- Home
- »
- Medical Devices
- »
-
U.S. Fluoroscopy Procedures Market, Industry Report, 2033GVR Report cover
![U.S. Fluoroscopy Procedures Market Size, Share & Trends Report]()
U.S. Fluoroscopy Procedures Market (2026 - 2033) Size, Share & Trends Analysis Report By Type (C-Arms {including Mobile}, Fixed Table-based, Others), By CPT (50010-50437, 50500-50593, 50600-50980), By End Use (Hospitals), And Segment Forecasts
- Report ID: GVR-4-68040-876-3
- Number of Report Pages: 80
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Fluoroscopy procedures Market Summary
The U.S. fluoroscopy procedures market size was estimated at USD 132.1 million in 2025 and is expected to reach a value of USD 182.7 million in 2033, growing at a CAGR of 4.1% from 2026 to 2033. The market growth is supported by the country’s advanced healthcare infrastructure and high utilization of medical imaging for both diagnostic and interventional procedures.
Key Market Trends & Insights
- By type, the C-arms {including mobile} segment accounted for the largest revenue share of 52.5% in 2025.
- By CPT, the 52300-52356 segment held the largest revenue share of 44.1% in 2025.
- By end use, the hospitals segment held the largest revenue share of 51.4% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 132.1 Million
- 2033 Projected Market Size: USD 182.7 Million
- CAGR (2026-2033): 4.1%
Fluoroscopy enables real-time X-ray imaging, allowing physicians to guide minimally invasive procedures across specialties such as cardiology, orthopedics, gastroenterology, urology, and interventional radiology. In the United States, hospitals, catheterization laboratories, and ambulatory surgical centers widely rely on fluoroscopic guidance to perform complex procedures including angiography, catheter placement, spinal interventions, and gastrointestinal examinations. The growing preference for image-guided minimally invasive treatments has strengthened the role of fluoroscopy within the broader medical imaging ecosystem.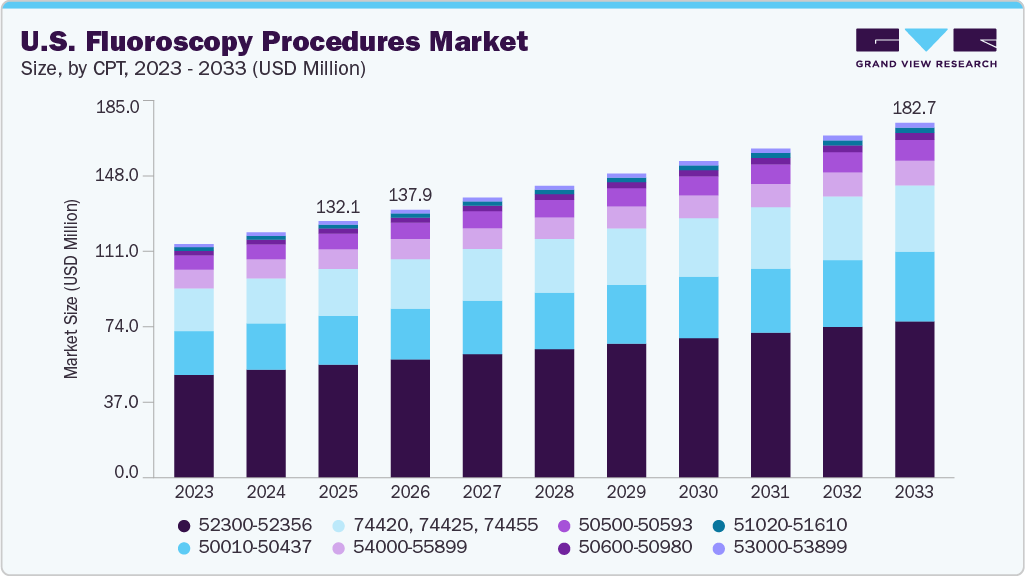
Technological advancements in imaging systems are playing a significant role in improving the efficiency, safety, and clinical outcomes of fluoroscopy-guided procedures. Hospitals and interventional imaging centers are increasingly replacing conventional fluoroscopy systems with advanced digital platforms that offer enhanced visualization, reduced radiation exposure, and improved workflow during minimally invasive procedures. According to the study published by National Library of Medicine in January 2026 evaluating fluoroscopy systems in clinical practice found that approximately 62% of surveyed fluoroscopy units now use flat-panel detectors (FPDs), which provide improved image quality and lower radiation dose compared with older image-intensifier systems. The study also noted that pulsed fluoroscopy capabilities are now nearly universal in modern systems, reflecting technological upgrades across hospitals and interventional imaging centers. These advancements support more precise image-guided interventions while improving radiation safety for both patients and healthcare professionals.
The rising prevalence of kidney and urinary tract diseases plays critical role in the growth of U.S. fluoroscopy procedures industry. Fluoroscopy provides real-time visualization during minimally invasive procedures, allowing physicians to guide catheters, stents, and other instruments with precision. As chronic kidney disease and urinary disorders continue to affect a significant portion of the U.S. population, healthcare facilities increasingly depend on fluoroscopy systems to support safe and efficient interventions. According to the Centers for Disease Control and Prevention (CDC) report “Chronic Kidney Disease in the United States, 2023,” published in May 2024, more than 1 in 7 U.S. adults (about 35.5 million people) are estimated to have chronic kidney disease, highlighting a significant and growing healthcare burden. The CDC also notes that many individuals remain undiagnosed, increasing the need for early detection and image-guided clinical interventions. This rising disease prevalence supports growing demand for advanced diagnostic and treatment technologies, including fluoroscopy systems used in minimally invasive kidney and urinary tract procedures.
Recent population estimates also show a continuing rise in the older age group, reinforcing long-term demand for imaging systems that support efficient and precise treatment workflows. According to a Pew Research Center report published in January 2024, the U.S. centenarian population (people aged 100 and older) is projected to grow from about 101,000 in 2024 to nearly 422,000 by 2054, highlighting a significant long-term demographic shift toward an older population. This trend reflects broader aging dynamics in the U.S., where the number of adults aged 65 and above is also expected to rise substantially over the coming decades. This growing elderly population increases demand for healthcare services, including minimally invasive and image-guided procedures supported by technologies such as fluoroscopy.
Market Characteristics & Concentration
The U.S. fluoroscopy procedures market is moderately concentrated, with procedure volumes largely driven by major hospitals, integrated health systems, specialized cardiovascular centers, and interventional radiology facilities. Large healthcare providers and hospital networks account for a significant share of fluoroscopy-guided procedures due to their advanced imaging infrastructure, availability of specialized physicians, and well-established catheterization laboratories and hybrid operating rooms. At the same time, ambulatory surgical centers and outpatient imaging facilities are increasingly performing fluoroscopy-guided procedures as healthcare providers shift toward cost-efficient outpatient care models. The market is characterized by continuous growth in image-guided minimally invasive procedures across specialties such as cardiology, orthopedics, gastroenterology, and pain management. Increasing adoption of catheter-based interventions, spinal procedures, gastrointestinal examinations, and vascular treatments is expanding the utilization of fluoroscopy in clinical practice further driving the growth of fluoroscopy procedures market.
The degree of innovation in the U.S. fluoroscopy procedures industry continues to advance steadily. For instance, in February 2026, Zimmer Biomet announced that it will showcase several digital and AI-driven orthopedic technologies at the American Academy of Orthopaedic Surgeons (AAOS) 2026 Annual Meeting. Among the highlighted technologies is OrthoGrid Hip AI, an AI-powered, fluoroscopy-based system designed to provide surgeons with intuitive and instantaneous intra-operative guidance during hip procedures. The technology assists surgeons in achieving accurate component positioning and improved surgical outcomes by delivering real-time imaging insights during surgery, reflecting the growing integration of artificial intelligence with fluoroscopy-guided orthopedic procedures in the U.S.
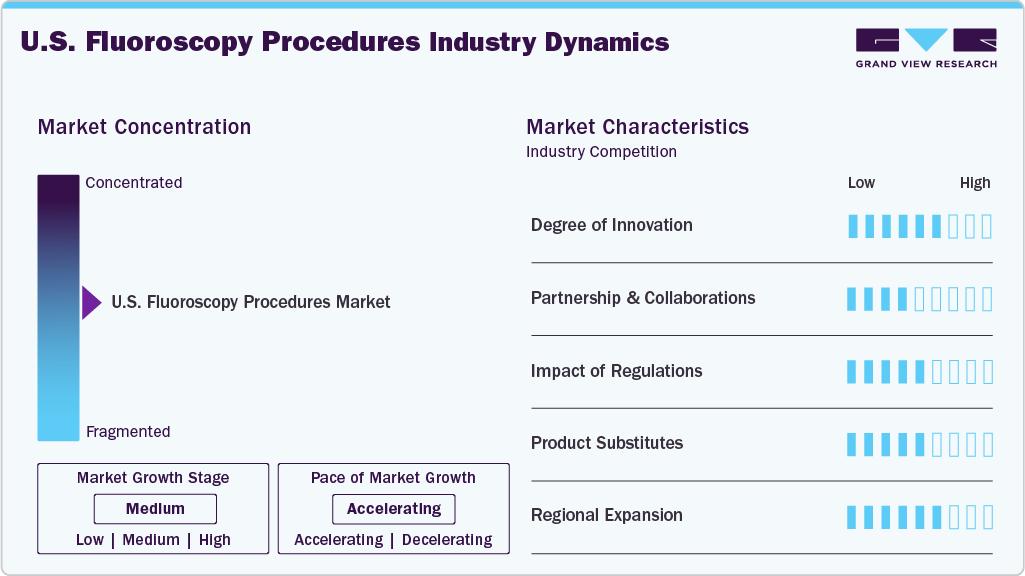
The market is experiencing a significant rise in partnerships and collaborations. Companies are increasingly adopting this strategy to expand service offerings and reach a broader customer base. For instance, in October 2023, GE HealthCare and University Hospitals announced a 10-year strategic collaboration to deploy advanced medical imaging technologies across the health system’s radiology and cardiovascular departments in the country. The partnership includes the implementation of imaging systems such as X-ray and fluoroscopy technologies, along with digital solutions aimed at improving workflow efficiency and clinical decision-making for image-guided procedures.
Stringent regulatory frameworks significantly influence the U.S. fluoroscopy procedures market by ensuring patient safety, procedural quality, and effective radiation management during image-guided interventions. Regulatory authorities such as the U.S. Food and Drug Administration establish safety requirements for fluoroscopy equipment and oversee device approvals that directly impact how fluoroscopy-guided procedures are performed in clinical settings. In addition, clinical practice standards developed by the American College of Radiology and radiation safety recommendations from the National Council on Radiation Protection and Measurements guide healthcare providers on appropriate imaging protocols, radiation dose limits, and patient protection measures during procedures.
Product substitutes in the U.S. fluoroscopy procedures industry mainly include alternative imaging-guided techniques that can perform diagnostic or interventional procedures with reduced radiation exposure or improved imaging capabilities. Imaging modalities such as Computed Tomography (CT), Magnetic Resonance Imaging (MRI), and Ultrasound Imaging are widely used alternatives depending on the clinical indication. CT enables image-guided biopsies and complex diagnostic evaluations, while MRI offers detailed soft-tissue visualization without ionizing radiation, making it suitable for several neurological and musculoskeletal assessments. Ultrasound is increasingly adopted for real-time guidance in procedures such as vascular access, biopsies, and certain pain management interventions due to its portability, safety profile, and absence of radiation exposure.
In terms of geographic expansion in the U.S. fluoroscopy procedures market, healthcare providers are increasingly adopting advanced imaging technologies to expand access to fluoroscopy-guided interventions across hospitals and specialized care centers. For instance, in April 2025, Siemens Healthineers and Tower Health announced a 10-year Value Partnership aimed at modernizing imaging infrastructure across Tower Health’s hospitals in Pennsylvania. The collaboration includes the deployment of advanced diagnostic and interventional imaging technologies, including systems used for fluoroscopy-guided procedures in radiology and cardiology departments, along with digital tools to enhance workflow efficiency and clinical decision-making. This highlights the expansion of image-guided procedures and improved access to advanced fluoroscopy-based interventions across U.S. healthcare facilities.
Type Insights
The C-arms {including mobile} segment held the largest market share of 52.5% in 2025 and is expected to witness the fastest CAGR of 4.2% over forecast period. C-arm fluoroscopy systems both fixed and mobile directly influence patient experience, access to minimally invasive care, and procedural outcomes in U.S. hospitals and ambulatory settings. In terms of clinical utilization, 51% of fixed C-arm procedures in 2023 were performed in radiology-related departments, 35% in cardiology, and 15% in surgery. From the patient’s standpoint, this distribution highlights the expanding role of C-arms in vascular, cardiac, and minimally invasive interventions. Notably, 53% of procedures are performed by interventional radiologists, while 47% involve cardiac/cardiovascular surgeons and interventional cardiologists, indicating strong growth in image-guided cardiovascular care. Looking ahead, 23% of surveyed facilities expect a shift from open surgery to fixed C-arm-guided procedures within the next two to three years (2024-2026). For patients, this translates into shorter recovery times, reduced hospital stays, and lower complication risks key drivers of consumer preference for image-guided therapies.
The fixed table-based segment is expected to grow at the second-fastest CAGR during the forecast period. Fixed table-based fluoroscopy systems play a vital role in shaping patient experience, diagnostic accuracy, and procedural comfort in U.S. hospitals and imaging centers. These systems are widely used for gastrointestinal studies, vascular imaging, orthopedic assessments, and interventional procedures, directly impacting patient outcomes through real-time imaging and minimally invasive diagnostics. A key market development occurred in March 2025, when Canon Medical Systems launched the Adora DRFi hybrid radiography/fluoroscopy system. The system received FDA clearance in December 2024, marking an important regulatory milestone for fixed table-based innovation in the U.S. market. From the patient’s perspective, FDA approval enhances trust in safety, performance standards, and radiation management protocols.
CPT Insights
The 52300-52356 segment dominated the U.S. fluoroscopy procedures industry, accounting for the largest share in 2025. The CPT code range 52300-52356 covers procedures involving the urethra and ureter performed primarily through endoscopic (minimally invasive) techniques. The segment represents highly patient-focused urological interventions that emphasize precision, reduced recovery time, and real-time imaging support. The code range broadly include procedures such as cystourethroscopy (endoscopic examination of the urethra and bladder), ureteroscopy (visualization of the ureter), removal or fragmentation of ureteral stones, laser lithotripsy procedures, ureteral dilation for stricturesand others. Many of these procedures are performed using fluoroscopy-guided imaging to help physicians accurately visualize stone location, confirm stent placement, and monitor contrast flow within the urinary tract. Fluoroscopy enhances procedural accuracy while reducing the need for open surgery.
The 50010-50437 segment is expected to grow during the forecast period. This CPT code covers surgical procedures involving the kidney and ureter. This category represents critical interventions that directly impact patients suffering from kidney stones, tumors, urinary obstruction, trauma, or severe infections. These procedures are performed by urologists and interventional specialists using open, laparoscopic, or minimally invasive percutaneous techniques often supported by fluoroscopic imaging for real-time guidance. The code range include different procedures including removal of kidney stones (including large or complex stones), incisions into the kidney or renal pelvis to relieve obstruction, partial or complete kidney removal for tumors or severe damage, removal of ureteral stones. In these procedures fluoroscopic imaging support improves precision, reduces complications, and enhances procedural safety. In the U.S., advanced digital fluoroscopy platforms emphasize lower radiation exposure, which is increasingly important to informed healthcare consumers.
End Use Insights
The hospitals segment accounted for the largest share of the U.S. fluoroscopy procedures market in 2025. Hospitals are where patients directly experience fluoroscopy technology for diagnostic and interventional procedures. Whether for gastrointestinal studies, cardiac catheterizations, orthopedic surgeries, urology interventions, or pain management procedures, hospitals rely heavily on fluoroscopy systems to deliver real-time imaging that enhances precision and safety. Hospitals also manage complex and high-risk cases that outpatient centers may not handle, making them essential for trauma care, advanced cardiovascular procedures, oncology interventions, and emergency gastrointestinal treatments. As chronic diseases such as cardiovascular disorders, kidney stones, and gastrointestinal conditions continue to rise in the U.S., the demand for fluoroscopy-guided interventions in hospital settings is growing steadily. Additionally, the shift toward value-based care models has pushed hospitals to invest in safer, more efficient imaging systems. Radiation dose monitoring, workflow optimization, and integration with electronic health records further improve patient outcomes.
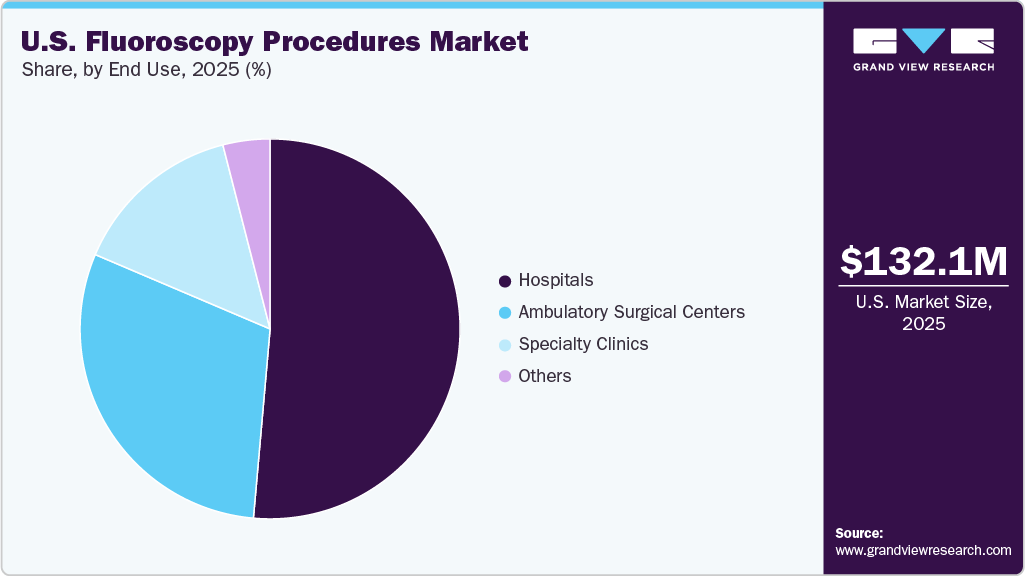
The ambulatory surgical centers segment is expected to register the fastest CAGR during the forecast period. These facilities align closely with modern patient preferences for convenience, affordability, faster service, and minimally invasive care. The healthcare industry has witnessed a notable shift from traditional hospital-based procedures to outpatient settings for non-emergency surgeries and diagnostics. Patients increasingly prefer ambulatory centers because they eliminate overnight stays, reduce wait times, and provide a more personalized care experience. Fluoroscopy plays a critical role in supporting this transition, particularly in procedures such as orthopedic interventions, pain management injections, gastrointestinal endoscopies, cardiac catheterizations, and minimally invasive spine treatments. Modern ASCs are equipped with compact, high-performance C-arm fluoroscopy systems that deliver real-time imaging with lower radiation doses. These systems allow physicians to perform image-guided procedures with precision while maintaining patient comfort. This highlights the rising number of fluoroscopy performed in these facilities which contributes to the segment growth.
Key U.S. Fluoroscopy Procedures Companies & Market Share Insights
The U.S. fluoroscopy procedures market is highly competitive, with several established players actively pursuing strategic initiatives to strengthen their market presence. Companies are increasingly focusing on partnerships, collaborations, mergers, and acquisitions to enhance fluoroscopy technologies and improve patient care solutions.
Key U.S. Fluoroscopy Procedures Market Companies:
- GE HealthCare
- Siemens Healthcare GmbH
- Koninklijke Philips N.V.
- Canon Medical Systems
- Shimadzu Corporation
- Dornier MedTech
- UMG / DEL MEDICAL
Recent Developments
-
In August 2025, GE HealthCare Paris Regional Health announced that it has added the advanced GE P180 Fluoroscopy System to its diagnostic imaging services, enhancing its ability to perform a wide range of procedures with superior imaging quality, lower radiation exposure, and improved workflow, marking a significant upgrade in patient care and diagnostic efficiency
-
In January 2024, Shimadzu Medical Systems USA announced that its FLUOROspeed X1 radiographic/fluoroscopy system has received HCAI Special Seismic Certification pre-approval, validating its earthquake-resistant design and enabling safer deployment in seismically active healthcare facilities. The certification supports the system’s use in environments where enhanced structural integrity is required for consistent clinical performance.
-
In July 2023, Canon Medical Systems announced the launch of its Zexira i9 digital X-ray RF system, a versatile multi-purpose fluoroscopy-capable imaging platform featuring a flat panel detector and advanced image processing to deliver high-quality images at low radiation dose and enhanced clinical workflow. The system allows field-of-view adjustment during procedures without moving the table or X-ray tube and includes optional tomosynthesis, DSA, and extended imaging functions for a broad range of fluoroscopic examinations.
U.S. Fluoroscopy Procedures Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 137.9 million
Revenue forecast in 2033
USD 182.7 million
Growth rate
CAGR of 4.1% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Type, CPT, end use
Country scope
U.S.
Key company profiled
GE HealthCare; Siemens Healthcare GmbH; Koninklijke Philips N.V.; Canon Medical Systems; Shimadzu Corporation; Dornier MedTech; UMG / DEL MEDICAL
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Fluoroscopy Procedures Market Report Segmentation
This report forecasts revenue growth at country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. fluoroscopy procedures market report based on product, application, and end use:
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
C-Arms {including Mobile}
-
Fixed Table-based
-
Others
-
-
CPT Outlook (Revenue, USD Million, 2021 - 2033)
-
54000-55899
-
50010-50437
-
50500-50593
-
50600-50980
-
51020-51610
-
52300-52356
-
53000-53899
-
74420, 74425, 74455
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Not-for-Profit
-
For Profit
-
-
Ambulatory Surgical Centers
-
Specialty Clinics
-
Others
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










