Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 6.88 billion
Revenue forecast in 2035
USD 10.99 billion
Growth rate
CAGR of 4.78% from 2025 to 2035
Base year
2024
Forecast period
2025 - 2035
Quantitative units
Revenue in USD Million/Billion and CAGR from 2025 to 2035
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
CPT Procedure Codes, End Use
Key companies profiled
Integra LifeSciences; Stryker; Baxter; Ethicon (J&J); Takeda Pharmaceutical; B. Braun Medical; Cook Medical; Polyganics
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Dural Sealants And Fibrin Glues Market Report Segmentation
This report forecasts revenue growth at the country level and analyzes the latest industry trends in each sub-segments from 2024 to 2035. For this study, Grand View Research has segmented the U.S. dural sealants and fibrin glues market report based on CPT procedure codes and end use:
-
CPT Procedure Codes Outlook (Revenue, USD Unit, 2024 - 2035)
-
61510 / 61512 - Craniotomy for supratentorial lesion removal
-
61320 - 61323 - Craniectomy or craniotomy, decompressive (with or without duraplasty)
-
61518- Craniotomy for infratentorial/posterior fossa lesion
-
62140 / 62141- Cranioplasty or dural reconstruction
-
63707- Repair of CSF leak, cranial or spinal, without laminectomy
-
63709 - Repair of CSF leak or pseudomeningocele with laminectomy
-
63710- Dural graft (spinal)
-
63030 - Laminotomy/laminectomy (lumbar), decompression of nerve root
-
63047 - Laminectomy, >1 vertebral segment, lumbar
-
22612 / 22630 - Posterior spinal fusion with decompression
-
-
End Use Outlook (Revenue, USD Unit, 2024 - 2035)
-
Hospitals
-
Ambulatory Surgical Centers
-
Specialty Clinics
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










