- Home
- »
- Medical Devices
- »
-
U.S. Hemodialysis Catheter Market, Industry Report, 2033GVR Report cover
![U.S. Hemodialysis Catheter Market Size, Share & Trends Report]()
U.S. Hemodialysis Catheter Market (2025 - 2033) Size, Share & Trends Analysis Report By Design (Single Lumen, Double Lumen,Triple Lumen), By Material, By Shape (Straight, Curved, Pre-Curved), By Component, By Usage, By Application, By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-644-6
- Number of Report Pages: 110
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Hemodialysis Catheter Market Summary
The U.S. hemodialysis catheter market size was estimated at USD 408.14 million in 2024 and is projected to reach USD 741.41 million by 2033, growing at a CAGR of 6.9% from 2025 to 2033. The demand for hemodialysis catheters is increasing in the U.S.
Key Market Trends & Insights
- By design, the double lumen segment led the market with the largest revenue share of 42.19% in 2024.
- Based on shape, the straight segment led the market with the largest revenue share of 45.99% in 2024.
- By application, the chronic kidney disease (CKD) segment is anticipated to grow at the fastest CAGR over the forecast period.
- By component, the catheter kits segment is anticipated to grow at the fastest CAGR over the forecast period.
- By end use, the hospitals segment led the market with the largest revenue share of 34.86% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 408.14 Million
- 2033 Projected Market Size: USD 741.41 Million
- CAGR (2025-2033): 6.9%
Due to the rising prevalence of kidney disorders such as chronic kidney disease (CKD) and acute kidney injury (AKI), along with supportive government regulations and continuous technological advancements in catheter design and functionality. The rising burden of kidney disorders is a significant driver for the expansion of the U.S. hemodialysis catheters market. According to the data published by the American Kidney Fund in June 2025, approximately 35.5 million Americans[ are affected by kidney disease, with around 815,000 living with kidney failure. This substantial disease burden, coupled with a growing number of patients requiring dialysis, is expected to significantly drive the demand for hemodialysis catheters across the U.S. in the coming years.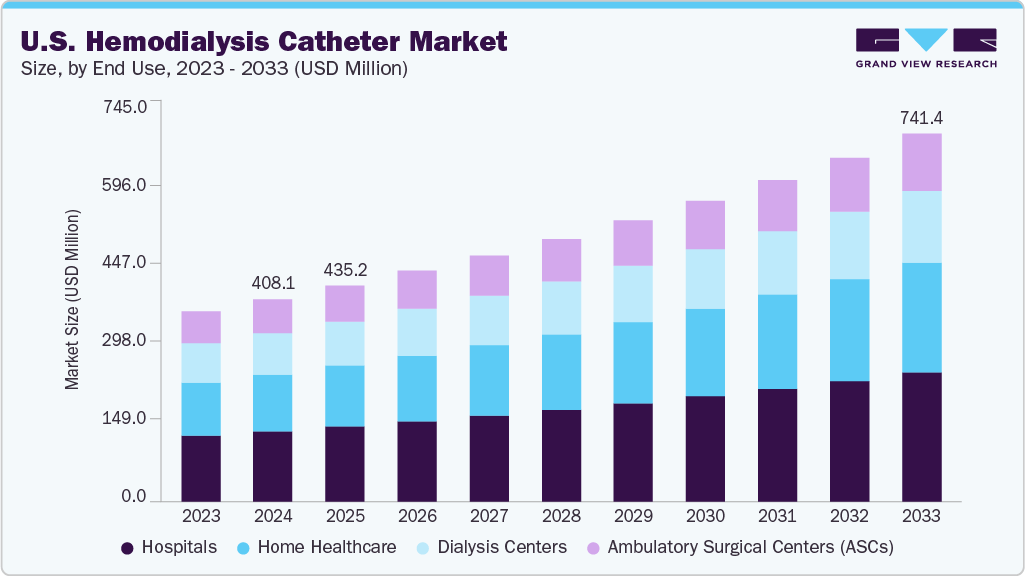
Furthermore, the widespread presence of dialysis centers across the U.S. and the high volume of hemodialysis procedures performed in these facilities are expected to boost demand for hemodialysis catheters significantly. This growing infrastructure ensures greater access to treatment and supports consistent utilization of catheters, reinforcing their essential role in renal care management nationwide.
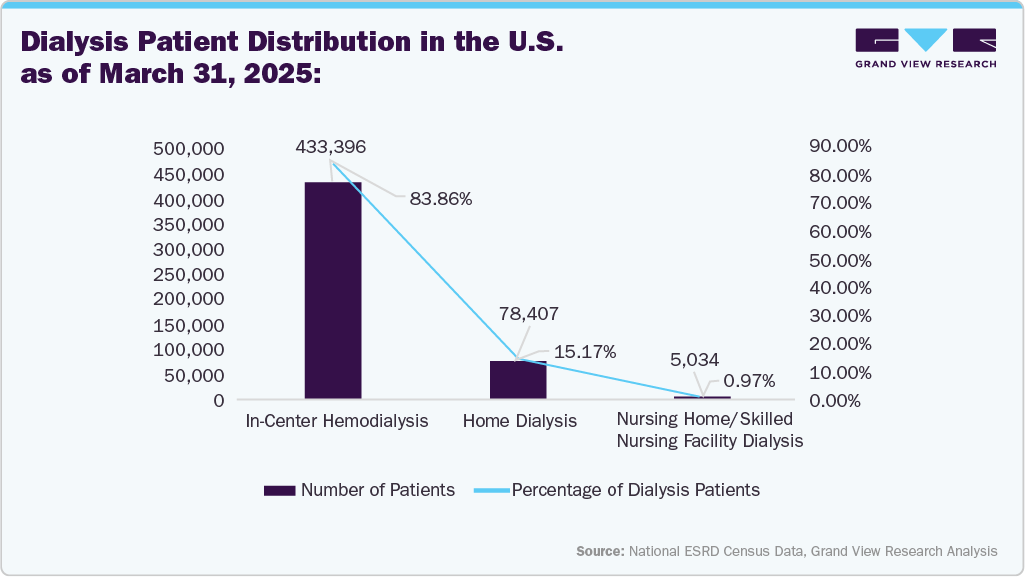
Note: These figures are based on data from the 18 ESRD Networks, which collectively support 7,556 dialysis centers and 223 transplant centers across the U.S. and its territories.
The above data indicates that the majority of dialysis treatments across the End Stage Renal Disease (ESRD) networks in the U.S. are performed in dialysis centers and primarily involve in-center hemodialysis, accounting for nearly 84% of all dialysis patients. This high preference for in-center hemodialysis is a major driver for the U.S. hemodialysis catheter market.
Furthermore, the rising focus of manufacturers on preventing catheter-associated infections, such as central line-associated bloodstream infections, is anticipated to support the market growth. Industry players have manufactured and designed novel catheters and solutions that help reduce such infections.
For instance, in November 2023, CorMedix Inc., a biopharmaceutical company dedicated to developing therapies for life-threatening conditions, announced that the U.S. Food and Drug Administration (FDA) authorized DefenCath-a catheter lock solution (CLS) containing taurolidine and heparin. This marks the first and only FDA-authorized antimicrobial CLS in the U.S. specifically indicated to decrease the burden of catheter-related bloodstream infections in adult patients with kidney failure undergoing chronic hemodialysis via a central venous catheter (CVC). In a pivotal Phase 3 clinical trial, DefenCath demonstrated a 71% reduction in CRBSI risk, underscoring its significant potential to improve safety in this high-risk patient population. Such developments are anticipated to propel market growth in the coming years.
Key Opinion Leaders
Company Name
KOLs
Growth Opportunities
Teleflex
“Max barrier kits like the Arrow ErgoPack complete system provide clinicians with the necessary components they need for their acute hemodialysis and large bore insertions. The intuitive configuration is designed to optimize their workflow, while also meeting the recommendations of CDC, SHEA, INS, and OSHA guidelines, coupling these benefits along with the protection from Arrowg+ard Blue catheters helps clinicians fight against central line-associated bloodstream infections, protecting their patients and improving their procedures."
- Customization & Flexibility
- Infection Prevention Focus
- Compliance with Safety Guidelines
- Streamlined Clinical Workflow Enhancements
CorMedix Inc
“Patients and their loved ones have faced many burdens related to kidney failure, including complications caused by catheter related bloodstream infections and associated loss of work, severe disability and death. Until now, patients who need hemodialysis via a central venous catheter have had little choice other than to accept high infection risks associated with the existing standard of care. The FDA’s approval of DefenCath is a meaningful moment for patients and their healthcare providers because they now have a new alternative to reduce the risks of CRBSIs.”
- FDA-Approved Antimicrobial Catheter Lock Solution (CLS)
- High-Need Target Population: CVC-Dependent Dialysis Patients
- Rising Clinical and Administrative Pressure to Reduce Infections
CorMedix
“The approval of DefenCath marks a major advancement in reducing life-threatening infections for patients receiving hemodialysis via central venous catheters and an important milestone for CorMedix. As the first FDA-approved antimicrobial catheter lock solution designed to prevent CRBSIs, DefenCath offers healthcare providers an option to reduce the risk of infections in a patient population already vulnerable due to underlying kidney failure. We thank all the patients, caregivers, clinical investigators, and our employees who have played an integral role in the development and regulatory approval of DefenCath. Our commercial team along with our broader organization is preparing for commercial launch, and we look forward to working with healthcare providers and facilities to give hemodialysis patients access to DefenCath in early 2024.”
- Enhanced Safety Profile for Catheter-Based Hemodialysis
- Support for Early-Stage Dialysis Patients
- Prevention of Catheter-Related Bloodstream Infection
Source: Grand View Research Analysis
Market Concentration & Characteristics
The market growth stage is high, and the pace of growth is accelerating. The U.S. hemodialysis catheter market is characterized by high growth due to the rising burden of kidney diseases, the growing launches of novel products, and the growing focus on preventing catheter-associated infections.
Industry players and researchers are increasingly focusing on developing advanced U.S. hemodialysis catheters. Companies are focusing on developing antimicrobial technology catheters to reduce catheter-associated infections. For instance, Arrowg+ard Blue Acute Hemodialysis Catheters, offered by Teleflex Incorporated, utilizes chlorhexidine-based antimicrobial technology to decrease central line-associated bloodstream infection (CLABSI), the second leading cause of death in hemodialysis patients. Such product availability and developments are anticipated to drive innovation in the U.S. hemodialysis catheter market.
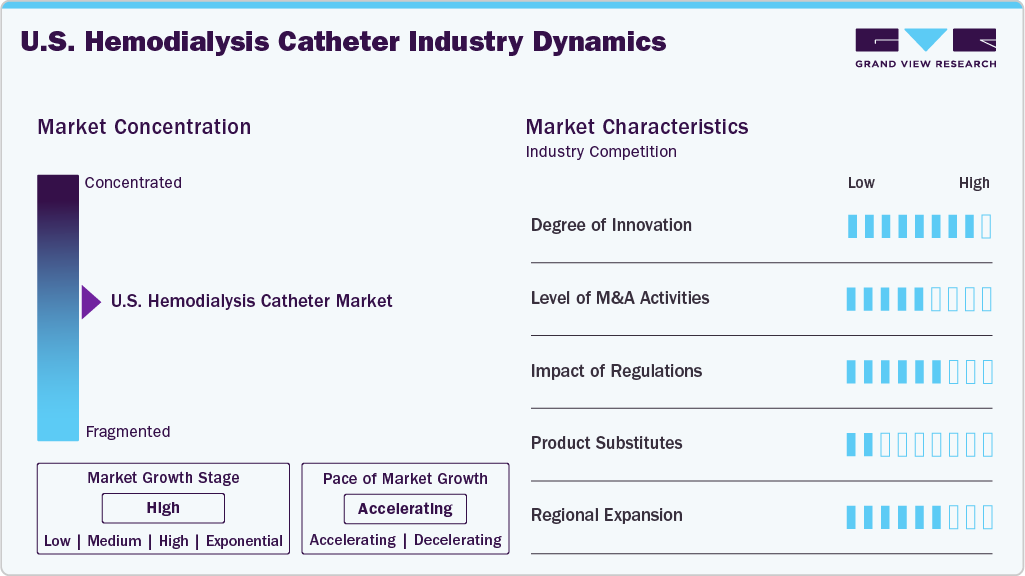
In the U.S., hemodialysis catheters are regulated primarily by the Food and Drug Administration (FDA) as Class II medical devices, requiring 510(k) premarket clearance before marketing. Manufacturers must demonstrate that their catheters are safe and effective. This includes providing data on biocompatibility, flow performance, tip design, sterilization, and labeling. Devices with antimicrobial coatings or infection prevention features-such as catheter lock solutions-must provide additional clinical evidence to support such claims, often requiring randomized clinical trial data. For example, DefenCath by CorMedix was approved under the FDA’s Limited Population Pathway (LPAD) and designated as a Qualified Infectious Disease Product (QIDP) due to its antimicrobial properties and focus on preventing catheter-related bloodstream infections (CRBSIs).
Mergers and acquisitions are actively employed in the U.S. hemodialysis catheter industry, enabling companies to expand their portfolios, accelerate innovation, and strengthen market presence. For instance, in 2023, Merit Medical acquired AngioDynamics’ dialysis catheter portfolio for USD 100 million. Through this strategic move, Merit Medical acquired DuraFlow, Evenmore, DuraMax, Schon XL, Trio-CT, and Vaxel Plus Hemodialysis Catheter brands.
Design Insights
The double-lumen segment dominated the U.S. hemodialysis catheter market, accounting for share of 42.19% of total revenue in 2024. Its dual channel structure allows for the simultaneous withdrawal and return of blood, enhancing dialysis efficiency and improving patient outcomes. Suitable for both temporary use and therapy transitions, double-lumen catheters offer versatility across patient needs. These advantages are expected to continue driving their adoption throughout the forecast period.
The triple lumen is expected to be the fastest-growing segment in the U.S. hemodialysis catheters market due to its enhanced functionality, offering an additional channel for simultaneous fluid administration, medication delivery, and blood sampling alongside dialysis. This versatility makes it ideal for critically ill and intensive care patients. Rising demand in acute care settings and technological advancements that improve safety and durability are driving increased adoption of triple-lumen catheters across healthcare facilities.
Material Insights
In 2024, the polyurethane segment accounted for the largest share of the U.S. hemodialysis catheter market. Polyurethane is widely used due to its excellent biocompatibility, flexibility, and resistance to kinking. In addition, it softens at body temperature after insertion, improving patient comfort and reducing the risk of vascular trauma, making it a preferred choice among clinicians for both short- and long-term use.
The silicone segment is expected to at the fastest CAGR in the U.S. hemodialysis catheter market from 2025 to 2033. Silicone offers outstanding biocompatibility, making it ideal for long-term use in chronic dialysis therapy. Radiopaque silicone enhances catheter visibility under imaging, facilitating accurate placement. Additionally, the growing availability of advanced silicone-based products, such as permanent hemodialysis catheters, is anticipated further to support the segment’s expansion over the forecast period.
Shape Insights
In 2024, the straight segment led the U.S. hemodialysis catheter market, accounting for the largest share of 45.99%. This dominance is driven by the widespread availability and clinical preference for straight-shaped catheters offered by key manufacturers like BD and Teleflex. Products such as the Power-Trialysis Dialysis Catheter from BD and Arrowg +ard Blue Acute Hemodialysis Catheters from Teleflex Incorporated are available in straight shape, promoting widespread adoption.
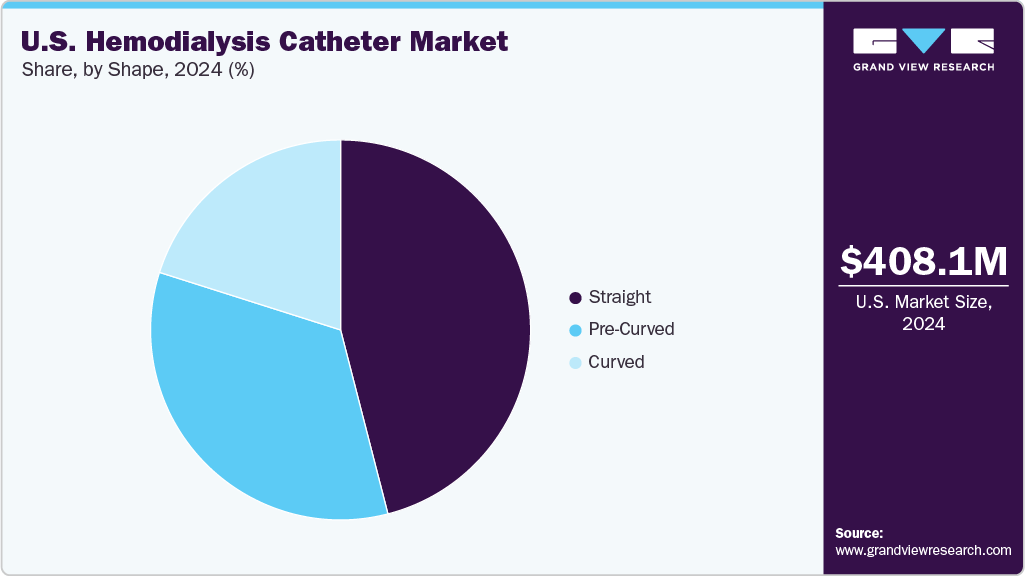
The pre-curved segment is projected to be the fastest growing in the U.S. hemodialysis catheter market from 2025 to 2033. This growth is driven by the increasing clinical preference for pre-curved designs, which offer greater patient comfort compared to straight catheters. Their anatomical shape also reduces the risk of kinking, ensuring better catheter function, stability, and overall treatment efficiency during hemodialysis procedures.
Component Insights
The catheters segment held the largest market share in 2024, driven by several critical factors. The rising prevalence of chronic kidney disease (CKD) and a growing dialysis population have significantly increased the demand for vascular access solutions. Catheters are commonly used as the initial access point for new dialysis patients, particularly in acute or emergencies. Additionally, the wide availability of various catheter types-such as double-lumen, triple-lumen, straight, and pre-curved-designed to meet diverse clinical needs continues to support the segment’s dominant position in the market.
The catheter kit segment is projected to witness the fastest CAGR of 12.4% from 2025 to 2033. This anticipated growth is fueled by the increasing demand for catheter-related supplies and the introduction of advanced product kits. For example, in August 2023, Teleflex launched its enhanced Arrow ErgoPack complete system- a Max barrier kit featuring Arrowg+ard Blue hemodialysis and large-bore catheters in multiple French sizes, lumens, and lengths-designed to streamline workflows and meet diverse clinical requirements.
Usage Insights
The long-term segment held the largest market share in 2024 and is expected to grow the fastest during the forecast period. This growth is driven by the increasing number of patients with chronic kidney disease (CKD) and end-stage renal disease (ESRD) who require prolonged dialysis treatment. According to the data published by the National Institute of Diabetes and Digestive and Kidney Diseases in September 2024, over 808,000 people in the U.S. live with ESRD, with 68% receiving dialysis.
The short-term segment is expected to experience significant growth from 2025 to 2033. Short-term catheters, which are designed for vascular access lasting less than 30 days, are primarily utilized in urgent and critical care settings for rapid hemodialysis access. They are also used for temporary dialysis before surgeries and for short-term renal replacement therapy. Companies in the market are supporting this segment's growth with products like BD's DuoGlide Short-Term Dialysis Catheter, specifically designed to address these immediate clinical needs.
Application Insights
The chronic kidney disease (CKD) segment held the largest market share in 2024, and it is anticipated to grow at the fastest CAGR during the forecast period. This growth is primarily due to the growing prevalence of CKD across the U.S. population. According to data published by the National Kidney Fund in August 2024, the number of patients with kidney failure (ESKD) is projected to exceed 1 million by 2030. This escalating disease burden is expected to be a key driver of segment growth over the forecast period.
The Acute Kidney Injury (AKI) segment is anticipated to witness significant growth in the U.S. hemodialysis catheter market from 2025 to 2033. This growth is primarily driven by the rising incidence of AKI among hospitalized and critically ill patients. According to data published by the National Library of Medicine in November 2023, AKI is present in approximately 1% of all hospital admissions in the U.S. Furthermore, during hospitalization, the incidence of AKI ranges from 2% to 5%, with rates reaching up to 67% among patients admitted to intensive care units (ICUs). These figures underscore the growing need for temporary hemodialysis access, as AKI frequently requires immediate renal replacement therapy. The increasing burden of AKI in clinical settings and the availability of specialized short-term dialysis catheters are expected to drive strong growth in this segment over the forecast period.
End Use Insights
The hospital segment dominated the U.S. hemodialysis catheter market in 2024, accounting for the largest share of 34.86%. This leading position is primarily driven by the high volume of dialysis procedures performed in hospital settings, particularly for patients with acute kidney injury (AKI) and those requiring emergency vascular access. Hospitals serve as primary points of care for critically ill patients, many of whom need immediate hemodialysis using short-term catheters. The availability of advanced infrastructure, skilled healthcare professionals, and access to specialized catheter technologies further strengthens hospitals' role in delivering effective renal replacement therapy. Additionally, increasing hospital admissions related to chronic kidney disease (CKD) contribute to the growing demand for catheter-based dialysis in these facilities.
The home care settings segment is projected to register the fastest CAGR in the U.S. hemodialysis catheter market from 2025 to 2033. This growth is driven by the rising preference for home-based dialysis due to its convenience, cost-effectiveness, and improved quality of life for patients with chronic kidney disease (CKD). Advances in portable dialysis equipment, increased patient education, and supportive reimbursement policies have made home dialysis a more accessible and viable option. Additionally, the growing burden on hospital infrastructure and the need to reduce infection risks-especially among immunocompromised patients-have further accelerated the shift toward home care. Government and healthcare initiatives promoting home hemodialysis and innovations in catheter design that enable safe and effective use in non-clinical settings are expected to fuel this segment’s expansion significantly over the forecast period.
Key U.S. Hemodialysis Catheter Company Insights
BD, Teleflex Incorporated, Mozarc Medical Holding LLC., Medical Components, Inc,Merit Medical Systems,Medtronic, Healthline Medical Products., and Integer Holdings Corporation, are some of the major players in the U.S. hemodialysis catheter market. Companies are expanding their portfolios of U.S. hemodialysis catheter and acquiring other smaller players to meet the growing demand. Moreover, industry players are also launching antimicrobial and innovative products to gain a competitive advantage.
Key U.S. Hemodialysis Catheter Companies:
- BD
- Teleflex Incorporated
- Mozarc Medical Holding LLC.
- Medical Components, Inc
- Merit Medical Systems
- Medtronic
- Healthline Medical Products.
- Integer Holdings Corporation
Recent Developments
-
In November 2023, CorMedix Inc. announced FDA approval for DefenCath, the first and only antimicrobial catheter lock solution in the U.S., indicated to reduce catheter-related bloodstream infections (CRBSIs) in adult patients with kidney failure undergoing chronic hemodialysis via central venous catheters.
-
In August 2023, Teleflex Incorporated launched the Arrow ErgoPack Complete Kits with Hemodialysis and Large Bore catheters in the U.S.
-
In June 2023, Merit Medical acquired AngioDynamics’ dialysis catheter portfolio for USD 100 million. Through this strategic move, Merit Medical acquired DuraFlow, Evenmore, DuraMax, Schon XL, Trio-CT, and Vaxel Plus Hemodialysis Catheter brands.
- In April 2021, BD announces 510(k) clearance of Pristine long-term hemodialysis catheter from the U.S. FDA. This catheter has a unique side-hole free symmetric Y-Tip distal lumen design.
U.S. Hemodialysis Catheter Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 435.18 million
Revenue forecast in 2033
USD 741.41 million
Growth rate
CAGR of 6.9% from 2025 to 2033
Base year for estimation
2024
Actual data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Design, material, shape, component, usage, application, end use
Country scope
U.S.
Key companies profiled
BD; Teleflex Incorporated; Mozarc Medical Holding LLC.; Medical Components, Inc; Merit Medical Systems; Medtronic; Healthline Medical Products.; Integer Holdings Corporation
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Hemodialysis Catheter Market Report Segmentation
This report forecasts revenue growth at country level and provides an analysis on the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. hemodialysis catheter market report based on design, material, shape, component, usage, application, and end use.
-
Design Outlook (Revenue, USD Million, 2021 - 2033)
-
Single Lumen
-
Double Lumen
-
Triple Lumen
-
-
Material Outlook (Revenue, USD Million, 2021 - 2033)
-
Polyurethane
-
Silicone
-
-
Shape Outlook (Revenue, USD Million, 2021 - 2033)
-
Straight
-
Curved
-
Pre-Curved
-
-
Component Outlook (Revenue, USD Million, 2021 - 2033)
-
Catheters
-
Catheter Kit
-
-
Usage Outlook (Revenue, USD Million, 2021 - 2033)
-
Long Term
-
Short Term
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Chronic Kidney Disease (CKD)
-
Acute Kidney Injury (AKI)
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Dialysis Centers
-
Ambulatory Surgical Centers (ASCs)
-
Home Healthcare
-
Frequently Asked Questions About This Report
b. The U.S. hemodialysis catheter market size was estimated at USD 408.14 million in 2024.
b. The U.S. hemodialysis catheter market is expected to grow at a compound annual growth rate of 6.9% from 2025 to 2033 to reach USD 741.41 million by 2033.
b. The double-lumen segment dominated the U.S. hemodialysis catheter market, accounting for 42.19% of total revenue in 2024.
b. Some key players operating in the U.S. hemodialysis catheter market include BD, Teleflex Incorporated, Mozarc Medical Holding LLC., Medical Components, Inc., Merit Medical Systems, Medtronic, Healthline Medical Products, and Integer Holdings Corporation.
b. Key factors driving the market growth include the rising prevalence of kidney disorders such as chronic kidney disease (CKD) and acute kidney injury (AKI), supportive government regulations, and continuous technological advancements in catheter design and functionality.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










