- Home
- »
- Medical Devices
- »
-
Aorto-iliac Occlusive Disease Treatment Market Report, 2033GVR Report cover
![Aorto-iliac Occlusive Disease Treatment Market Size, Share & Trends Report]()
Aorto-iliac Occlusive Disease Treatment Market (2026 - 2033) Size, Share & Trends Analysis Report, By Device Type (Endovascular Devices, Surgical Devices), By Procedure (Endovascular Procedures, Hybrid Procedures), By End-use (Hospitals), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-602-6
- Number of Report Pages: 140
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Aorto-iliac Occlusive Disease Treatment Market Size & Trends
The global aorto-iliac occlusive disease treatment market size was estimated at USD 2.26 billion in 2025 and is projected to reach USD 3.36 billion by 2033, growing at a CAGR of 5.02% from 2026 to 2033. Aorto-Iliac Occlusive Disease (AIOD) is a condition where the large arteries in the abdomen and pelvis, specifically the aorta and iliac arteries, become narrowed or blocked, reducing blood flow to the legs.
Key Market Trends & Insights
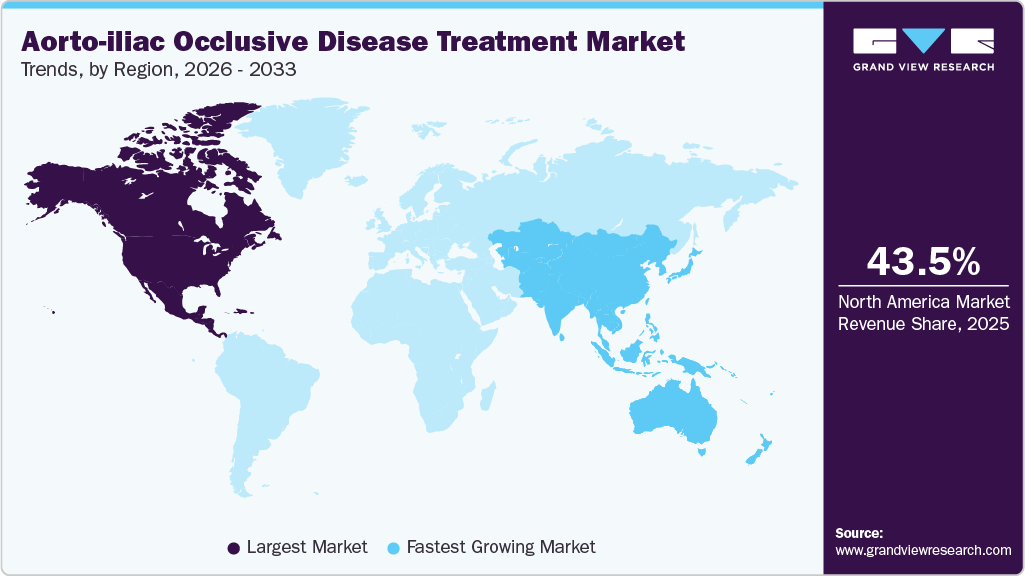
- North America aorto-iliac occlusive disease (iliac disease) treatment market dominated global market in 2025 and accounted for the largest revenue share of 43.49%.
- U.S. aorto-iliac occlusive disease (iliac disease) treatment market is anticipated to register the fastest growth rate during the forecast period.
- In terms of device type, the endovascular devices segment held the largest revenue share in 2025.
- In terms of procedure, the endovascular procedures segment held the largest revenue share in 2025.
- In terms of end use, the hospitals segment held the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 2.26 Billion
- 2033 Projected Market Size: USD 3.36 Billion
- CAGR (2026-2033): 5.02%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
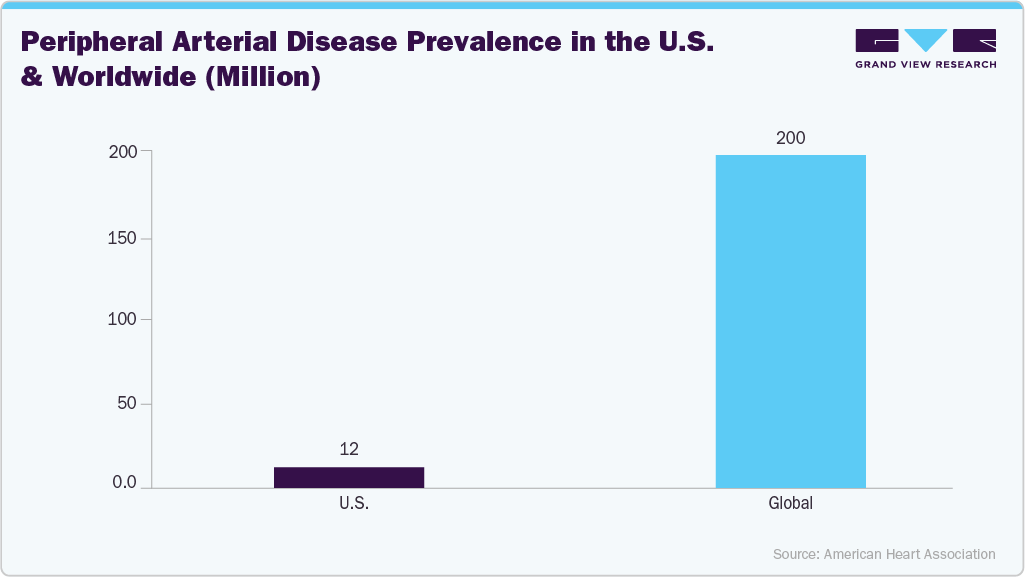
The increasing prevalence of Peripheral Artery Disease (PAD) is one of the key factors driving the market's growth. According to the American Heart Association, Inc. article published in June 2023, Peripheral artery disease (PAD) affects an estimated 200 million people worldwide. It significantly contributes to developing Aorto-iliac Occlusive Disease (AIOD). The growing prevalence of PAD, driven by factors such as aging populations, diabetes, and smoking, has increased the need for effective treatments for AIOD. This rising demand drives the adoption of minimally invasive procedures, including iliac artery stenting and endovascular therapies, which help restore proper blood flow and alleviate symptoms like leg pain and limited mobility.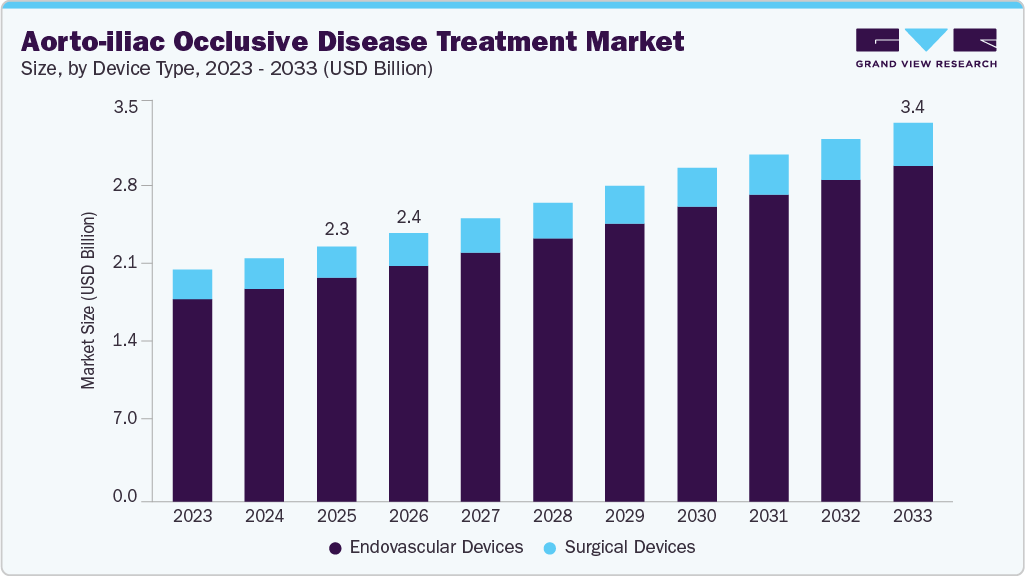
Increasing government initiatives drive the growth of the market. According to the Congress.Gov article published in June 2023, the U.S. government took a significant step toward enhancing early detection and management of Peripheral Artery Disease (PAD), a primary contributor to Aorto-Iliac Occlusive Disease, through the introduction of the Arterial and Venous Comprehensive (ARC) Act. This legislation proposes amendments to the Social Security Act, aiming to ensure that Medicare and Medicaid beneficiaries identified as high-risk can access PAD screening without incurring out-of-pocket costs. Covered diagnostic procedures include ankle-brachial index (ABI) tests and arterial duplex scans, critical for identifying PAD and assessing disease severity in the aorto-iliac region. Authorizing USD 6 million in annual funding from 2024 to 2028, the ARC Act seeks to improve early diagnosis and timely intervention, potentially reducing the burden of advanced occlusive conditions and boosting the demand for targeted treatment options such as iliac stenting and revascularization therapies. This initiative promotes preventive care and supports broader access to essential vascular health services.
Moreover, as per the American College of Cardiology Foundation. article published in February 2025, the Sustainable Cardiopulmonary Rehabilitation Services in the Home Act (H.R. 783), recently reintroduced, proposes to make the Medicare coverage for in-home cardiopulmonary rehabilitation services permanent. This legislative move is designed to enhance access to essential rehabilitation, especially for patients with mobility limitations or those residing in remote areas. Facilitating easier access to rehabilitation care, this initiative supports the effective management of peripheral artery disease (PAD) and related conditions, including Aorto-Iliac Occlusive Disease, ultimately improving patient outcomes and driving demand for advanced treatment options in the market.

Technological advancements in aorto-iliac occlusive disease (iliac disease) treatment drive the market's growth. For instance, in January 2025, Bentley Medical introduced the BeFlow iliac covered stent system at the Leipzig Interventional Course (LINC) 2025, marking a significant advancement in treating aorto-iliac occlusive disease. Designed to address complex iliac lesions, the BeFlow stent offers enhanced long-term patency and durability, aligning with European Society for Vascular Surgery guidelines that recommend covered stents for heavily stenosed or occluded iliac arteries. Available in diameters ranging from 7 to 10 mm and lengths of 37 and 57 mm, the BeFlow stent aims to provide a cost-effective alternative to bare metal stents, improving patient access to advanced vascular interventions.

The regulatory framework for aorto-iliac occlusive disease treatment in North America is shaped by country-specific agencies, with the U.S. Food and Drug Administration (FDA), Health Canada, and Mexico’s COFEPRIS overseeing product approvals, safety monitoring, and clinical evidence requirements. While harmonization is limited, shared reliance on global clinical data and increasing regional collaboration under frameworks like the USMCA influence market entry strategies. Regulatory delays or variations across the region can affect the availability of newer technologies for iliac disease interventions.

Advances in delivery systems have dramatically improved procedural success rates for balloon expandable covered endoprostheses. Low-profile catheters and refined trackability enable interventionalists to access complex lesions more easily and precisely. These innovations not only shorten procedure times but also reduce the incidence of vascular trauma and complications, which is especially crucial in the treatment of advanced aorto-iliac occlusive lesions. In January 2025, W. L. Gore & Associates received regulatory clearance in the EU for a redesigned version of its GORE VIABAHN VBX Balloon Expandable Endoprosthesis, now compatible with smaller 6 Fr introducer sheaths. This advancement stems from updates to the delivery system, not the stent itself, allowing for broader patient applicability and smoother procedures in complex vascular cases.
The market is divided into three anatomical regions: aortoiliac, femoropopliteal, and tibial. This structure mirrors the arterial tree from the pelvis to the foot, where disease can appear alone or in combination. Anatomical site influences symptoms, treatment choices, and procedural outcomes. In the femoropopliteal segment, rising adoption of drug-coated balloons, supported by evidence of improved vessel patency and reduced reintervention needs, reflects how clinical data shapes device uptake. Understanding PAD distribution by anatomical territory remains key to evaluating patient flows, technology positioning, and interventional complexity.
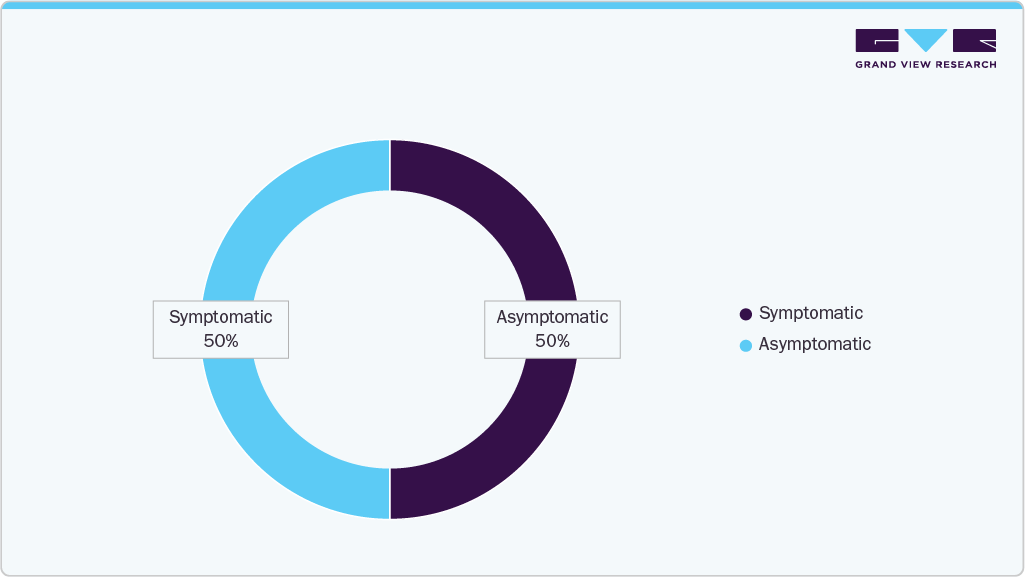
Aortoiliac occlusive disease (AIOD) is a common presentation of peripheral arterial disease (PAD), particularly affecting older adults and often progressing without symptoms. In July 2023, according to content published under Drugs & Diseases > Vascular Surgery by Medscape, PAD was estimated to affect up to 25% of the U.S. population by age 70, though more than 50% of patients may remain asymptomatic. Occlusion in the aortoiliac segment is the second most frequently affected site after the superficial femoral artery.AIOD often presents as intermittent claudication or critical limb ischemia, warranting timely diagnosis and intervention.
In North America, the burden of aortoiliac occlusive disease continues to rise in parallel with aging demographics, diabetes, and smoking prevalence. Clinical data suggest that AIOD accounts for a substantial proportion of lower extremity revascularization procedures, particularly in patients presenting with lifestyle-limiting claudication or critical limb ischemia. Although precise AIOD-specific prevalence estimates are limited, its contribution to symptomatic PAD cases is significant, often necessitating endovascular or surgical intervention in tertiary care settings. Despite increasing use of minimally invasive methods, aortofemoral bypass remains a benchmark for long-term patency in complex cases.
Market Concentration & Characteristics
The aorto-iliac occlusive disease (iliac disease) treatment industry is experiencing notable innovation driven by next-generation endovascular technologies. Advancements include image-guided stent deployment systems, drug-eluting stents tailored for iliac anatomy, and precision-engineered balloon-expandable and self-expanding stents that improve flexibility and radial strength. Hybrid techniques integrating real-time 3D imaging and robotic-assisted navigation improve procedural precision and outcomes in complex cases. In May 2025, Boston Scientific initiated the MAJESTIC clinical trial in New Zealand to evaluate the safety and performance of its Innova Drug-Eluting Stent (DES) System, which performed the first patient implant at Auckland City Hospital.
Key players in the aorto-iliac occlusive disease (AIOD) treatment industry, including Medtronic, Abbott, and Boston Scientific Corporation, are actively pursuing mergers, acquisitions, and strategic collaborations to strengthen their market position. These efforts aim to accelerate the development of advanced endovascular devices, expand global distribution networks, and integrate innovative technologies such as drug-coated balloons and next-generation stent systems. In February 2025, Teleflex Incorporated announced it has entered into a definitive agreement to acquire substantially all of the Vascular Intervention business of BIOTRONIK SE & Co. KG for an estimated cash payment of approximately USD 839.04 million. This acquisition aims to enhance Teleflex's capabilities in the vascular intervention market.
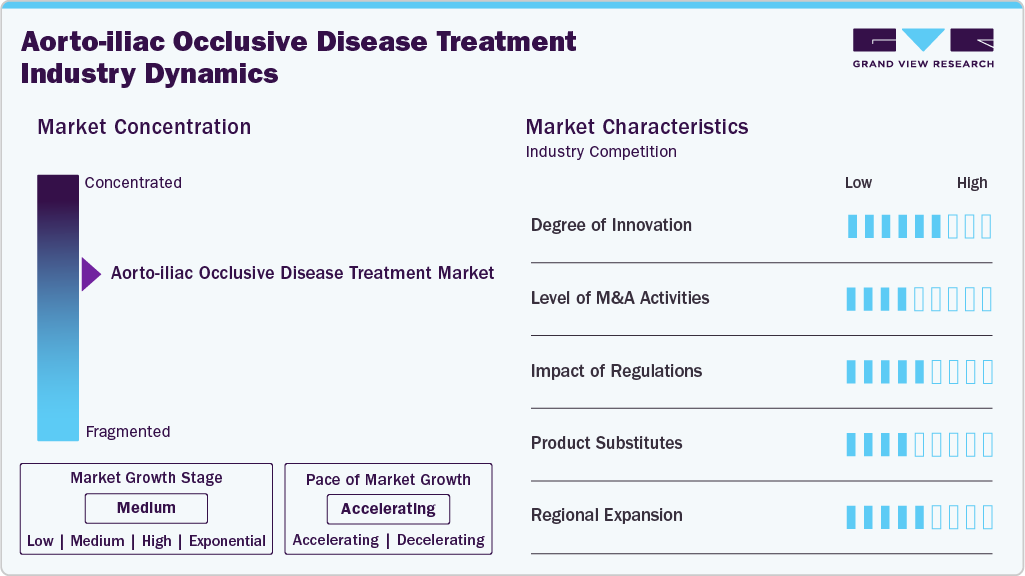
Regulatory bodies like the FDA and EMA play a key role in shaping the aorto-iliac occlusive disease treatment industry by ensuring the safety and effectiveness of medical devices. While approval processes can be lengthy, initiatives such as fast-track approvals and innovation-friendly policies are helping accelerate the entry of advanced technologies such as drug-eluting stents and imaging-guided systems, supporting market growth.
There are no direct substitutes for aorto-iliac occlusive disease treatment procedures, as they are essential for restoring adequate blood flow to the lower limbs. Although lifestyle changes and pharmacological therapies can help manage symptoms, they do not offer the revascularization benefits of endovascular or surgical interventions.
Key players in the aorto-iliac occlusive disease treatment industry are expanding their presence by offering specialized training for clinicians, partnering with regional health institutions, and tailoring intervention strategies to address the unique clinical demands of underserved populations. These efforts aim to improve access to advanced endovascular and surgical solutions, enhance treatment outcomes, and support early diagnosis and intervention in regions with limited vascular care infrastructure.
Device Type Insights
The endovascular devices segment held the largest share of 87.61% in 2025. Endovascular devices are specialized medical tools used to treat vascular diseases by accessing and repairing blood vessels from within, typically through minimally invasive catheter-based techniques. Endovascular devices play a crucial role in the minimally invasive treatment of AIOD, offering alternatives to traditional open surgery. These devices include balloon angioplasty systems, various stent types (such as self-expanding, balloon-expandable, and covered stents), and atherectomy systems, all designed to restore proper blood flow through narrowed or blocked arteries. Technological advancements have significantly improved the safety and efficacy of these devices. For instance, in April 2024, Getinge’s Advanta V12 covered stent system recently received certification under the European Union Medical Device Regulation (EU MDR), confirming its suitability for treating patients with aortoiliac occlusive disease, including complex lesions at the aortic bifurcation.
The balloon angioplasty devices segment is expected to show lucrative growth during the forecast period, owing to the increasing incidence of PAD and the technological advances in product launches, which drive the development of the market. Balloon-expandable stents are specialized vascular implants delivered to the target vessel using a balloon catheter. Once properly positioned, the balloon is inflated to expand the stent, pressing it against the arterial wall to restore blood flow. For instance, in February 2024, W. L. Gore & Associates received FDA approval for its low-profile VBX balloon-expandable stent graft. Designed for treating de novo or restenotic lesions in the iliac artery, this stent accommodates vessel diameters from 5 to 13 mm and lesion lengths up to 110 mm. The device's reduced profile allows compatibility with 6-French sheaths, facilitating procedures in patients with challenging anatomies.
Procedure Insights
The endovascular procedures segment held the largest share of 61.20% in 2025 due to the increasing incidence of PAD diseases, government initiatives, and technological advancements. Artificial Intelligence (AI) is increasingly being integrated into endovascular procedures, aiming to enhance precision, reduce procedural risks, and improve overall outcomes. Recent studies have explored AI-driven systems that assist in the autonomous navigation of catheters and guidewires, particularly utilizing reinforcement learning (RL) and imitation learning techniques. According to the School of Biomedical Engineering & Imaging Sciences article published in May 2024, analyzed 14 studies focusing on AI applications in autonomous endovascular navigation. The review found that 64% of these studies employed reinforcement learning, while 50% utilized learning from expert demonstrations. These AI models were primarily tested on physical phantoms and in-silico models, demonstrating the potential to decrease operation times, enhance decision-making during surgery, and reduce operator radiation exposure.
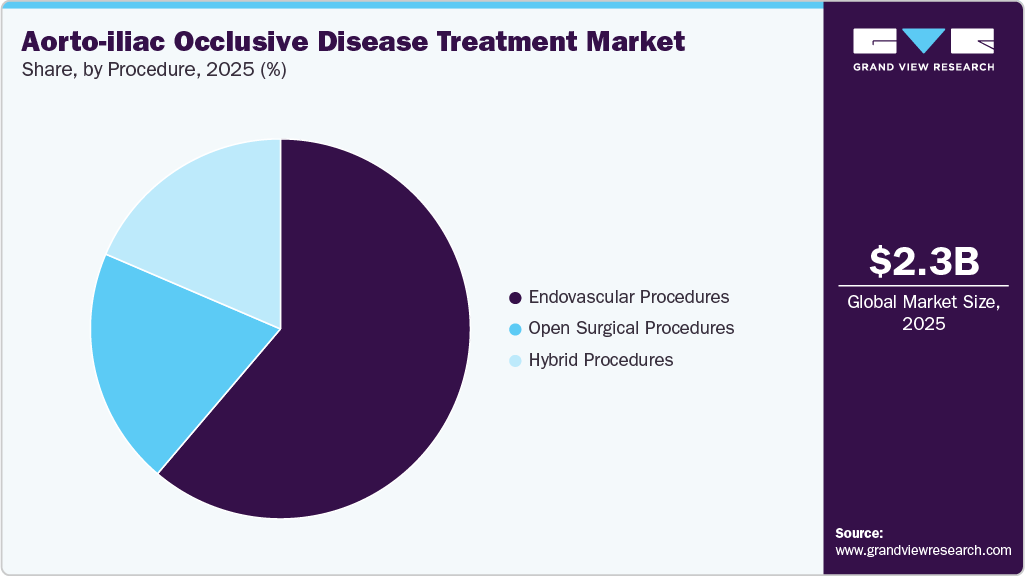
The hybrid procedures segment will show lucrative growth during the forecast period, owing to the increasing prevalence of PAD and technological advancements, which drive the market's growth. Hybrid procedures integrate open femoral endarterectomy with endovascular recanalization and stenting of iliac occlusions to treat complex iliofemoral artery blockages. According to the Elsevier Inc. article published in January 2023, a study involving 147 patients and 163 iliofemoral occlusions, this combined approach showed a high technical success rate of 88.3% and excellent primary patency rates of 98.7% at 30 days, 98.1% at 6 months, 96.6% at 1 year, and 93.7% at 2 years. Minimizing surgical trauma and reducing recovery time compared to traditional aortobifemoral bypass, hybrid procedures are especially beneficial for patients with significant comorbidities and complex lesions. The use of covered stents in nearly 34% of cases and the successful management of long occlusions highlight its versatility.
End Use Insights
The hospitals segment held the largest share of 48.83% in 2025. Hospitals are increasingly expanding their treatment portfolios to include advanced and minimally invasive procedures for managing complex aorto-iliac occlusive disease. For instance, NYU Langone Health has implemented minimally invasive procedures such as atherectomy and balloon angioplasty with stenting. These treatments are performed under local anesthesia, often allowing patients to return home the same day, thereby reducing hospital stays and accelerating recovery times. In January 2025, GE HealthCare signed a USD 249 million AI imaging deal with a 31-hospital system in the U.S. to enhance diagnostic accuracy and clinical workflows, highlighting hospitals’ commitment to innovation in vascular care.
U.S. Hospital Database 2024
Hospital Category
Number of Hospitals
Total Number of All U.S. Hospitals
6,120
Number of U.S. Community Hospitals
5,129
Number of Nongovernment Not-for-Profit Community Hospitals
2,987
Number of Investor-Owned (For-Profit) Community Hospitals
1,219
Number of State and Local Government Community Hospitals
923
Number of Federal Government Hospitals
207
Number of Nonfederal Psychiatric Hospitals
659
Other Hospitals
125
Source: American Hospital Association & GVR
The outpatient facilities segment is expected to show lucrative growth during the forecast period. Outpatient refers to a type of medical care or treatment that does not require an overnight stay in a hospital or medical facility. Outpatients visit a clinic, hospital, or healthcare center for diagnosis, treatment, or minor procedures and return home the same day. Outpatient facilities are also expanding their capabilities to treat AIOD, offering patients convenient access to effective therapies without the need for hospitalization. For instance, in June 2023, a study published in Seminars in Interventional Radiology examined the safety and efficacy of treating peripheral arterial disease (PAD) in non-hospital settings, such as office-based labs (OBLs) and ambulatory surgical centers.
Regional Insights
North America dominated the aorto-iliac occlusive disease (iliac disease) treatment market, with a share of 43.49% in 2025. The rising incidence of PAD, increased government initiatives, and technological advancements drive the market's growth. According to the Science Direct article published in March 2025, it is estimated that around 6.5 million individuals aged 40 years and above are affected, representing approximately 5.8% of this population group. Additionally, the annual incidence rate of PAD is approximately 2.69%, reflecting the steady rise in new cases each year. These trends highlight the increasing need for effective diagnostic and treatment solutions in the U.S. aorto-iliac occlusive disease treatment market, as healthcare systems work to address the complications and risks associated with age-related vascular conditions.
The U.S. accounted for the largest share of North America's aorto-iliac occlusive disease (iliac disease) treatment market in 2025. Increasing PAD incidence and technological advancements drive the growth of the market. For instance, in November 2024, Philips has launched a clinical trial in the U.S. to evaluate a novel device that merges laser atherectomy with intravascular lithotripsy in a single catheter system. The first successful procedure was performed on a 78-year-old male patient at the Cardiovascular Institute of the South in Louisiana, marking a promising advancement in minimally invasive treatment strategies. Integrating plaque modification technologies into one device, the system has the potential to simplify procedures, reduce operating time, and improve outcomes particularly in challenging iliac artery cases.
Europe Aortoiliac Occlusive Disease Treatment Market Trends
Europe's aorto-iliac occlusive disease (iliac disease) treatment market held the second-largest revenue market share in 2025. Increasing incidence of the PAD and government initiatives drives the growth of the market. According to NCBI article published in December 2024, PAD was diagnosed in 941 individuals, the prevalence of PAD was significantly higher among European patients, affecting 8.1% of this group. This elevated PAD burden in Europe directly emphasizes the growing demand for effective aorto-iliac occlusive disease treatments.
Germany's aorto-iliac occlusive disease (iliac disease) treatment market is growing over the forecast period. Increasing incidence of the PAD and technologically advanced product offered by the key players drives the growth of the market. According to a Hogrefe article published in October 2023, Peripheral artery disease (PAD) impacts an estimated 15-20% of the German population, with prevalence rates steadily increasing in recent years. This growing incidence reflects broader trends in cardiovascular health challenges within the country, driving a heightened need for effective management of related conditions such as aorto-iliac occlusive disease.
The UK's aorto-iliac occlusive disease (iliac disease) treatment market held the second-largest market share in 2025. The rising incidence of PAD and technological advancement drives the growth of the market. According to the British Heart Foundation article published in August 2024, in the UK, approximately 20% of individuals aged 60 and above are affected by peripheral artery disease (PAD), highlighting a significant health concern among the aging population. The primary risk factors driving PAD smoking, diabetes, obesity, and hypertension closely align with those linked to cardiovascular diseases such as heart disease and stroke.
The France aorto-iliac occlusive disease (iliac disease) treatment market is anticipated to witness a significant CAGR during the forecast period. The increasing incidence of PAD, and technological advances drive the market's growth. According to the Elsevier B.V. article published in June 2024, Peripheral artery disease (PAD) affects up to one-fifth of the population in France, reflecting a substantial public health challenge. Studies indicate that among individuals with one or more cardiovascular risk factors, approximately 12.2% have asymptomatic PAD, often detected through ankle-brachial index screening.
Asia Pacific Aorto-iliac Occlusive Disease Treatment Market Trends
The Asia Pacific region is expected to grow fastest during the forecast period. Key aspects such as rapidly increasing prevalence of peripheral artery disease (PAD), rising awareness about vascular health, expanding healthcare infrastructure, and improving access to advanced medical technologies are driving this growth. In January 2024, the ZYLOX Peripheral Venous Stent System by Zylox-Tonbridge Medical Technology Co., Ltd. received NMPA approval. This stent system features a tapered gradient and integrated structure to reduce thrombosis risk and align with the natural vessel diameter.
China accounted for the most extensive AIOD treatment market share in the Asia Pacific region in 2025. Rising PAD cases and technological advancements drive the growth of the market. According to the NCBI article published in August 2023, in China, peripheral artery disease (PAD) affects approximately 45.3 million people, underscoring a significant and growing healthcare challenge. For instance, in February 2025, the Grency Iliac Vein Stent System by Hangzhou Weiqiang Medical Technology Co., Ltd., approved by the National Medical Products Administration (NMPA), is a self-expanding nickel-titanium alloy stent designed for treating non-thrombotic iliac vein compression syndrome.
The Japan aorto-iliac occlusive disease (iliac disease) treatment market held the largest market share in the Asia Pacific region. Increasing PAD cases, and strategic initiatives by the key players drive the market's growth. According to the NCBI article published in June 2024, In Japan, a study involving 3,906 individuals with diabetes revealed that peripheral artery disease (PAD) was present in approximately 7.6% of the participants, with the mean age being 60.8 years. In November 2023, Penumbra Inc.’s Indigo system, designed for effective thrombus removal, received regulatory approval under the Japanese Pharmaceutical & Medical Device Act in April 2023.
The Indian aorto-iliac occlusive disease (iliac disease) treatment market is experiencing significant growth driven by the rising number of PAD cases in India and technological advancements. In May 2025, the Sanjay Gandhi Postgraduate Institute of Medical Sciences (SGPGIMS) in Lucknow has recently introduced an AI-powered intravascular optical coherence tomography (OCT) system. This technology provides high-resolution, real-time imaging of arterial plaques, enabling cardiologists to tailor angioplasty procedures more precisely. The incidence is particularly elevated in older populations and individuals with underlying risk factors, such as diabetes and tobacco use conditions that are becoming increasingly common in the country.
Latin America Aortoiliac Occlusive Disease Treatment Market Trends
The Latin America aorto-iliac occlusive disease (iliac disease) treatment market is growing due to several factors. Rising PAD cases and technological advancements drive the growth of the market. According to the Pan American Health Organization article published in October 2024, In Latin America, particularly in countries like Argentina, the rising elderly population is significantly influencing the aorto-iliac occlusive disease (iliac disease) treatment market. In 2024, individuals aged 65 and older accounted for approximately 12.4% of Argentina’s total population, up from 9.6% in 2000 an increase of 2.8 percentage points. This demographic trend is contributing to a growing burden of age-related vascular conditions, including peripheral artery disease (PAD), thereby driving demand for effective treatment solutions and expanding the market for aorto-iliac disease interventions across the region.
Brazil's aorto-iliac occlusive disease (iliac disease) treatment market is witnessing steady growth, fueled by an increasing prevalence of peripheral artery disease (PAD) and a rising demand for minimally invasive treatment options. The country faces a growing burden of vascular diseases, particularly among aging populations and individuals with common risk factors such as diabetes, hypertension, and smoking. According to MDPI article published in January 2024, in 2021, Brazil ranked sixth globally in the number of adults living with diabetes mellitus (DM), with around 16 million individuals affected. More recently, the Vigitel 2023 survey reported that 10.2% of the Brazilian population is currently living with diabetes, emphasizing its growing burden as a primary public health concern with significant economic implications.
MEA Aorto-iliac Occlusive Disease Treatment Market Trends
The MEA aorto-iliac occlusive disease (iliac disease) treatment market is expected to grow lucratively due to the economic development and improved healthcare facilities in the region have led to increased demand for advanced medical treatments. Countries such as Saudi Arabia are witnessing a rise in peripheral artery disease (PAD) prevalence, which is contributing to the expansion of the iliac stents market. According to a BioMed Central Ltd article published in March 2024, a recent study conducted in Saudi Arabia found that peripheral artery disease (PAD) affects approximately 11.7% of patients aged 45 years and older within a cohort of 471 individuals.
Saudi Arabia's aorto-iliac occlusive disease (iliac disease) treatment market is growing over the forecast period. Rising incidence of the PAD and technological advancement drives the growth of the market. According to a BioMed Central Ltd article published in March 2024, a recent study conducted in Saudi Arabia found that peripheral artery disease (PAD) affects approximately 11.7% of patients aged 45 years and older within a cohort of 471 individuals. The relatively high prevalence reported may be influenced by the study’s focused sample size and the older age group, which is generally at greater risk for vascular disorders. This highlights a significant patient population in need of effective aorto-iliac occlusive disease treatment options, underscoring the growing demand for advanced therapeutic interventions in Saudi Arabia.
Key Aorto-iliac Occlusive Disease Treatment Company Insights
Some of the key players operating in the industry include Boston Scientific Corporation, BD, and Abbott. The company’s key strategies include understanding the strengths and weaknesses of major market participants, anticipating future market trends, opportunities, and challenges, and making proactive decisions based on insights into emerging technologies and changing consumer preferences. For instance, Biotronik SE & Co KG and Getinge AB are some of the emerging players in aorto-iliac occlusive disease (iliac disease) treatment.
Key Aorto-iliac Occlusive Disease Treatment Companies:
The following are the leading companies in the aorta-iliac occlusive disease (iliac disease) treatment market. These companies collectively hold the largest market share and dictate industry trends.
- BD.
- Bentley InnoMed
- Scitech Medical
- Abbott
- Boston Scientific Corporation
- W. L. Gore & Associates, Inc.
- Medtronic
- Cook
- Terumo Corporation
- iVascular
- Getinge AB
- Biotronik SE & Co KG
Recent Developments
-
In May 2025, the Saudi Food and Drug Authority (SFDA) initiated digital awareness campaigns to inform the public about the safe and proper use of medical devices, which are crucial for managing conditions like PAD.
-
In March 2025, the Washington Healthcare Authority noted that balloon angioplasty remains a central treatment for lower extremity PAD, especially in femoro-popliteal and infra-popliteal segments. The report emphasized its use for improving blood flow and limb preservation, pointing to growing adoption of drug-coated balloon technologies.
-
In January 2025, Bentley launched its BeFlow iliac-covered stent system, CE-certified and available in Europe, in February 2025. Designed for iliac occlusive and stenotic diseases, the BeFlow offers four diameters (7-10 mm) and two lengths (37 and 57 mm) to provide a cost-effective alternative to bare metal stents. Dr. Koen Deloose performed the first-in-human procedure on a 78-year-old patient with severe claudication.
-
In January 2025, Boston Scientific Corporation has signed a definitive agreement to acquire Bolt Medical, Inc., a company specializing in an advanced laser-based intravascular lithotripsy (IVL) platform designed to treat both coronary and peripheral artery diseases.
-
In December 2024, Biotronik concluded the BIO-OSCAR SOC trial, a prospective, multicenter, observational study evaluating current standard of care practices in treating peripheral artery disease (PAD). The study aimed to assess procedural outcomes and device use in endovascular treatments for atherosclerotic lesions, providing insights into real-world practices for managing PAD.
-
In March 2024, Cook Medical entered into an exclusive distribution agreement with Getinge for the iCast covered stent system in the U.S. This collaboration aims to enhance the availability and distribution of the device, which is indicated for the treatment of iliac arterial occlusive disease.
-
In January 2024, Zylox-Tonbridge Medical Technology Co., Ltd. has received marketing approval from the National Medical Products Administration (NMPA) for its ZYLOX® Peripheral Venous Stent System. This innovative device is designed to address lower limb and pelvic venous return disorders caused by iliac vein compression, such as May-Thurner Syndrome.
Aorto-iliac Occlusive Disease Treatment Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 2.38 billion
Revenue forecast in 2033
USD 3.36 billion
Growth rate
CAGR of 5.02% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Device type, procedure, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Mexico; Canada; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
BD.; Abbott; Boston Scientific Corporation; W. L. Gore & Associates, Inc.; Medtronic; Cook; Terumo Corporation; iVascular; Getinge AB; Biotronik SE & Co KG
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options Global Aorto-iliac Occlusive Disease Treatment Market Report Segmentation
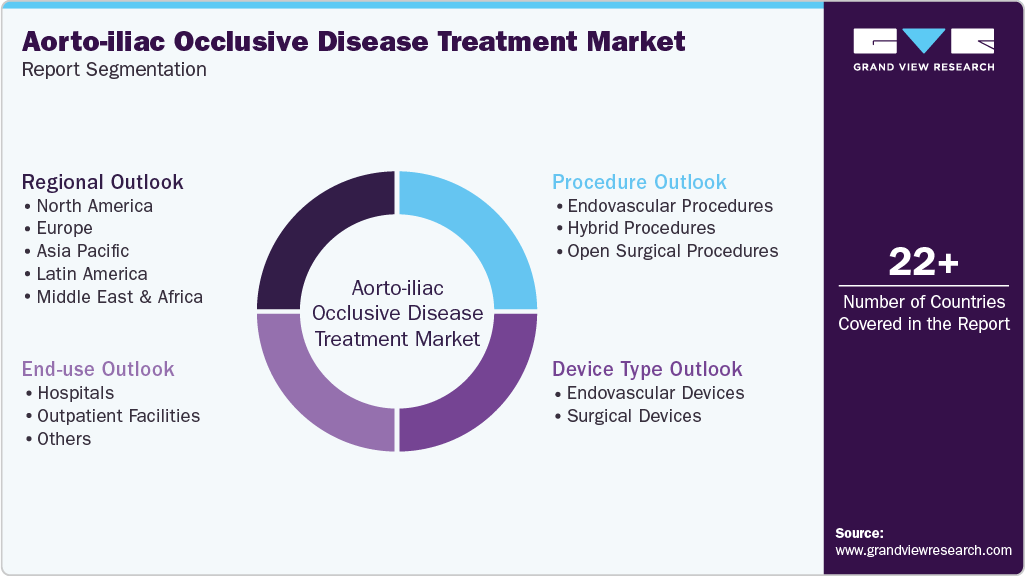
This report forecasts revenue growth at global, regional & country levels and analyzes the industry trends in each sub-segment from 2021 to 2033. for this study, Grand View Research has segmented the global aorto-iliac occlusive disease treatment market report based on device type, procedure, end use, and region:
-
Device Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Endovascular Devices
-
Balloon Angioplasty Devices
-
Atherectomy Systems
-
Stents
-
Self-Expanding Stents
-
Balloon-Expandable Stents
-
Covered Stent Grafts
-
Bifurcated Aortic Stent Grafts
-
-
-
Surgical Devices
-
-
Procedure Outlook (Revenue, USD Million, 2021 - 2033)
-
Endovascular Procedures
-
Balloon Angioplasty
-
Primary Stenting
-
Kissing Stents
-
Covered Stent Grafts
-
Endovascular Aortic Repair (EVAR)
-
-
Hybrid Procedures
-
Open Surgical Procedures
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Outpatient Facilities
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global aorto-iliac occlusive disease treatment market size was estimated at USD 2.26 billion in 2025 and is expected to reach USD 2.38 billion in 2026.
b. The global aorto-iliac occlusive disease (iliac disease) treatment market is expected to grow at a compound annual growth rate of 5.02% from 2026 to 2033 to reach USD 3.36 billion by 2033.
b. North America dominated the aortoiliac occlusive disease (iliac disease) treatment market, with a share of 43.49% in 2025. The rising incidence of PAD, increased government initiatives, and technological advancements drive the market's growth.
b. Some of the major participants in the aorto-iliac occlusive disease (iliac disease) treatment market include BD.; Abbott; Boston Scientific Corporation; W. L. Gore & Associates, Inc.; Medtronic; Cook; Terumo Corporation; iVascular; Getinge AB; Biotronik SE & Co KG
b. The increasing prevalence of Peripheral Artery Disease (PAD) is one of the key factors driving the market's growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










