- Home
- »
- Clinical Diagnostics
- »
-
U.S. Molecular Diagnostics Market Size, Share Report, 2030GVR Report cover
![U.S. Molecular Diagnostics Market Size, Share & Trends Report]()
U.S. Molecular Diagnostics Market (2023 - 2030) Size, Share & Trends Analysis Report By Disease (Healthcare-associated Infections, Respiratory), By End-use (Hospital Core Laboratory, Decentralized Test Sites), And Segment Forecasts
- Report ID: GVR-4-68040-126-4
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2018 - 2021
- Forecast Period: 2023 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Report Overview
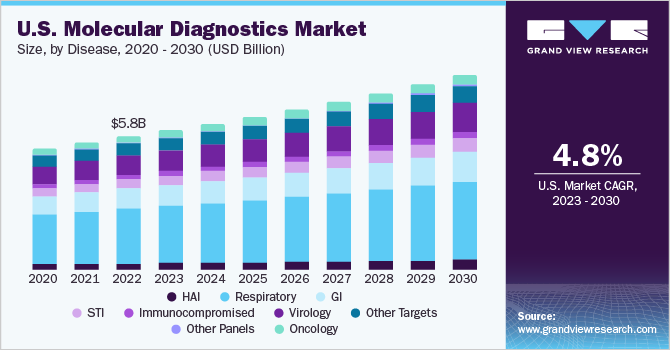
The U.S. molecular diagnostics market size was estimated at USD 5.83 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 4.82% from 2023 to 2030. The increasing prevalence of infections and technological advancements in molecular diagnostic tests drive the market. Furthermore, the presence of favorable reimbursement policies coupled with rising government support in the form of funding and initiatives for the development of diagnostics tests is anticipated to drive the U.S. molecular diagnostics growth over the forecast period.
The COVID-19 pandemic has positively impacted the market by encouraging players to develop novel and rapid diagnostic kits capable of detecting coronavirus. This has resulted in positive revenue growth for major market players operating in the infectious disease segment. For instance, Thermo Fisher Scientific reported 150% revenue growth owing to the rising demand for diagnostics offerings in April 2021. However, the rising adoption of vaccination in the region is anticipated to reduce the demand for COVID-19 testing over the forecast period.
Moreover, increasing incidences of infections in the country primarily drive the demand for cost-effective and efficient diagnostics techniques. According to a study published by the Journal of Clinical Virology in March 2023, enterovirus infection incidents increased by 21.7% compared to the previous year. Furthermore, enterovirus majorly affects the population with ages less than 5 years, of which around 41% of the infected toddlers require supplemental oxygen. In addition, according to the article published by the National Library of Medicine in May 2021, around 40 % of the U.S. population in the U.S. is affected by H. pylori infection every year.
The presence of government support in the form of approvals and funding is anticipated to fuel the market growth over the forecast period. For instance, in May 2022, Abbot received approval for its Alinity m STI Assay from the U.S. FDA. The test is capable of differentiating and detecting STIs. In addition, in 2020, ASPR’s Biomedical Advanced Research and Development Authority and NIH’s National Institute of Allergy and Infectious Diseases organized the Antimicrobial Resistance Diagnostic Challenge in which Visby Medical’s rapid PCR test won USD 19 million in federal funding.
High prices associated with molecular tests are one of the major factors impeding the market for U.S. molecular diagnostics. Furthermore, the scarcity of comparable tests is another reason for price hikes. The significant variations in prices for different applications of each molecular diagnostic product further compound this problem. However, the presence of favorable reimbursement policies in the country is anticipated to fuel the market demand. For instance, in January 2022, the U.S. Department of Health and Human Services announced that individuals with group health plans or covered by private health insurance who purchase at-home COVID-19 tests will get benefits of cost coverage from the insurance or plan.
Disease Insights
The respiratory disease segment dominated the market with a share of 40.79% in 2022. The growth is attributed to the increasing prevalence of respiratory infections such as HPIVs, MDR-TB, and group A streptococcal, among others, coupled with the increasing awareness regarding the availability of diagnostics tests for diagnosing and treatment of infections. According to a study conducted by the CDC, there has been a significant increase in the number of parainfluenza infections till December 2022. This is further contributing to the growing number of hospitalizations caused by HPIVs.
The healthcare-associated infections segment is anticipated to witness the fastest growth from 2023 to 2030. The growth is attributed to the increasing incidences of HAI and technological advancements in diagnostic techniques are driving the growth. According to a study published by the National Library of Medicine in December 2022, around 4% of the total hospitalizations are affected by HAI in the U.S. every year. Furthermore, rising government support in the form of initiatives to reduce the incidence of infections during hospitalizations is anticipated to fuel the growth over the forecast period. For instance, in 2022, the Centers for Medicare & Medicaid Services introduced the Hospital-Acquired Condition (HAC) Reduction Program to encourage hospitals to reduce incidences of infection during hospitalization and improve overall patient safety.
End-use Insights
The hospital core laboratory held the largest share of 38.43% of the market for U.S. molecular diagnostics in 2022 and is anticipated to grow at the fastest CAGR over the forecast period. The segment growth is attributed to the increasing prevalence of infections, such as MRSA, Staphylococcus aureus (SA), and enterovirus. According to a study published by NCBI in May 2022, the prevalence of SA carriage was found to be 30.2% among individuals aged between 18 years and 64 years, 20.7% among children, and 16.7% among people aged more than 65 years.
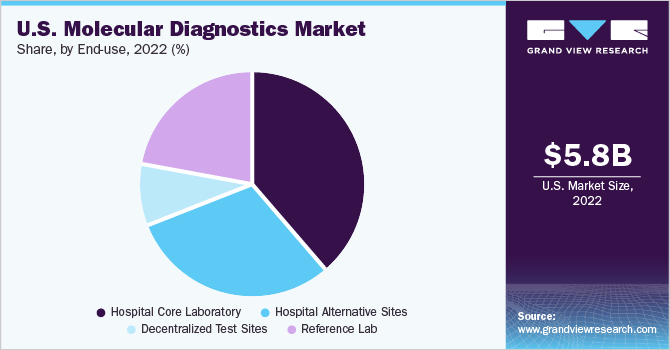
Reference labs are also expected to register a considerable CAGR during the forecast period. Increasing government support through initiatives and reimbursement policies is anticipated to drive market growth over the forecast period. For instance, in March 2023, the U.S. government launched the AHRQ Safety Program for MRSA Prevention. The program is anticipated to boost the demand for molecular diagnostic techniques by creating awareness regarding multidrug-resistant organisms and MRSA transmission.
Key Companies & Market Share Insights
Major players in the market are adopting various strategies such as new product launches, acquisitions, partnerships, collaborations, and expansions to enhance their product penetration and expand their customer base. For instance, in January 2023, QIAGEN N.V. and Helix entered into a strategic partnership for the development of hereditary disease companion diagnostics. This partnership is anticipated to assist QIAGEN N.V. in expanding critical access to providers and patients for genomic testing. Moreover, in May 2021, F. Hoffmann-La Roche Ltd announced the acquisition of GenMark Diagnostics for USD 1.8 billion. The goal of this acquisition is to expand Roche’s syndromic testing portfolio with the help of GenMark’s expertise. Some prominent players in the U.S. molecular diagnostics market include:
-
Abbott
-
Siemens Healthcare GmbH
-
QIAGEN
-
F. Hoffmann-La Roche Ltd
-
Quest Diagnostics Incorporated
-
Danaher
-
Thermo Fisher Scientific, Inc.
-
Hologic Inc.
-
BD
-
Charles River Laboratories
-
Bio-Rad Laboratories, Inc.
-
Illumina, Inc.
-
Agilent Technologies, Inc.
U.S. Molecular Diagnostics Market Report Scope
Report Attribute
Details
Revenue forecast in 2030
USD 8.46 billion
Growth rate
CAGR of 4.82% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Quantitative units
Revenue in USD million/billion, and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Disease, end-use
Key companies profiled
Abbott; Siemens Healthcare GmbH; QIAGEN; F. Hoffmann-La Roche Ltd; Quest Diagnostics Incorporated; Danaher; Thermo Fisher Scientific, Inc.; Hologic Inc.; BD; Charles River Laboratories; Bio-Rad Laboratories, Inc.; Illumina, Inc.; Agilent Technologies, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Molecular Diagnostics Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2018 to 2030. For this report, Grand View Research has segmented the U.S. molecular diagnostics market report based on disease, and end-use:
-
Disease Outlook (Revenue, USD Million, 2018 - 2030)
-
Healthcare-associated Infections (HAI)
-
Methicillin-resistant Staphylococcus Aureus
-
Staphylococcus Aureus
-
Enterovirus
-
Clostridioides Difficile
-
Van A
-
Sepsis Panel
-
-
Respiratory
-
Flu
-
Respiratory Syncytial Virus
-
Paraflu
-
Multidrug-resistant Tuberculosis
-
Mycobacterium Avium-intracellulare
-
Group A Streptococcal
-
Adenovirus
-
Human Metapneumovirus
-
Rhinoviruses
-
Middle East Respiratory Syndrome Coronavirus
-
Tuberculosis
-
Severe Acute Respiratory Syndrome Coronavirus 2
-
Human Bocavirus-Infection
-
Bordetella
-
Mycoplasma Pneumonia
-
Respiratory Panels
-
-
Gastrointestinal (GI)
-
Norovirus
-
Helicobacter Pylori
-
Shiga Toxin
-
GI Parasite Panel
-
GI Bacterial Panel
-
GI Viral Panel
-
GI Panels
-
-
Women’s Health/STI
-
Chlamydia trachomatis (CT)/Neisseria Gonorrhoeae (NG)
-
Trichomonas Vaginalis
-
Mycoplasma Genitalium
-
Human Papillomavirus
-
Bacterial Vaginosis
-
Gonorrhoea Colloquially
-
Mycoplasma Genitalium Resistance
-
Herpes Simplex Virus
-
Group B Streptococcus
-
-
Immunocompromised
-
Cytomegalovirus
-
Epstein-barr Virus
-
Varicella-zoster Virus
-
BK virus
-
-
Virology
-
Human Immunodeficiency Viruses
-
Hepatitis C Virus
-
Hepatitis B Virus
-
Hepatitis E Virus
-
-
Other Targets
-
Babesia
-
Zika
-
West Nile Virus
-
Chikungunya Virus/dengue Virus
-
Enhancer of Zeste Homolog 2
-
PIK3CA
-
Malaria
-
Monkeypox Virus
-
-
Other Panels
-
Meningitis Panel
-
Lyme Panel
-
UTI Panel
-
-
Oncology
-
Microsatellite Instability
-
B-raf
-
Epidermal Growth Factor Receptor
-
Kirsten RAT SARCOMA VIRUS
-
Others
-
-
-
End-use Outlook (Revenue, USD Million, 2018 - 2030)
-
Hospital Core Laboratory
-
Hospital Alternative Sites
-
Decentralized Test Sites
-
Reference Lab
-
Frequently Asked Questions About This Report
b. Some key players operating in the U.S. molecular diagnostics market includeAbbott; Siemens Healthcare GmbH; QIAGEN; F. Hoffmann-La Roche Ltd; Quest Diagnostics Incorporated; Danaher; Thermo Fisher Scientific, Inc.; Hologic Inc.; BD; Charles River Laboratories; Bio-Rad Laboratories, Inc.; Illumina, Inc.; Agilent Technologies, Inc.
b. Key factors that are driving the market growth include increasing prevalence of infections and technological advancements in molecular diagnostic tests.
b. The U.S. molecular diagnostics market was valued at USD 5.83 billion in 2022.
b. The U.S. molecular diagnostics market is expected to expand at a compound annual growth rate (CAGR) of 4.82% from 2023 to 2030 to reach USD 8.46 billion by 2030.
b. The respiratory disease segment dominated the U.S. molecular diagnostics market with a share of 40.79% in 2022. The growth is attributed to the increasing prevalence of respiratory infections such as HPIVs, MDR-TB, and group A streptococcal, among others, coupled with the increasing awareness regarding availability of diagnostics tests for diagnosing and treatment of infections
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










