- Home
- »
- Medical Devices
- »
-
U.S. Pressure Infusor Market Size, Industry Report, 2033GVR Report cover
![U.S. Pressure Infusor Market Size, Share & Trends Report]()
U.S. Pressure Infusor Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Reusable Pressure Infusors, Disposable Pressure Infusors), By Capacity (500 ml, 1000 ml, 3000 ml), By Application, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-869-9
- Number of Report Pages: 90
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Pressure Infusor Market Summary
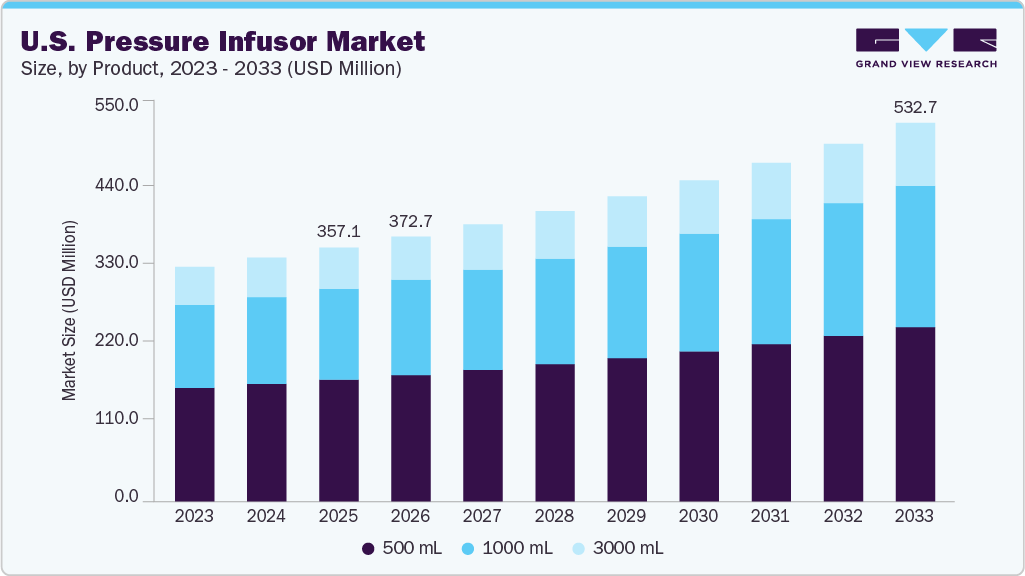
The U.S. pressure infusor market size was estimated at USD 357.11 million in 2025 and is projected to reach USD 532.71 million by 2033, growing at a CAGR of 5.23% from 2026 to 2033. This growth is attributed to the increasing demand for rapid fluid resuscitation in emergency and critical care settings, rising adoption of disposable pressure infusors for infection control, and the growing number of surgical procedures requiring efficient fluid management.
Key Market Trends & Insights
- By product, disposable pressure infusors segment dominated the market with the largest revenue share of 79.97% in 2025.
- By capacity, the 500 mL segment held the largest market share in 2025.
- By application, the emergency & critical care resuscitation segment held the largest market share in 2025
- By end use, the hospitals and clinics segment led the market in 2025 and accounted for the largest market share.
Market Size & Forecast
- 2025 Market Size: USD 357.11 Million
- 2033 Projected Market Size: USD 532.71 Million
- CAGR (2026-2033): 5.23%
The increasing volume of surgical procedures in the U.S. is a key driver for the pressure infusors industry. According to a study published by ScienceDirect in June 2024, the average total cardiac surgical volume in the U.S. reached 271.5 procedures per 100,000 population per year, reflecting the high procedural burden within hospital settings. Cardiac and other major surgeries often require rapid and controlled fluid or blood administration to maintain hemodynamic stability, particularly during intraoperative blood loss or postoperative complications. As surgical volumes continue to rise, hospitals and surgical centers are expected to increase procurement of essential perioperative devices, including pressure infusor bags, thereby supporting market growth in the U.S.The growing burden of hemorrhagic conditions in the U.S. is contributing to increased demand for rapid fluid and blood administration devices. According to the American Heart Association, hemorrhagic strokes account for approximately 13% of all stroke cases, representing a significant subset of patients who may require urgent hemodynamic stabilization and blood product transfusion. Management of hemorrhagic stroke and other acute bleeding emergencies often involves rapid volume replacement to maintain cerebral perfusion and systemic stability. As the incidence of such critical bleeding events persists, hospitals and emergency departments continue to rely on pressure infusor bags for immediate and controlled transfusion support, thereby driving growth in the U.S. pressure infusor industry.
Ongoing product innovation and material advancements are further supporting the growth by improving safety, visibility, and clinical efficiency. Manufacturers are increasingly introducing latex-free materials to minimize allergy risks, along with clear or transparent pressure infusor bags that allow clinicians to visually monitor fluid levels, detect air bubbles, and quickly identify leaks without removing the IV bag. Reinforced seams enhance durability, while upgraded pressure gauges and controlled release valves provide more accurate and stable pressure delivery. Moreover, lightweight, high-strength polymer materials improve product longevity while maintaining cost-effectiveness. These innovations enhance usability in emergency and surgical settings, reduce operational risks, and encourage greater adoption across U.S. healthcare facilities.
Market Concentration & Characteristics
The chart below illustrates the relationship between market concentration, industry characteristics, and industry participants. The x-axis represents the level of industry concentration, ranging from low to high. The y-axis represents various industry characteristics, including industry competition, impact of regulations, level of partnerships & collaborations activities, degree of innovation, and regional expansion.
The degree of innovation in the U.S. pressure infusors industry is moderate, focused on improving safety, infection control, and usability. Manufacturers are introducing latex-free materials, clear bag designs for better fluid visibility, reinforced construction, and accurate pressure gauges. For instance, the Infu-Surg Pressure Infusion Bag is a disposable, latex-free, single-patient multiple-use device, supporting infection prevention and operational efficiency in the U.S. hospitals.
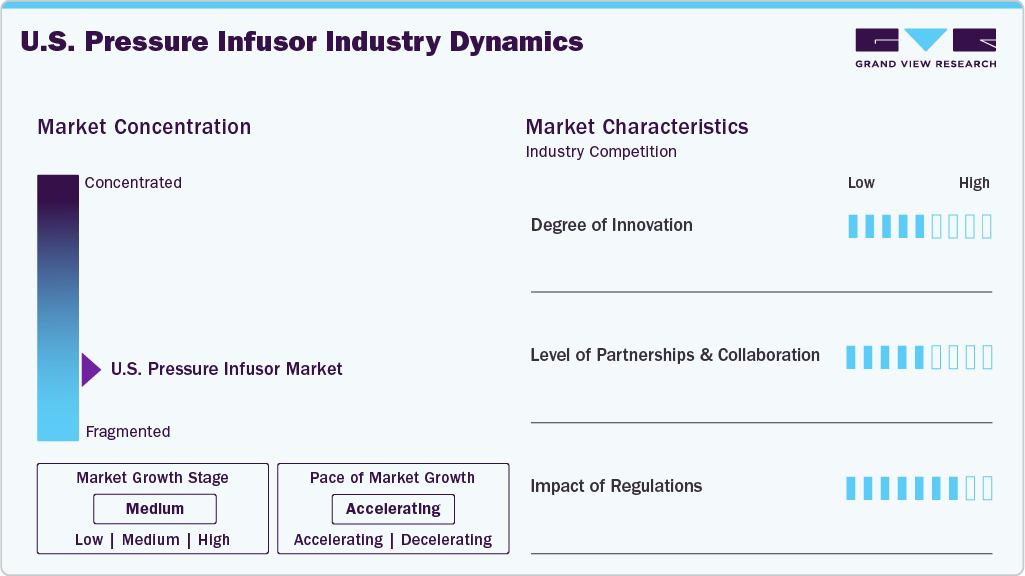
The level of partnerships and collaborations in the U.S. pressure infusors industry is moderate, primarily focused on distribution expansion and supply chain efficiency. Manufacturers partner with medical distributors and group purchasing organizations to improve hospital access, ensure product availability, and strengthen market penetration across emergency and surgical care settings.
Regulations have a high impact on the U.S. pressure infusors industry, as devices must comply with requirements set by the U.S. Food and Drug Administration, including quality system, labeling, and applicable premarket regulations. These standards increase compliance costs and create entry barriers but ensure product safety, reliability, and infection control adherence, strengthening overall market credibility and stability.
Product Insights
The disposable pressure infusors segment held the largest revenue share of 79.97% in 2025 and it is expected to grow at the fastest CAGR over the forecast period. This growth is attributed to their ability to reduce cross-contamination risks, eliminate the need for sterilization, offer ease of handling, and comply with modern hospital infection-control protocols, making them the preferred choice in most clinical settings. In addition, rising awareness of hospital-acquired infections and stringent regulatory guidelines are accelerating their adoption, while improvements in material durability and cost-effectiveness further support market growth.
Moreover, the reusable segment is expected to grow at a significant CAGR over the forecast period. This growth is driven by the cost-sensitive healthcare settings and resource-limited hospitals, particularly where sterilization infrastructure already exists. They remain preferred in military, emergency medical services, and rural facilities due to their durability and lower per-use cost. However, growth is moderate due to increasing regulatory scrutiny and rising infection control standards, which are shifting procurement preferences toward single-use devices.
Capacity Insights
Based on capacity, the 500 ml segment held the largest revenue share in 2025. This growth is driven by its widespread use in emergency departments, ICUs, and routine surgical procedures, where rapid but controlled fluid infusion is required. Its compact size, ease of handling, and compatibility with blood and drug infusion protocols make it a standard volume across acute care settings, supporting consistent demand growth.
The 1000 mL segment is expected to grow at the fastest CAGR over the forecast period, due to increasing surgical procedures, blood transfusions, and prolonged infusion therapies, where moderate-to-large fluid volumes are required. It offers an optimal balance between volume and usability, leading to broad adoption in operating rooms and critical care units.
Application Insights
Based on application, the emergency & critical care resuscitation segment dominated the market in 2025. This segment is driven by the rising incidence of trauma, cardiovascular emergencies, and critical illness cases where rapid fluid delivery is essential for patient stabilization. Pressure infusors are integral to resuscitation protocols in emergency departments, ambulances, and trauma centers, ensuring precise and rapid fluid administration.
However, surgical fluid management is expected to register the fastest CAGR over the forecast period. This segment is driven by growth in elective and minimally invasive surgeries, orthopedic and cardiovascular procedures, and perioperative fluid management requirements. The shift toward outpatient and ambulatory surgeries further boosts demand for compact and efficient pressure infusion devices.
End Use Insights
Based on end use, hospitals and clinics held the largest share in 2025 among all end-use segments, as they account for the highest volume of emergency, critical care, and surgical procedures, have well-established infrastructure for pressure infusor use, and prioritize rapid fluid resuscitation and patient safety. In addition, their larger budgets and access to advanced medical devices support the widespread adoption of both disposable and reusable pressure infusors.
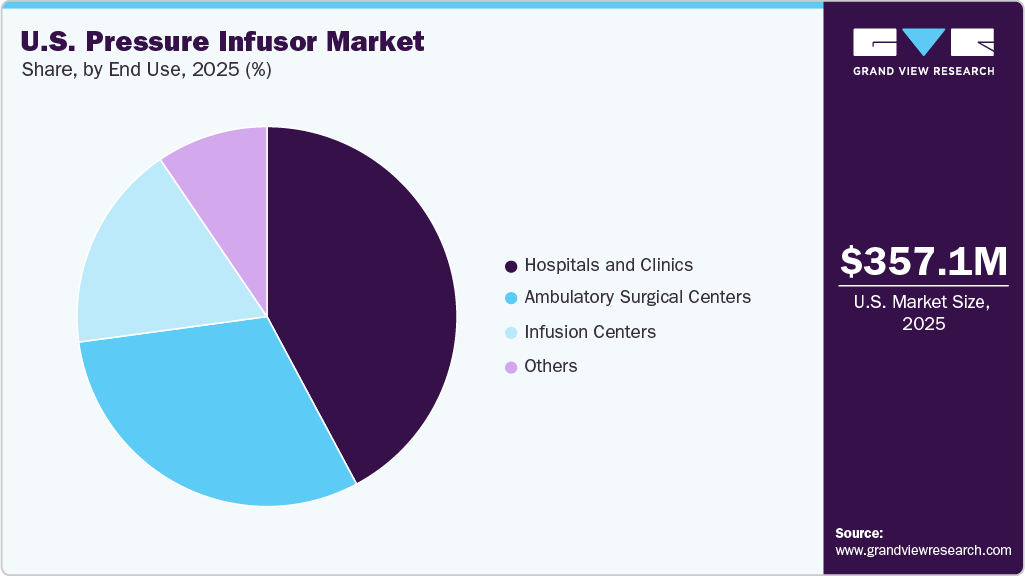
ASCs are a fast-growing end-use segment, driven by the shift from inpatient to outpatient surgeries, cost containment strategies, and minimally invasive procedures. ASCs increasingly use disposable pressure infusion systems integrated into surgical kits, improving workflow efficiency and compliance with infection control standards.
Key U.S. Pressure Infusor Company Insights
The market is fragmented, with a few players dominating the U.S. pressure infusor industry. These key vendors leverage strategies such as product innovation, strategic partnerships, and acquisitions to strengthen their market position, drive overall growth, and set industry benchmarks for safety, efficiency, and clinical adoption.
Key U.S. Pressure Infusor Companies:
- ICU Medical, Inc.
- Medline
- Merit Medical Systems.
- ASP Global (acquired by Platinum Equity)
- Tapmedic LLC
- Spacelabs Healthcare
- AirLife
- VBM Medical Inc.
Recent Developments
- In August 2024, Platinum Equity announced it has acquired ASP Global, a strategic healthcare partner that develops, sources and distributes consumable medical products for providers and distributors.
U.S. Pressure Infusor Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 372.73 million
Revenue forecast in 2033
USD 532.71 million
Growth rate
CAGR of 5.23% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, capacity, application, end use
ICU Medical, Inc.; Medline; Merit Medical Systems; ASP Global (acquired by Platinum Equity); Tapmedic LLC; Spacelabs Healthcare; AirLife; VBM Medical Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Pressure Infusor Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. Pressure infusor market report based on product, capacity, application, and end use.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Reusable pressure infusors
-
Disposable pressure infusors
-
-
Capacity Outlook (Revenue, USD Million, 2021 - 2033)
-
500 mL
-
1000 mL
-
3000 mL
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Emergency & Critical Care Resuscitation
-
Blood and Blood Product Transfusion
-
Surgical Fluid Management
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals and Clinics
-
Ambulatory Surgical Centers
-
Infusion Centers
-
Others
-
Frequently Asked Questions About This Report
b. The U.S pressure infusor market size was estimated at USD 357.11 million in 2025 and is expected to reach USD 372.73 million in 2026.
b. The U.S pressure infusor market is expected to grow at a compound annual growth rate of 5.23% from 2026 to 2033 to reach USD 532.71 million by 2033.
b. Based on product type the disposable pressure infusors segment held a larger revenue share of 79.97% in 2025 and are expected to grow at fastest CAGR. This growth is attributed to their ability to reduce cross-contamination risks, eliminate the need for sterilization, offer ease of handling, and comply with modern hospital infection-control protocols, making them the preferred choice in most clinical settings.
b. Some key players operating in the U.S pressure infusor market include ICU Medical, Inc., Medline, Merit Medical Systems, ASP Global (acquired by Platinum Equity), Tapmedic LLC, Spacelabs Healthcare, AirLife, VBM Medical Inc.
b. Key factors that are driving the market growth include the increasing demand for rapid fluid resuscitation in emergency and critical care settings, rising adoption of disposable pressure infusors for infection control, and the growing number of surgical procedures requiring efficient fluid management
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










