- Home
- »
- Clinical Diagnostics
- »
-
Blood-based Biomarker For Parkinson’s Disease Market Report, 2033GVR Report cover
![Blood-based Biomarker For Parkinson’s Disease Market Size, Share & Trends Report]()
Blood-based Biomarker For Parkinson’s Disease Market (2025 - 2033) Size, Share & Trends Analysis Report By Biomarker (Protein Biomarkers, Inflammatory Markers), By Technology (ELISA/Immunoassay, Multiplex platforms), By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-667-9
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Blood-based Biomarker For Parkinson’s Disease Market Summary
The global blood-based biomarker for Parkinson’s disease market size was estimated at USD 117.79 million in 2024 and is projected to reach USD 350.87 million by 2033, growing at a CAGR of 19.25% from 2025 to 2033. The market is exhibiting significant growth driven by rising prevalence of Parkinson’s disease worldwide, increasing investment in neurology diagnostics and biomarker research and advances in proteomics and genomic technologies.
Key Market Trends & Insights
- North America blood-based biomarker for Parkinson’s disease market held the largest share of 43.32% of the global market in 2024.
- The blood-based biomarker for Parkinson’s disease market in the U.S. is expected to grow significantly over the forecast period.
- By biomarker, the protein biomarkers segment held the largest market share of 55.10% in 2024.
- Based on technology, the ELISA/Immunoassay segment held the highest market share in 2024.
- By end use, academic/research labs segment held the highest market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 117.79 Million
- 2033 Projected Market Size: USD 350.87 Million
- CAGR (2025-2033): 19.25%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
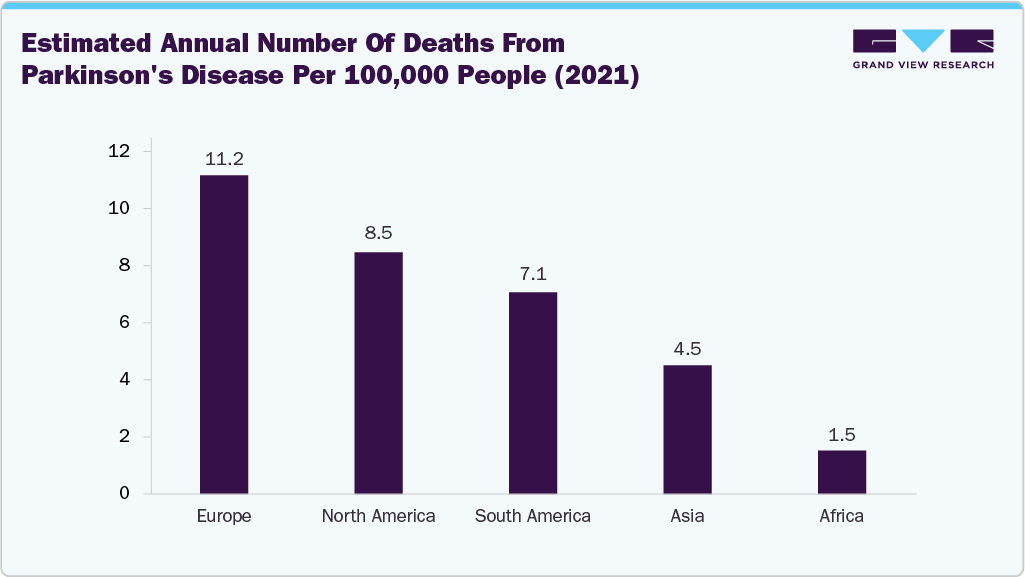
The data from the global burden of disease study 2021, researchers project that by 2050, Parkinson’s disease will affect approximately 25.2 million people worldwide across all ages and sexes, an increase of 112% compared to the numbers recorded in 2021. The global incidence of Parkinson’s disease (PD) is increasing due to geriatric populations, particularly in North America, Europe, and parts of Asia-Pacific. Parkinson’s disease (PD) incidence rates increase significantly with age, particularly among individuals aged 65 and older, making age the primary risk factor for the condition. Some studies consistently show that PD is more prevalent in males than females across all age groups. The rising incidence of PD closely correlates with the expanding aging population worldwide, suggesting that demographic shifts will continue to drive up the number of cases in the coming decades. As life expectancy increases, the burden of Parkinson’s disease is expected to grow. The graph below depicts cases of Parkinson’s disease in various continents according to the IHME Global Burden of Disease (2024):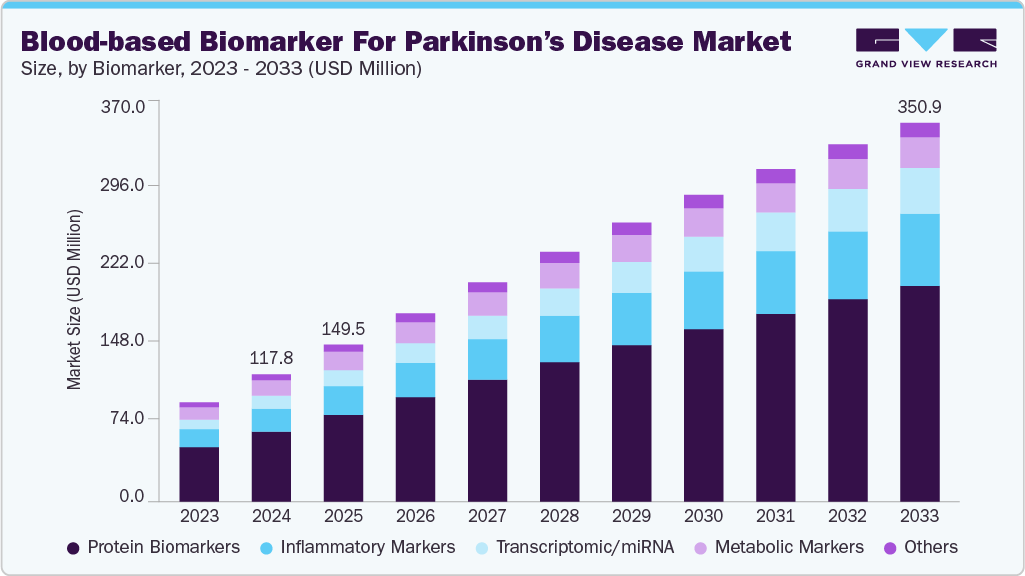
This growing patient pool drives urgent demand for early, accurate and minimally invasive diagnostic tools. Blood-based biomarkers offer a promising alternative to traditional clinical and imaging-based diagnosis, which often detects PD only in later stages. As awareness of neurodegenerative diseases increases globally, so does the need for accessible diagnostics, positioning blood-based biomarkers as a critical solution to meet the rising burden of Parkinson’s and support earlier therapeutic intervention.
Governments, biotech firms, and non-profit foundations are significantly increasing fundingfor Parkinson’s biomarker research. Initiatives like the Michael J. Fox Foundation’s Parkinson’s Progression Markers Initiative (PPMI) and EU-funded projects such as PD-MitoQUANT have helped establish large biobanks and longitudinal cohorts, enabling biomarker validation on a global scale. For instance, in May 2024, the Michael J. Fox Foundation for Parkinson's Research (MJFF) announced a total of USD 9.75 million in funding awarded to five research teams focused on developing biological tools to measure and monitor the progression of Parkinson’s disease (PD). Such teams work on creating quantitative biomarkers biological indicators that can accurately track PD from its earliest stages to more advanced progression. The goal is to have these biomarkers ready for clinical drug trials within the next two to three years, ultimately accelerating the development of effective therapies and improving disease management strategies. Pharmaceutical companies also invest in companion diagnostics to align with emerging blood-based therapies. This funding surge supports translational research, encourages public-private partnerships, and accelerates clinical trials, creating a robust ecosystem for blood-based biomarker development, regulatory approval and eventual commercialization in the global Parkinson’s diagnostics market.
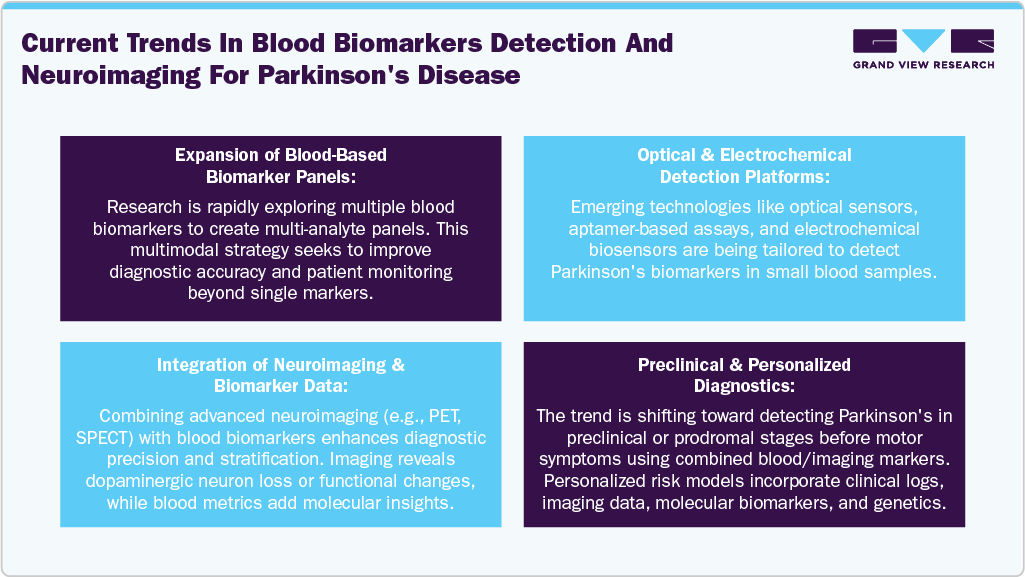
Technological advancements in proteomics and genomics are key drivers for the development of blood-based biomarkers for Parkinson’s disease. Some of the techniques, such as next-generation sequencing and mass spectrometry, enable the identification of disease-specific genetic variations, protein expression patterns and molecular signatures in blood samples. These technologies allow for early, non-invasive detection and monitoring of Parkinson’s disease, improving diagnostic accuracy and personalized treatment strategies. Integration of omics data enhances our understanding of disease mechanisms, facilitating the discovery of reliable biomarkers. As these tools become more precise and accessible, they significantly accelerate biomarker discovery and validation in Parkinson’s research and clinical applications. Some of the current trends in blood-based biomarkers are highlighted below:
Market Concentration & Characteristics
The market exhibits a high degree of innovation driven by rapid advances in proteomics, genomics, and AI-based data analysis. Continuous R&D is leading to the identification of novel blood-based biomarkers, enabling early, non-invasive Parkinson’s diagnosis and personalized treatment, positioning innovation as a critical growth accelerator. For instance, in June 2024, scientists from UCL and University Medical Center Göttingen developed an AI-driven blood test capable of detecting Parkinson’s disease up to seven years before visible symptoms emerge, enabling earlier diagnosis and intervention.
Mergers and acquisitions are increasingly common as biotech firms and diagnostic companies seek to consolidate technologies and expand biomarker pipelines. For example, in November 2024, Alamar Biosciences partnered with The Michael J. Fox Foundation to advance Parkinson’s disease research by using its precision proteomics platform to develop protein assays targeting biomarkers linked to synuclein-related disorders. These strategic collaborations help accelerate clinical validation, regulatory approvals, and market entry.

The market is heavily influenced by regulatory frameworks set by agencies like the FDA and EMA. Biomarker-based diagnostics must meet stringent clinical validation, safety, and efficacy standards. While regulations ensure reliability, they also extend product development timelines and costs, posing challenges for smaller players and startups.
Imaging techniques (like PET and MRI scans) and CSF-based biomarker tests serve as substitutes but are more invasive, expensive, and less scalable. Blood-based biomarkers offer a non-invasive, cost-effective alternative, although clinical adoption depends on diagnostic accuracy, regulatory approval and integration into existing diagnostic workflows.
The market is moderately concentrated among neurologists, research institutions and specialized diagnostic labs. Academic medical centers and biotech companies are the primary adopters. As awareness grows, diagnostic usage may expand to broader healthcare settings but current adoption remains largely focused within expert-driven environments.
Biomarker Insights
Protein biomarkers represent the largest segment in the market for Parkinson’s disease, with a market share of 55.10% in 2024, owing to their high specificity, stability, and relevance to disease mechanisms. These biomarkers are crucial for early diagnosis, monitoring disease progression, and evaluating treatment response. Advances in proteomics technologies, including mass spectrometry and immunoassays, have enhanced the detection and quantification of low-abundance proteins in blood samples. Their non-invasive nature and potential to reflect neurodegenerative changes make protein biomarkers highly valuable. For example, in September 2023, AXIM Biotechnologies has developed the first rapid, non-invasive point-of-care test for detecting abnormal alpha-synuclein, using a tear sample instead of a costly and invasive spinal tap procedure.
Transcriptomic/miRNA biomarkers are anticipated to register the fastest CAGR, over the forecast period, especially circulating microRNAs (miRNAs) that are driving rapid advances in blood-based Parkinson’s diagnostics. These small, non-coding RNAs are dysregulated in PD patients and are detectable in minimally invasive blood samples. They are remarkably stable in biofluids and can reflect brain changes, making them ideal for early detection and disease monitoring. Exosomal miRNAs further enhance signal fidelity by crossing the blood-brain barrier. The combination of transcriptomic profiling and advanced biosensors positions miRNA panels as the fastest-growing blood biomarkers in PD research.
Technology Insights
Enzyme-linked immunosorbent assays (ELISA) dominated the market for Parkinson’s disease with a revenue share of 44.87% in 2024, comprising the largest technology segment. ELISAs offer exceptional specificity, sensitivity, and high-throughput capacity, making them the gold standard for confirming protein biomarkers like α‑synuclein and phosphorylated variants (pS129). Innovations such as chemiluminescent or electrochemiluminescent ELISAs further enhance detection limits into picogram/mL or even femtogram/mL ranges, enabling earlier and more accurate PD diagnosis. Their robust performance, versatility across sample types, and suitability for clinical and research settings underpin their continued growth and widespread adoption in Parkinson’s biomarker development.
Multiplex platforms are the anticipated to witness the fastest CAGR over the forecast period, offering unparalleled efficiency and diagnostic depth. These systems, such as multiplex mass spectrometry assays, can simultaneously analyze multiple proteins, metabolites, or nucleic acids, enabling early detection of PD up to seven years before symptom onset. By measuring panels of eight proteins or more, these platforms enhance sensitivity and specificity, capturing complex, multi-pathway disease signatures. Their high-throughput nature accelerates biomarker discovery and supports precision medicine. In addition, advances in machine learning and microfluidics streamline data interpretation and facilitate scalable, cost-effective deployment in clinical and point-of-care settings.
End Use Insights
Academic and research laboratories held the largest share of 43.30% in 2024, driven by robust R&D investment and cutting-edge discovery tools. These institutions, including universities and public research centers, pioneer biomarker validation and translational studies, securing a growing share of funding. They leverage next‑generation platforms such as Simoa, mass spectrometry, and advanced immunoassays to identify and assess novel biomarkers for early PD detection. Their high-throughput analytical capabilities and collaborative networks expedite preclinical validation and standardizationpositioning academic labs at the forefront of the PD biomarker pipeline. This surge in activity establishes them as a critical growth engine in the neurological biomarker landscape.
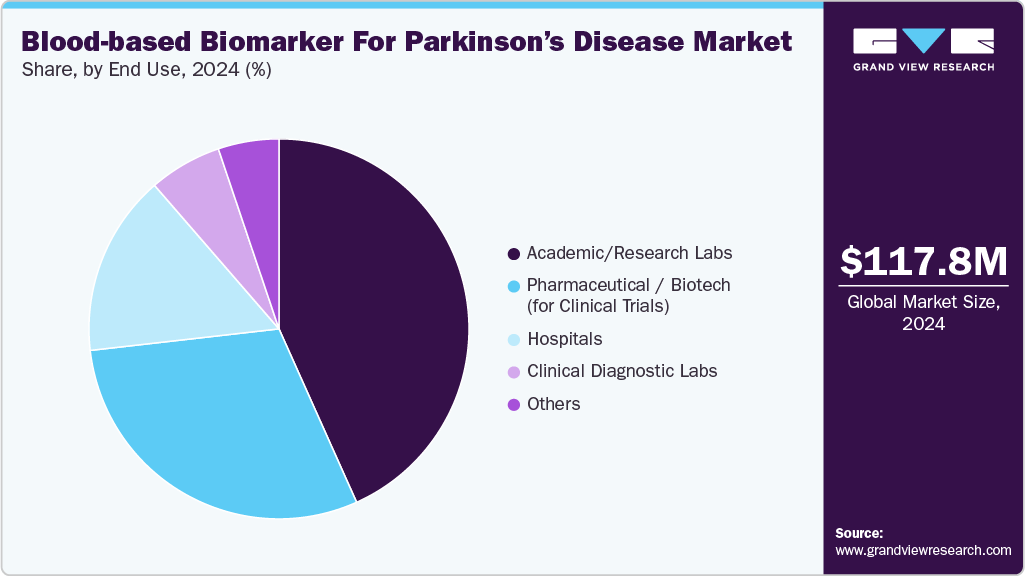
Pharmaceutical and biotech firms are anticipated to be the fastest-growing segment over the forecast period. These companies are increasingly investing in diagnostic tools to support the development of personalized, disease-modifying therapies, aiming to identify early-stage or prodromal PD populations. They are collaborating with academic institutions and research consortia, such as Berg Pharma’s work with the Parkinson’s Institute and the PPMI to advance biomarker discovery and validation. The growing regulatory support and rising R&D funding, pharma and biotech players are accelerating translational pipelines, integrating multi-omics platforms and digital diagnostics to bolster drug trials and improve therapeutic efficacy measurement.
Regional Insights
North America blood-based biomarkers for Parkinson’s disease industry dominated globally with the largest revenue share of 43.32% in 2024, owing to robust R&D funding, advanced healthcare infrastructure, and strong presence of pharma and biotech firms competing in ultra-sensitive, multiplex assays. For instance, in August 2024, the Parkinson’s Foundation has committed nearly USD 3 million in funding through 33 research grants aimed at advancing innovative and high-impact studies focused on accelerating progress in Parkinson’s disease (PD) research.
U.S. Blood-based Biomarker For Parkinson’s Disease Market Trends
The blood-based biomarkers for Parkinson’s disease industry in the U.S. is evolving beyond traditional protein targets to encompass inflammatory and immune biomarkers, reflecting the growing recognition of PD as an inflammatory neurodegenerative disorder. Emerging evidence suggests that chronic neuroinflammation mediated by activated microglia, cytokines, and CD4+ T cell subsets plays a central role in PD pathogenesis even before clinical symptoms emerge. Academic institutions such as Massachusetts General Hospital and Mayo Clinic are investigating inflammatory signatures in PD patients undergoing surgical procedures, noting that anesthesia and perioperative stress may temporarily amplify neuroinflammatory markers, potentially accelerating disease progression.
Europe Blood-based Biomarker For Parkinson’s Disease Market Trends
Europe blood-based biomarkers for Parkinson’s disease industry is experiencing robust growth. Some of the players like Roche, Quanterix, Thermo Fisher and BioMérieux are intensifying competition in ultra‑sensitive blood assays, supported by strong regulatory frameworks and collaborative research initiatives. In February 2025, Mair Therapeutics announced launch and secures pre-seed funding to develop innovative therapies for Parkinson’s disease, marking a significant step toward advancing novel treatment solutions for neurodegenerative disorders.
The UK blood-based biomarkers for Parkinson’s disease industry is advancing with competition because of NHS-backed R&D funding and growing demand for early diagnostics drive innovation. In December 2023, Researchers at Oxford's Nuffield Department of Clinical Neurosciences developed a groundbreaking blood-based test capable of detecting the underlying pathology of Parkinson’s disease before the onset of major symptoms. Some of the players like Roche, Quanterix, and Oxford Nuffield are advancing ultra‑sensitive multiplex assays for pre‑symptomatic detection.
Asia Pacific Blood-based Biomarker For Parkinson’s Disease Market Trends
Asia‑Pacific blood-based biomarkers for Parkinson’s disease industry is anticipated to be the fastest-growing region over the forecast period, fueled by rapid healthcare investment, expanding clinical trial activity, and rising neurological disorder prevalence. Key countries such as China, India, and Japan have fiercely competingcompanies like Roche, Thermo Fisher, and Abbott, and regional biotech innovators are intensifying R&D in cost-effective, sensitive multiplex assays.
The blood-based biomarkers for Parkinson’s disease industry in India is gaining momentumdriven by growing PD prevalence and demand for non-invasive early diagnostics. India, through DBT and ICMR, actively promotes Parkinson’s research via genomics, epidemiology, and traditional medicine. Funding supports public-private collaboration and international partnerships like IUSSTF to advance neuroscience innovation.
Latin America Blood-based Biomarker For Parkinson’s Disease Market Trends
The Latin America blood-based biomarkers for Parkinson’s disease industry is expanding, driven by rising neurological disease awareness, improved healthcare infrastructure, and public-private R&D initiatives. Institutions like Brazil’s Fiocruz and Argentina’s CONICET, alongside Thermo Fisher and Abbott, are advancing proteomics and genomics multiplex assays.
The blood-based biomarkers for Parkinson’s disease industry in Brazil is witnessing significant advancements with rising demand for non-invasive diagnostics and omics advancements. In October 2024, ADx NeuroSciences partnered with Alamar Biosciences to develop custom biomarker assays using NULISA technology and ARGO HT System, to enable precise detection and measurement of key biomarkers that support the development of therapies for neurological diseases.
Middle East & Africa Blood-based Biomarker For Parkinson’s Disease Market Trends
The blood-based biomarkers for Parkinson’s disease industry in the Middle East & Africa is witnessing increasing competition fueled by rising demand for noninvasive diagnostics, healthcare investment, and AI integration. In April 2024, researchers at the Hebrew University of Jerusalem innovated a RNA-based blood test to detect Parkinson’s early, targeting two biomarkers such as an accumulating RNA repeat and decreasing mitochondrial RNA linked to disease progression.
South Africa blood-based biomarkers for Parkinson’s disease industry growth is driven by rising neurological research investment and growing PD awareness. Institutions like the University of Cape Town and Fiocruz collaborate on plasma alpha-synuclein assays and mass spectrometry metabolomics. Emerging tools such as ultra-sensitive Simoa and immunoassays are under trial, supported by partnerships with Newcastle University via ESP Diagnostics.
Key Blood-Based Biomarkers For Parkinson’s Disease Company Insights
The blood-based biomarker for Parkinson’s industry is dominated by QIAGEN, Quanterix, Thermo Fisher, Bio-Rad Laboratories and Abbott, with emerging players expanding through innovative platforms and collaborations. Their combined efforts are accelerating early diagnostics and clinical adoption.
Key Blood-Based Biomarkers For Parkinson’s Disease Companies:
The following are the leading companies in the blood-based biomarker for Parkinson’s disease market. These companies collectively hold the largest market share and dictate industry trends.
- Abbott
- ACOBIOM
- Alseres Pharmaceuticals
- Banyan Biomarkers
- Bio-Rad Laboratories
- DiaGenic ASA
- Johnson & Johnson Services
- Merck KGaA
- QIAGEN
- Quanterix
- Rules-Based Medicine
- Thermo Fisher Scientific
Recent Developments
-
In May 2025, Rune Labs and the Parkinson’s Foundation launched the first clinical initiative aimed at linking genetic biomarkers with digital health data to enhance understanding and management of Parkinson’s disease.
-
In April 2025, FYR and Mayo Clinic have partnered to enhance Parkinson’s research by using blood-based biomarkers, aiming to better understand disease progression and subtypes to improve patient care and clinical trial success.
-
In January 2025, C2N Diagnostics, supported by a $1.5 million grant from The Michael J. Fox Foundation, will link fluid samples with novel biomarker tests to advance understanding of Parkinson’s disease mechanisms.
Blood-based Biomarker For Parkinson’s Disease Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 145.49 million
Revenue forecast in 2033
USD 350.87 million
Growth Rate
CAGR of 19.25% from 2025 to 2033
Historical Period
2021 - 2023
Actual data
2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Technology, biomarker, end use, and region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Abbott Laboratories; ACOBIOM; Alseres Pharmaceuticals; Banyan Biomarkers; Bio-Rad Laboratories; DiaGenic ASA; Johnson & Johnson; Merck KGaA; QIAGEN; Quanterix; Thermo Fisher Scientific.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Blood-based Biomarker For Parkinson’s Disease Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the blood-based biomarker for Parkinson’s disease market report based on technology, biomarker, end use, and region:
-
Biomarker Outlook (Revenue, USD Million, 2021-2033)
-
Protein biomarkers
-
Inflammatory markers
-
Metabolic markers
-
Transcriptomic/miRNA
-
Others
-
-
Technology Outlook (Revenue, USD Million, 2021-2033)
-
ELISA/Immunoassay
-
Multiplex platforms
-
NGS/qPCR (genetic)
-
Mass spectrometry
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021-2033)
-
Academic/Research Labs
-
Pharmaceutical / Biotech (for Clinical Trials)
-
Hospitals
-
Clinical Diagnostic Labs
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
India
-
China
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global blood-based biomarker for Parkinson’s disease market is expected to grow at a compound annual growth rate of 19.25% from 2025 to 2033 to reach USD 350.87 million by 2033.
b. Protein biomarkers segment dominates the market with a revenue share of 55.10% in 2024 by biomarker type, driven by their strong biological relevance, diagnostic utility, and clinical validation. Proteins like α-synuclein, neurofilament light chain (NfL), and DJ-1 are directly associated with Parkinson’s disease pathology, making them highly sought after for early detection and monitoring. Technological advancements in ELISA, mass spectrometry, and multiplex immunoassays have improved sensitivity and specificity for detecting these markers in blood. Additionally, pharmaceutical companies increasingly use protein biomarkers in clinical trials for patient stratification and therapeutic monitoring, accelerating their integration into research and clinical workflows.
b. Some key players operating in the blood-based biomarker for Parkinson’s disease market include Amprion, C2N Diagnostics, Olink Proteomics, Denali Therapeutics, Biogen, Roche / Genentech. Verily Life Sciences, AC Immune, SomaLogic, Quanterix
b. The blood-based biomarker for Parkinson’s disease market is driven by a growing patient population, with 90,000 new diagnoses annually, and increasing demand for early, non-invasive diagnostic tools. Expanding research efforts, especially in academic and pharmaceutical settings, are accelerating biomarker discovery and validation. Technological advancements in multiplex assays and AI-based platforms are further supporting adoption. Additionally, collaborations involving the NIH, MJFF, and Verily are helping standardize biomarker use. As assays like α-synuclein and NfL progress toward regulatory approval, their integration into clinical practice is becoming increasingly viable.
b. The global blood-based biomarker for Parkinson’s disease market size was estimated at USD 117.79 million in 2024 and is expected to reach USD 145.49 million in 2025.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










