- Home
- »
- Medical Devices
- »
-
Antibody CDMO Market Size & Share, Industry Report, 2033GVR Report cover
![Antibody Contract Development & Manufacturing Organization Market Size, Share & Trends Report]()
Antibody Contract Development & Manufacturing Organization Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Monoclonal Antibodies (mAbs)), By Service, By Source, By Workflow, By Therapeutic Area, By End-use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-116-7
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Antibody CDMO Market Summary
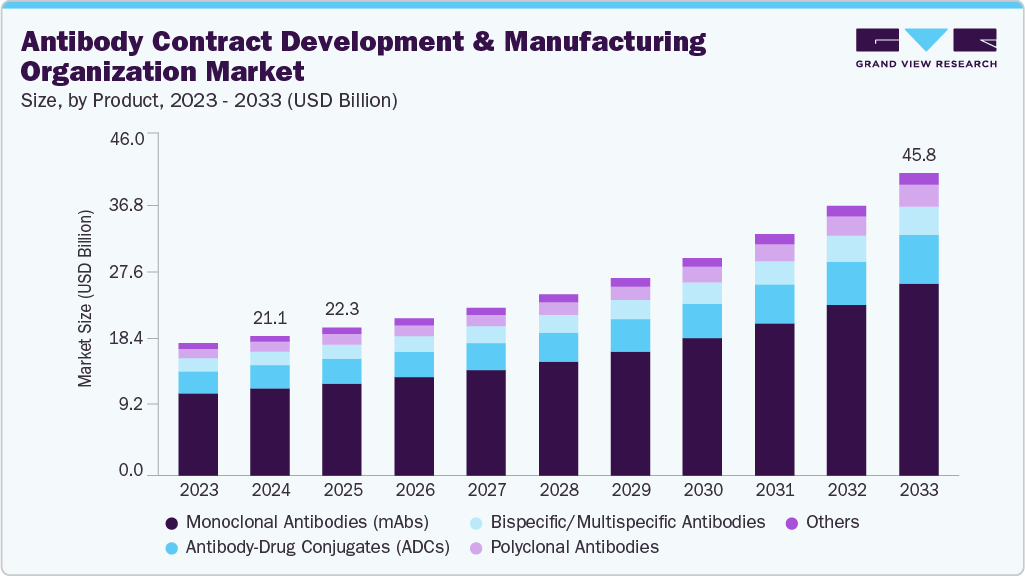
The global antibody contract development & manufacturing organization market size was estimated at USD 21.1 billion in 2024 and is projected to reach USD 45.8 billion by 2033, growing at a CAGR of 9.38% from 2025 to 2033. The antibody contract development & manufacturing organization (CDMO) industry is driven by increasing global demand for monoclonal antibodies and biologics.
Key Market Trends & Insights
- The North America antibody CDMO market held the largest share of 43.40% of the global market in 2024.
- The antibody CDMO market in the U.S. is expected to grow significantly over the forecast period.
- Based on product, the monoclonal antibodies (mAbs) segment held the largest market share in 2024.
- Based on service, the contract development segment held the largest market share in 2024.
- Based on source, the microbial segment held the largest market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 21.1 Billion
- 2033 Projected Market Size: USD 45.8 Billion
- CAGR (2025-2033): 9.38%
- North America: Largest market in 2024
Besides, rising demand for outsourcing services, Rapid advancements in antibody engineering and high-throughput screening, stringent regulatory requirements, and increasing cost effectiveness within CDMO services have led most biopharmaceutical manufacturers to outsource their production capabilities. Such factors are expected to drive the market growth over the estimated time period.Furthermore, the demand for monoclonal antibodies (mAbs) and biologics is emerging due to the rising prevalence of chronic and autoimmune diseases, such as cancer, rheumatoid arthritis, and multiple sclerosis, which has led to an increased need for targeted therapies. Besides, the monoclonal antibodies offer high specificity, efficacy, and fewer side effects than traditional drugs, making them preferred treatment options. In addition, advances in biotechnology and antibody engineering have expanded the therapeutic applications of antibodies. Also, growing patient awareness, favorable reimbursement policies, and developing healthcare infrastructure in emerging markets further fuel the adoption and demand for mAbs and biologics across the globe.
Furthermore, the rapid expansion of oncology and autoimmune disease treatment pipelines is driving a growing need for antibody CDMOs services in the market. Besides, rising R&D focused on targeted therapies and biologics requires specialized manufacturing capabilities that support the market. As more drugs advance through clinical and commercial phases, CDMOs are essential in delivering efficient, scalable, and compliant antibody production to meet the market requirements.
Moreover, growing stringent regulatory requirements have further led regulatory agencies, such as the FDA and EMA, to enforce strict guidelines for developing, manufacturing, and quality control of biologics, including monoclonal antibodies. Besides, compliance with Good Manufacturing Practices (GMP), validation protocols, and safety standards is essential to ensure product efficacy and patient safety. These standards drive biopharmaceutical companies to collaborate with experienced antibody CDMOs with the expertise and infrastructure to meet regulatory expectations efficiently, reducing the risk of delays or non-compliance during drug development and commercialization. Such factors are expected to drive market growth.
Opportunity Analysis
The global antibody CDMO market is fueled by increasing demand for monoclonal antibodies, antibody-drug conjugates, and biologics across therapeutic fields such as oncology, autoimmune diseases, and infectious diseases. Pharmaceutical and biotech companies are increasingly outsourcing to minimize costs, speed up development timelines, and access specialized expertise, which creates opportunities for CDMOs that provide comprehensive services such as cell line development, process optimization, analytical testing, and commercial manufacturing. In addition, technological advancements, including single-use bioreactors, continuous processing, and innovative methods enhance operational efficiency & scalability. In addition, CDMOs are expanding into emerging markets and forming strategic collaborations with regional partners to tap into new client bases and navigate different regulatory environments. Moreover, growing emphasis on personalized medicine and complex biologics drives the demand for adaptable, high-quality manufacturing solutions. CDMOs can distinguish themselves and secure a larger portion of the evolving antibody therapeutics market by focusing on regulatory compliance, sustainability efforts, and optimizing capacity. Thus, technological innovation, geographic expansion, and service diversification position antibody CDMOs for sustained growth and strategic importance in the global biopharmaceutical landscape.
Impact of U.S. Tariffs on the Global Antibody Contract Development & Manufacturing Organization Market
The U.S. tariffs on pharmaceuticals, especially the 100% levy on imported branded drugs, have affected the global antibody CDMO market. These tariffs disrupt supply chains, increasing production costs and creating uncertainty for CDMOs and their clients. Most companies that depend on imported components face rising expenses, which could result in delays in drug development and manufacturing. While some CDMOs with significant operations in the U.S. may find ways to adapt, smaller companies and those without domestic facilities are at a disadvantage. In addition, tariffs also push for reshoring initiatives, encouraging CDMOs to invest in U.S.-based manufacturing to alleviate the impacts of the tariffs. However, such a transition requires considerable capital investment and time, potentially straining resources and affecting the timeliness of service delivery. Ultimately, the tariffs introduce complexities that CDMOs need to navigate to remain competitive and ensure the prompt delivery of antibody-based therapies.
Technological Advancements
The antibody CDMO industry is witnessing rapid technological advancements that enhance efficiency, quality, and scalability. Single-use bioreactors are increasingly adopted, reducing contamination risk, enabling flexible scale-up, streamlining cleaning and validation, and supporting clinical and commercial antibody production. Besides, continuous manufacturing integrates upstream and downstream processes, improving yield consistency, cost-effectiveness, and responsiveness to market demand. In addition, advanced cell line engineering, such as CRISPR and synthetic biology, optimizes antibody yield, stability, and post-translational modifications, accelerating development timelines and meeting complex therapeutic needs.

High-throughput screening and automation allow rapid evaluation of cell lines, process conditions, and product quality, minimizing errors, enhancing reproducibility, and managing multiple projects simultaneously. Additionally, advanced analytical and process characterization tools such as mass spectrometry, HPLC, and real-time monitoring provide critical insight into antibody structure, purity, and stability, ensuring regulatory compliance and product consistency. Thus, these technological trends enable CDMOs to deliver high-quality, scalable antibody therapeutics, reduce development timelines, and maintain competitiveness in an increasingly demanding global biopharmaceutical landscape, further fueling the industry's innovation and operational excellence.
Pricing Model Analysis
Antibody CDMOs utilize pricing models to align services with client requirements and project goals. Milestone-based pricing is mainly linked to achieving specific project milestones, such as cell line development or regulatory approval, promoting timely delivery and reducing risk. Besides, the fixed-fee model charges a predetermined amount for defined services, offering clients budget predictability while CDMOs ensure operational efficiency. In addition, value-based pricing sets fees according to the value or impact of the antibody project, reflecting complexity, innovation, and market potential, fostering strategic collaboration.
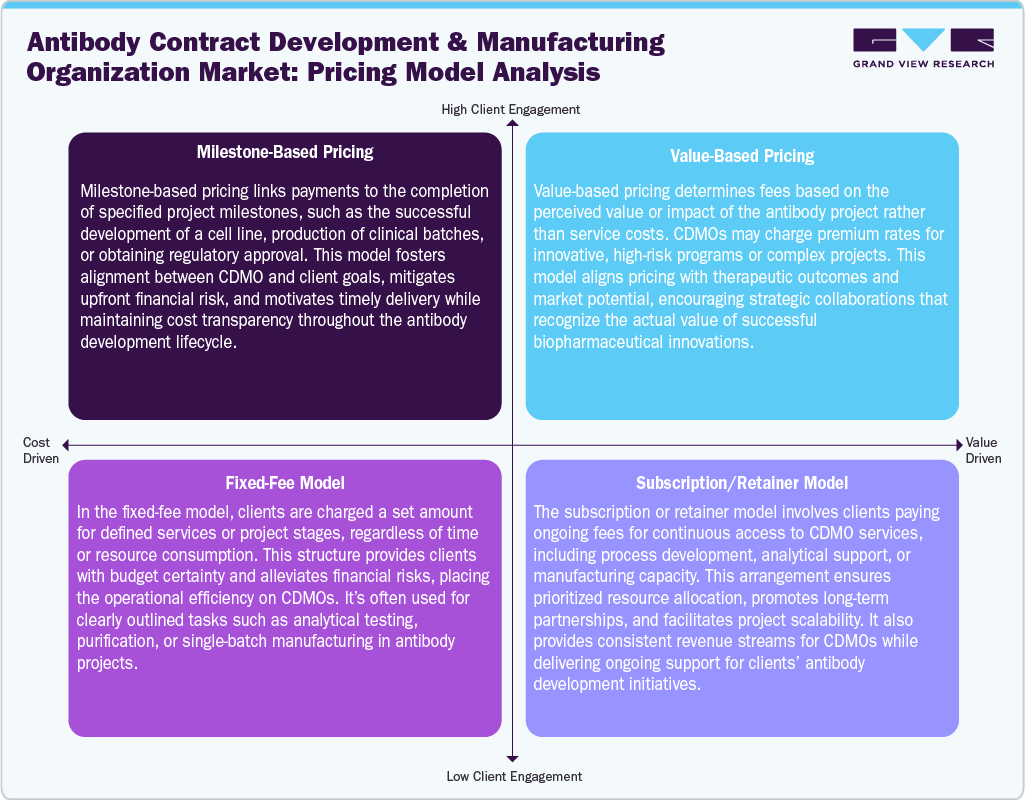
Moreover, the subscription model supports clients to pay recurring fees for ongoing access to CDMO capabilities, including process development, analytical support, or manufacturing capacity. This ensures priority resource allocation, long-term collaboration, and flexibility to scale projects. Thus, by adopting these pricing approaches, antibody CDMOs can provide transparent, client-focused solutions, balance financial risk, and support end-to-end antibody development and manufacturing, ultimately enhancing strategic partnerships and operational efficiency in a competitive biopharmaceutical market.
Market Concentration & Characteristics
The growth stage of the antibody contract development & manufacturing organization industry is medium, and growth is accelerating. The market is characterized by the degree of innovation, level of M&A activities, regulatory impact, service expansion, and regional expansion.
The market increasingly focuses on technologies such as bispecific antibodies, antibody-drug conjugates, and advanced cell line engineering. In addition, R&D and process optimization enable the development of next-generation therapeutics, improving production efficiency and enhancing product stability to meet evolving clinical demands.

M&A is accelerating as companies seek to expand capabilities, increase manufacturing capacity, and access advanced technologies. In addition, strategic activities enhance offerings, strengthen the global presence, integrate solutions for clients, and drive competitive advantage and market growth in antibody development and manufacturing.
Stringent regulatory frameworks influence CDMO operations. Compliance with cGMP standards, FDA, EMA, and local regulatory requirements ensures safety, efficacy, and quality. In addition, CDMOs must maintain robust documentation, validation, and quality control systems, affecting timelines, costs, and service offerings while safeguarding patient safety.
Antibody CDMOs are broadening their offerings beyond basic production to include full-service solutions such as cell line development, process optimization, analytical testing, formulation, fill-finish, and regulatory support. This expansion allows clients to outsource end-to-end development, reducing operational burdens, accelerating timelines, and accessing specialized expertise for clinical and commercial antibody programs.
Antibody CDMOs are expanding operations across North America, Europe, and the Asia Pacific to meet growing global demand. Regional expansion enhances market reach, ensures proximity to clients, provides flexible manufacturing capacity, and facilitates regulatory approvals in local markets. Strategic geographic growth strengthens partnerships and improves accessibility for global pharmaceutical and biotech companies.
Product Insights
On the basis of product, the monoclonal antibodies (mAbs) segment dominated the market with the largest revenue share in 2024. The segment is driven by rising demand for therapies and treatments for chronic and rare diseases. The CDMO services offer end-to-end support for developing and manufacturing therapeutic monoclonal antibodies (mAbs). The services include cell line development, upstream and downstream process optimization, purification, formulation, analytical characterization, and regulatory support. Besides leveraging scalable manufacturing platforms, CDMOs support biopharma companies to accelerate timelines, ensure consistent product quality, and meet cGMP standards. Moreover, they cater to preclinical, clinical, and commercial production needs, enabling efficient, cost-effective delivery of high-quality mAbs while reducing operational burdens and capital investment for their clients. Such factors are expected to drive segment growth over the estimated time period.
The antibody-drug conjugate (ADC) segment is expected to grow significantly during the forecast period. The growth is fueled by the rising need for specialized support for developing and manufacturing ADC therapeutics, combining monoclonal antibodies with potent cytotoxic drugs. Besides, growing advances in antibody production, linker chemistry, payload conjugation, purification, analytical characterization, formulation, and regulatory compliance further make CDMOs essential production partners. Moreover, CDMOs ensure scalable, high-quality manufacturing for preclinical, clinical, and commercial supply, while maintaining the safety and stability of these complex molecules, contributing to segment growth. Thus, such factors are expected to drive the segment growth over the estimated period.
Service Insights
In terms of service, the contract development segment accounted for the largest share in 2024, largely attributed to the increasing complexity of drug development, the rising need for specialized expertise, stringent regulatory requirements, and increased focus on early-stage development of antibodies. The contract development offers a range of services such as lead optimization, cell line development, process development, and analytical characterization, which is expected to drive the market growth over the estimated time period. Besides, the services support companies to accelerate timelines by designing scalable, robust manufacturing processes while ensuring regulatory compliance, which is expected to drive the market growth over the estimated time period.
The contract manufacturing segment is expected to grow significantly during the forecast period. Contract manufacturing plays a crucial role in supporting antibody CDMO services by enabling scalable, high-quality production of antibodies without requiring pharma or biotech companies to invest in their own manufacturing infrastructure. In addition, ensures compliance with cGMP standards, streamlines the transition from development to clinical or commercial supply, reduces costs and operational risks, and provides flexible capacity to meet varying project demands, making CDMOs full-service partners in antibody development and commercialization, which is anticipated to drive the segment. Such factors are expected to drive the market over the estimated time period.
Source Insights
Based on source, the microbial segment dominated the market with the largest revenue share in 2024. The segment is driven by the growing importance of microbes in drug development, as this source uses microorganisms to produce enzymes, vaccines, antibiotics, and therapeutic proteins. Besides, CDMOs specializing in developing and manufacturing antibodies using microbial expression systems like E. coli or yeast are expected to support the segment growth. Moreover, biopharmaceutical CDMOs increasingly opt for microbial sources for strain engineering, fermentation, purification, and quality control. Furthermore, these CDMOs cater to pharma and biotech companies, enabling cost-effective, scalable production of monoclonal antibodies and recombinant proteins for research, preclinical, and commercial applications, which are expected to drive the segment growth over the estimated period.
On the other hand, the mammalian segment is projected to grow at a notable CAGR duringthe forecast period. Mammalian cells are used in the biopharmaceutical market to produce complex biologics, including recombinant proteins, monoclonal antibodies, and vaccines. This source develops and produces antibodies using mammalian cell systems, such as CHO or HEK cells, to ensure proper folding and post-translational modifications. Besides, the cells support cell line development, upstream and downstream processing, quality control, and regulatory support, enabling biopharma companies to manufacture high-quality monoclonal antibodies for preclinical and commercial use efficiently.
Workflow Insights
On the basis of workflow, the commercial segment accounted for the largest share in 2024. The segment growth is driven by the shifting trends towards outsourcing companies to streamline operations and accelerate product launches. Some other factors contributing to segment growth are rising global trends and an expanding product pipeline. Besides, most CDMOs provide end-to-end services for the large-scale production of therapeutic antibodies, including cell line development, upstream and downstream manufacturing, analytical testing, and regulatory support. Thus, by offering specialized expertise, advanced technologies, and flexible manufacturing capacities, commercial CDMOs accelerate time-to-market, support global supply, and help companies meet growing demand for monoclonal antibodies and other antibody-based therapeutics.
The clinical segment is expected to grow significantly during the forecast period. The growth is attributed to increasing focus of CDMOs on supporting early- to mid-stage antibody development, providing services such as cell line development, small- to mid-scale GMP manufacturing, analytical testing, and regulatory guidance for clinical trials. Besides, CDMOs enable pharmaceutical and biotech companies to efficiently produce high-quality antibodies for Phase I–III studies without heavy infrastructure investment. In addition, the growing complexity of personalized medicines and antibodies has increased the requirement for specialized expertise and adaptable manufacturing solutions. Moreover, by partnering with CDMOs, biopharma companies can accelerate development timelines, reduce operational risks, and help companies advance novel therapeutic antibodies from preclinical research to clinical evaluation. Thus, the expanding clinical trial activities globally are expected to drive the market.
Therapeutic Area Insights
On the basis of therapeutic area, the oncology segment accounted for the largest share in 2024. The segment growth is driven by the rising prevalence of cancer, advancing cancer therapeutics by providing specialized services for monoclonal antibodies, bispecific antibodies, and antibody-drug conjugates (ADCs). For instance, the Cancer Atlas mentioned that the number of cancer cases is expected to reach 29 million globally by 2040. This is expected to drive the need for new oncology drugs across the globe, further contributing to market growth. Besides, CDMOs offer end-to-end support, including cell line development, process optimization, GMP manufacturing, analytical testing, and regulatory guidance tailored to oncology programs, further supporting market growth. In addition, for oncology drug development, offer end-to-end support, including cell line development, process optimization, GMP manufacturing, analytical testing, and regulatory guidance tailored to oncology programs, further supporting the market growth. Moreover, by partnering with CDMOs, pharmaceutical and biotech companies can accelerate the development of targeted cancer therapies, ensure consistent quality and scalability, and reduce production costs, which is expected to drive the segment growth over the estimated time period.
The autoimmune diseases segment is expected to grow at the second-fastest CAGR during the forecast period. The growth outlook is strengthened by rising autoimmune diseases, which have led pharmaceutical and biotech companies to shift towards CDMOs for their expertise in developing therapeutic antibodies targeting conditions such as rheumatoid arthritis, lupus, and multiple sclerosis. Moreover, most CDMO companies offer a range of advanced capabilities such as antibody discovery support, cell line development, upstream and downstream manufacturing, analytical characterization, and regulatory compliance. In addition, these companies support accelerating the production of high-quality, scalable antibodies while minimizing infrastructure investment and operational risks. Thus, such factors are expected to drive the segment growth over the estimated period.
End-use Insights
On the basis of end use, the large pharma & biotech companies segment accounted for the largest share in 2024. Large pharma and biotech companies are increasingly partnering with antibody CDMOs to accelerate biologics production while reducing capital investment and operational complexity. In addition, outsourcing provides end-to-end services, including cell line development, upstream and downstream manufacturing, analytical testing, and regulatory support. Besides, the services enable companies to focus on expanding monoclonal antibody and other therapeutic antibody production efficiently, meet global demand, and focus on core R&D activities. Moreover, strategic collaborations with CDMOs support faster time-to-market, risk mitigation, and access to specialized technologies and expertise. Thus, such factors are expected to drive the segment growth over the estimated period.
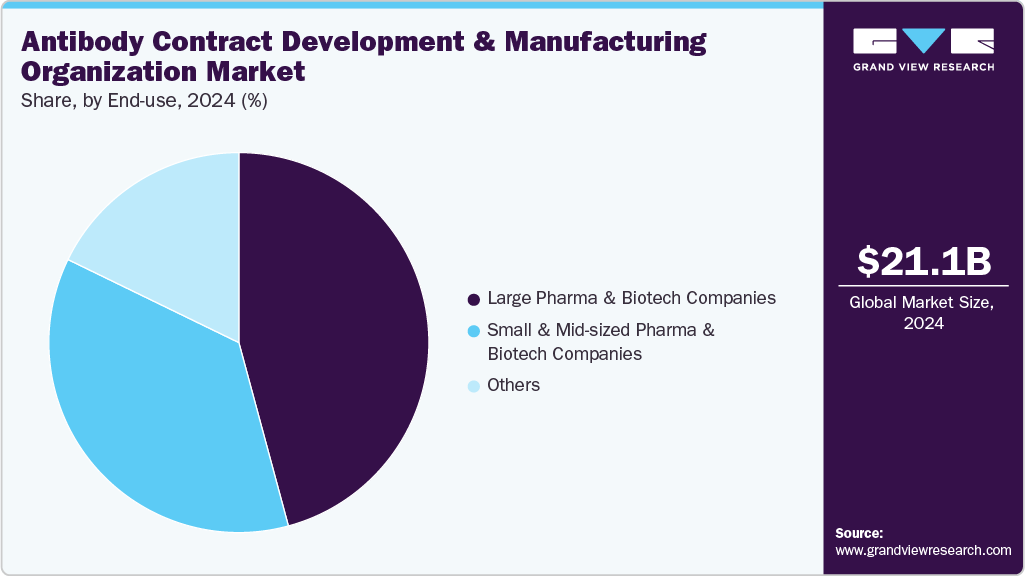
The small & mid-sized pharma & biotech companies’ segment is expected to grow at the second-fastest CAGR during the forecast period. Small and mid-sized pharma and biotech companies increasingly depend on CDMOs as these service providers offer comprehensive services, including cell line development, process optimization, analytical testing, and regulatory support, enabling smaller companies to efficiently develop and scale monoclonal antibodies and other therapeutic antibodies. Besides outsourcing to CDMOs, these companies can focus on research and innovation, accelerate development timelines, and reduce operational risks. Moreover, growing strategic partnerships are expected to boost the specialized expertise and state-of-the-art technologies, enabling companies to focus on core activities. Such factors are expected to drive segment growth.
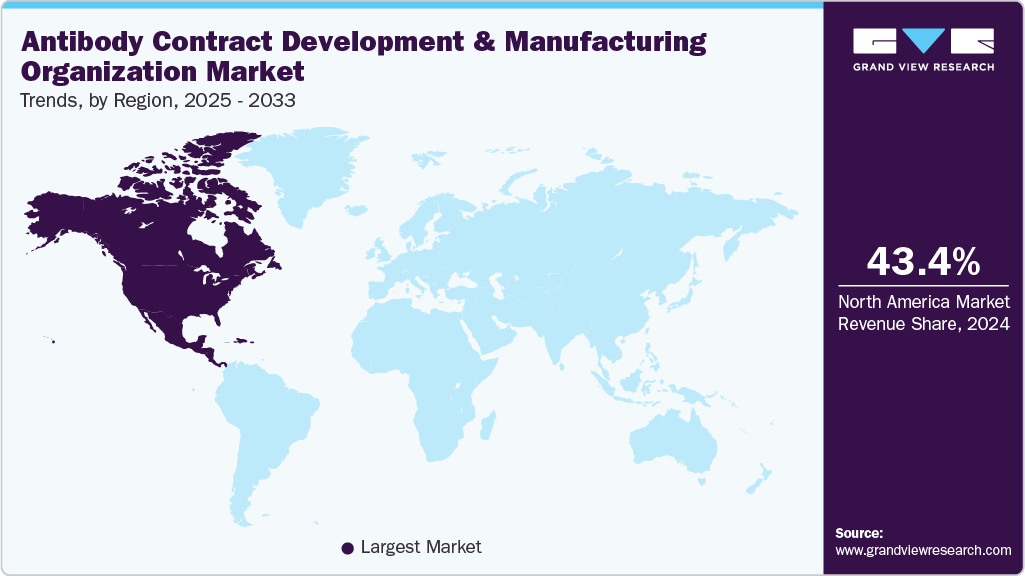
Regional Insights
North America antibody contract development & manufacturing organization market dominatedthe market in 2024, holding a revenue share of 43.40%.The market growth is attributed to strong biopharmaceutical innovation, advanced manufacturing infrastructure, and significant R&D investments. Besides, the region benefits from the presence of leading CDMOs and biopharma companies, coupled with robust regulatory frameworks that enhance product quality and market reliability. In addition, increasing demand for monoclonal antibodies, biosimilars, and novel biologics across the U.S. and Canada has created a rising need for CDMO expertise. Moreover, strong manufacturing capabilities and technological advancements further strengthen regional capabilities. Moreover, strategic collaborations, government support for biologics manufacturing, and expanding pipelines of antibody therapeutics are expected to drive the market growth.
U.S. Antibody Contract Development & Manufacturing Organization Market Trends
The antibody contract development & manufacturing organization industry in the U.S.accounted for the largest market share in North America, owing to increasing outsourcing practices by biopharmaceutical companies, rising demand for mAbs and ADCs, and rising investment in analytical and cell-line development, which drives the market growth. In addition, increased biosimilar production, regulatory emphasis on quality-by-design, and strategic partnerships between biotech and CDMOs have led to a rise in antibody innovation, further supporting the market growth.
The Canada antibody contract development & manufacturing organization industryis expected to grow significantly during the forecast period. The market is driven by rising investments in biologics and biosimilar production, supportive government funding for biomanufacturing, and the expansion of advanced R&D infrastructure. Besides, increasing collaborations along with a growing pipeline of therapeutic antibodies are expected to drive the segment growth. In addition, the rising advancements in process optimization and single-use technologies support the market. Moreover, growing focus on antibody innovation, cost-efficiency, and high-quality production is expected to drive the market over the estimated period.
Europe Antibody Contract Development & Manufacturing Organization Market Trends
The Europe antibody contract development & manufacturing organization industry is witnessing strong growth, driven by increasing demand for monoclonal antibodies, biosimilars, and antibody-drug conjugates. In addition, strong emphasis on advanced bioprocessing capabilities and stringent regulatory frameworks ensuring high-quality production across Germany, the UK, and France is expected to drive the market. Moreover, growing investments in R&D, expansion of single-use and continuous bioprocessing technologies, and rising outsourcing by small and mid-sized biotech companies are expected to drive the market over the estimated period.
The antibody contract development & manufacturing organization industry in Germany held the largest share in 2024. This growth can be attributed to strong biopharmaceutical infrastructure, advanced manufacturing technologies, and high R&D spending. Besides, the growing adoption of technology, strong focus on biologics production, presence of major CDMOs, and expertise in cell-line development and process optimization are expected to drive the market. Moreover, government support for biotech innovation and the expansion of antibody-based therapeutic pipelines are expected to support the industry over the estimated period.
The UK antibody contract development & manufacturing organization industryis expected to grow significantly over the forecast period. The country's growth is fueled by the presence of CDMOs in the country, strong biopharma innovation, and expanding antibody and biosimilar pipelines. Besides, increasing adoption of single-use technologies, strategic partnerships, and investment in automation are expected to support the market.
Asia Pacific Antibody Contract Development & Manufacturing Organization Market Trends
Asia Pacific antibody contract development & manufacturing organization industry is expected to grow at a significant CAGR over the forecast period. The market is driven by emerging opportunities for rising biologics demand, increasing biopharmaceutical R&D investments, and expansion of manufacturing infrastructure. Besides, several biopharma companies focus on advanced single-use technologies, process optimization, and capacity expansion to cater to domestic and global needs. Moreover, countries such as China, Japan, and India are strengthening their capabilities with growing outsourcing by small and mid-sized biotech companies, and the development of novel antibody therapeutics is accelerating market growth across the region.
The antibody contract development & manufacturing organization industry in Chinaiswitnessing new growth opportunities due to new investments in state-of-the-art manufacturing facilities, increased adoption of single-use and continuous bioprocessing technologies, and strategic collaborations with global biopharma companies.
The Japan antibody contract development & manufacturing organization industryis driven bystrong government support for biopharmaceutical innovation, advanced manufacturing infrastructure, and high R&D investments. Moreover, growing demand for monoclonal antibodies, biosimilars, and antibody-drug conjugates, coupled with strategic collaborations between domestic and global CDMOs, contributes to market growth.
The antibody contract development & manufacturing organization industry in Indiais experiencingrapid expansion, attributed to increasing investments in biologics R&D, expanding manufacturing infrastructure, and rising demand for monoclonal antibodies and biosimilars. Moreover, favorable government initiatives, cost-effective production capabilities, and strategic partnerships by global biopharmaceutical companies are expected to fuel market growth.
Latin America Antibody Contract Development & Manufacturing Organization Market Trends
The antibody contract development & manufacturing organization industry in Latin Americais expected to grow significantly over the estimated period. The market is driven by rising demand for a range of therapies, increasing investments in biopharmaceutical R&D, and rising demand for monoclonal antibodies and biosimilars. Besides, countries like Brazil and Argentina emerge as regional hubs offering regional expansion, expanding manufacturing infrastructure, and favorable regulatory reforms.
Brazil antibody contract development & manufacturing organization industryis driven by supportive government policies, increasing investments in biopharmaceutical R&D, and expanding manufacturing infrastructure. The adoption of advanced bioprocessing technologies and process optimization enhances the country's competitiveness in antibody development and manufacturing. Such factors are expected to fuel the market over the forecast period.
Middle East & Africa Antibody Contract Development & Manufacturing Organization Market Trends
The antibody contract development & manufacturing organization industry in the Middle East & Africa is expected to experience steady growth over the forecast period. The market is driven by growing healthcare investments, rising demand for monoclonal antibodies and biosimilars, and expansion of manufacturing infrastructure. Besides, strategic partnerships with global CDMOs, government support for biotechnology initiatives, and adoption of advanced single-use and continuous bioprocessing technologies contribute to market growth.
The UAE antibody contract development & manufacturing organization industry is witnessing growth, underpinned by growing demand for biologics, government initiatives supporting biotechnology and biopharmaceutical innovation, and the expansion of advanced manufacturing infrastructure. In addition, increasing investments in R&D are expected to drive the market over the estimated period.
Key Antibody Contract Development & Manufacturing Organization Company Insights
The key players operating across the market are adopting strategic initiatives such as service launches, mergers & acquisitions, partnerships & agreements, and expansions to gain a competitive edge in the market. For instance, in October 2025, Takara Bio Inc. announced the launch of its CDMO services for antibody drug development and production. The initiative will leverage one of Japan’s largest single-use bioreactor systems, with capacities from several dozen liters up to 3,000 liters, and will also encompass CDMO services for regenerative medicine, cell therapy, and gene therapy products.
Key Antibody Contract Development & Manufacturing Organization Companies:
The following are the leading companies in the antibody CDMO market. These companies collectively hold the largest market share and dictate industry trends.
- Lonza Group
- Catalent, Inc
- Samsung Biologics
- WuXi Biologics
- AGC Biologics
- AbbVie Inc.
- Boehringer Ingelheim International GmbH
- Charles River Laboratories
- FUJIFILM Holdings Corporation
- mAbxience
Recent Developments
-
In September 2025, KBI Biopharma, Inc. (KBI), announced collaboration with Infinimmune, Inc., a biotechnology firm specializing in human-first antibody discovery and design. The partnership aims to support the manufacturing and development of Infinimmune’s lead human monoclonal antibody candidate, IFX-101.
-
In June 2025, LOTTE BIOLOGICS mentioned contract manufacturing partnership with Ottimo Pharma, a biopharmaceutical firm focused on developing novel PD1/VEGFR2 dual-pathway antibody therapies aimed at improving outcomes for cancer patients. Under the Under the agreement, the company will manufacture the antibody drug substance for Ottimo Pharma’s Jankistomig at its Syracuse Bio Campus facility located in New York.
Antibody Contract Development & Manufacturing Organization Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 22.3 billion
Revenue forecast in 2033
USD 45.8 billion
Growth rate
CAGR of 9.38% from 2025 to 2033
Actual Data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, service, source, workflow, therapeutic area, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Thailand; South Korea; Australia; Brazil; Argentina; South Africa; UAE; Saudi Arabia; Kuwait; Qatar; Oman
Key companies profiled
Lonza Group; Catalent, Inc; Samsung Biologics; WuXi Biologics; AGC Biologics; AbbVie Inc.; Boehringer Ingelheim International GmbH; Charles River Laboratories; FUJIFILM Holdings Corporation; mAbxience
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Antibody Contract Development & Manufacturing Organization Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global antibody contract development & manufacturing organization market report based on product, service, source, workflow, therapeutic area, end-use and region.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Monoclonal Antibodies (mAbs)
-
Antibody-Drug Conjugates (ADCs)
-
Bispecific/ Multispecific Antibodies
-
Polyclonal Antibodies
-
Others
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Contract Development
-
Cell Line Development
-
Process Development
-
Upstream
-
Downstream
-
-
Analytical Testing & Method Validation
-
Scale-Up & Tech Transfer
-
-
Contract Manufacturing
-
API Manufacturing
-
Finished Drug Products Manufacturing
-
-
Packaging and Labelling
-
Regulatory Affairs
-
Logistics & Storage
-
Others
-
-
Source Outlook (Revenue, USD Million, 2021 - 2033)
-
Mammalian
-
Microbial
-
-
Workflow Outlook (Revenue, USD Million, 2021 - 2033)
-
Clinical
-
Commercial
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
Infectious Diseases
-
Neurological Disorders
-
Cardiovascular Disease
-
Metabolic Disorders
-
Autoimmune Diseases
-
Respiratory Diseases
-
Ophthalmology
-
Gastrointestinal Disorders
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Large Pharma & Biotech Companies
-
Small & Mid-sized Pharma & Biotech Companies
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
UAE
-
Saudi Arabia
-
Kuwait
-
Qatar
-
Oman
-
-
Frequently Asked Questions About This Report
b. The global antibody contract development & manufacturing organization market size was estimated at USD 21.1 billion in 2024 and is expected to reach USD 22.3 billion in 2025.
b. The global antibody contract development & manufacturing organization market is expected to grow at a compound annual growth rate of 9.38% from 2025 to 2033 to reach USD 45.8 billion by 2033.
b. The oncology segment held a market share of 24.46% by therapeutic area in 2024. The growing investment in R&D activities for cancer-based antibody therapeutics is one of the major factors supporting the segment's growth.
b. Some key players operating in the market include Lonza, Catalent, Inc, Samsung Biologics, WuXi Biologics, AGC Biologics, AbbVie Inc., Boehringer Ingelheim International GmbH, Charles River Laboratories, FUJIFILM Holdings Corporation, and mAbxience, among others. others
b. The market is driven by increasing global demand for monoclonal antibodies and biologics. In addition, rising demand for outsourcing services, rapid advancements in antibody engineering & high-throughput screening, stringent regulatory requirements, and increasing cost effectiveness within CDMO services have led most biopharmaceutical manufacturers to outsource their production capabilities. Such factors are expected to drive the market growth over the estimated time period.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










