- Home
- »
- Medical Devices
- »
-
U.S. Ultrasound Probe Disinfection Market Size Report, 2033GVR Report cover
![U.S. Ultrasound Probe Disinfection Market Size, Share & Trends Report]()
U.S. Ultrasound Probe Disinfection Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Instruments, Consumables), By Method (High-Level Disinfection, Intermediate/Low-Level Disinfection), By End Use (Hospitals & Clinics), And Segment Forecasts
- Report ID: GVR-4-68040-667-1
- Number of Report Pages: 90
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Ultrasound Probe Disinfection Market Summary
The U.S. ultrasound probe disinfection market size was estimated at USD 202.04 million in 2024 and is projected to reach USD 414.94 million by 2033, growing at a CAGR of 8.38% from 2025 to 2033. The demand for ultrasound probe disinfection in the U.S. is on the rise, driven by the growing prevalence of chronic conditions such as heart disease and cancer, increasing reliance on medical imaging, a surge in product approvals, and ongoing technological advancements in probe disinfection methods.
Key Market Trends & Insights
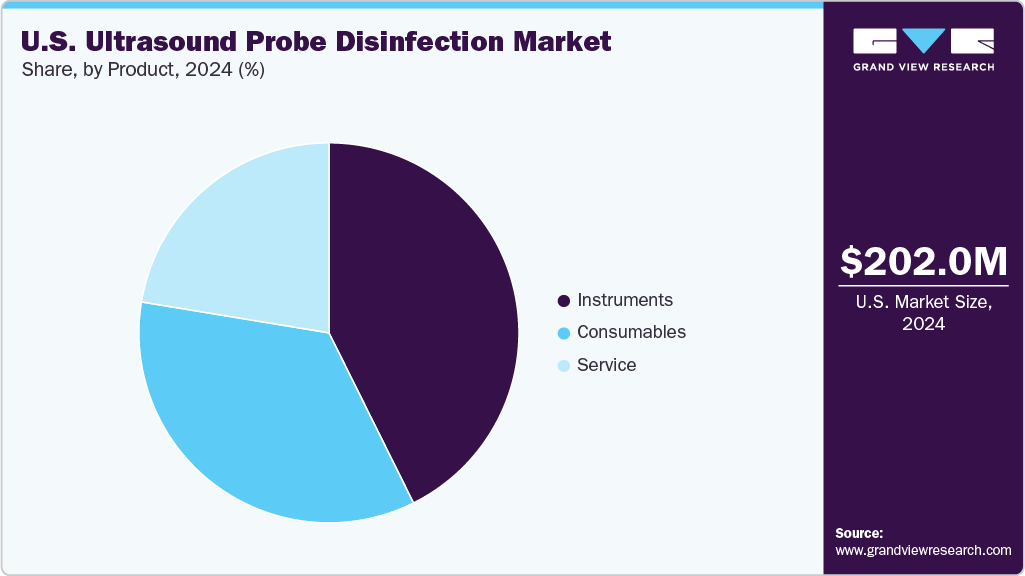
- By product, the instruments segment led the market with the largest revenue share of 42.66% in 2024.
- By method, the High-Level Disinfection (HLD) segment is anticipated to grow fastest over the forecast period.
- By end-use, the hospitals and clinics segment led the market with the largest revenue share of 43.46% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 202.04 Million
- 2033 Projected Market Size: USD 414.94 Million
- CAGR (2025-2033): 8.38%
The rising burden of chronic disorders is a significant driver for the market. The growing prevalence of cardiovascular diseases is fueling the demand for echocardiograms-ultrasound procedures of the heart that use phased array probes. According to data released by MedStar Health in January 2024, over 7 million echocardiograms are conducted annually in the U.S., underscoring the increasing dependence on cardiac ultrasound for diagnostic purposes. This substantial volume of procedures necessitates regular and thorough disinfection of ultrasound probes to prevent cross-contamination and safeguard patient health. As the use of phased array probes continues to grow in cardiac procedures, stringent disinfection protocols become even more critical. Therefore, the expanding adoption of cardiac ultrasound is expected to drive the growth of the ultrasound probe disinfection market.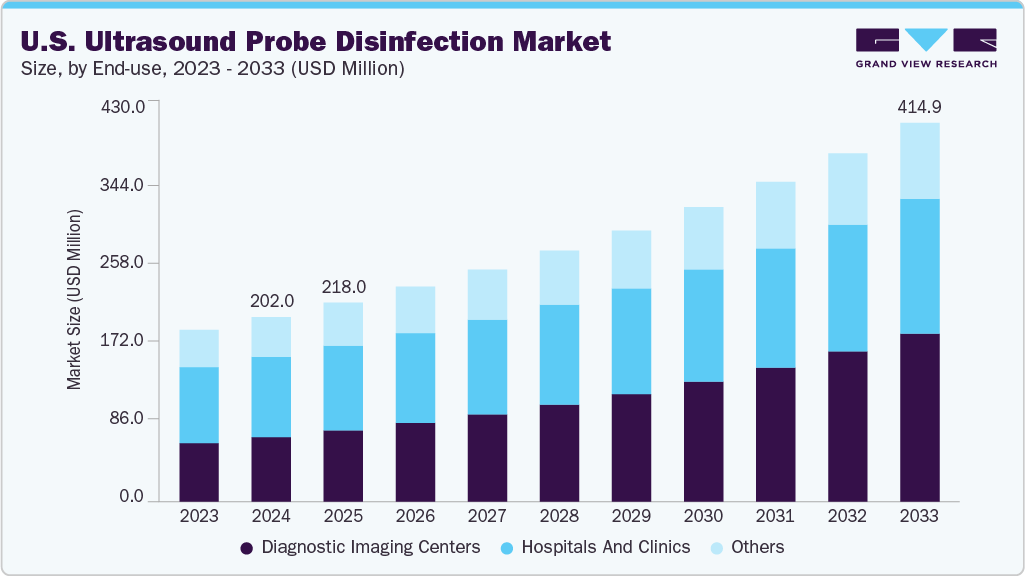
Furthermore, the growing burden of health conditions like rectal cancer, prostate cancer, infertility, and heart disease is expected to significantly increase the demand for high-level disinfection solutions that can disinfect semi critical probes in the coming years. As semi-critical probes come into direct contact with mucous membranes (such as the vagina, rectum, and pharynx), they require high-level disinfection to ensure patient safety and prevent the transmission of infections.
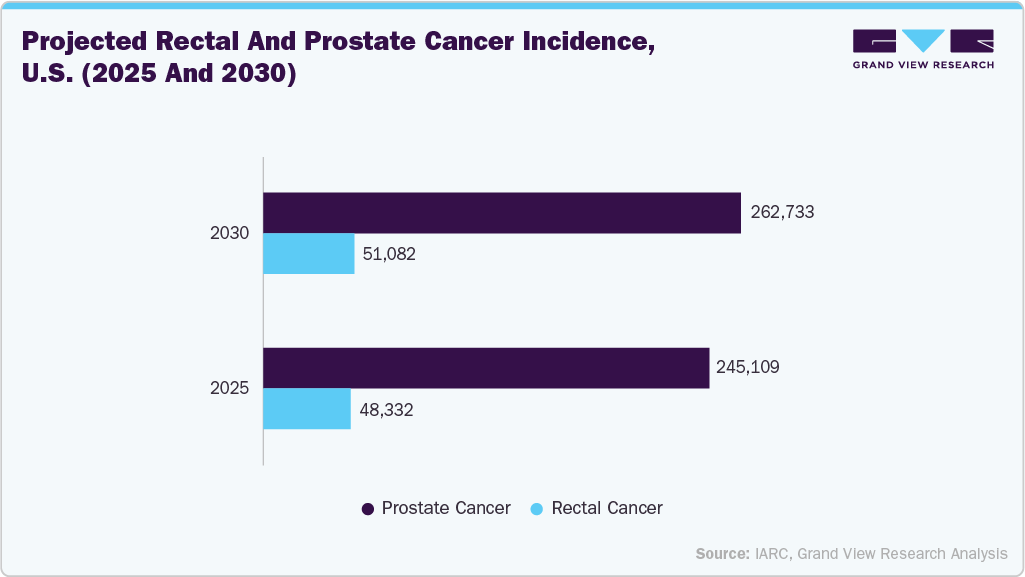
The above data indicates the growing burden of rectal and prostate cancer in the U.S. The rising prevalence of rectal and prostate cancer in the U.S. is contributing to a growing demand for diagnostic procedures, particularly transrectal ultrasound. As the use of these procedures increases, effective disinfection of ultrasound probes becomes critically important to prevent cross-infection and maintain patient safety. Compliance with infection control standards is essential in clinical settings. Therefore, the growing cancer burden is expected to significantly boost the growth of the U.S. ultrasound probe disinfection market in the coming years.
Moreover, the rising introduction of regulations and guidelines by government bodies and healthcare organizations is anticipated to drive the demand for the ultrasound probe disinfection market. These directives encourage developing and adopting advanced disinfection products that meet updated safety standards. For example, in April 2024, the Association for the Advancement of Medical Instrumentation (AAMI) released a comprehensive guideline on the sterile processing of ultrasound probes in healthcare settings. Such documents provide clear protocols for effective probe disinfection. They are expected to accelerate market growth by guiding manufacturers in developing compliant, innovative solutions and encouraging broader adoption across clinical environments.
The growing incidence of healthcare-associated infections (HAIs) in the U.S. is expected to drive the demand for ultrasound probe disinfection solutions significantly. According to data published by the CDC in May 2024, approximately one in 31 hospital patients acquires at least one HAI on any given day. This alarming statistic underscores the urgent need for infection prevention strategies. As a result, there is increasing emphasis on the use of advanced disinfection technologies for medical devices, including ultrasound probes, to reduce cross-contamination and transmission risks. This heightened focus on infection control is anticipated to propel the growth of the U.S. ultrasound probe disinfection market in the coming years.
Key Opinion Leaders
Company Name
KOLs
Growth Opportunities
CS Medical
“Ethos is another example of our continued commitment to working with health care professionals and other professional organizations in the reduction of HAIs and increasing awareness for a better and safer health care system. In addition to our employees, I am thankful and appreciative of the cooperation and assistance given to us by various ultrasound probe manufacturers, without whom Ethos would not be the product it is today.”
- Industry collaborations
- Growing focus on HAIs reduction
- Development of automated disinfector
Parker Laboratories
“More than 215 million ultrasound scans are performed in the United States each year, with approximately 20% of them requiring high-level disinfection. Tristel ULT is the first product of its kind in the US, a high-level disinfectant foam for reprocessing ultrasound probes. As the exclusive manufacturer of Tristel ULT and Tristel DUO in the US, Parker Laboratories now offers a complete line of disinfecting solutions for ultrasound departments. These new products are just a few of the innovative and cost-effective solutions we are committed to bringing to market to increase patient safety and improve workflow in the healthcare setting."
- Distribution partnerships
- Development of cost effective solutions
- Growing demand for high level disinfectants
Source: Grand View Research Analysis
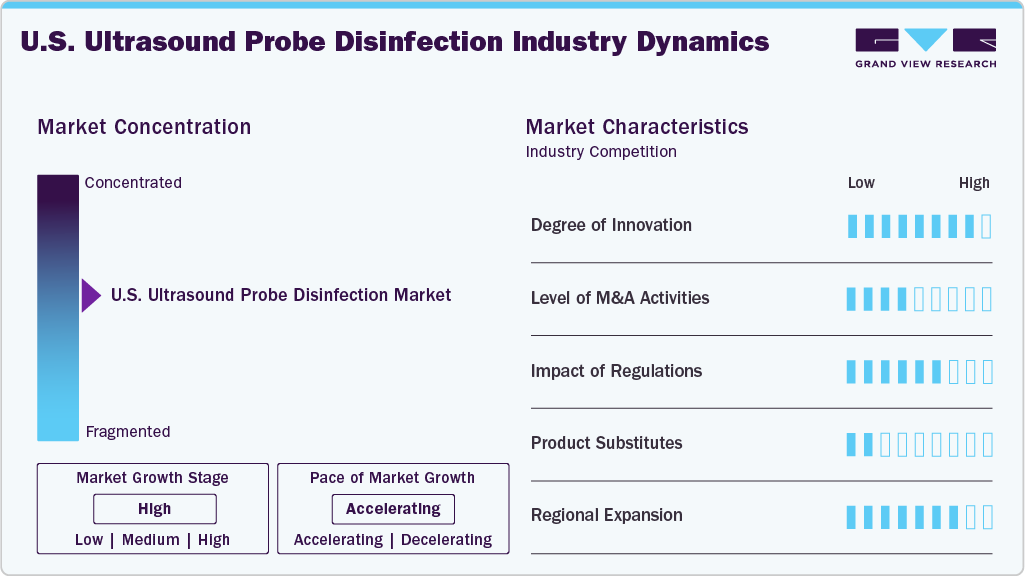
Market Concentration & Characteristics
The market growth stage is high, and the pace of growth is accelerating. The U.S. ultrasound probe disinfection market is characterized by high growth due to the rising burden of chronic diseases, increasing focus on infection controls, supportive government and regulatory initiatives, and the growing launches of novel products.
Industry players are innovating their offerings by integrating novel technologies in probe disinfection instruments. Emerging technologies within the ultrasound probe disinfection space include chemical-free UV-C disinfection, automated disinfection systems, and digital traceability & infection control management systems. Industry participants are actively designing automated disinfection solutions to fulfill this growing demand, with regulatory bodies encouraging market expansion through timely authorizations of innovative products. For instance, in June 2023, CS Medical introduced the FDA-approved Ethos Automated Ultrasound Probe Cleaner Disinfector, the first North American system to integrate high-level disinfection (HLD) and automated cleaning for surface and endocavity probes.
In the U.S., ultrasound probe disinfection products are regulated primarily by the Food and Drug Administration (FDA) as medical devices. FDA oversees the marketing authorization of these products, including instruments and consumables. For example, in October 2023, the Germitec Chronos, a UV radiation disinfection chamber designed for high-level disinfection of transvaginal, external, and transrectal ultrasound probes, received De Novo approval from the U.S. FDA.
Table 1 Some of the FDA-cleared sterilants and high level disinfectants used for probe disinfection are mentioned below:
Product / Approval Number
Manufacturer
Active Ingredients
High Level Disinfectant Contact Conditions
TD-5 High-level Disinfectant (K051305)
CS Medical, L.L.C.
2.65% Glutaraldehyde
5 min at 37.8ºC
Single use to be used exclusively with the TD-100 Transesophageal Probe Disinfector.
Contact conditions established by simulated use testing with endoscopes.
Tristel Duo ULT High Level Disinfectant Foam for Ultrasound Probes (DEN220041)
Tristel Solutions Limited
320 ppm Chlorine dioxide
For Manual application using DUO Wipes: 2 min at 20°C
Single use - foam generated on site
For processing of endocavity transvaginal, transrectal and skin surface transducers only.
Contact conditions established by simulated use testing with transducer.
Source: Food and Drug Administration (U.S. FDA)
Industry players are increasingly focusing on expanding their product reach within the U.S. market through strategic distribution partnerships with OEMs, e-commerce platforms, and distributors. For example, in September 2022, Parker Labs partnered with Tristel to exclusively manufacture and distribute Tristel DUO disinfectant foam for ultrasound transducer and medical surface disinfection in the U.S. These partnerships are expected to play a key role in driving the U.S. ultrasound probe disinfection market growth.
Method Insights
The intermediate level disinfection/low-level disinfection segment accounted for the largest share of the market in 2024. The ease of use, lower cost, and availability of disinfectant wipes and sprays support the growth of the intermediate-level or low-level disinfection segment. Additionally, increasing adoption in outpatient settings and routine diagnostic procedures continues to drive demand for this disinfection method.
The high-level disinfection segment is projected to be the fastest-growing segment in the U.S. ultrasound probe disinfection market from 2025 to 2033. This growth is driven by the increasing availability of FDA-approved, cost-effective high-level disinfectants tailored for use with ultrasound probes. For example, in June 2023, the U.S. FDA granted clearance for Tristel ULT, a novel disinfecting foam approved for high-level disinfection of ultrasound probes used in body cavities and skin surfaces. Such approvals are expected to support segment expansion significantly.
End-use Insights
The hospitals & clinics segment dominated the market in 2024, accounting for the largest share of 43.46%. The high procedure volume, availability of advanced equipment, trained healthcare staff, and strong patient preference for hospital-based care drive this dominance. Additionally, strict infection control protocols and higher adoption of disinfection technologies further support growth in this segment.
The diagnostic imaging centers segment is projected to register the highest CAGR from 2025 to 2033. This growth is driven by increasing investment in diagnostic imaging infrastructure, a high volume of ultrasound procedures, and the rising demand for outpatient imaging services. Factors such as faster patient turnaround, cost-effective services, and growing awareness of infection control practices are further expected to boost segment growth.
Product Insights
The instruments segment dominated the market, accounting for 42.66% of total revenue in 2024. It is also anticipated to grow fastest over the forecast period. The availability of various advanced instruments, including automated reprocessors, UV-C disinfectors, manual reprocessors/soaking stations, and ultrasound probe storage cabinets, drives this growth. Additionally, increasing FDA approvals for innovative disinfection instruments are expected to propel segment growth further. For example, in June 2023, CS Medical received U.S. FDA clearance for its new Class II medical device, the Ethos Automated Ultrasound Probe Cleaner Disinfector. Such approvals for advanced disinfection instruments are expected to boost the segment’s expansion significantly.
The service segment is expected to grow significantly in the U.S. ultrasound probe disinfection market. This growth is driven by the increasing adoption of disinfection services tailored to meet the rising demands of healthcare facilities, ensuring compliance with infection control regulations and enhancing patient safety.
Key U.S. Ultrasound Probe Disinfection Companies Insights
Key players operating in the U.S. ultrasound probe disinfection market are undertaking various initiatives to strengthen their presence and increase the reach of their products and services. Strategies such as expansion activities and partnerships are key in propelling the market growth.
Key U.S. Ultrasound Probe Disinfection Companies:
- ASP (Fortive)
- Nanosonics
- CIVCO Medical Solutions
- Tristel Plc
- Ecolab Inc.
- Germitec
- CS Medical LLC
- Steelco S.p.A
- STERIS
- EDM Medical Solutions
- GE HealthCare
- UV Smart
- The Clorox Company
- Ruhof Corporation
Recent Developments
-
In February 2025, Germitec announced the successful completion of a USD 30 million financing round. This funding will support the company's efforts to accelerate growth in the U.S. market and further advance innovations in UV-C infection prevention technologies.
-
In November 2024, Nanosonics announced that the trophon Wireless Ultrasound Probe Holder is available in the U.S.It is specifically designed for HLD of wireless ultrasound probes.
-
In September 2024, the U.S. FDA granted its first-ever De Novo classification for an ultraviolet radiation disinfection chamber device to Germitec’s Chronos. This chemical-free UV-C High-level Disinfection (HLD) device is designed to disinfect endocavitary and external ultrasound probes.
U.S. Ultrasound Probe Disinfection Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 218.03 million
Revenue forecast in 2033
USD 414.94 million
Growth rate
CAGR of 8.38% from 2025 to 2033
Base year for estimation
2024
Actual data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, method, end-use
Country scope
U.S.
Key companies profiled
ASP (Fortive); Nanosonics; CIVCO Medical Solutions; Tristel Plc; Ecolab Inc.; Germitec; CS Medical LLC; Steelco S.p.A; STERIS; EDM Medical Solutions; GE HealthCare; The Clorox Company; UV Smart; Ruhof Corporation
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Ultrasound Probe Disinfection Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. ultrasound probe disinfection market report based on product, method, and end-use:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Instruments
-
Automated Reprocessors
-
UV-C Disinfectors
-
Manual Reprocessors/Soaking Stations
-
Ultrasound Probe Storage Cabinets
-
-
Consumables
-
Consumables, By Formulations
-
Disinfectant Liquids
-
Disinfectant Wipes
-
Disinfectant Sprays
-
-
Consumables, By Detergent
-
Enzymatic Detergents
-
Non-enzymatic Detergents
-
-
-
Service
-
-
Method Outlook (Revenue, USD Million, 2021 - 2033)
-
High-Level Disinfection
-
Intermediate Level Disinfection/Low-Level Disinfection
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals and Clinics
-
Diagnostic Imaging Centers
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. ultrasound probe disinfection market size was estimated at USD 202.04 million in 2024 and is expected to reach USD 218.03 million in 2025.
b. The U.S. ultrasound probe disinfection market is expected to grow at a compound annual growth rate of 8.38% from 2025 to 2033 to reach USD 414.94 million by 2033.
b. The instruments segment dominated the U.S. ultrasound probe disinfection market, accounting for 42.66% of total revenue in 2024. This dominance can be attributed to the availability of various advanced instruments, including automated reprocessors, UV-C disinfectors, manual reprocessors/soaking stations, and ultrasound probe storage cabinets.
b. Some key players operating in the U.S. ultrasound probe disinfection market include ASP (Fortive), Nanosonics, CIVCO Medical Solutions, Tristel Plc, Ecolab Inc., Germitec, CS Medical LLC, Steelco S.p.A, STERIS, EDM Medical Solutions, GE HealthCare, The Clorox Company, UV Smart, and Ruhof Corporation.
b. Key factors that are driving the market growth include the growing prevalence of chronic conditions such as heart disease and cancer, increasing reliance on medical imaging, a surge in product approvals, and ongoing technological advancements in probe disinfection methods.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










