- Home
- »
- Medical Devices
- »
-
U.S. Renal Denervation Catheter Market Size Report, 2033GVR Report cover
![U.S. Renal Denervation Catheter Market Size, Share & Trends Report]()
U.S. Renal Denervation Catheter Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Radiofrequency (RF) Ablation Catheters, Ultrasound Ablation Catheters), By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-682-8
- Number of Report Pages: 150
- Format: PDF
- Historical Range:
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Renal Denervation Catheter Market Summary
The U.S. renal denervation catheter market size was estimated at USD 23.20 million in 2025 and is projected to reach USD 182.70 million by 2033, growing at a CAGR of 29.43% from 2025 to 2033. The growth is driven by the rising prevalence of hypertension and growing awareness about the limitations of pharmacological treatments, increasing clinical evidence supporting the safety and efficacy of renal denervation, and favorable regulatory advancements, such as the recent FDA approval, are further accelerating market adoption.
Key Market Trends & Insights
- By product, the radiofrequency ablation catheters segment led the market with the largest revenue share in 2025 and is expected to grow at 28.86% CAGR.
- By product, the ultrasound ablation catheters segment is expected to register the fastest CAGR from 2025 to 2033.
- By end use, the hospital segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 23.20 million
- 2033 Projected Market Size: USD 182.70 million
- CAGR (2025-2033): 29.43%
According to data released by the National Center for Health Statistics and the National Health and Nutrition Examination Survey in October 2024, the prevalence of hypertension among U.S. adults was 23.4% for those aged 18-39, rising to 52.5% for individuals aged 40-59, and reaching 71.6% for those aged 60 and older during the period from August 2021 to August 2023.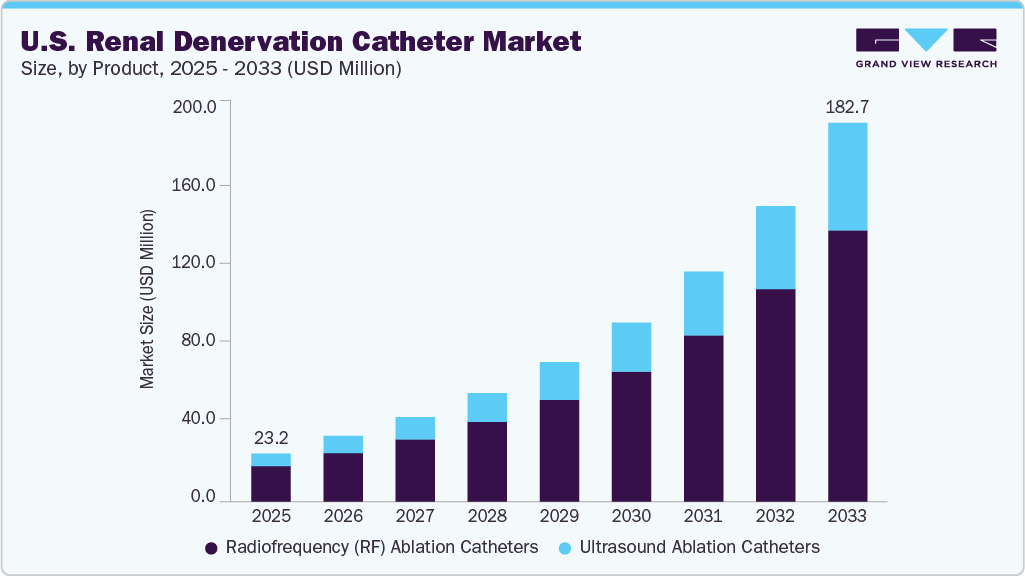
The high prevalence of hypertension is one of the primary drivers for the U.S. renal denervation catheter industry. In addition, a growing subset of patients suffers from treatment-resistant hypertension, which does not respond adequately to multiple antihypertensive medications. This widespread burden of disease creates a strong clinical need for alternative therapeutic interventions, such as renal denervation, which offers a non-pharmacological approach to long-term blood pressure control. As awareness and diagnosis rates improve, the eligible patient pool for RDN procedures is expected to expand, further propelling market demand.
According to a CDC update in January 2025:
-
In 2022, high blood pressure was a primary or contributing cause of 685,875 deaths in the U.S.
-
About 1 in 5 deaths related to COVID-19 occurred in people with a history of hypertensive disease.
-
Nearly half of U.S. adults (48.1%)-approximately 119.9 million people-have high blood pressure, defined as:
-
Systolic BP >130 mm Hg
-
Diastolic BP >80 mm Hg
-
Or are taking medication for hypertension.
-
Only 1 in 4 adults (22.5%) with high blood pressure have their condition under control (approx. 27.0 million people).
-
About 45% of adults with uncontrolled high blood pressure have readings of 140/90 mm Hg or higher, representing 37 million adults.
-
34 million U.S. adults who should be on antihypertensive medications are either untreated or not adhering to prescribed treatment.
-
Among them, 19 million have blood pressure ≥140/90 mm Hg.
On March 25, 2025, the CDC Foundation, in partnership with AstraZeneca, launched a new National Hypertension Control Program under its Million Hearts Alliance. This two-year initiative targets the critical issue of uncontrolled hypertension, which affects nearly half of U.S. adults and contributes to around 75% of cardiovascular deaths.
The program aims to reduce mortality through a multi-pronged strategy:
-
National awareness campaigns to educate the public on hypertension risks and management.
-
Community outreach and capacity-building, including resources for local clinics and healthcare teams.
-
Strengthened clinician-community linkages, piloted through initiatives like the Atlanta Hypertension Initiative.
The CDC Foundation’s National Hypertension Control Program is expected to boost the U.S. renal denervation catheter market by increasing hypertension awareness, improving diagnosis rates, and strengthening clinical care pathways. By targeting uncontrolled and treatment-resistant hypertension, the program expands the eligible patient pool for RDN procedures.
“We are excited to join the CDC Foundation in raising awareness about uncontrolled and hard-to-treat hypertension and its impact on public health. By accelerating diagnosis and treatment, we can reduce the unnecessary risk of heart attacks, strokes, heart failure, and kidney failure- improving long-term health outcomes and, ultimately, patient lives.”- Sarah Walters, vice president, U.S. Cardiovascular, Renal and Metabolism.
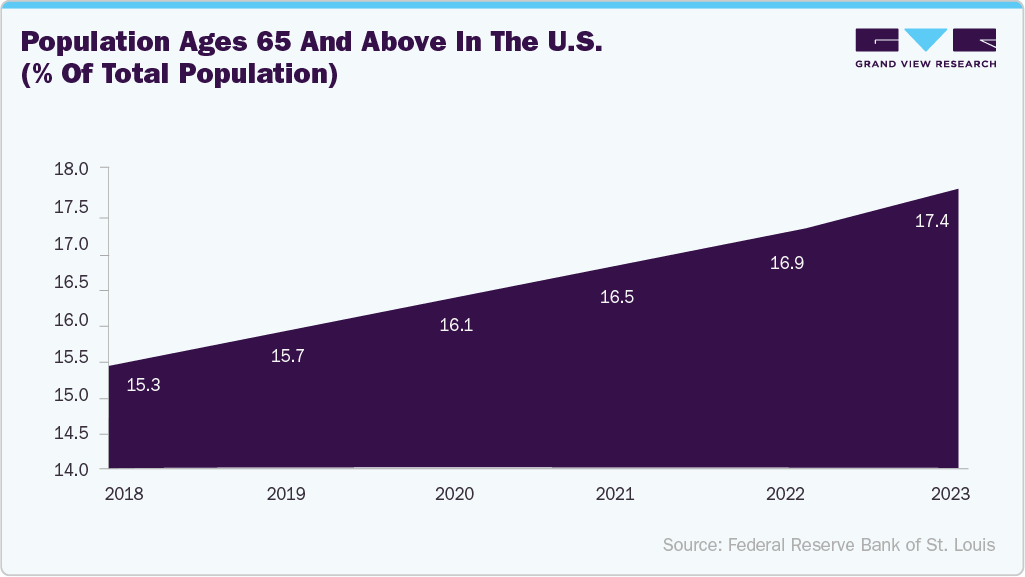
Furthermore, the aging population in the U.S. is a key driver of the renal denervation catheter market due to the strong correlation between age and hypertension. As individuals age, physiological changes, such as arterial stiffness, reduced kidney function, and increased sympathetic nervous system activity, contribute to a higher risk of developing high blood pressure. According to the Population Reference Bureau, the number of Americans ages 65 and older is projected to increase from 58 million in 2022 to 82 million by 2050 (a 47% increase), and the 65-and-older age group’s share of the total population is projected to rise from 17% to 23%. Older adults are also more likely to have resistant hypertension and multiple comorbidities, making medication management more complex and less effective. Renal denervation offers a minimally invasive, long-term treatment option for this group, especially for those who cannot tolerate or adhere to multiple antihypertensive drugs. As the elderly population grows, the demand for durable, low-maintenance solutions such as RDN is expected to increase, thereby driving market growth.
The figure below shows the growth of the U.S. aging population, rising from 16.47% in 2021 to 17.93% by 2024. This upward trend reflects increasing awareness, adoption of advanced hypertension treatments, and supportive national health initiatives.
Market Concentration & Characteristics
The market growth stage is high, and the pace of the market growth is accelerating. The U.S. renal denervation catheter industry is characterized by increasing clinical adoption, strong regulatory support, and a growing prevalence of treatment-resistant hypertension. Rising investments in innovative RDN technologies, favorable reimbursement developments, and national health programs targeting hypertension control are further propelling market expansion.
The U.S. renal denervation catheter market exhibits a high degree of innovation, marked by the development of advanced technologies such as ultrasound-based, radiofrequency, and chemical ablation systems. Companies are investing heavily in clinical trials and next-generation devices to improve efficacy, safety, and ease of use.
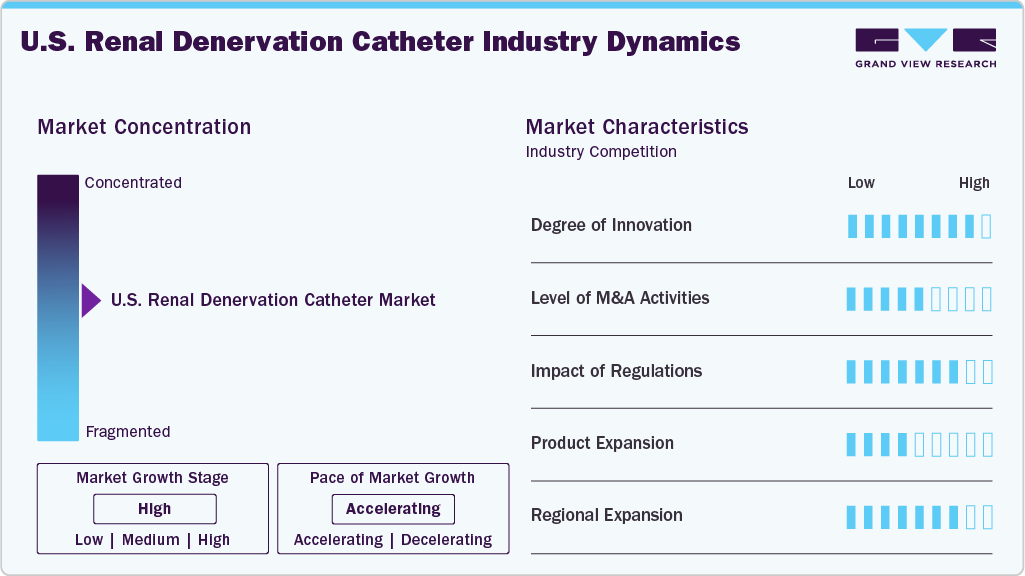
The U.S. renal denervation catheter industry has seen significant mergers and acquisitions (M&A) activity. While not highly saturated, the market attracts strategic investments as larger medtech companies seek to strengthen their cardiovascular portfolios and access promising RDN technologies. Partnerships between device manufacturers and clinical research organizations are growing to support trial expansion and accelerate market entry. As the market matures and reimbursement pathways become clearer, M&A activity is expected to increase.
Regulations play a significant role in shaping the U.S. renal denervation catheter market. The recent FDA approval of innovative RDN systems, such as Recor’s Paradise Ultrasound Renal Denervation System, signals growing regulatory confidence in their safety and efficacy. Stringent clinical trial requirements ensure high standards but can delay market entry for new players. However, supportive regulatory pathways like the FDA’s Breakthrough Device Designation help expedite approval processes and boost innovation.
Product expansion in the U.S. renal denervation catheter industry is accelerating as key players introduce next-generation devices with improved precision, safety, and ease of use. Companies are expanding their RDN portfolios by developing technologies across different ablation methods, such as radiofrequency and ultrasound, to target diverse patient populations. Furthermore, ongoing clinical trials and FDA approvals are paving the way for broader indications and adoption, strengthening the market presence of existing players and attracting new entrants.
Regional expansion is gaining momentum as companies move beyond major urban centers to target underserved and high-risk populations across various states. With the support of national hypertension control programs and growing awareness among healthcare providers, manufacturers are collaborating with regional health systems and academic institutions to expand clinical trials and device availability. This geographic outreach enhances market penetration and ensures broader access to innovative RDN therapies across the country.
Product Insights
The Radiofrequency (RF) ablation catheters segment dominated the U.S. renal denervation catheter market in 2024, owing to their established clinical efficacy, procedural familiarity, and broad physician acceptance. These systems deliver controlled thermal energy to ablate the renal sympathetic nerves, effectively reducing blood pressure in patients with treatment-resistant hypertension. With its Symplicity Spyral system, companies such as Medtronic have led extensive clinical studies showing RF-based renal denervation's safety and effectiveness, supporting physician confidence and adoption. The maturity of RF technology, combined with a growing number of FDA-approved devices, makes it the most commercially viable option in the current landscape. Furthermore, RF systems offer the advantage of real-time feedback and precision, making them suitable for varied anatomical presentations. RF catheters continue to hold a leading position due to their proven track record, robust clinical data, and widespread integration into interventional cardiology practices.
The ultrasound ablation catheters segment is anticipated to register the fastest CAGR during the forecast period. This rapid growth is driven by the increasing adoption of non-thermal, circumferential energy delivery systems that offer enhanced precision and reduced procedural time. Devices such as Recor Medical’s Paradise Ultrasound Renal Denervation System, which received FDA approval, present the market’s shift toward innovative technologies that address the limitations of radiofrequency systems. Ultrasound-based RDN offers a less operator-dependent technique with consistent energy delivery, making it particularly attractive to new users and institutions aiming for procedural standardization. Furthermore, strong clinical trial results, such as those from the RADIANCE program, support the efficacy and safety of ultrasound-based RDN, enhancing its credibility in clinical practice. As awareness grows and reimbursement pathways evolve, the adoption of ultrasound ablation catheters is expected to surge, positioning them as the fastest-growing segment in the forecast years.
"Despite the longstanding availability of dozens of affordable anti-hypertensive medications, blood pressure control rates in the U.S. are alarmingly low and falling. Given the significant blood pressure reductions seen in trials, ultrasound renal denervation offers a much-needed advancement in our currently available options to control hypertension. uRDN has proven efficacy in patients with truly resistant hypertension, a population for whom medication therapy often fails. It is also effective in patients with mild to moderate hypertension who cannot tolerate enough medication to control their blood pressure."- Naomi Fisher, MD, Associate Professor of Medicine, Harvard Medical School, and Director of Hypertension Service and Hypertension Innovation, Division of Endocrinology, Diabetes and Hypertension at Brigham and Women's Hospital.
End Use Insights
The hospital segment held the dominant share in the U.S. renal denervation catheter industry in 2024, driven by the high volume of complex cardiovascular procedures performed in these settings. Hospitals have advanced imaging technologies, trained interventional cardiologists, and the infrastructure necessary to support minimally invasive procedures such as renal denervation. Moreover, the availability of emergency care and post-procedural monitoring makes hospitals the preferred choice for both patients and healthcare providers. As leading institutions adopt innovative technologies such as the Paradise Ultrasound RDN System, hospitals play a central role in the early adoption and diffusion of renal denervation therapies. In addition, favorable reimbursement frameworks and clinical trial participation further solidify hospitals as the primary revenue-generating end users in this market. For instance, in March 2024, Physicians at the Tampa General Hospital (TGH) Heart & Vascular Institute became the first in the region to implement a technology for managing hypertension. The Paradise Ultrasound Renal Denervation System, developed by Recor Medical, utilizes ultrasound energy to ablate renal nerves and effectively reduce high blood pressure.
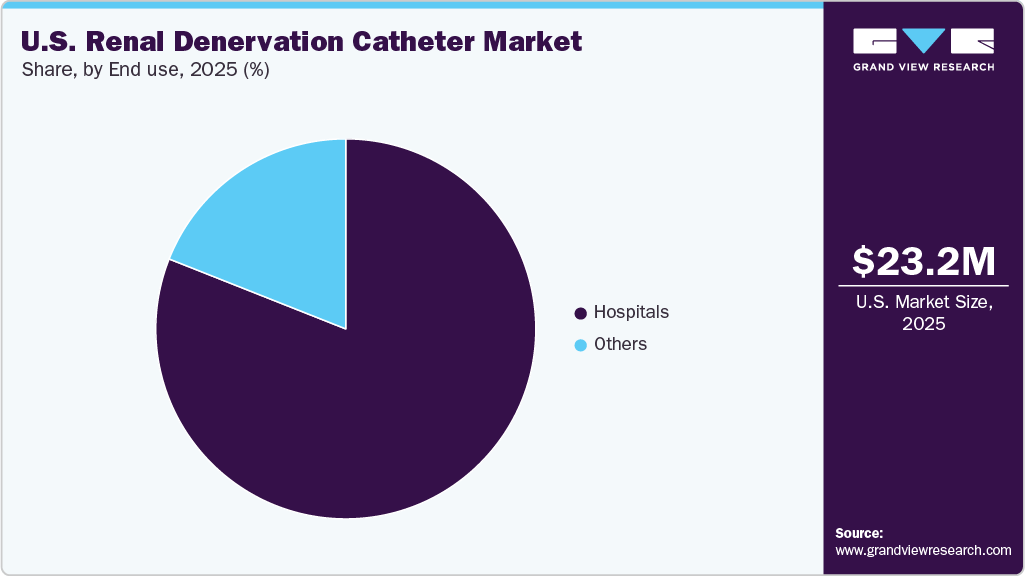
The other renal denervation catheter market segment is expected to register the fastest CAGR during the forecast period. This expansion is primarily attributed to the increasing decentralization of healthcare services, where procedures that required inpatient hospital care are safely and effectively conducted in outpatient or specialized facilities. Ambulatory surgical centers offer reduced procedural costs, shorter wait times, and faster recovery environments, making them an attractive option for providers and patients seeking renal denervation therapies. Specialty clinics, particularly those focused on hypertension and cardiovascular care, are integrating renal denervation into their treatment offerings, supported by growing awareness of its clinical benefits and the availability of compact, minimally invasive technologies. Furthermore, academic and research institutions are pivotal in accelerating renal denervation adoption by conducting clinical trials, generating real-world evidence, and enabling innovation. These settings often serve as early adopters of advanced medical technologies and are critical in shaping clinical guidelines and best practices.
Key U.S. Renal Denervation Catheter Company Insights
The key U.S. renal denervation catheter market players are adopting various strategies to gain revenue share, such as product launches, product approvals, mergers and acquisitions, and geographic expansions.
Key U.S. Renal Denervation Catheter Companies:
- Recor Medical, Inc. (Otsuka Medical Devices Co., Ltd.)
- Medtronic
- Boston Scientific Corporation
- MicroPort Scientific Corporation
- EnligHTN, St. Jude Medical (Abbott Laboratories)
- Ablative Solutions, Inc.
Recent Developments
-
In March 2025, Boston Scientific Corporation announced that it has entered into a definitive agreement to acquire SoniVie Ltd., a privately held medical device company. SoniVie has developed the TIVUS Intravascular Ultrasound System, an investigational technology designed to denervate nerves surrounding blood vessels to address various hypertensive conditions, including renal artery denervation (RDN) for hypertension.
-
In January 2025, Medtronic, one of the global leaders in healthcare technology, announced that the Centers for Medicare & Medicaid Services (CMS) has initiated a national coverage analysis (NCA) for renal denervation. This process will enable CMS to review and establish a nationwide Medicare coverage policy for renal denervation procedures in patients with hypertension.
-
In January 2025, Recor Medical, Inc. and its parent company, Otsuka Medical Devices Co., Ltd. (“Otsuka Medical Devices”), announced that the Centers for Medicare & Medicaid Services (CMS) had initiated a National Coverage Analysis (NCA) for RDN, a device-based treatment for hypertension.
U.S. Renal Denervation Catheter Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 23.20 million
Revenue forecast in 2033
USD 182.70 million
Growth rate
CAGR of 29.43% from 2025 to 2033
Actual data
2025
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends, and volume analysis
Segments covered
Product, end use
Regional scope
U.S.
Key companies profiled
Recor Medical, Inc. (Otsuka Medical Devices Co., Ltd.); Medtronic; Boston Scientific Corporation; MicroPort Scientific Corporation; EnligHTN; St. Jude Medical (Abbott Laboratories); Ablative Solutions, Inc.
Customization scope
Free report customization (equivalent to 8 analysts working days) with purchase. Addition or alteration to country, & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Renal Denervation Catheter Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2025 to 2033. For this study, Grand View Research has segmented the U.S. renal denervation catheter market report based on product and end use:
-
Product Outlook (Revenue, USD Million, 2025 - 2033)
-
Radiofrequency (RF) Ablation Catheters
-
Ultrasound Ablation Catheters
-
-
End Use Outlook (Revenue, USD Million, 2025 - 2033)
-
Hospitals
-
Other
-
Frequently Asked Questions About This Report
b. The U.S. renal denervation catheter market size was estimated at USD 23.20 million in 2025.
b. The U.S. renal denervation catheter market is expected to grow at a compound annual growth rate of 29.43% from 2025 to 2033 to reach USD 182.70 million by 2033.
b. Based on product, the radiofrequency ablation catheters segment led the market with the largest revenue share in 2025 and is expected to grow at 28.86% CAGR.
b. Some key players operating in the U.S. renal denervation catheter market include Recor Medical, Inc. (Otsuka Medical Devices Co., Ltd.), Medtronic, Boston Scientific Corporation, MicroPort Scientific Corporation, EnligHTN, St. Jude Medical (Abbott Laboratories), Ablative Solutions, Inc.
b. The U.S. renal denervation catheter market is driven by the rising prevalence of treatment-resistant hypertension, technological advancements in catheter design, and recent FDA approvals that have boosted physician confidence. Growing awareness of non-drug alternatives, an aging population with comorbidities, and supportive healthcare infrastructure further support market growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










