- Home
- »
- Medical Devices
- »
-
U.S. Umbilical Vessel Catheters Market, Industry Report 2033GVR Report cover
![U.S. Umbilical Vessel Catheters Market Size, Share & Trends Report]()
U.S. Umbilical Vessel Catheters Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Umbilical Arterial Catheters, Umbilical Venous Catheters), By Material, By Channel, By Indication/Use, By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-822-9
- Number of Report Pages: 300
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Umbilical Vessel Catheters Market Summary
The U.S. umbilical vessel catheters market size was estimated at USD 165.39 million in 2024 and is projected to reach USD 258.83 million by 2033, growing at a CAGR of 5.21% from 2025 to 2033. This growth is attributed to the rising prevalence of preterm births and neonatal complications that require immediate vascular access for life-saving interventions.
Key Market Trends & Insights
- By product, the umbilical venous catheters (UVCs) segment led the market with the largest revenue share of 58.63%in 2024.
- By material, the polyurethane-based catheters segment led the market with the largest revenue share of 52.90% in 2024.
- Based on channel, the single lumen segment led the market with the largest revenue share of 67.05% in 2024.
- Based on indication/use, the parenteral nutrition segment led the market with the largest revenue share of 22.93% in 2024.
- By end use, the hospitals (NICUs) segment led the market with the largest revenue share of 79.85% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 165.39 Million
- 2033 Projected Market Size: USD 258.83 Million
- CAGR (2025-2033): 5.21%
Growing investments in NICU infrastructure, coupled with the expansion of Level III and Level IV neonatal care units, further strengthen demand for the product. Technological improvements, such as biocompatible materials, kink-resistant designs, and enhanced catheter tip positioning, also enhance adoption. Furthermore, heightened clinical emphasis on infection prevention and safe neonatal vascular access supports the continued use of advanced UVC and UAC systems.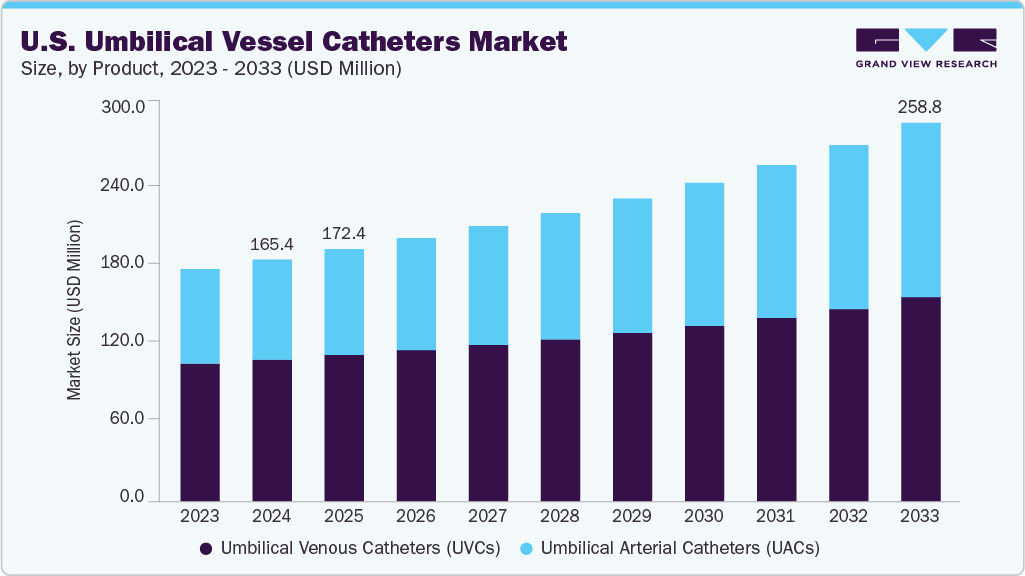
The rising incidence of preterm births is a primary factor driving the growth of the U.S. umbilical vessel catheters industry. Preterm infants often require immediate and reliable vascular access for administering fluids, medications, and parenteral nutrition in neonatal intensive care units (NICUs). Umbilical vessel catheters (UVCs and UACs) play a crucial role in stabilizing these infants during the critical early stages of life. With increasing global preterm birth rates and improved survival outcomes, the demand for advanced, safe, and infection-resistant catheters continues to rise. Moreover, growing awareness among healthcare professionals and the expansion of neonatal care infrastructure further support market growth worldwide. According to the Reproductive Health Journal, the number of preterm births remained high globally, with an estimated 13.4 million cases in 2020. According to the Centers for Disease Control and Prevention (CDC), both early and late preterm birth rates increased by 4% from 2020 to 2021 but later declined by 1%-2% from 2021 to 2022.
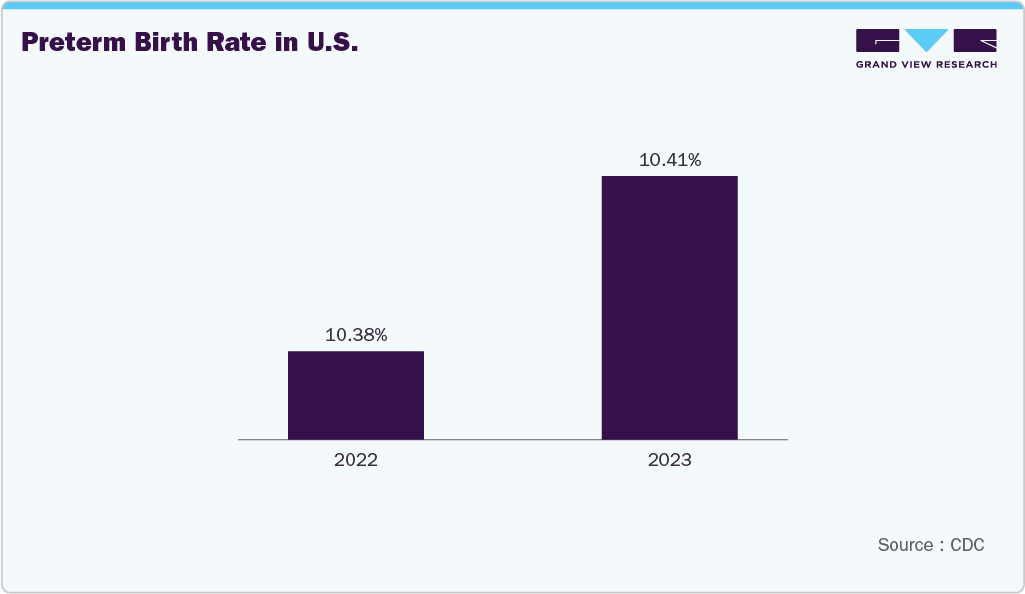
The growing need for advanced neonatal intensive care is a major factor driving the U.S. umbilical vessel catheters industry. As the number of preterm and low-birth-weight infants continues to rise, the demand for reliable and safe vascular access solutions has increased significantly. Umbilical vessel catheters play a crucial role in delivering essential medications, fluids, and nutrition while allowing continuous monitoring of critically ill newborns. The expansion of neonatal intensive care units (NICUs) and the adoption of advanced medical technologies are further enhancing their use. In addition, the focus on improving neonatal survival rates and reducing complications is accelerating the adoption of high-performance, infection-resistant catheters across hospitals worldwide.
The expansion of Level III and Level IV neonatal care units is another driver of the U.S. umbilical vessel catheter industry, as these advanced facilities manage the highest volume of premature and critically ill newborns requiring immediate vascular access. As more hospitals upgrade their NICU capabilities, the demand for reliable UVCs and UACs increases to support life-saving interventions, including fluid resuscitation, parenteral nutrition, medication delivery, and continuous monitoring. These units follow stringent clinical protocols that standardize the use of umbilical catheters, ensuring consistent utilization. As investment in specialized neonatal infrastructure increases, the need for high-quality catheters becomes even more crucial in delivering advanced newborn care.
Market Characteristics & Concentration
The U.S. umbilical vessel catheters industry is characterized by a moderately structured market, with a few established medical device companies dominating the neonatal vascular-access products market. High regulatory standards, strong brand loyalty among hospitals, and the need for proven clinical safety limit rapid shifts in market share.
The degree of innovation in the U.S. umbilical vessel catheters industry is steadily increasing, driven by the need for safer and more reliable neonatal vascular access. Manufacturers are focusing on advanced materials, such as polyurethane and silicone, to enhance flexibility, biocompatibility, and catheter longevity. Innovations in tip design, depth markings, and compatibility with imaging technologies also enhance placement accuracy. In addition, efforts to reduce infection risk through improved sterilization and securement solutions continue to shape new product development.

The level of mergers and acquisitions in the U.S. umbilical vessel catheters industry is relatively moderate, primarily driven by larger medical device companies expanding their portfolios in neonatal care. Established players often acquire smaller firms specializing in vascular access technologies to strengthen product offerings and integrate innovative designs. These strategic moves help companies enhance market presence and streamline distribution channels. Consolidation activity supports competitive positioning but remains measured due to the niche size of the market.
Regulations have a significant impact on the U.S. umbilical vessel catheters industry, as manufacturers must comply with strict FDA standards for safety, biocompatibility, and device performance. These requirements increase development timelines and costs but also ensure high product reliability for neonatal use. Hospitals (NICUs) tend to favor vendors with strong regulatory track records, which supports trust and fosters consistent demand. Regulatory oversight supports market stability while encouraging continuous quality improvement.
Product substitutes in the U.S. umbilical vessel catheters industry are limited, as UVCs and UACs remain the standard for rapid neonatal vascular access in critical care settings. Peripheral IV lines or peripherally inserted central catheters (PICCs) act as alternatives, but they are not suitable for immediate post-birth interventions. Substitutes cannot match the speed, placement site, or functionality required in emergencies; their impact on reducing UVC/UAC demand is minimal.
Product Insights
The umbilical venous catheters (UVCs) segment led the market with the largest revenue share of 58.63%in 2024, due to their critical role in providing rapid central venous access immediately after birth. They are widely used for administering fluids, medications, parenteral nutrition, and emergency interventions, making them indispensable in Level III and IV NICUs. Their versatility, ease of placement, and compatibility with essential neonatal procedures further strengthen their adoption across hospitals. As neonatal care standards continue to advance, UVCs remain the preferred choice for early and effective vascular management in newborns.
The umbilical arterial catheters (UACs) segment is expected to grow at the fastest CAGR during the forecast period, driven by their essential role in continuous hemodynamic monitoring and frequent blood gas sampling in critically ill neonates. As NICUs prioritize real-time physiological assessment and tighter clinical management, the demand for reliable arterial access has accelerated. Advances in catheter design, improved biocompatibility, and reduced complication rates are also supporting the wider adoption of this technology. With the expansion of high-acuity neonatal care capabilities, UAC usage is expected to rise steadily across U.S. hospitals.
Material Insights
The polyurethane-based catheters segment led the market with the largest revenue share of 52.90% in 2024 and is expected to grow at the fastest CAGR during the forecast period, driven by their superior flexibility, biocompatibility, and reduced risk of vascular irritation compared to traditional PVC options. Their ability to soften at body temperature ensures safer long-term placement, making them preferred in advanced NICUs. As clinicians prioritize improved patient comfort and lower complication rates, adoption of polyurethane catheters continues to accelerate. Ongoing material innovation and demand for premium neonatal devices further support their rapid market expansion.
The PVC-coated catheters segment is anticipated to grow at a significant CAGR during the forecast period, driven by their long-established clinical use, affordability, and reliable performance in neonatal care. Hospitals continue to prefer PVC-based designs because they provide adequate stiffness for easier insertion while maintaining acceptable biocompatibility for short-term use. Their cost-effectiveness makes them especially attractive for high-volume NICUs and routine emergency procedures. As a result, PVC-coated catheters remain a widely trusted and consistently utilized option across U.S. neonatal centers.
Channel Insights
The single lumen segment led the market with the largest revenue share of 67.05% in 2024, due to its widespread use in routine neonatal care and its suitability for most early-life interventions. Clinicians prefer these devices for their simplicity, ease of placement, and lower risk of complications compared to multi-lumen options. They are commonly used for fluid administration, medication delivery, and initial stabilization in NICUs. Their cost-effectiveness and strong clinical familiarity further support their leading position across U.S. hospitals.
The triple-lumen segment is expected to grow at the fastest CAGR during the forecast period, due to its ability to support multiple therapies simultaneously. They enable clinicians to administer medications, nutrition, and blood products without repeated line changes, which is especially valuable in critically ill neonates. As NICUs handle more complex and high-acuity cases, the demand for multi-channel access solutions continues to rise. Improved designs that enhance safety and reduce line complications are further accelerating the adoption of triple-lumen systems.
Indication/Use Insights
The parenteral nutrition segment led the market with the largest revenue share of 22.95% in 2024, due to its vital intervention for preterm and critically ill newborns who cannot tolerate enteral feeding immediately after birth. UVCs are widely used to deliver essential nutrients, electrolytes, and glucose, supporting early metabolic stability and growth. As NICUs prioritize optimal nutritional management to reduce complications and improve outcomes, reliance on catheter-based parenteral nutrition remains high. This consistent clinical need strengthens its leading position within the market.
The segment of neonates with difficult venous access (DVA) for emergency resuscitation is expected to grow at the fastest CAGR during the forecast period, driven by the critical need for rapid and reliable vascular access in life-threatening situations. In these cases, UVCs provide immediate central access when peripheral veins are inaccessible or collapse. As hospitals strengthen their emergency response protocols and high-acuity neonatal admissions increase, demand for quick-placement catheters continues to rise. This trend is further supported by the growing reliance of clinicians on standardized resuscitation guidelines that prioritize umbilical access in DVA scenarios.
End Use Insights
The hospitals (NICUs) segment led the market with the largest revenue share of 79.85% in 2024, due to their high volume of preterm and critically ill newborns requiring immediate vascular access. These facilities rely heavily on UVCs and UACs for administering medications, nutrition, and monitoring vital parameters during the first hours of life. Their advanced infrastructure, skilled neonatal teams, and adherence to standardized care protocols drive consistent catheter utilization. As NICUs continue to expand and adopt more sophisticated neonatal care practices, hospitals remain the primary end users fueling market demand.
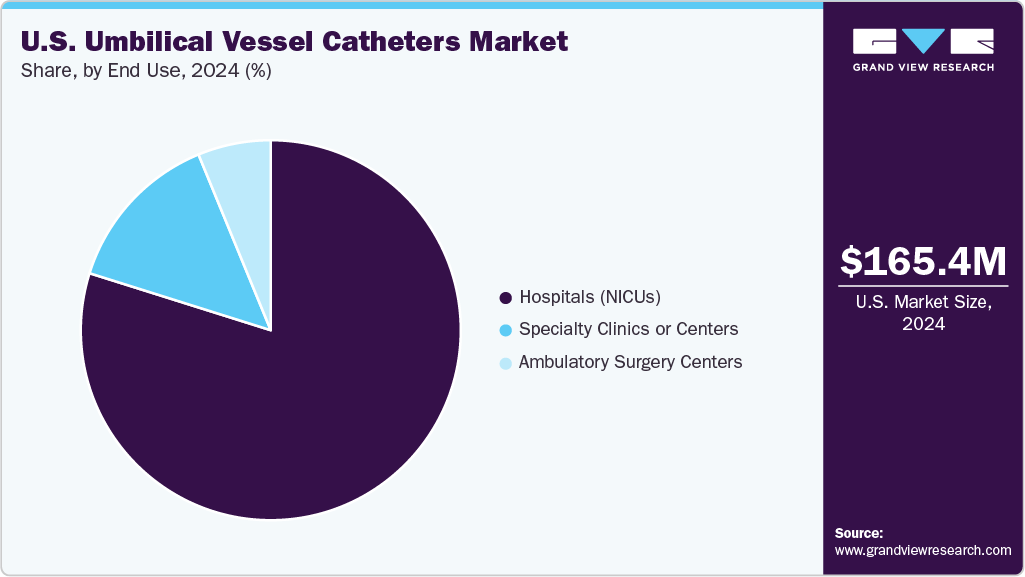
The specialty clinics or centers segment is expected to grow at the fastest CAGR during the forecast period, driven by their increasing role in providing focused, high-acuity care for premature and at-risk newborns. These centers are expanding their capabilities with dedicated neonatal teams and advanced equipment, enabling them to manage complex cases that previously required hospital NICUs. As referral networks strengthen and more clinics adopt standardized neonatal resuscitation and vascular-access protocols, their use of UVCs and UACs is rising steadily. This growth reflects a broader shift toward specialized, decentralized neonatal services across the U.S.
Key U.S. Umbilical Vessel Catheters Company Insights
The U.S. umbilical vessel catheter industry is led by a mix of established medical device manufacturers that hold strong positions due to long-standing relationships with hospitals and proven product reliability. Market share is concentrated among companies offering high-quality, biocompatible catheters supported by solid regulatory compliance. Competition focuses on performance, neonatal safety features, and clinician trust rather than aggressive pricing.
Key U.S. Umbilical Vessel Catheters Companies:
- Vygon
- Utah Medical Products, Inc.
- Cardinal Health
- Laborie
- AMECATH
- Neotech Products
Recent Developments
- In February 2023, Laborie Medical Technologies Corp., a leading diagnostic and therapeutic medical technology company, announced the acquisition of Novonate, Inc. and its innovative LifeBubble technology. This device is designed to enhance the protection and security of umbilical catheters, thereby improving safety and reducing complications for neonates in intensive care environments.
U.S. Umbilical Vessel Catheters Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 172.43 million
Revenue forecast in 2033
USD 258.83 million
Growth rate
CAGR of 5.21% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends, and volume analysis
Segments covered
Product, material, channel, indication/use, end use
Regional scope
U.S.
Key companies profiled
Vygon; Utah Medical Products, Inc.; Cardinal Health; Laborie; AMECATH; Neotech Products
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Umbilical Vessel Catheters Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. umbilical vessel catheters market report based on product, material, channel, indication/use and end use:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Umbilical Arterial Catheters (UACs)
-
Umbilical Venous Catheters (UVCs)
-
-
Material Outlook (Revenue, USD Million, 2021 - 2033)
-
Polyurethane-based Catheters
-
Silicone-based Catheters
-
PVC-coated Catheters
-
Others
-
-
Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Single Lumen
-
Double Lumen
-
Triple Lumen
-
-
Indication/Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Neonates with Difficult Venous Access (DVA) for Emergency Resuscitation
-
Exchange Blood Transfusion
-
Cardiac Catheterisation
-
Parenteral Nutrition
-
Drug Administration
-
Frequent Blood Sampling/Arterial Blood Gas Monitoring
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals (NICUs)
-
Specialty Clinics or Centers
-
Ambulatory Surgery Centers
-
Frequently Asked Questions About This Report
b. The U.S. umbilical vessel catheters market size was valued at USD 165.39 million in 2024 and is expected to reach the value of USD 172.43 million by 2025.
b. The U.S. umbilical vessel catheters market is projected to grow at a CAGR of 5.21% from 2025 to 2033 to reach USD 258.83 million by 2033.
b. The umbilical venous catheters (UVCs) segment dominates the U.S. umbilical vessel catheters market in 2024 due to their critical role in providing rapid central venous access immediately after birth. They are widely used for administering fluids, medications, parenteral nutrition, and emergency interventions, making them indispensable in Level III and IV NICUs.
b. Some of the key players operating in the market include Vygon, Utah Medical Products, Inc., Cardinal Health, Laborie, and AMECATH, among others.
b. The U.S. umbilical vessel catheters market is driven by increasing neonatal intensive care, rising preterm birth rates, technological advancements, and growing awareness of minimally invasive procedures. Additionally, the demand for better neonatal vascular access solutions and expanding healthcare infrastructure contribute to market growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










