- Home
- »
- Plastics, Polymers & Resins
- »
-
Bioplastics In Diagnostic Devices Market Size Report, 2033GVR Report cover
![Bioplastics In Diagnostic Devices Market Size, Share & Trends Report]()
Bioplastics In Diagnostic Devices Market (2025 - 2033) Size, Share & Trends Analysis Report By Material (PLA, PHA, PBS/PBAT), By Application (Rapid Test Kits, Instrument Housings, Cartridges & Microfluidic Consumables), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-828-0
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Bulk Chemicals
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Bioplastics In Diagnostic Devices Market Summary
The global bioplastics in diagnostic devices market size was estimated at USD 273.5 million in 2024 and is projected to reach USD 668.5 million by 2033, growing at a CAGR of 10.6% from 2025 to 2033. Product adoption began in lower-risk, high-volume disposable components where regulatory barriers are lower, and cost sensitivity is high, notably lateral-flow assay (LFA) housings, rapid test kits, and non-sterile ancillary components.
Key Market Trends & Insights
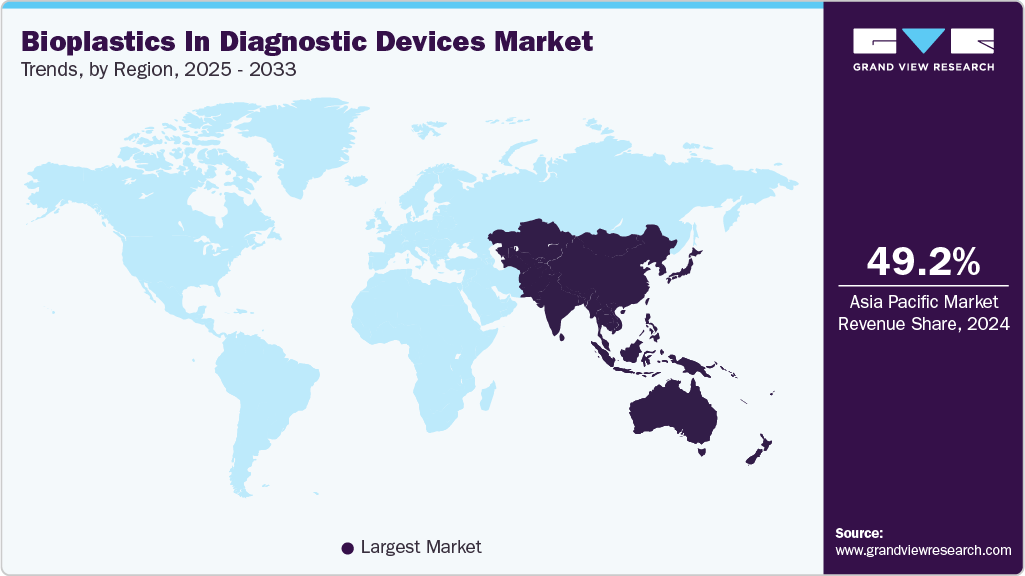
- Asia Pacific dominated the bioplastics in diagnostic devices market with the largest revenue share of 49.23% in 2024.
- The bioplastics in diagnostic devices market in China is becoming a major production hub for bioplastic diagnostic components.
- By material, the PHA segment dominated the market across the material segmentation in terms of revenue, accounting for a market share of 51.24% in 2024.
- By application, the rapid test kits segment dominated the market in terms of revenue, accounting for a market share of 61.89% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 273.5 Million
- 2033 Projected Market Size: USD 668.5 Million
- CAGR (2025-2033): 10.6%
- Asia Pacific: Largest market in 2024
The market is an intersection of two dynamics: rapid growth in point-of-care testing and increasing procurement pressure on healthcare providers to reduce single-use plastic waste. Geographically, the market is strongest in regions with mature IVD industries and sustainability procurement, while emerging markets trail in adoption due to price sensitivity and the availability of medical-grade biopolymers. Market value today is still small relative to the broader IVD consumables market, but growth rates are materially higher, driven by OEM sustainability commitments, corporate ESG targets, and increasing availability of medical-grade bioplastic resins and qualified converters.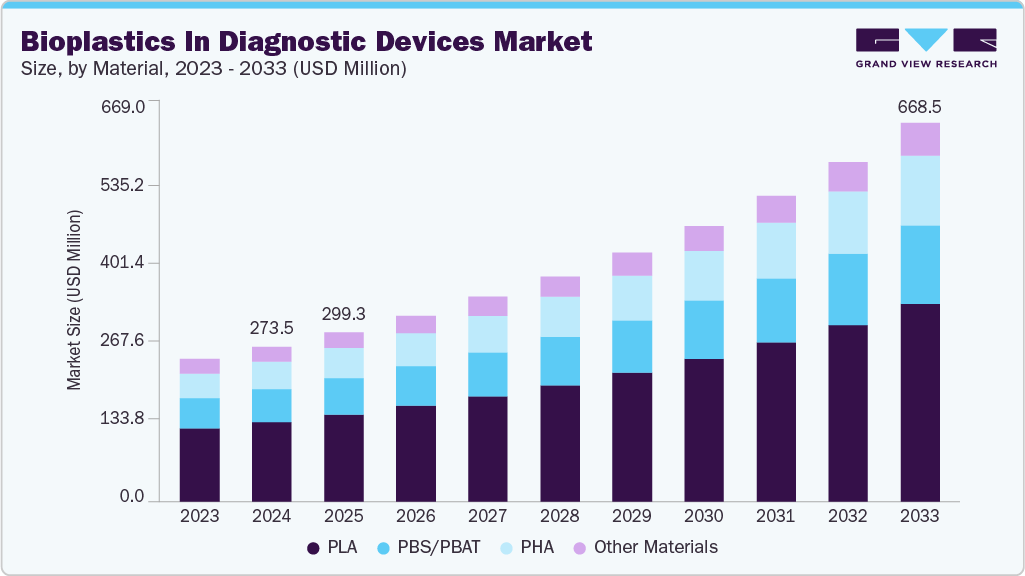
Drivers, Opportunities & Restraints
The major driver is the surge in point-of-care testing (POCT), especially lateral-flow assays and rapid diagnostics, which are predominantly single-use, high-volume products well suited to substitution with bioplastic housings and components. Healthcare systems and public procurement agencies increasingly favor suppliers that can demonstrate reduced lifecycle impacts; replacing commodity petrochemical housings with PLA or PHA variants allows test manufacturers and buyers to show measurable circularity or biodegradability benefits. This creates a strong commercial incentive for diagnostics OEMs to trial and, where feasible, adopt bioplastic components.
The lateral-flow and rapid-test housings, where material substitution delivers high ESG value at modest technical risk; these parts are typically low-complexity injection-molded housings or casings where PLA and PBS blends meet performance and sterilization requirements. There is also a clear commercial opening to offer full-kit sustainability: converting not only housings but trays, blister Rapid Test Kits, and secondary Rapid Test Kits to mono-material bioplastics or compostable pack designs creates differentiated product offerings for large buyers and retailers. OEMs that can demonstrate validated compatibility with membranes, reagents, and sterilization workflows stand to win preferred-supplier status.
Regulatory and clinical validation hurdles are the largest restraint. Diagnostic devices must often be validated for biocompatibility, extractables/leachables, sterilization stability, and long-term storage interactions with reagents. The time and cost to qualify a new polymer grade deters many manufacturers from switching materials unless there is a clear commercial imperative or regulatory requirement. This slows migration from petrochemical resins, especially for higher-complexity cartridges and reagent-contact parts.
Market Concentration & Characteristics
The market growth stage of the bioplastics in diagnostic devices industry is medium, and the pace is accelerating. Degree of innovation is concentrated in materials formulation, compatibilizers for blends, and process adaptation. Most innovation today is incremental and application-driven, but some breakthrough work in scalable PHA production and bio-based high-performance polyesters could be a step-change if costs come down.
Strategic M&A is visible mainly where biopolymer producers seek capacity or market access, and among contract manufacturers aiming to secure medical-grade capability. Large diagnostics OEMs tend to prefer supplier partnerships and qualification agreements over outright acquisitions, but expect consolidation among specialty compounders and converters as the market matures and scale becomes essential to supply validated medical grades.
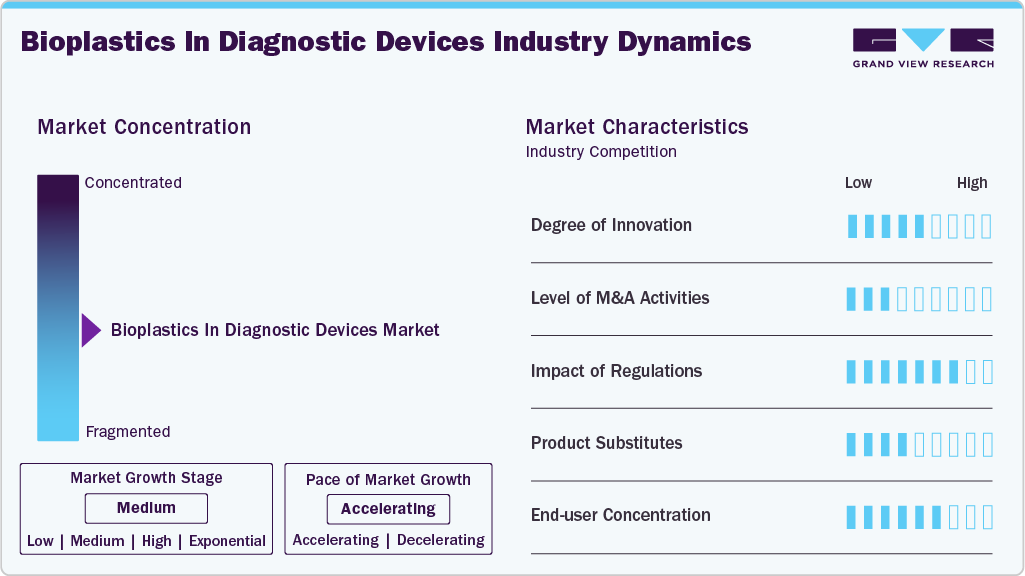
Impact of regulation is significant and mixed as procurement and waste-management regulations favor substitution and create commercial pull, while medical/regulatory requirements impose strict technical barriers that slow adoption. Net effect: regulations create demand incentives but also raise the qualification bar that bioplastic suppliers must clear.
Conventional petrochemical polymers remain the primary substitutes; they are cheaper, well-qualified, and fully standardized across OEM supply chains. Alternative non-polymer options are emerging for some Rapid Test Kits, but are not broadly applicable for reagent-contact or dimensional-precision diagnostic components.
A relatively small number of large diagnostics OEMs and kit manufacturers account for a substantial share of volume, including rapid-test market leaders and major instrument platform providers, which means winning a few strategic customers can rapidly scale a supplier. At the same time, a long tail of smaller test manufacturers and regional suppliers offers multiple entry points for bioplastic converters and niche players.
Material Insights
PLA dominated the market across the material segmentation in terms of revenue, accounting for a market share of 51.24% in 2024. PLA is one of the widely adopted bioplastics in diagnostic devices due to its cost-effectiveness, clarity, moldability, and compatibility with microfluidic fabrication. It is increasingly used in housings, disposable test kits, and microchannels for rapid diagnostics, though improvements in heat resistance and sterilization performance continue to expand its use. PBS and PBAT are favored for applications where flexibility, toughness, and heat-resistance are needed, making them ideal for rapid-test components, sample-collection consumables, and certain cartridge formats. These blends offer improved performance over pure PLA in sterilization processes and durability-driven testing workflows.
The PHA segment is anticipated to grow at a substantial CAGR of 11.2% through the forecast period. PHA is gaining traction as a premium bioplastic for diagnostic applications requiring higher biodegradability, excellent biocompatibility, and chemical resistance. Its suitability for sensitive microfluidic channels and biological assays makes it attractive for next-generation diagnostic consumables, despite higher cost challenges.
Application Insights
Rapid test kits dominated the market across the application segmentation in terms of revenue, accounting for a market share of 61.89% in 2024, and is forecasted to grow at the fastest CAGR of 11.0% from 2025 to 2033. Bioplastics are increasingly used in rapid test kits as manufacturers seek lightweight, low-carbon materials that maintain durability, chemical resistance, and precision during testing. PLA and PBS/PBAT are becoming preferred materials for test housings, cassettes, and sample wells due to their molding accuracy and environmental performance. Cartridges and microfluidic devices represent one of the most promising growth areas for bioplastics, as diagnostic companies aim to reduce waste from disposable testing consumables. PHA and PLA are especially suited for high-precision channels, reagent reservoirs, and single-use lab-on-chip platforms.
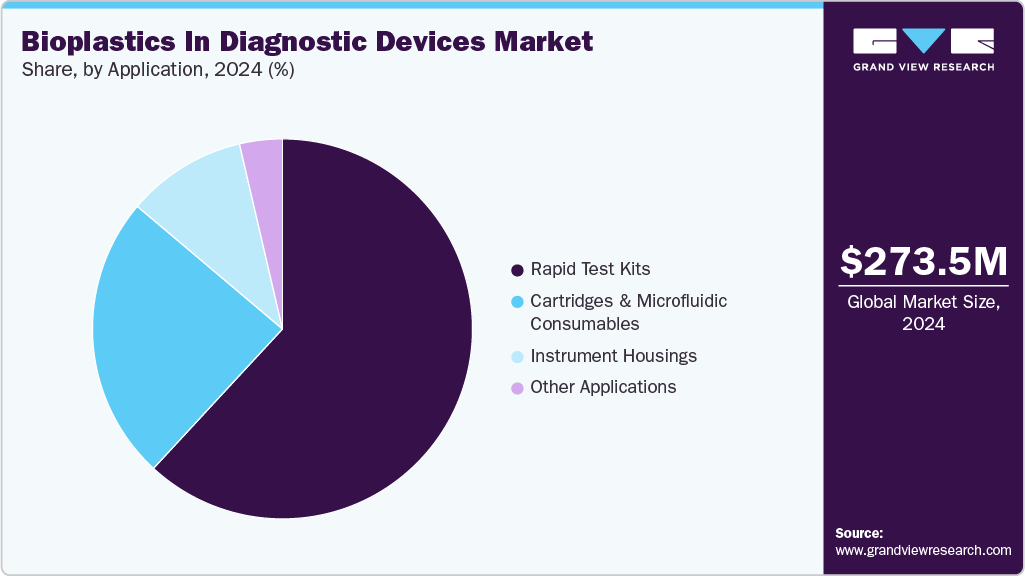
Bioplastic-based housings are being increasingly used in smaller benchtop instruments and portable diagnostic devices where sustainability credentials and design flexibility matter. PLA and PBS/PBAT provide a balance of aesthetics, structural strength, and environmental benefits, supporting greener device manufacturing.
Regional Insights
Asia Pacific held the largest share of 49.23% in terms of revenue of the bioplastics in diagnostic devices market in 2024 and is expected to grow at the fastest CAGR of 11.0% over the forecast period. Asia Pacific shows rapidly increasing demand driven by large-scale diagnostic manufacturing hubs in China, Japan, South Korea, and India, which are increasingly exploring bio-based materials to reduce carbon footprint and comply with upcoming regional plastics restrictions. Rising mass-diagnostics production-particularly for infectious disease testing-creates strong incentives for cost-efficient, high-volume bioplastic solutions.
China Bioplastics In Diagnostic Devices Market Trends
The bioplastics in diagnostic devices market in China is becoming a major production hub for bioplastic diagnostic components as the government accelerates green manufacturing and medical-waste reduction policies. High-volume diagnostic producers are actively exploring PLA and PBS/PBAT blends to reduce reliance on petroleum-based plastics.
India bioplastics in diagnostic devices market is led by rapid test manufacturers and microfluidic start-ups, and is beginning to integrate bioplastics to align with national sustainability mandates and reduce import dependence on virgin polymers. Growing domestic innovation in low-cost point-of-care devices is boosting material experimentation.
North America Bioplastics In Diagnostic Devices Market Trends
The bioplastics in diagnostic devices market in North America is driven by stringent sustainability goals across healthcare systems, rising procurement of eco-friendly consumables, and the strong presence of biotech OEMs developing rapid diagnostics and point-of-care devices. Regulatory encouragement toward reducing single-use petroleum plastics is accelerating the transition, while major diagnostic manufacturers are beginning to test bioplastic-based cartridges and microfluidic components for compliance, sterilizability, and scalability.
The U.S. bioplastics in diagnostic devices market is seeing strong traction toward bioplastic diagnostic components, supported by federal sustainability initiatives, large diagnostic OEM presence, and increasing use of disposable microfluidic devices in point-of-care testing. Hospitals and labs are evaluating bio-based materials to reduce landfill loads associated with single-use polymers.
Europe Bioplastics In Diagnostic Devices Market Trends
The bioplastics in diagnostic devices market in Europe remains the global leader in bioplastics adoption for diagnostic devices due to strict environmental regulations, the EU Green Deal, and active investment in compostable and bio-based polymers that can replace legacy plastics. Diagnostic manufacturers across Germany, France, and the Nordics are rapidly integrating PLA, PHA, and blended biopolymers into housings, microfluidic chips, and test-kit cartridges as part of hospital sustainability mandates and waste-management targets.
Germany bioplastics in diagnostic devices market leads Europe in bioplastic adoption for diagnostic devices due to its advanced polymer R&D ecosystem, strong circular-economy legislation, and the presence of major diagnostics exporters. Manufacturers are increasingly incorporating PLA and PHA in casings, test cartridges, and microfluidics.
Latin America Bioplastics In Diagnostic Devices Market Trends
The bioplastics in diagnostic devices market in Latin America is gradually adopting bioplastics in diagnostic devices, supported by the growing presence of regional medical-device assemblers and rising healthcare modernization efforts in Brazil, Mexico, and Chile. Sustainability policies remain uneven, but hospital networks are beginning to shift toward eco-friendly consumables, especially in public health programs targeting infectious diseases.
Middle East & Africa Bioplastics In Diagnostic Devices Market Trends
The bioplastics in diagnostic devices market in the Middle East & Africa is currently at an early adoption stage, largely driven by imports of diagnostic devices rather than regional production. However, sustainability-focused healthcare development in the UAE, Saudi Arabia, and South Africa is creating interest in bioplastic-based diagnostic consumables, especially for high-volume testing, where medical waste management is a rising concern.
Saudi Arabia bioplastics in diagnostic devices market is growing significantly as the healthcare modernization under Vision 2030 is encouraging hospitals and diagnostic providers to explore environmentally preferable materials, including bioplastics for rapid-test consumables and device housings. Increased procurement of advanced diagnostics also opens opportunities for bio-based alternatives.
Key Bioplastics In Diagnostic Devices Company Insights
Market competition is layered, including upstream resin producers competing to supply medical-grade, low-extractable PLA/PHA/PBS grades and certification support, while downstream converters and contract manufacturers compete on clean-room capability, sterilization experience, and regulatory dossiers. Adoption is driven by partnerships and pilot programs, with stronger incumbency for suppliers that can offer validated medical grades, stable supply, and documented sterilization compatibility.
Key Bioplastics In Diagnostic Devices Companies:
The following are the leading companies in the bioplastics in diagnostic devices market. These companies collectively hold the largest market share and dictate industry trends.
- NatureWorks
- Total Corbion PLA
- Corbion
- Danimer Scientific
- BASF SE
- Mitsubishi Chemical
- Kaneka Corporation
- Novamont
- FKuR Kunststoff GmbH
Recent Developments
-
In December 2024, GreenworksBio partnered with CSIR‑IICT to develop high-performance, eco-friendly bioplastics and biocomposites derived from agricultural waste such as crop residues and biomass. Their innovation uses nano-cellulose and starch-based feedstocks to create compostable plastics and reinforced biocomposites - including blends of biodegradable polymers such as PLA, PHA, PBS/PBAT - that offer strong mechanical strength, good barrier and thermal properties, and rapid biodegradation. They’ve also set up a production facility to manufacture compostable granules, eco-friendly tableware, hygiene solutions, and other products, demonstrating a real-world pathway to replace conventional single-use plastics with sustainable, bio-based alternatives.
Bioplastics In Diagnostic Devices Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 299.3 million
Revenue forecast in 2033
USD 668.5 million
Growth rate
CAGR of 10.6% from 2025 to 2033
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion, volume in tons and CAGR from 2025 to 2033
Report coverage
Revenue forecast, competitive landscape, growth factors, and trends
Report Segmentation
Material, application, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country Scope
U.S.; Canada; Mexico; Germany; France; UK; Spain; Italy; China; Japan; India; South Korea; Brazil; Argentina; Saudi Arabia; South Africa; UAE
Key companies profiled
NatureWorks; Total Corbion PLA; Corbion; Danimer Scientific; BASF SE; Mitsubishi Chemical; Kaneka Corporation; Novamont; FKuR Kunststoff GmbH
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Bioplastics In Diagnostic Devices Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global bioplastics in diagnostic devices market report based on material, application, and region:
-
Material Outlook (Revenue, USD Million, Volume, Tons, 2021 - 2033)
-
PLA
-
PHA
-
PBS/PBAT
-
Other Materials
-
-
Application Outlook (Revenue, USD Million, Volume, Tons, 2021 - 2033)
-
Rapid Test Kits
-
Cartridges & Microfluidic Consumables
-
Instrument Housings
-
Other Applications
-
-
Regional Outlook (Revenue, USD Million, Volume, Tons, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
-
Asia Pacific
-
China
-
India
-
Japan
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
Saudi Arabia
-
South Africa
-
UAE
-
-
Frequently Asked Questions About This Report
b. The global bioplastics in diagnostic devices market size was estimated at USD 273.5 million in 2024 and is expected to reach USD 299.3 million in 2025.
b. The global bioplastics in diagnostic devices market is expected to grow at a compound annual growth rate of 10.6% from 2025 to 2033 to reach USD 668.5 million by 2033.
b. PLA dominated the bioplastics in diagnostic devices market in terms of revenue, accounting for a market share of 51.24% in 2024 as it is widely adopted bioplastics in diagnostic devices due to its cost-effectiveness, clarity, moldaility, and compatibility with microfluidic fabrication.
b. Some key players operating in the bioplastics in diagnostic devices market include NatureWorks, Total Corbion PLA, Corbion, Danimer Scientific, BASF SE, Mitsubishi Chemical, Kaneka Corporation, Novamont, and FKuR Kunststoff GmbH.
b. The major driver driving the bioplastics in diagnostic devices market is the surge in point-of-care testing (POCT), especially lateral-flow assays and rapid diagnostics, which are predominantly single-use, high-volume products well suited to substitution with bioplastic housings and components.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










