- Home
- »
- Medical Devices
- »
-
Biotechnology & Pharmaceutical Services Outsourcing Market, 2030GVR Report cover
![Biotechnology And Pharmaceutical Services Outsourcing Market Size, Share & Trends Report]()
Biotechnology And Pharmaceutical Services Outsourcing Market Size, Share & Trends Analysis Report By Services (Consulting, Auditing and Assessment, Regulatory Affairs), By End-use (Pharma, Biotech), By Region, And Segment Forecasts, 2024 - 2030
- Report ID: GVR-1-68038-326-3
- Number of Pages: 133
- Format: Electronic (PDF)
- Historical Range: 2018 - 2023
- Industry: Healthcare
Market Size & Trends
The global biotechnology and pharmaceutical services outsourcing market was valued at USD 75.13 billion in 2023 and is anticipated to grow at a compound annual growth rate (CAGR) of 5.2% from 2024 to 2030.The rising drug development costs, diminishing internal capabilities, increasing regulatory frameworks, and low-cost service deployment to contract research organizations (CROs), and contract manufacturing organizations (CMOs) are some of the major factors driving the market growth.
Post pandemic, demand for innovative and effective therapies is increasing and driving growth. The rising drug development costs coupled with higher failure rates and growing regulatory pressure also have a significant impact on market dynamics, accelerating the growth rate. Furthermore, well-established CROs, regulatory outsourcing firms, management consulting firms, and contract manufacturers are catering to the complex demand of the pharmaceutical and biotechnological sectors. Contract research and manufacturing companies are investing in personnel, infrastructure, and technology to acquire a significant share of the healthcare outsourcing market.
The increasing demand due to the ongoing patent cliff of biotechnology drugs is anticipated to fuel demand. An increasing number of end-to-end service providers, to meet the rising demand for low-cost drug development and manufacturing, is further anticipated to propel the market growth. Moreover, novel drug delivery mechanisms and new product launches are anticipated to drive the outsourcing demand. Many companies are opting for outsourcing owing to the increasing competition in the healthcare industry. For instance, in February 2023, Lonza completed the expansion of its conjugation facility in Visp, Switzerland.
The expansion included the addition of manufacturing suites & supporting infrastructure, increasing the development & manufacturing capacity for bioconjugates & antibody-drug conjugates for preclinical, clinical, & commercial supply. An increasing number of R&D activities for new drug development, combination products, and other advanced medicines have increased the demand for contract biotechnology and pharmaceutical services. Currently, the global pharmaceutical industry has the second-highest R&D intensity measures of any sector, which indicates that the spending on R&D is increasing, and the overall spending is likely to grow during the forecast period. For instance, in 2022, the 11 major pharmaceutical companies invested USD 104 billion in research and development (R&D), constituting 42% of the overall research expenditure in the global biopharmaceutical sector for the previous year.
Market Concentration & Characteristics
Market growth stage is medium, and pace of the market growth is accelerating. The biotechnology and pharmaceutical services outsourcing market is characterized by a high degree of innovation. The pharmaceutical and biotechnology industry is driven by continuous advancements in technology. Novel manufacturing processes, analytical techniques, and research methodologies are developed to improve efficiency, reduce costs, and enhance the quality of biopharmaceutical products.

Biotechnology and pharmaceutical services outsourcing market is also characterized by a medium level of merger and acquisition (M&A) activity by a leading player. Companies are merge or acquire others in the biotechnology and pharmaceutical services outsourcing sector to expand their service offerings, geographic reach, or client base.
Biotechnology and pharmaceutical services outsourcing market is characterized by a high impact of regulations. The pharmaceutical and biotechnology sectors are subject to strict quality and safety standards to ensure the efficacy and safety of drugs and biologics. Regulatory agencies establish and enforce these standards, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Biotechnology and pharmaceutical services outsourcing market is characterized by a high impact of regional expansion. Companies in the biotechnology and pharmaceutical sectors look for opportunities to access emerging markets with growing healthcare needs. Regional expansion allows them to establish a presence in these markets, collaborate with local partners, and tap into new patient populations.
Biotechnology and pharmaceutical services outsourcing market is characterized by a high impact on service expansion.Biotechnology and pharmaceutical companies prefer outsourcing partners that offer integrated solutions, covering a wide spectrum of services. Service expansion allows outsourcing providers to offer end-to-end solutions, encompassing discovery, clinical trials, preclinical development, regulatory affairs, manufacturing, and post-market support.
Service Insights
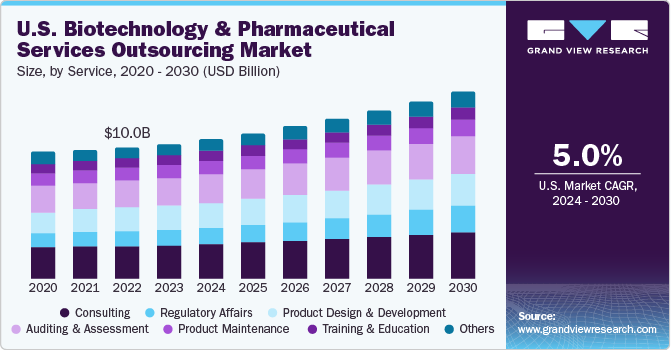
The consulting services segment dominated the market in 2023 and accounted for the largest share of over 19% of the overall revenue. The segment is expected to maintain its position over the forecast period. This can be attributed to the increasing M&A activities and constantly changing regulatory protocols. However, the pharma and biotech market has witnessed a continuous entry of new players, such as Signa Medical Writing, bioSyntagma, and Fieve Clinical Research, Inc. These players need to be compliant with set standards and norms, for which consulting is essential as these new entrants lack such capabilities.
However, the other services segment is expected to witness a higher CAGR of 6.1% over the forecast period owing to the rising outsourcing of generics and biosimilar manufacturing as CMOs are offering services at lower cost. The other segment is inclusive of contract manufacturing, product upgrade, and IT consulting. Biostatistics outsourcing is increasing owing to the growing concerns about biotechnology and pharmaceutical companies with regard to competition in product commercialization, clinical trial complexity, and the quality of clinical trials that have boosted the importance of biostatistics.
End-use Insights
The pharma end-use segment dominated the global market in 2023 and account for the largest share of over 55% of the overall revenue. The segment is expected to register a CAGR of 5.4% over the forecast period. An increase in R&D spending by pharmaceutical companies for the development of potential novel products and a rise in investments by CROs for the development of core capabilities are expected to drive the market in the region in the coming years. Contract service providers are recognized as an effective strategic decision to curb the issues of drug shortfall and high production costs, as well as meet the growing demand.
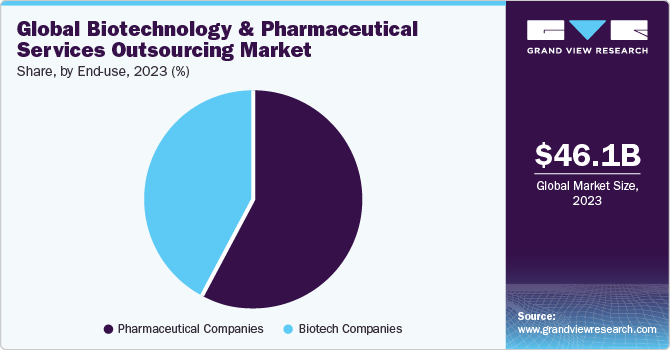
On the other hand, the biotechnology end-use segment is expected to have significant growth during the forecast period. This growth can be attributed to the rapidly expanding biotech product pipelines, along with a trend toward biotechnology entities retaining their assets longer. Moreover, CROs concentrate on adapting their services to smaller biotechnology firms, which have emerged as prominent players in the clinical development of novel medicines and are responsible for driving innovation and expansion.
Regional Insights
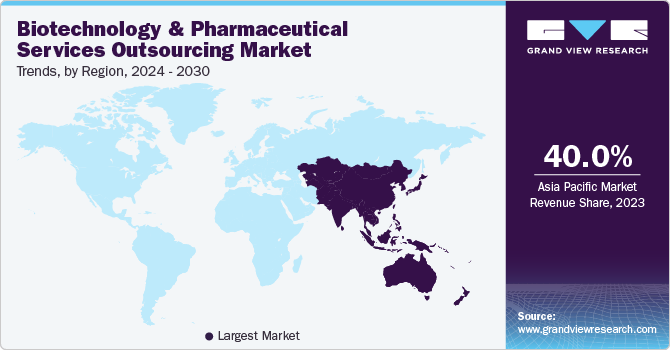
North America dominated the global market in 2023 and accounted for the largest share of over 53% of the overall revenue. This growth is attributed to the presence of several established CROs and CMOs, such as Covance Inc., IQVIA, Catalent, and Samsung Biologics, and growing R&D investments by life sciences and pharmaceutical companies in the region. The presence of stringent regulatory policies and a rise in R&D expenditure are among the factors that are likely to boost the demand for outsourcing of services by pharmaceutical and biotechnology companies in the region. In addition, the expansion of several CMOs and CROs into the country is projected to propel the market growth. For instance, in May 2022, Lonza expanded its inhalation facility at its Tampa (the U.S.) site to improve its capabilities in the development and manufacture of inhaled formulations.
The U.S. held dominant share in North America biotechnology/pharmaceutical services outsourcing market in 2023. Pharmaceutical and biotechnology companies outsource part of their regulatory functions, such as report writing & publishing, clinical trial application services, and product design & maintenance, to regulatory service providers, thereby contributing to market growth. Moreover, outsourcing helps a company focus more on its core capabilities and improve its service delivery, thereby providing competitive advantage.
Asia Pacific is expected to witness a CAGR of 5.6% over the forecast period owing to a rise in investments by developed countries and various regulatory reforms in clinical trial evaluation to align with the standards of various countries investing in the region. The low cost of drug development & manufacturing and the availability of a skilled workforce is likely to foster contract development and manufacturing in this region. Moreover, economic policy reforms in countries, such as China, are anticipated to create an open and balanced economy, which presents ample growth opportunities for market players to invest in this region. For instance, in August 2022, Porton Advanced Solutions, a Chinese CDMO entity, raised USD 80 million to advance the cell and gene therapy platform.
China biotechnology/pharmaceutical services outsourcing market is expected to grow in recent years owing to entry of new players and service providers in the country, which can be attributed to stringent standards around patient safety in conducting clinical trials, easy access to a large patient pool, and cheap labor. In addition, low operating cost, increasing investments to improve technology, high R&D capacity, and rising adoption of global R&D standards to meet international requirements are some of the key factors responsible for the growing demand for regulatory & consulting services in China.
Key Companies & Market Share Insights
The market is highly competitive and companies undertake various strategies, such as merger & acquisition, collaboration, regional expansion, service portfolio expansion, and competitive pricing, to sustain in the competitive environment and acquire a higher market share. For instance, in May 2023, Vetter announced plans for the expansion of its production capacities and services at its Austrian site. This strategic move comes in response to the rising demand from pharmaceutical clients, aiming to meet their evolving needs and address the increased requirements for services, such as drug development, aseptic filling, and packaging.
Key Biotechnology and Pharmaceutical Services Outsourcing Companies:
The following are the leading companies in the biotechnology and pharmaceutical services outsourcing market. These companies collectively hold the largest market share and dictate industry trends. Financials, strategy maps & products of these biotechnology and pharmaceutical services outsourcing companies are analyzed to map the supply network.
- Parexel International Corporation
- The Quantic Group
- IQVIA
- Lachman Consultant Services, Inc.
- GMP Pharmaceuticals Pty Ltd.
- Concept Heidelberg GmbH
- Covance Inc. (LabCorp)
- Charles River Laboratories
- ICON plc.
- Syneos Health
Recent Developments
-
In May 2023, LabCorp declared its commitment to enhancing the enduring strategic collaboration with Providence through a newly established agreement. As part of this arrangement, LabCorp will acquire the outreach laboratory business of Providence Oregon and specific assets situated in Oregon.
-
In September 2023, Charles River Laboratories International, Inc. declared a collaboration agreement encompassing multiple programs to deploy Logica across various targets within the RS portfolio previously unexplored for drug development. Logica is an Artificial Intelligence (AI)-driven drug solution that converts biological insights into refined assets.
Biotechnology and Pharmaceutical Services Outsourcing Market Report Scope
Report Attribute
Details
Market size value in 2024
USD 79.72 billion
Revenue forecast in 2030
USD 107.97 billion
Growth rate
CAGR of 5.2% from 2024 to 2030
Historical data
2018 - 2023
Forecast period
2024 - 2030
Quantitative units
Revenue in USD Million and CAGR from 2024 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Service, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; U.K.; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; India; Japan; Australia; Thailand; South Korea; Brazil; Mexico; Argentina; Saudi Arabia; UAE; South Africa, Kuwait
Key companies profiled
The Quantic Group; IQVIA; Parexel International Corporation; Lachman Consultant Services, Inc.; GMP Pharmaceuticals Pty Ltd.; Concept Heidelberg GmbH; Covance, Inc. (LabCorp); Charles River Laboratories; ICON plc; Syneos Health
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Biotechnology and Pharmaceutical Services Outsourcing Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For the purpose of this study, Grand View Research has segmented the global biotechnology and pharmaceutical services outsourcing market report on the basis of service, end use, and region:
-
Service Outlook (Revenue, USD Million, 2018 - 2030)
-
Consulting
-
Regulatory compliance
-
Remediation
-
Quality management systems consulting
-
Others
-
-
Auditing and assessment
-
Regulatory affairs
-
Clinical trial applications & product registration
-
Regulatory writing & publishing
-
Legal representation
-
Others
-
-
Product maintenance
-
Product design & development
-
Product testing & validation
-
Training & education
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2018 - 2030)
-
Pharma
-
Biotech
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Europe
-
U.K.
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
Asia Pacific
-
India
-
China
-
Japan
-
Australia
-
Thailand
-
South Korea
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Frequently Asked Questions About This Report
b. Key factors that are driving the biotechnology and pharmaceutical services outsourcing market growth include growing drug development costs coupled with high clinical development failure rates and increasing competition within the healthcare drug manufacturers.
b. The global biotechnology and pharmaceutical services outsourcing market size was estimated at USD 70.48 billion in 2022 and is expected to reach USD 75.12 billion in 2023.
b. The global biotechnology and pharmaceutical services outsourcing market is expected to grow at a compound annual growth rate of 5.3% from 2023 to 2030 to reach USD 107.96 billion by 2030.
b. North America dominated the biotechnology and pharmaceutical services outsourcing market with a share of 53.5% in 2022. This is attributable to the presence of several multinational and local consulting firms in this region.
b. Some key players operating in the biotechnology and pharmaceutical services outsourcing market include The Quantic Group; IQVIA; Parexel International Corporation; Lachman Consultant Services, Inc.; GMP Pharmaceuticals Pty Ltd.; Concept Heidelberg GmbH; Covance, Inc.; Charles River Laboratories; PRA Health Sciences; and ICON plc.
Table of Contents
Chapter 1. Biotechnology And Pharmaceutical Services Outsourcing Market: Methodology and Scope
1.1. Market Segmentation and Scope
1.2. Research Methodology
1.2.1. Information Procurement
1.3. Information or Data Analysis
1.4. Methodology
1.5. Research Scope and Assumptions
1.6. Market Formulation & Validation
1.6.1.1. Country Market: CARG Calculation
1.7. Country Based Segment Share Calculation
1.8. List of Data Sources
Chapter 2. Biotechnology And Pharmaceutical Services Outsourcing Market: Executive Summary
2.1. Market Outlook
2.2. Segment Outlook
2.3. Regional Insights
2.4. Competitive Insights
2.5. Biotechnology and Pharmaceutical Services Outsourcing Market Snapshot
2.6. Biotechnology and Pharmaceutical Services Outsourcing Market Segment Snapshot
2.7. Biotechnology and Pharmaceutical Services Outsourcing Market Competitive Landscape Snapshot
Chapter 3. Biotechnology and Pharmaceutical Services Outsourcing Market: Variables, Trends, & Scope
3.1. Market Lineage Outlook
3.2. Market Dynamics
3.2.1. Market Driver Analysis
3.2.1.1. Increasing outsourcing of R&D activities
3.2.1.2. Changing regulatory landscape
3.2.1.3. Increasing focus on core competencies by pharmaceutical and biotechnology companies
3.2.1.4. Growing drug development costs coupled with high clinical development failure rates
3.2.1.5. Increasing mergers and collaborations
3.2.2. Market Restraint Analysis
3.2.2.1. Monitoring issues and lack of standardization
3.2.2.2. Loss of control
3.2.2.3. Data security issues
3.3. Biotechnology and Pharmaceutical Services Outsourcing Market Analysis Tools
3.3.1. Industry Analysis - Porter’s
3.3.1.1. Bargaining power of the suppliers
3.3.1.2. Bargaining power of the buyers
3.3.1.3. Threats of substitution
3.3.1.4. Threats from new entrants
3.3.1.5. Competitive rivalry
3.3.2. PESTEL Analysis
3.3.2.1. Political landscape
3.3.2.2. Economic and Social landscape
3.3.2.3. Technological landscape
3.3.3. End-use Perception Analysis
3.3.3.1. Why do you outsource Biotechnology and Pharmaceutical services to CROs?
3.3.3.2. Percentage of outsourcing of different services.
3.3.3.3. Importance of pharmaceutical & biotechnology CRO selection.
3.3.4. Service Price Analysis
3.3.4.1. Pricing strategy
3.3.4.1.1. Transactional business models
3.3.4.1.2. Performance-based business models
3.3.4.1.3. Outcome-based business models
3.3.4.2. Consulting rates
3.3.4.3. Heat map for pricing analysis
Chapter 4. Biotechnology and Pharmaceutical Services Outsourcing Market: Service Estimates & Trend Analysis
4.1. Segment Dashboard
4.2. Biotechnology and Pharmaceutical Services Outsourcing Market: Service Movement Analysis, USD Million, 2022 & 2030
4.3. Consulting
4.3.1. Consulting Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.3.2. Regulatory Compliance
4.3.2.1. Regulatory Compliance Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.3.3. Remediation
4.3.3.1. Remediation Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.3.4. Quality Management Systems Consulting
4.3.4.1. Quality Management Systems Consulting Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.3.5. Others
4.3.5.1. Others Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.4. Auditing and Assessment
4.4.1. Auditing and Assessment Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.5. Regulatory affairs
4.5.1. Regulatory affairs Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.5.2. Clinical Trial Applications & Product Registration
4.5.2.1. Clinical Trial Applications & Product Registration Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.5.3. Remediation
4.5.3.1. Remediation Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.5.4. Regulatory Writing & Publishing
4.5.4.1. Regulatory Writing & Publishing Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.5.5. Others
4.5.5.1. Others Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.6. Product Maintenance
4.6.1. Product Maintenance Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.7. Product Design & Development
4.7.1. Product Design & Development Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.8. Product Testing & Validation
4.8.1. Product Testing & Validation Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.9. Training & Education
4.9.1. Training & Education Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
4.10. Others
4.10.1. Others Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
Chapter 5. Biotechnology and Pharmaceutical Services Outsourcing Market: End-Use Estimates & Trend Analysis
5.1. Segment Dashboard
5.2. Biotechnology and Pharmaceutical Services Outsourcing Market: End-Use Movement Analysis, USD Million, 2022 & 2030
5.3. Pharmaceutical Companies
5.3.1. Pharmaceutical Companies Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.4. Biotech Companies
5.4.1. Biotech Companies Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
Chapter 6. Biotechnology and Pharmaceutical Services Outsourcing Market: Regional Estimates & Trend Analysis
6.1. Biotechnology and Pharmaceutical Services Outsourcing Market: Regional Movement Analysis, 2022 & 2030
6.2. North America
6.2.1. Regulatory Framework
6.2.2. North America Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.2.3. U.S.
6.2.3.1. Key Country Dynamics
6.2.3.2. Competitive Scenario
6.2.3.3. U.S. Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.2.4. Canada
6.2.4.1. Key Country Dynamics
6.2.4.2. Competitive Scenario
6.2.4.3. Canada Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.3. Europe
6.3.1. Regulatory Framework
6.3.2. Europe Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.3.3. UK
6.3.3.1. Key Country Dynamics
6.3.3.2. Competitive Scenario
6.3.3.3. UK Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.3.4. Germany
6.3.4.1. Key Country Dynamics
6.3.4.2. Competitive Scenario
6.3.4.3. Germany Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.3.5. France
6.3.5.1. Key Country Dynamics
6.3.5.2. Competitive Scenario
6.3.5.3. Prevalence Insights
6.3.5.4. France Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.3.6. Italy
6.3.6.1. Key Country Dynamics
6.3.6.2. Competitive Scenario
6.3.6.3. Prevalence Insights
6.3.6.4. Italy Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.3.7. Spain
6.3.7.1. Key Country Dynamics
6.3.7.2. Competitive Scenario
6.3.7.3. Prevalence Insights
6.3.7.4. Spain Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.3.8. Denmark
6.3.8.1. Key Country Dynamics
6.3.8.2. Competitive Scenario
6.3.8.3. Prevalence Insights
6.3.8.4. Denmark Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.3.9. Sweden
6.3.9.1. Key Country Dynamics
6.3.9.2. Competitive Scenario
6.3.9.3. Prevalence Insights
6.3.9.4. Sweden Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.3.10. Norway
6.3.10.1. Key Country Dynamics
6.3.10.2. Competitive Scenario
6.3.10.3. Prevalence Insights
6.3.10.4. Norway Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.4. Asia Pacific
6.4.1. Regulatory Framework
6.4.2. Asia Pacific Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.4.3. Japan
6.4.3.1. Key Country Dynamics
6.4.3.2. Competitive Scenario
6.4.3.3. Prevalence Insights
6.4.3.4. Japan Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.4.4. China
6.4.4.1. Key Country Dynamics
6.4.4.2. Competitive Scenario
6.4.4.3. Prevalence Insights
6.4.4.4. China Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.4.5. India
6.4.5.1. Key Country Dynamics
6.4.5.2. Competitive Scenario
6.4.5.3. Prevalence Insights
6.4.5.4. India Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.4.6. Australia
6.4.6.1. Key Country Dynamics
6.4.6.2. Competitive Scenario
6.4.6.3. Prevalence Insights
6.4.6.4. Australia Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.4.7. Thailand
6.4.7.1. Key Country Dynamics
6.4.7.2. Competitive Scenario
6.4.7.3. Prevalence Insights
6.4.7.4. Thailand Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.4.8. South Korea
6.4.8.1. Key Country Dynamics
6.4.8.2. Competitive Scenario
6.4.8.3. Prevalence Insights
6.4.8.4. South Korea Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.5. Latin America
6.5.1. Regulatory Framework
6.5.2. Latin America Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.5.3. Brazil
6.5.3.1. Key Country Dynamics
6.5.3.2. Competitive Scenario
6.5.3.3. Brazil Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.5.4. Mexico
6.5.4.1. Key Country Dynamics
6.5.4.2. Competitive Scenario
6.5.4.3. Mexico Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.5.5. Argentina
6.5.5.1. Key Country Dynamics
6.5.5.2. Competitive Scenario
6.5.5.3. Argentina Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.6. Middle East & Africa
6.6.1. Regulatory Framework
6.6.2. Middle East & Africa Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.6.3. South Africa
6.6.3.1. Key Country Dynamics
6.6.3.2. Competitive Scenario
6.6.3.3. South Africa Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.6.4. Saudi Arabia
6.6.4.1. Key Country Dynamics
6.6.4.2. Competitive Scenario
6.6.4.3. Saudi Arabia Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.6.5. UAE
6.6.5.1. Key Country Dynamics
6.6.5.2. Competitive Scenario
6.6.5.3. UAE Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
6.6.6. Kuwait
6.6.6.1. Key Country Dynamics
6.6.6.2. Competitive Scenario
6.6.6.3. Kuwait Biotechnology and Pharmaceutical Services Outsourcing Market Estimates And Forecasts, 2018 - 2030 (USD Million)
Chapter 7. Competitive Landscape
7.1. Company Categorization
7.2. Company Market Share Analysis, 2022
7.3. Strategy Mapping
7.4. Company Profiles/Listing
7.4.1. The Quantic Group
7.4.1.1. Overview
7.4.1.2. Financial Performance (Net Revenue/Sales/EBITDA/Gross Profit)
7.4.1.3. Product Benchmarking
7.4.1.4. Strategic Initiatives
7.4.2. IQVIA HOLDINGS, INC.
7.4.2.1. Overview
7.4.2.2. Financial Performance (Net Revenue/Sales/EBITDA/Gross Profit)
7.4.2.3. Product Benchmarking
7.4.2.4. Strategic Initiatives
7.4.3. Parexel International Corporation
7.4.3.1. Overview
7.4.3.2. Financial Performance (Net Revenue/Sales/EBITDA/Gross Profit)
7.4.3.3. Product Benchmarking
7.4.3.4. Strategic Initiatives
7.4.4. Lachman Consultant Services
7.4.4.1. Overview
7.4.4.2. Financial Performance (Net Revenue/Sales/EBITDA/Gross Profit)
7.4.4.3. Product Benchmarking
7.4.4.4. Strategic Initiatives
7.4.5. GMP Pharmaceuticals
7.4.5.1. Overview
7.4.5.2. Financial Performance (Net Revenue/Sales/EBITDA/Gross Profit)
7.4.5.3. Product Benchmarking
7.4.5.4. Strategic Initiatives
7.4.6. Concept Heidelberg GmbH
7.4.6.1. Overview
7.4.6.2. Financial Performance (Net Revenue/Sales/EBITDA/Gross Profit)
7.4.6.3. Product Benchmarking
7.4.6.4. Strategic Initiatives
7.4.7. Covance, INC.
7.4.7.1. Overview
7.4.7.2. Financial Performance (Net Revenue/Sales/EBITDA/Gross Profit)
7.4.7.3. Product Benchmarking
7.4.7.4. Strategic Initiatives
7.4.8. Charles River
7.4.8.1. Overview
7.4.8.2. Financial Performance (Net Revenue/Sales/EBITDA/Gross Profit)
7.4.8.3. Product Benchmarking
7.4.8.4. Strategic Initiatives
7.4.9. PRA Health Sciences
7.4.9.1. Overview
7.4.9.2. Financial Performance (Net Revenue/Sales/EBITDA/Gross Profit)
7.4.9.3. Product Benchmarking
7.4.9.4. Strategic Initiatives
7.4.10. ICON plc.
7.4.10.1. Overview
7.4.10.2. Financial Performance (Net Revenue/Sales/EBITDA/Gross Profit)
7.4.10.3. Product Benchmarking
7.4.10.4. Strategic Initiatives
List of Tables
Table 1. List of secondary sources
Table 2. List of abbreviations
Table 3. Global Biotechnology and Pharmaceutical Services Outsourcing Market, By Region, 2018 - 2030 (USD Million)
Table 4. Global Biotechnology and Pharmaceutical Services Outsourcing Market, By Services, 2018 - 2030 (USD Million)
Table 5. Global Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use, 2018 - 2030 (USD Million)
Table 6. North America Biotechnology and Pharmaceutical Services Outsourcing Market, By Country, 2018 - 2030 (USD Million)
Table 7. North America Biotechnology and Pharmaceutical Services Outsourcing Market, By Services, 2018 - 2030 (USD Million)
Table 8. North America Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use, 2018 - 2030 (USD Million)
Table 9. U.S. Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 10. U.S. Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 11. Canada Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 12. Canada Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 13. Europe Biotechnology and Pharmaceutical Services Outsourcing Market, by Country, 2018 - 2030 (USD Million)
Table 14. Europe Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 15. Europe Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 16. U.K. Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 17. U.K. Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 18. Germany Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 19. Germany Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 20. France Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 21. France Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 22. Italy Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 23. Italy Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 24. Spain Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 25. Spain Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 26. Denmark Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 27. Denmark Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 28. Sweden Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 29. Sweden Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 30. Norway Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 31. Norway Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 32. Asia Pacific Biotechnology and Pharmaceutical Services Outsourcing Market, by Country, 2018 - 2030 (USD Million)
Table 33. Asia Pacific Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 34. Asia Pacific Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 35. Japan Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 36. Japan Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 37. China Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 38. China Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 39. India Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 40. India Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 41. Australia Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 42. Australia Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 43. Thailand Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 44. Thailand Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 45. South Korea Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 46. South Korea Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 47. Latin America Biotechnology and Pharmaceutical Services Outsourcing Market, by Country, 2018 - 2030 (USD Million)
Table 48. Latin America Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 49. Latin America Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 50. Brazil Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 51. Brazil Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 52. Mexico Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 53. Mexico Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 54. Argentina Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 55. Argentina Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 56. MEA Biotechnology and Pharmaceutical Services Outsourcing Market, by Country, 2018 - 2030 (USD Million)
Table 57. MEA Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 58. MEA Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 59. South Africa Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 60. South Africa Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 61. South Arabia Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 62. South Arabia Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 63. UAE Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 64. UAE Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 65. Kuwait Biotechnology and Pharmaceutical Services Outsourcing Market, by Services, 2018 - 2030 (USD Million)
Table 66. Kuwait Biotechnology and Pharmaceutical Services Outsourcing Market, by End Use, 2018 - 2030 (USD Million)
Table 67. Participant’s overview
Table 68. Financial performance
Table 69. Key companies undergoing expansions
Table 70. Key companies undergoing acquisitions
Table 71. Key companies undergoing collaborations
Table 72. Key companies launching new products/services
Table 73. Key companies undergoing partnerships
Table 74. Key companies undertaking other strategies
List of Figures
Fig. 1 Market segmentation
Fig. 2 Market research process
Fig. 3 Data triangulation techniques
Fig. 4 Primary research pattern
Fig. 5 Market research approaches
Fig. 6 Value-chain-based sizing & forecasting
Fig. 7 QFD modeling for market share assessment
Fig. 8 Market formulation and validation
Fig. 9 Commodity flow analysis
Fig. 10 Biotechnology and Pharmaceutical Services Outsourcing Market Outlook, 2018 - 2030 (USD Million)
Fig. 11 Biotechnology and Pharmaceutical Services Outsourcing Market Summary, 2021 (USD Million)
Fig. 12 Market trends & outlook
Fig. 13 Parent Market Outlook
Fig. 14 Ancillary Market Outlook
Fig. 15 Market driver analysis (Current & future impact)
Fig. 16 Market restraint analysis (Current & future impact)
Fig. 17 Company market position analysis
Fig. 18 Biotechnology and Pharmaceutical Services Outsourcing market, service outlook key takeaways (USD Million)
Fig. 19 Biotechnology and Pharmaceutical Services Outsourcing market: Service movement analysis (USD Million)
Fig. 20 Consulting market, 2018 - 2030 (USD Million)
Fig. 21 Regulatory compliance market, 2018 - 2030 (USD Million)
Fig. 22 Remediation market, 2018 - 2030 (USD Million)
Fig. 23 Quality management systems market, 2018 - 2030 (USD Million)
Fig. 24 Other consulting market, 2018 - 2030 (USD Million)
Fig. 25 Auditing and assessment market, 2018 - 2030 (USD Million)
Fig. 26 Regulatory affairs market, 2018 - 2030 (USD Million)
Fig. 27 Clinical trial applications & product registration market, 2018 - 2030 (USD Million)
Fig. 28 Regulatory writing & publishing market, 2018 - 2030 (USD Million)
Fig. 29 Legal representation market, 2018 - 2030 (USD Million)
Fig. 30 Other regulatory affairs market, 2018 - 2030 (USD Million)
Fig. 31 Product maintenance market, 2018 - 2030 (USD Million)
Fig. 32 Product design & development market, 2018 - 2030 (USD Million)
Fig. 33 Product testing & validation market, 2018 - 2030 (USD Million)
Fig. 34 Training & education market, 2018 - 2030 (USD Million)
Fig. 35 Others market, 2018 - 2030 (USD Million)
Fig. 36 Biotechnology and pharmaceutical services outsourcing market: End Use movement analysis
Fig. 37 Biotechnology and pharmaceutical services outsourcing market End Use outlook: Key takeaways
Fig. 38 Pharma market, 2018 - 2030 (USD Million)
Fig. 39 Biotech market, 2018 - 2030 (USD Million)
Fig. 40 Regional outlook, 2018 & 2030
Fig. 41 Regional market dashboard
Fig. 42 Regional market place: Key takeaways
Fig. 43 North America biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 44 U.S. biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 45 Canada biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 46 Europe biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 47 UK biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 48 Germany biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 49 France biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 50 Italy biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 51 Spain biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 52 Denmark biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 53 Sweden biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 54 Norway biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 55 Asia Pacific biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 56 Japan biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 57 China (Mainland) biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 58 India biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 59 Australia biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 60 Thailand biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 61 South Korea biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 62 Latin America biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 63 Brazil biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 64 Mexico biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 65 Argentina biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 66 MEA biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 67 South Africa biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 68 Saudi Arabia biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 69 UAE biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)
Fig. 70 Kuwait biotechnology and pharmaceutical services outsourcing market, 2018 - 2030 (USD Million)What questions do you have? Get quick response from our industry experts. Request a Free ConsultationMarket Segmentation
- Biotechnology & Pharmaceutical Services Outsourcing Service Outlook (Revenue, USD Million, 2018 - 2030)
- Consulting
- Regulatory Compliance
- Remediation
- Quality Management Systems Consulting
- Others
- Auditing & Assessment
- Regulatory Affairs
- Clinical Trial Applications & Product Registration
- Regulatory Writing & Publishing
- Legal Representation
- Others
- Product Maintenance
- Product Design & Development
- Product Testing & Validation
- Training & Education
- Others
- Consulting
- Biotechnology & Pharmaceutical Services Outsourcing End-Use Outlook (Revenue, USD Million, 2018 - 2030)
- Pharma
- Biotech
- Biotechnology & Pharmaceutical Services Outsourcing Regional Outlook (Revenue, USD Million, 2018 - 2030)
- North America
- North America Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- North America Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- U.S.
- U.S. Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- U.S. Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- U.S. Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Canada
- Canada Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Canada Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Canada Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- North America Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Europe
- Europe Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Europe Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- UK
- UK Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- UK Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- UK Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Germany
- Germany Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Germany Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Germany Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- France
- France Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- France Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- France Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Italy
- Italy Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Italy Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Italy Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Spain
- Spain Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Spain Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Spain Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Denmark
- Denmark Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Denmark Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Denmark Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Sweden
- Sweden Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Sweden Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Sweden Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Norway
- Norway Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Norway Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Norway Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Europe Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Asia Pacific
- Asia Pacific Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Asia Pacific Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Japan
- Japan Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Japan Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Japan Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- China
- China Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- China Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- China Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- India
- India Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- India Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- India Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Australia
- Australia Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Australia Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Australia Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Thailand
- Thailand Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Thailand Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Thailand Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- South Korea
- South Korea Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- South Korea Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- South Korea Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Latin America
- Latin America Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Latin America Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Brazil
- Brazil Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Brazil Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Brazil Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Mexico
- Mexico Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Mexico Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Mexico Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Argentina
- Argentina Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Argentina Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Argentina Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Latin America Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- MEA
- MEA Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- MEA Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- South Africa
- South Africa Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- South Africa Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- South Africa Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Saudi Arabia
- Saudi Arabia Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Saudi Arabia Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- UAE
- UAE Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- UAE Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- UAE Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Kuwait
- Kuwait Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- Consulting
- Regulatory compliance
- Remediation
- Quality management systems consulting
- Others
- Auditing and assessment
- Regulatory affairs
- Clinical trial applications & product registration
- Regulatory writing & publishing
- Legal representation
- Others
- Product maintenance
- Product design & development
- Product testing & validation
- Training & education
- Others
- Consulting
- Kuwait Biotechnology and Pharmaceutical Services Outsourcing Market, By End Use
- Pharma
- Biotech
- Kuwait Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- MEA Biotechnology and Pharmaceutical Services Outsourcing Market, By Service
- North America
Biotechnology And Pharmaceutical Services Outsourcing Market Dynamics
Drivers: Increasing Outsourcing Of R&D Activities
Industry is witnessing outsourcing; many patents of many drugs are expected to expire soon, which is anticipated to drive the biosimilar market. This, in turn, is expected to boost R&D activities of companies for commercializing generic versions of drugs. Launching generics in the market in a feasible timeline and cost is anticipated to drive the demand for global Contract Research Organizations (CROs). In 2015, the top 30 sponsor companies had allied with at least one CRO. It has been observed that significant players have three alliances on average to maintain their product portfolios. A pharmaceutical company engages multiple CROs, each providing a unique service. Companies invest in R&D to gain a competitive advantage; pharmaceutical and biotechnology companies allocate a significant portion of their revenue to R&D activities.
Changing Regulatory Landscape
The regulatory approval process is becoming more stringent and time-consuming, and to gain a more significant market share, market players are striving to receive product approvals at the first attempt. Pharmaceutical and biotechnology companies must have an in-house regulatory department or outsource their regulatory affairs functions due to stringent regulatory requirements in developed countries and changing regulations in developing countries. Companies are outsourcing biotech/pharmaceutical services based on project size and priority due to the difficulty of establishing in-house regulatory affairs departments in offshore countries.
Pharmaceutical companies must manage continuous changes in regulatory requirements, spanning different business activities and multiple geographies. Noncompliance with changing regulatory requirements may result in penalties and delays, which may lead to loss of revenue. Medical device companies' geographic expansion aimed at speedy approvals in global markets is expected to further contribute to an increase in the adoption of outsourcing models for regulatory affairs and consulting services.
Restraints: Monitoring Issues And Lack Of Standardization
Managing and controlling outsourced services performed by Contract Research Organizations (CROs) is complex. There is a lack of standardization as not all CROs comply with international regulatory requirements, which may adversely affect the quality of services. Therefore, pharmaceutical and biotechnology companies are now outsourcing only lower-end functions and prefer performing high-end functions in-house. Standardization and authenticity of documented data are significant issues, as many small-scale companies need digital documentation capabilities. However, service providers are trying to comply with international standards of regulatory authorities such as the U.S. FDA to overcome this difficult challenge.
What Does This Report Include?
This section will provide insights into the contents included in this biotechnology and pharmaceutical services outsourcing market report and help gain clarity on the structure of the report to assist readers in navigating smoothly.
Biotechnology and pharmaceutical services outsourcing market qualitative analysis
-
Industry overview
-
Industry trends
-
Market drivers and restraints
-
Market size
-
Growth prospects
-
Porter’s analysis
-
PESTEL analysis
-
Key market opportunities prioritized
-
Competitive landscape
-
Company overview
-
Financial performance
-
Product benchmarking
-
Latest strategic developments
-
Biotechnology and pharmaceutical services outsourcing market quantitative analysis
-
Market size, estimates, and forecast from 2018 to 2030
-
Market estimates and forecast for product segments up to 2030
-
Regional market size and forecast for product segments up to 2030
-
Market estimates and forecast for application segments up to 2030
-
Regional market size and forecast for application segments up to 2030
-
Company financial performance
What questions do you have? Get quick response from our industry experts. Request a Free ConsultationResearch Methodology
A three-pronged approach was followed for deducing the biotechnology and pharmaceutical services outsourcing market estimates and forecasts. The process has three steps: information procurement, analysis, and validation. The whole process is cyclical, and steps repeat until the estimates are validated. The three steps are explained in detail below:
Information procurement: Information procurement is one of the most extensive and important stages in our research process, and quality data is critical for accurate analysis. We followed a multi-channel data collection process for biotechnology and pharmaceutical services outsourcing market to gather the most reliable and current information possible.
- We buy access to paid databases such as Hoover’s and Factiva for company financials, industry information, white papers, industry journals, SME journals, and more.
- We tap into Grand View’s proprietary database of data points and insights from active and archived monitoring and reporting.
- We conduct primary research with industry experts through questionnaires and one-on-one phone interviews.
- We pull from reliable secondary sources such as white papers and government statistics, published by organizations like WHO, NGOs, World Bank, etc., Key Opinion Leaders (KoL) publications, company filings, investor documents, and more.
- We purchase and review investor analyst reports, broker reports, academic commentary, government quotes, and wealth management publications for insightful third-party perspectives.
Analysis: We mine the data collected to establish baselines for forecasting, identify trends and opportunities, gain insight into consumer demographics and drivers, and so much more. We utilized different methods of biotechnology and pharmaceutical services outsourcing market data depending on the type of information we’re trying to uncover in our research.
-
Market Research Efforts: Bottom-up Approach for estimating and forecasting demand size and opportunity, top-down Approach for new product forecasting and penetration, and combined approach of both Bottom-up and Top-down for full coverage analysis.
-
Value-Chain-Based Sizing & Forecasting: Supply-side estimates for understanding potential revenue through competitive benchmarking, forecasting, and penetration modeling.
-
Demand-side estimates for identifying parent and ancillary markets, segment modeling, and heuristic forecasting.
-
Qualitative Functional Deployment (QFD) Modelling for market share assessment.
Market formulation and validation: We mine the data collected to establish baselines for forecasting, identify trends and opportunities, gain insight into consumer demographics and drivers, and so much more. We utilize different methods of data analysis depending on the type of information we’re trying to uncover in our research.
-
Market Formulation: This step involves the finalization of market numbers. This step on an internal level is designed to manage outputs from the Data Analysis step.
-
Data Normalization: The final market estimates and forecasts are then aligned and sent to industry experts, in-panel quality control managers for validation.
-
This step also entails the finalization of the report scope and data representation pattern.
-
Validation: The process entails multiple levels of validation. All these steps run in parallel, and the study is forwarded for publishing only if all three levels render validated results.
Biotechnology And Pharmaceutical Services Outsourcing Market Categorization:
The biotechnology and pharmaceutical services outsourcing market was categorized into three segments, namely service (Consulting, Auditing & Assessment, Regulatory Affairs, Product Maintenance, Product Design & Development, Product Testing & Validation, Training & Education), end-use (Pharma, Biotech), regions (North America, Europe, Asia Pacific, Latin America, Middle East & Africa).
Segment Market Methodology:
The biotechnology and pharmaceutical services outsourcing market was segmented into service, end-use, and regions. The demand at a segment level was deduced using a funnel method. Concepts like the TAM, SAM, SOM, etc., were put into practice to understand the demand. We at GVR deploy three methods to deduce market estimates and determine forecasts. These methods are explained below:
Market research approaches: Bottom-up
-
Demand estimation of each product across countries/regions summed up to from the total market.
-
Variable analysis for demand forecast.
-
Demand estimation via analyzing paid database, and company financials either via annual reports or paid database.
-
Primary interviews for data revalidation and insight collection.
Market research approaches: Top-down
-
Used extensively for new product forecasting or analyzing penetration levels.
-
Tool used invoice product flow and penetration models Use of regression multi-variant analysis for forecasting Involves extensive use of paid and public databases.
-
Primary interviews and vendor-based primary research for variable impact analysis.
Market research approaches: Combined
- This is the most common method. We apply concepts from both the top-down and bottom-up approaches to arrive at a viable conclusion.
Regional Market Methodology:
The biotechnology and pharmaceutical services outsourcing market was analyzed at a regional level. The globe was divided into North America, Europe, Asia Pacific, Latin America, Middle East & Africa, keeping in focus variables like consumption patterns, export-import regulations, consumer expectations, etc. These regions were further divided into twenty-three countries, namely, the U.S.; Canada; the UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Mexico; Argentina; South Africa; Saudi Arabia; UAE; Kuwait.
All three above-mentioned market research methodologies were applied to arrive at regional-level conclusions. The regions were then summed up to form the global market.
Smart home market companies & financials:
The biotechnology and pharmaceutical services outsourcing market was analyzed via companies operating in the sector. Analyzing these companies and cross-referencing them to the demand equation helped us validate our assumptions and conclusions. Key market players analyzed include:
-
THE QUANTIC GROUP - The Quantic Group provides industry and government biotechnology and pharmaceutical consulting services. It operates through various segments, such as regulatory affairs, research, development, manufacturing, and quality operations. Quantic has supportive clients from all over the globe that mainly operate in regulatory affairs. It also provides services in other segments such as marketing & manufacturing strategies, clinical trial practices, research & development, product, manufacturing & quality systems methodologies, and others.
-
IQVIA HOLDINGS, INC. - IQVIA Holdings, Inc. is one of the leading companies that provides contract research services, technology solutions, and advanced analytics to the global life sciences industry. The company operates through three main business areas- R&D solutions, commercialization solutions, and technology solutions. It serves biotechnology, consumer health, pharmaceuticals, and device & diagnostics companies in North America, Europe, Asia Pacific, and Africa. IQVIA also serves pharmacies, pharmaceutical distributors, payers, and government & regulatory agencies.
-
PAREXEL INTERNATIONAL CORPORATION - Parexel International Corporation is a healthcare company that provides biopharmaceutical and pharmaceutical services worldwide. It provides biopharmaceutical services to various clients across the globe and also offers new treatment options for various rare disorders. It serves biotechnology, consumer health companies, pharmaceutical, and device & diagnostic companies in North America, Europe, Asia Pacific, and Africa. It operates through different segments, such as clinical trials, regulatory, consulting, and market access. It provides services to more than 100 countries. They also outsource their services to different pharmacies, pharmaceutical distributors, payers, & regulatory agencies.
-
LACHMAN CONSULTANT SERVICES - Lachman Consultant Services provides technical and regulatory affairs services to clients worldwide. It also consults other organizations to solve their problems and develop effective strategies for approval of new drugs and devices. It also provides consulting services to global clients such as pharmaceutical, biotechnology, medical devices, and dietary supplement companies. It provides services in North America, Europe, and Asia Pacific regions.
-
GMP PHARMACEUTICALS - GMP Pharmaceuticals is one of the leading manufacturers of medicines, natural health products, and infant formulas. It serves health food companies across the globe. For example, it has around 20 years of experience providing worldwide nutritional and health food services. GMP Pharmaceuticals manufactures various pharmaceutical products such as coated & uncoated tablets, liquids, capsules, creams, ointments, and powders. It is the only contract manufacturer in Australia and New Zealand.
-
CONCEPT HEIDELBERG GMBH - Concept Heidelberg GmbH is one of the leading organizations in Europe that provides training and information services in pharmaceutical quality assurance and drug quality. It organizes more than 350 seminars and conferences at least in 10 European countries every year. It provides GMP/GDP services to various pharmaceutical & biopharmaceutical industries, such as in-house training, e-learning, and consulting services.
-
COVANCE, INC. - Covance, Inc. is a contract research organization that provides clinical and commercialization services to the biopharmaceutical and pharmaceutical industries. LabCorp owns the company. It provides its services over 60 countries. Covance is the world's most wide-ranging drug development company that delivers solutions to pharmaceutical industries. It also offers laboratory testing services to chemical industries and is a market leader in discovery, toxicology, & central laboratory services. It is one of the top companies that provides management services in phase III clinical trials to pharmaceutical industries.
-
CHARLES RIVER - Charles River Laboratory provides clinical laboratory and preclinical services for the pharmaceutical and biotechnology industries. Charles operates through three segments: Research Models and Services (RMS), Discovery and Safety Assessment (DSA), and Manufacturing Support. Its DSA segment offerings include bioanalysis, drug metabolism, toxicology, and pharmacokinetics services. It caters its products and services to biotechnology and pharmaceutical companies, medical device companies, hospitals, clinics, and academic institutions. The company provides its products in about 20 countries and has a presence in Canada, Finland, Belgium, France, Hungary, Germany, Italy, China, Italy, Japan, Spain, the Netherlands, and the UK.
-
PRA HEALTH SCIENCES - PRA Health Sciences provides outsourced clinical development and data solution services to biopharmaceutical & pharmaceutical industries globally. It operates in different therapeutic areas, such as immunology, inflammation, and infectious diseases. The company provides services to various geographic regions such as North America, Europe, Asia, Latin America, South Africa, Australia, and the Middle East.
-
ICON PLC - ICON provides outsourced development services to the pharmaceutical, biotechnology, and medical device industries globally. The company specializes in the strategic development, management, and analysis of programs that support Clinical Development from compound selection to Phase I-IV clinical studies. The company offers a complete range of services, including consulting, development, and commercialization, from a global network of offices in 37 countries.
Value chain-based sizing & forecasting
Supply Side Estimates
-
Company revenue estimation via referring to annual reports, investor presentations, and Hoover’s.
-
Segment revenue determination via variable analysis and penetration modeling.
-
Competitive benchmarking to identify market leaders and their collective revenue shares.
-
Forecasting via analyzing commercialization rates, pipelines, market initiatives, distribution networks, etc.
Demand side estimates
-
Identifying parent markets and ancillary markets
-
Segment penetration analysis to obtain pertinent
-
revenue/volume
-
Heuristic forecasting with the help of subject matter experts
-
Forecasting via variable analysis
Biotechnology And Pharmaceutical Services Outsourcing Market Report Objectives:
-
Understanding market dynamics (in terms of drivers, restraints, & opportunities) in the countries.
-
Understanding trends & variables in the individual countries & their impact on growth and using analytical tools to provide high-level insights into the market dynamics and the associated growth pattern.
-
Understanding market estimates and forecasts (with the base year as 2022, historic information from 2018 to 2021, and forecast from 2023 to 2030). Regional estimates & forecasts for each category are available and are summed up to form the global market estimates.
Biotechnology And Pharmaceutical Services Outsourcing Market Report Assumptions:
-
The report provides market value for the base year 2022 and a yearly forecast till 2030 in terms of revenue/volume or both. The market for each of the segment outlooks has been provided on region & country basis for the above-mentioned forecast period.
-
The key industry dynamics, major technological trends, and application markets are evaluated to understand their impact on the demand for the forecast period. The growth rates were estimated using correlation, regression, and time-series analysis.
-
We have used the bottom-up approach for market sizing, analyzing key regional markets, dynamics, & trends for various products and end-users. The total market has been estimated by integrating the country markets.
-
All market estimates and forecasts have been validated through primary interviews with the key industry participants.
-
Inflation has not been accounted for to estimate and forecast the market.
-
Numbers may not add up due to rounding off.
-
Europe consists of EU-8, Central & Eastern Europe, along with the Commonwealth of Independent States (CIS).
-
Asia Pacific includes South Asia, East Asia, Southeast Asia, and Oceania (Australia & New Zealand).
-
Latin America includes Central American countries and the South American continent
-
Middle East includes Western Asia (as assigned by the UN Statistics Division) and the African continent.
Primary Research
GVR strives to procure the latest and unique information for reports directly from industry experts, which gives it a competitive edge. Quality is of utmost importance to us, therefore every year we focus on increasing our experts’ panel. Primary interviews are one of the critical steps in identifying recent market trends and scenarios. This process enables us to justify and validate our market estimates and forecasts to our clients. With more than 8,000 reports in our database, we have connected with some key opinion leaders across various domains, including healthcare, technology, consumer goods, and the chemical sector. Our process starts with identifying the right platform for a particular type of report, i.e., emails, LinkedIn, seminars, or telephonic conversation, as every report is unique and requires a differentiated approach.
We send out questionnaires to different experts from various regions/ countries, which is dependent on the following factors:
-
Report/Market scope: If the market study is global, we send questionnaires to industry experts across various regions, including North America, Europe, Asia Pacific, Latin America, and MEA.
-
Market Penetration: If the market is driven by technological advancements, population density, disease prevalence, or other factors, we identify experts and send out questionnaires based on region or country dominance.
The time to start receiving responses from industry experts varies based on how niche or well-penetrated the market is. Our reports include a detailed chapter on the KoL opinion section, which helps our clients understand the perspective of experts already in the market space.
What questions do you have? Get quick response from our industry experts. Request a Free ConsultationShare this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





