- Home
- »
- Medical Devices
- »
-
Breast Biopsy Devices Market Size, Industry Report, 2033GVR Report cover
![Breast Biopsy Devices Market Size, Share & Trends Report]()
Breast Biopsy Devices Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Biopsy Guidance Systems, Biopsy Drivers & Handpieces), By Procedure, By Guidance Technology, By End Use (Hospitals, Ambulatory Surgery Centers), By Region, And Segment Forecasts
- Report ID: 978-1-68038-981-4
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Breast Biopsy Devices Market Summary
The global breast biopsy devices market size was estimated at USD 3.52 billion in 2025 and is projected to reach USD 5.60 billion by 2033, growing at a CAGR of 6.04% from 2026 to 2033. The market is primarily driven by the rising global incidence of breast cancer and increasing demand for early and accurate diagnosis.
Key Market Trends & Insights
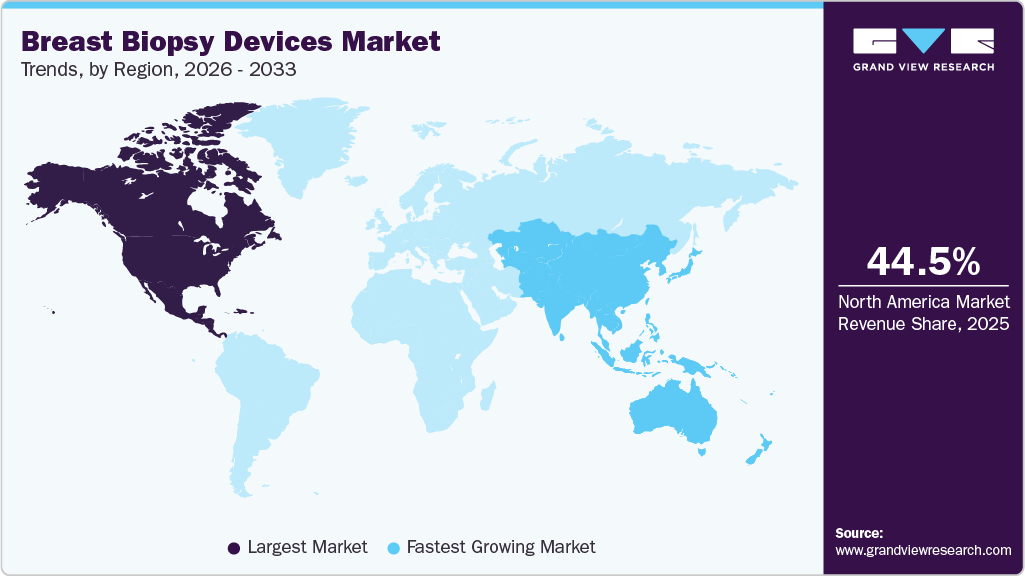
- North America dominated the breast biopsy devices industry with the largest revenue share of 44.50% in 2025.
- The breast biopsy devices industry in the U.S. accounted for the largest market revenue share in North America in 2025.
- By product, the biopsy consumables segment led the market with the largest revenue share in 2025.
- By procedure, the core needle biopsy (CNB) segment led the market with the largest revenue share in 2025.
- By guidance technology, the ultrasound-guided segment led the market with the largest revenue share in 2025.
- By end use, the hospitals segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 3.52 Billion
- 2033 Projected Market Size: USD 5.60 Billion
- CAGR (2026-2033): 6.04%
- North America region: Largest market in 2025
- Asia Pacific: Fastest growing market
The expansion of national screening programs and greater awareness of routine mammography are leading to more abnormalities being detected, requiring biopsy confirmation. Technological advancements in minimally invasive and image-guided biopsy techniques are improving diagnostic precision and patient comfort.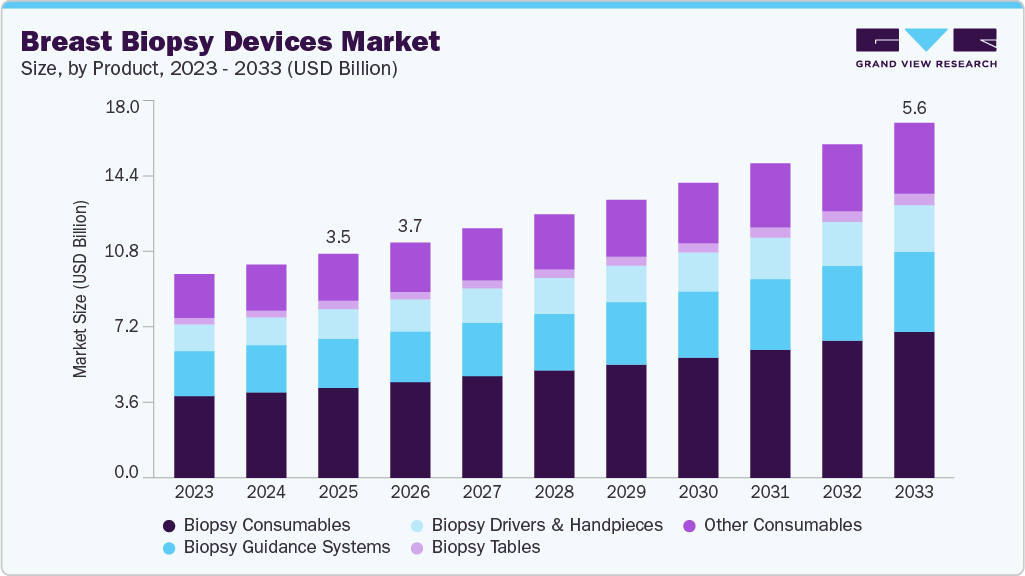
In addition, the expansion of healthcare infrastructure and outpatient care facilities is driving higher procedural volumes worldwide. According to Breastcancer.org, in 2026, it is estimated that about 42,140 women in the U.S. will die from breast cancer, highlighting the continuing mortality burden of the disease despite advances in early detection and treatment.
The increasing global burden of breast cancer is a major driver of the breast biopsy devices industry. As the number of newly diagnosed cases continues to rise worldwide, the demand for accurate and timely diagnostic procedures has grown significantly. Breast biopsy devices play a critical role in confirming malignancy in cases with abnormal imaging findings. Consequently, higher incidence rates directly contribute to increased procedure volumes, driving sustained growth in the breast biopsy devices industry. According to Breastcancer.org, about 1 in 8 women in the U.S. (approximately 13%) will be diagnosed with invasive breast cancer during their lifetime. Each year, breast cancer represents roughly 30% of all newly diagnosed cancers among U.S. women, making it one of the most common cancers affecting women. These statistics highlight the significant impact and ongoing burden of breast cancer nationwide. The table below shows key breast cancer statistics.
U.S. Breast Cancer Statistics (2026)
Category
Statistic
Most Common Cancer
Most common cancer diagnosed among women in the U.S.
Share of New Cancer Cases
~32% of all newly diagnosed cancers in women
New Invasive Cases (2026)
321,910 women
New DCIS Cases (2026)
60,730 women
Patients Younger Than 50
~16% of breast cancer cases
Localized Stage Diagnosis
~66% diagnosed before spreading outside the breast
Women Living with History of Breast Cancer
Over 4 million women in the U.S.
Estimated Deaths (2026)
42,140 women
Male Breast Cancer Cases
<1% of all breast cancer cases
Source: BreastCancer.Org
(Note: DCI - Ductal Carcinoma In-situ)
Breast cancer risk increases with age, with most cases diagnosed in women aged 50 years and older. Incidence and mortality rates are significantly higher in older age groups compared to younger women. The table below shows breast cancer cases by age.
Breast Cancer Cases and Deaths by Age Group (2024)
Age Group
DCIS Cases (Number)
DCIS (%)
Invasive Cases (Number)
Invasive (%)
Deaths (Number)
Deaths (%)
<40
1,360
2%
13,180
4%
990
2%
40-49
8,750
15%
37,650
12%
2,620
6%
50-59
13,760
24%
67,310
22%
6,800
16%
60-69
17,660
31%
89,540
29%
10,010
24%
70-79
11,890
21%
69,130
22%
10,140
24%
80+
3,080
5%
33,910
11%
11,690
28%
All
56,500
98%
310,720
100%
42,250
100%
Source: American Cancer Society
The growing preference for minimally invasive procedures is a significant driver for the breast biopsy market. Techniques such as core needle biopsy and vacuum-assisted biopsy are favored over traditional surgical biopsies due to reduced pain, minimal scarring, shorter recovery time, and lower overall procedure costs. These approaches can often be performed in outpatient settings under local anesthesia, improving patient comfort and workflow efficiency. As both patients and healthcare providers prioritize safer and less invasive diagnostic options, demand for advanced minimally invasive breast biopsy devices continues to rise.
Furthermore, advancements in imaging guidance, such as ultrasound, stereotactic, and MRI techniques, have further accelerated the adoption of minimally invasive breast biopsies. These technologies improve targeting accuracy, reduce the need for repeat procedures, and enhance diagnostic reliability. As clinical guidelines favor image-guided needle biopsies as the standard of care, demand for advanced breast biopsy devices continues to grow. For instance, in February 2025, Cancer Network reported that imaging-guided large-core needle breast biopsy accurately diagnoses most breast pathologies, achieving at least 90% accuracy in stereotactic core biopsy, offering a faster and more cost-effective alternative to surgical biopsy. Major complications are rare, and minimal tissue removal reduces cosmetic deformity. While most lesions are suitable for stereotactic techniques, some cases, such as ductal atypia or limited tissue sampling, still require surgical excision. Given that 70-80% of breast biopsies assess benign lesions, minimally invasive approaches provide effective diagnosis while preserving breast contour.
The expansion of breast cancer screening programs significantly drives the breast biopsy market by increasing the detection of suspicious breast abnormalities at earlier stages. As more women undergo routine mammography and advanced imaging, the number of lesions requiring diagnostic confirmation increases. This directly increases the volume of image-guided and minimally invasive biopsy procedures. Consequently, broader screening coverage leads to sustained demand for breast biopsy devices and technologies.
For instance, in February 2026, The Times of India reported that approximately 11,500 women in both urban and rural areas of Varanasi were screened for breast cancer through the Namo Shakti Rath initiative. Launched on January 14, the campaign was operated by the government in collaboration with the ITV Foundation, utilizing mobile units equipped with advanced cancer screening technology. During the drive, arrangements were made with a cancer hospital for further examination and treatment of suspected cases. The ITV Foundation deployed 20 Namo Shakti Rath vehicles in the Varanasi district to facilitate the screening program. Similarly, in March 2025, the American Cancer Society reported that the U.S. House of Representatives introduced the Screening for Communities to Receive Early and Equitable Needed Services (SCREENS)for Cancer Act of 2025. This bipartisan legislation aims to reauthorize the National Breast and Cervical Cancer Early Detection Program (NBCCEDP) for fiscal years 2026 through 2030. For more than three decades, the NBCCEDP has provided essential breast and cervical cancer screening, diagnostic testing, and treatment services to underserved, low-income, underinsured, and uninsured populations across the U.S. The American Cancer Society Cancer Action Network (ACS CAN) has strongly supported the swift passage of the bill to ensure continued access to lifesaving cancer screening and care.
Market Concentration & Characteristics
The breast biopsy devices industry demonstrates a high level of innovation, driven by advancements in minimally invasive technologies and the integration of precision imaging. Manufacturers are developing vacuum-assisted systems, automated core needle devices, and MRI-compatible biopsy tools to improve targeting accuracy and patient comfort. Integration with digital breast tomosynthesis and AI-based lesion localization further enhances procedural efficiency. Continuous improvements in device ergonomics and sample quality are strengthening diagnostic reliability and expanding clinical adoption.
The breast biopsy devices industry is witnessing a moderate-to-high level of mergers and acquisitions, driven by the need to expand product portfolios and strengthen technological capabilities. Large medical device companies are acquiring smaller, innovative firms to gain access to advanced, imaging-guided, and minimally invasive biopsy technologies. These strategic consolidations enhance global distribution networks and competitive positioning. M&A activity also supports faster commercialization of next-generation biopsy solutions.
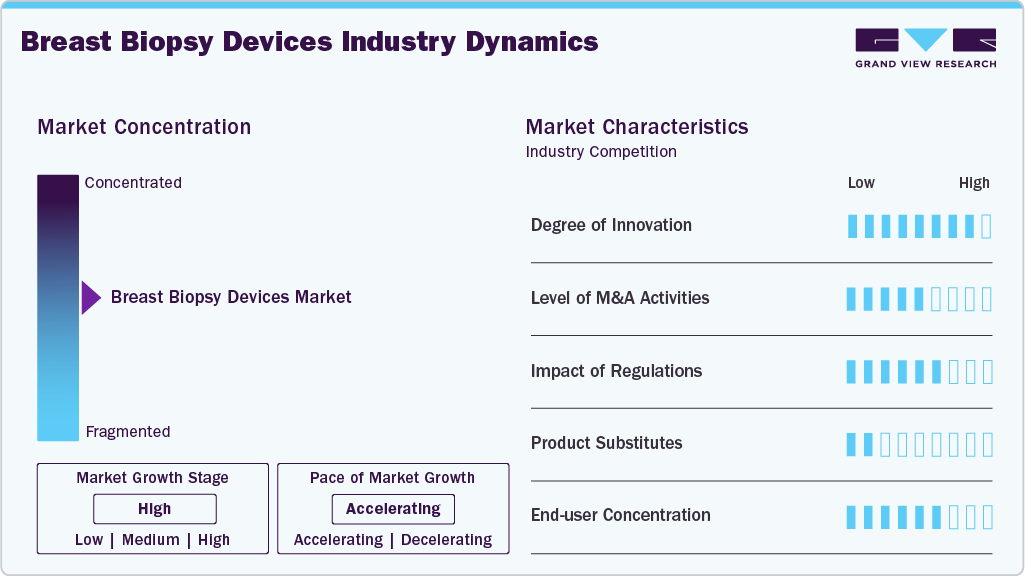
Regulations have a significant impact on the breast biopsy devices industry by ensuring product safety, clinical effectiveness, and quality standards. Stringent regulatory approval processes increase development timelines and compliance costs for manufacturers. However, regulatory oversight also enhances clinician and patient confidence in biopsy technologies. Clear guidelines and reimbursement policies further influence market adoption and commercialization strategies.
The breast biopsy devices industry shows moderate end-user concentration, with hospitals accounted for the largest share due to advanced imaging infrastructure and the availability of specialized radiologists. Diagnostic imaging centers and ambulatory surgical centers also represent significant segments, driven by the shift toward outpatient and minimally invasive procedures. Large urban healthcare facilities tend to dominate procedure volumes, while smaller and rural centers have comparatively lower adoption. This concentration reflects differences in access to technology, expertise, and patient referral patterns.
Product Insights
The biopsy consumables segment held the largest revenue share in 2025, primarily due to the repeated use of disposable components in every procedure. Items such as core needles, vacuum-assisted probes, localization markers, and procedure kits generate continuous demand across healthcare settings. The increasing adoption of minimally invasive and image-guided biopsies has further strengthened this segment’s dominance. As the number of breast biopsy procedures continues to grow, consumables remain a significant contributor to overall market growth.
The biopsy guidance systems segment is expected to grow fastest during the forecast period, driven by rising demand for precise, image-guided procedures. Continued advancements in imaging modalities such as 3D tomosynthesis, MRI, and AI-assisted targeting are enhancing lesion localization and biopsy accuracy. Increasing adoption of minimally invasive techniques in outpatient and diagnostic centers is also supporting this rapid growth. As healthcare providers focus on improved diagnostic outcomes and workflow efficiency, investment in advanced guidance systems is accelerating.
Procedure Insights
The core needle biopsy (CNB) segment dominated the market in 2025, driven by its widespread clinical acceptance as the standard diagnostic technique for suspicious breast lesions. CNB offers high diagnostic accuracy while being minimally invasive, cost-effective, and suitable for outpatient settings. Its compatibility with multiple imaging guidance methods, including ultrasound and stereotactic systems, further supports its strong adoption. The growing preference for reliable, tissue-based diagnosis continues to drive the dominance of the CNB segment.
Vacuum-assisted biopsy (VAB) is expected to grow fastest during the forecast period, driven by its ability to obtain larger tissue samples with fewer insertions, thereby improving diagnostic confidence. Its expanding clinical use for difficult-to-access lesions and microcalcifications, along with integration into advanced imaging systems, is driving adoption. VAB’s minimally invasive nature and suitability for both diagnostic and therapeutic procedures further enhance its appeal. As healthcare providers seek more efficient and precise biopsy solutions, demand for VAB technologies is rising rapidly.
Guidance Technology Insights
The ultrasound-guided segment dominated the market in 2025 due to its widespread availability, real-time imaging capability, and cost-effectiveness. Ultrasound guidance allows precise needle placement without radiation exposure, making it a preferred option for many clinicians. The technique is highly efficient for sampling palpable and visible lesions and is commonly performed in outpatient settings. Its accessibility and ease of use have contributed significantly to its leading market position.
The mammography/stereotactic-guided segment is expected to grow at the fastest rate during the forecast period. Increasing detection of microcalcifications and non-palpable lesions through digital mammography and tomosynthesis is driving demand for stereotactic biopsy procedures. Advancements in 3D imaging and targeting accuracy are further enhancing clinical adoption. As screening programs expand globally, the need for precise stereotactic-guided biopsies is projected to rise significantly.
End Use Insights
The hospitals segment dominated the market in 2025, driven by the availability of advanced imaging infrastructure and skilled multidisciplinary teams. Hospitals handle a high volume of diagnostic procedures, particularly complex, image-guided biopsies that require specialized equipment. Their capacity to manage complications and provide comprehensive cancer care further supports patient preference for hospital-based procedures. As a result, hospitals accounted for the largest share of breast biopsy device utilization.
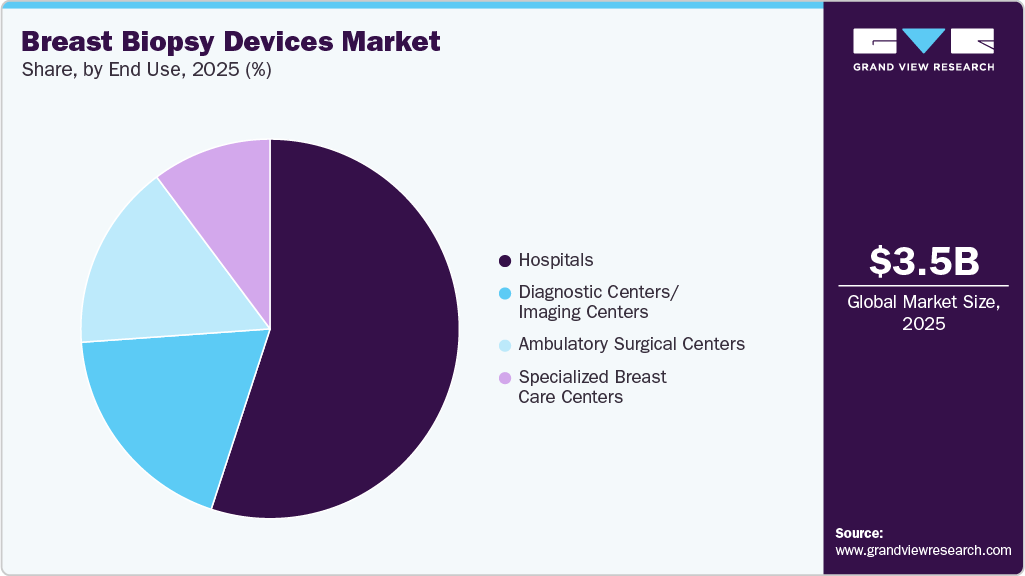
The ambulatory surgical centers (ASCs) segment is expected to grow at the fastest rate during the forecast period. The shift toward minimally invasive procedures and same-day diagnostics has increased the preference for outpatient settings. ASCs offer cost-effective treatment, shorter waiting times, and streamlined workflows, making them attractive to both patients and providers. As healthcare systems focus on efficiency and reducing hospital burden, biopsy procedures in ASCs are projected to rise significantly.
Regional Insights
North America Breast Biopsy Devices Market Trends
The breast biopsy devices industry in North America held the largest share and accounted for 44.50% of global revenue in 2025. The market is characterized by high adoption of minimally invasive and image-guided biopsy technologies. Increasing breast cancer screening participation, supported by widespread mammography programs, is contributing to higher diagnostic procedure volumes. The region is also witnessing a shift toward outpatient and ambulatory care settings for cost-effective treatment. Continuous technological advancements and integration of advanced imaging systems are further supporting market growth across the region.
U.S. Breast Biopsy Devices Market Trends
The U.S. breast biopsy devices industry is driven by increasing breast cancer screening rates and strong adoption of advanced biopsy technologies such as image-guided and minimally invasive systems. Healthcare providers are increasingly using ultrasound, stereotactic, and MRI-guided biopsy devices to improve diagnostic accuracy and patient comfort. The shift toward outpatient and ambulatory care settings is supporting broader use of efficient biopsy procedures. Continued innovation and favorable reimbursement policies are further shaping market growth in the U.S.
Europe Breast Biopsy Devices Market Trends
The Europe breast biopsy devices industry is experiencing significant growth driven by increased breast cancer awareness and widespread implementation of national screening programs across the region. Adoption of minimally invasive, image-guided biopsy technologies, such as ultrasound and stereotactic systems, is increasing as clinicians prioritize accuracy and patient comfort. Investments in healthcare infrastructure and outpatient care facilities are also supporting broader use of advanced biopsy solutions. In addition, technological innovation and favorable healthcare policies continue to shape market development in key European countries.
The UK breast biopsy devices industry is seeing growth supported by national breast screening programs and increased investment in early diagnostic infrastructure. There is a rising uptake of minimally invasive, image-guided biopsy techniques, such as ultrasound and stereotactic systems, to improve diagnostic accuracy and patient outcomes. The shift toward outpatient care and the integration of advanced imaging technologies are further driving demand. Continued emphasis on early detection and supportive healthcare policies is shaping market expansion in the UK.
The France breast biopsy devices industry is growing significantly as national cancer screening efforts and public health awareness campaigns drive early detection initiatives. There is increasing adoption of minimally invasive, image-guided biopsy technologies, especially ultrasound and stereotactic systems, to improve diagnostic precision and the patient experience. Investments in modern diagnostic facilities and outpatient care settings are expanding access to advanced biopsy procedures. Continued focus on early diagnosis and supportive reimbursement policies is shaping market growth in France.
Asia Pacific Breast Biopsy Devices Market Trends
The Asia Pacific breast biopsy devices industry is witnessing rapid growth driven by rising breast cancer incidence and expanding screening programs in countries such as China, India, and Japan. Increasing healthcare expenditure and improvements in diagnostic infrastructure are boosting the adoption of image-guided and minimally invasive biopsy technologies. There is also a growing shift toward outpatient and ambulatory care settings, enhancing access to advanced procedures. Continued investments in modern medical facilities and rising patient awareness are further accelerating market expansion across the region.
The China breast biopsy devices industry is experiencing strong growth as the country expands national breast cancer screening initiatives and improves early detection programs. Rising breast cancer incidence, combined with growing healthcare investment, is driving the adoption of advanced, minimally invasive, and image-guided biopsy technologies. Increased availability of outpatient diagnostic centers and modern imaging equipment supports broader procedural access. In addition, heightened patient awareness and supportive healthcare policies are further boosting market development in China.
Latin America Breast Biopsy Devices Market Trends
The Latin America breast biopsy devices industry is gradually expanding as awareness of breast cancer screening increases, and more countries implement organized early detection programs. Growing healthcare infrastructure and investment in diagnostic imaging technologies are supporting the uptake of minimally invasive and image-guided biopsy procedures. There is also increased demand for cost-effective and efficient diagnostic solutions in both urban and emerging markets. Continued efforts to improve access to breast care services are helping drive market growth across the region.
MEA Breast Biopsy Devices Market Trends
The MEA breast biopsy devices industry is gradually emerging as awareness of breast cancer and early detection efforts increase across the region. Investments in healthcare infrastructure and diagnostic imaging capabilities, particularly in GCC and key African markets, are supporting the adoption of image-guided and minimally invasive biopsy technologies. Limited access in rural areas remains a challenge, but expanding outpatient care facilities is improving procedural availability. Continued focus on screening programs and rising patient awareness are expected to drive market growth in the MEA region.
Key Breast Biopsy Devices Company Insights
Key companies in the breast biopsy devices industry include established medical device manufacturers known for imaging-guided and minimally invasive technologies, with leaders such as Hologic, BD, and GE Healthcare holding significant market share. These players maintain strong positions through broad product portfolios, global distribution networks, and ongoing innovation in biopsy and imaging systems.
Key Breast Biopsy Devices Companies:
The following key companies have been profiled for this study on the breast biopsy devices market.
- Hologic, Inc.
- Mammotome (Danaher)
- BD
- Argon Medical
- Merit Medical Systems
- GE HealthCare
- Cook Medical
- MOLLI Surgical (Stryker)
- Planmed Oy
- Metaltronica Spa
- Trivitron Healthcare
- Siemens Healthineers
- Fujifilm Healthcare
- INRAD
- SteryLab
- Resitu Medical
- TransMed7, LLC.
Recent Developments
-
In January 2026, BD (Becton, Dickinson and Company) announced that the U.S. Food and Drug Administration (FDA) granted 510(k) clearance for the EnCor EnCompass Breast Biopsy and Tissue Removal System. The advanced multi-modality breast biopsy system is designed to provide clinicians with flexibility across various breast imaging modalities for the diagnosis of breast disease. This clearance strengthens BD’s portfolio in minimally invasive breast biopsy solutions.
"This milestone for our new breast biopsy system marks a meaningful advancement in breast health, playing a critical role in aiding the early detection and diagnosis of breast disease," said Rima Alameddine, worldwide president, Peripheral Intervention at BD. "This innovation underscores our commitment to partnering with clinical leaders to deliver patient-centered solutions. Guided by our vision to transform breast health, we remain focused on developing technologies that empower providers and inspire confidence in care."
"The FDA clearance of the EnCor EnCompass Biopsy System demonstrates our ongoing focus on addressing the evolving needs of clinicians and patients in breast health," said Stacie Watson, vice president and general manager of the Oncology Platform at BD Interventional-Peripheral Intervention. "This multi-modality platform is engineered to provide flexibility, control, and ease of use, with features designed to support both clinician confidence and patient experience."
-
In September 2025, Resitu announced that it received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its first breast biopsy device, RESL09. The company stated that the milestone marks a significant achievement in advancing its innovative breast biopsy technology for clinical use in the United States.
-
In January 2025, Siemens Healthineers announced the first U.S. installation of its Mammomat B.brilliant mammography system. Marking the company’s first fully redesigned mammography platform in over a decade, the system features advanced 3D image acquisition and reconstruction technology. It also supports full-field digital mammography (2D imaging), breast biopsy procedures, and titanium-contrast-enhanced mammography, thereby enhancing diagnostic capabilities in breast imaging.
“At GRACE, our primary goal is to provide exceptional breast care to women,” said Andrea Lamphiear, MD, Founder of GRACE Breast Imaging & Medical Spa. “Our decision to acquire the nation’s first Mammomat B.brilliant mammography system was driven by the cutting-edge technology of Siemens Healthineers, which offers unparalleled image quality for early breast cancer detection. We believe this will best help us expand our vision of providing individualized breast care in a wellness-based and serene environment and making healthcare feel like self-care, while also allowing us, as radiologists, to have the best chance of finding breast cancer at its earliest stage.”
-
In March 2023, TransMed7, LLC announced the first clinical use of VacuPac, a new self-contained external vacuum-assist attachment compatible with the SpeedBird Universal and Concorde US devices from its SpeedBird and Concorde families of vacuum-assisted, Single Insertion - Multiple Collection (SIMC) breast biopsy systems. This milestone followed earlier announcements in August 2022 and February 2023 regarding the initial clinical use of the SpeedBird and Concorde devices as part of their commercial launch. The procedures utilizing VacuPac were performed by Dr. Michael Berry at the Margaret West Comprehensive Breast Center in Germantown, Tennessee.
Dr. Ed Staren, President of TransMed7, stated, “It has been my wish to add a portable, self-contained vacuum source to the already very handy SpeedBird and Concorde devices as an option for a completely tetherless procedure, while preserving the ability to use external vacuum sources when desired. I give credit to the technical team for enabling this option by developing the VacuPac concept into a successful reality. Therefore it gives me great satisfaction to announce the successful clinical utilization of our VacuPac attachment for our minimally invasive breast biopsy devices. This milestone demonstrates that an attachable, disposable, vacuum-assist device may be utilized to support any of our Zero5 technology-based biopsy instruments in a convenient, tetherless form for the care and benefit of patients. We anticipate that VacuPac will provide a useful option to support Zero5® technology in all of our next generation device platforms in their various applications all over the body. These include: Heron, Cardinal, Thunderbird and Phoenix in addition to its demonstrated use in SpeedBird and Concorde.”
Dr. James W. Vetter, TransMed7 Co-Founder and Chairman stated, “We specifically developed the single use, disposable VacuPac as a separate device to keep the biopsy devices themselves simple and cost-effective, while giving specialists freedom to choose suction sources for any given case. Leveraging VacuPac’s clinical successes and enthusiastic reception, we are expanding the lineup to include an even smaller version to match our Cardinal Fine Needle Core Biopsy Platform. VacuPac and its variants in the pipeline demonstrate our commitment to provide full fluid management in all current and future Zero5-based SIMC devices whether totally untethered with VacuPac or connected by tubing to wall suction or other sources including our own rechargeable battery-powered VacuTower.”
Breast Biopsy Devices Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 3.71 billion
Revenue forecast in 2033
USD 5.60 billion
Growth rate
CAGR of 6.04% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, procedure, guidance technology, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Norway; Italy; Denmark; Sweden; Spain; China; Japan; India; South Korea; Australia; Argentina; Brazil; UAE; South Africa; Thailand; Saudi Arabia; Kuwait
Key companies profiled
Hologic, Inc.; Mammotome (Danaher); BD; Argon Medical; Merit Medical Systems; GE HealthCare; Cook Medical; MOLLI Surgical (Stryker); Planmed Oy; Metaltronica Spa; Trivitron Healthcare; Siemens Healthineers; Fujifilm Healthcare; INRAD; SteryLab; Resitu Medical; TransMed7, LLC.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Breast Biopsy Devices Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis on the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global breast biopsy devices market report based on product, procedure, guidance technology, end use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Biopsy Guidance Systems
-
Biopsy Drivers & Handpieces
-
Vacuum-Assisted Biopsy (VAB) Drivers
-
Core Needle Biopsy (CNB) Guns
-
Automated
-
Semi-Automated
-
-
-
Biopsy Tables
-
Prone Tables
-
Upright/Add-on Tables
-
-
Biopsy Consumables
-
Needles & Probes
-
Site Markers & Clips
-
-
By Shape/Design
-
Standard Geometric Markers
-
Ring
-
Cross
-
Ribbon
-
Others
-
-
Advanced Anchoring Markers
-
Anchor
-
Wing, "U" Shape
-
Q" Shape
-
-
3D / Expandable Markers
-
3D Ribbon
-
3D Sphere
-
3D Mesh
-
-
Localization Systems
-
Traditional Wires
-
Wireless Seeds-RFID/Magnetic
-
-
Other Consumables
-
-
Procedure Outlook (Revenue, USD Million, 2021 - 2033)
-
Vacuum-Assisted Biopsy (VAB)
-
Core Needle Biopsy (CNB)
-
Fine Needle Aspiration Biopsy (FNAB)
-
Surgical (Open) Biopsy
-
Liquid Biopsy
-
-
Guidance Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Ultrasound-Guided
-
Mammography / Stereotactic-Guided
-
MRI-Guided
-
-
End Use Outlook (Revenue, USD Million; 2021 - 2033)
-
Hospitals
-
Ambulatory Surgical Centers (ASCs)
-
Specialized Breast Care Centers
-
Diagnostic Centers/Imaging Centers
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Sweden
-
Denmark
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Key factors that are driving the market growth include rising prevalence of breast cancer in women, global awareness and cancer screening programs, and demand for alternative, less-invasive and accurate procedures.
b. The global breast biopsy device market size was estimated at USD 3.52 billion in 2025 and is expected to reach USD 3.71 billion in 2026.
b. The global breast biopsy device market is expected to grow at a compound annual growth rate of 6.0% from 2026 to 2033 to reach USD 5.60 billion by 2033.
b. North America dominated the breast biopsy device market with a share of 44.50% in 2025. This is attributable to rising prevalence of breast cancer and presence of key players in the region.
b. Some key players operating in the breast biopsy device market include Hologic, Inc., Mammotome (Danaher), BD, Argon Medical, Merit Medical Systems, GE HealthCare, Cook Medical, MOLLI Surgical (Stryker), Planmed Oy, Metaltronica Spa, Trivitron Healthcare, Siemens Healthineers, Fujifilm Healthcare, INRAD, SteryLab, Resitu Medical, TransMed7, LLC.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










