- Home
- »
- Biotechnology
- »
-
Clinical Oncology Next Generation Sequencing Market, 2033GVR Report cover
![Clinical Oncology Next Generation Sequencing Market Size, Share & Trends Report]()
Clinical Oncology Next Generation Sequencing Market (2025 - 2033) Size, Share & Trends Analysis Report By Workflow (NGS Pre-Sequencing, NGS Sequencing, NGS Data Analysis), By Technology, By Application, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-3-68038-791-9
- Number of Report Pages: 125
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Clinical Oncology Next Generation Sequencing Market Summary
The global clinical oncology next generation sequencing market size was estimated at USD 482.3 million in 2024 and is projected to reach USD 1,930.2 million by 2033, growing at a CAGR of 16.77% from 2025 to 2033. The key drivers responsible for the growth of the clinical oncology next generation sequencing (NGS) industry are increasing technological advancements, growing competition among players, increasing healthcare spending, and the rise in the prevalence of cancer.
Key Market Trends & Insights
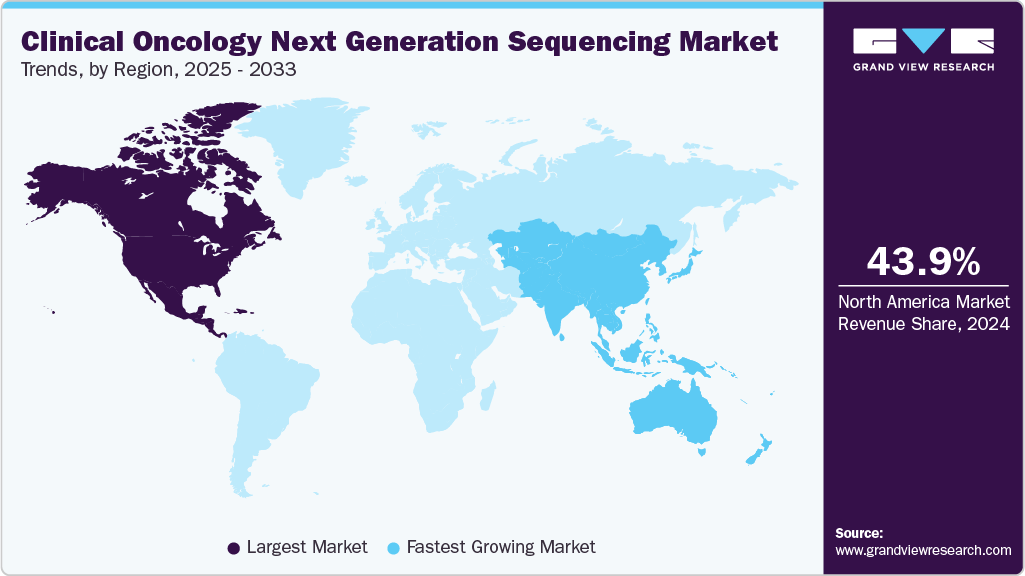
- North America clinical oncology NGS market held the largest share of 43.97% of the global market in 2024.
- The clinical oncology NGS industry in the U.S. is expected to grow significantly over the forecast period.
- By technology, the targeted sequencing & resequencing segment held the highest market share of 64.02% in 2024.
- Based on workflow, the NGS sequencing segment held the highest market share of 56.43% in 2024.
- By application, the companion diagnostics segment held the highest market share of 71.15% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 482.3 Million
- 2033 Projected Market Size: USD 1,930.2 Million
- CAGR (2025-2033): 16.77%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
In addition, augmenting research & development activities to invent solutions is expected to fuel the market growth over the forecast period.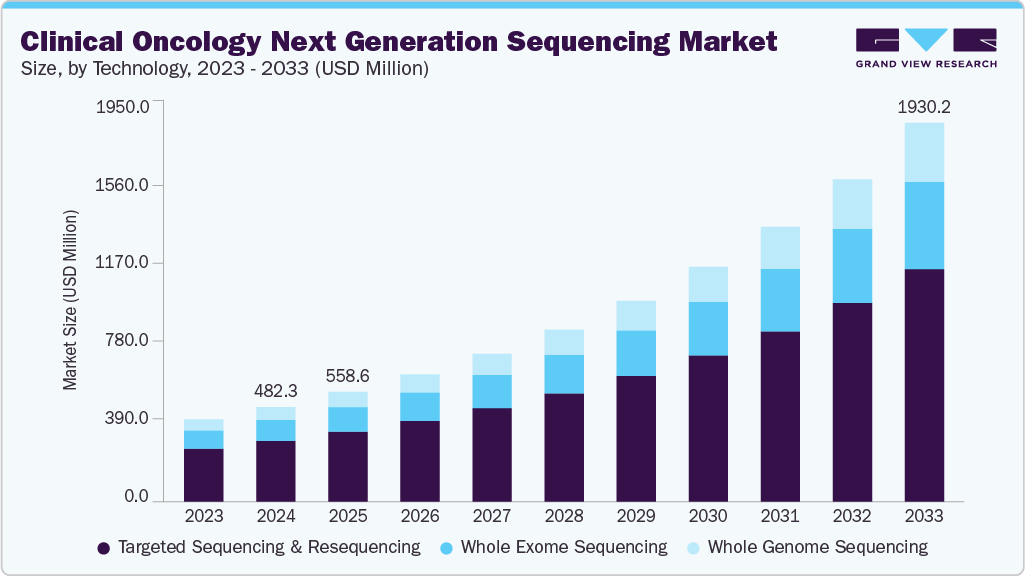
Escalating Cancer Rates Propel Adoption of NGS in Oncology
The global surge in cancer incidence has emerged as a significant public health challenge, significantly influencing diagnostic and treatment strategies. According to the World Health Organization, cancer is one of the leading causes of death globally, with millions of new cases reported each year. This alarming rise is attributed to aging populations, environmental factors, and changing lifestyle habits. As cancer becomes increasingly prevalent, healthcare systems are shifting focus toward early and accurate detection to improve treatment outcomes and reduce mortality. This trend fuels demand for advanced molecular diagnostic tools, particularly next-generation sequencing (NGS), which offers a comprehensive understanding of cancer genetics.
NGS technology enables the detection of multiple genetic mutations in a single test, making it a powerful tool in oncology. Its ability to analyze tumor heterogeneity and provide detailed genomic insights is critical for identifying cancer subtypes and developing targeted therapies. The rising incidence of complex and rare cancers has further amplified the need for precision diagnostics, where NGS plays a vital role in guiding clinicians toward personalized treatment approaches. As a result, hospitals, diagnostic laboratories, and cancer centers are increasingly integrating NGS platforms into their diagnostic workflows, recognizing their value in enhancing diagnostic accuracy and therapeutic decision-making.
Furthermore, as cancer cases continue to climb across both developed and emerging markets, governments and private stakeholders are investing heavily in genomic infrastructure. National genomic programs, increased reimbursement for NGS tests, and ongoing clinical trials using NGS-driven biomarkers reinforce the technology's role in routine cancer diagnostics. This upward trend in cancer incidence is expected to sustain and accelerate the adoption of NGS in clinical oncology, establishing it as a cornerstone of modern cancer care.
Advancing NGS Platforms Fuel Clinical Oncology Integration
The clinical oncology NGS market is witnessing robust growth driven by continuous technological advancements that enhance sequencing accuracy, speed, and usability. Innovations such as high-throughput platforms, error-correction algorithms, and improved chemistry have significantly increased the sensitivity and specificity of NGS, making it highly suitable for clinical oncology applications. Platforms such as Illumina’s NovaSeq and Thermo Fisher’s Ion Torrent systems have enabled comprehensive genomic profiling at lower turnaround times, facilitating faster diagnosis and treatment planning for cancer patients.
The emergence of integrated, automated workflows and cloud-based data interpretation tools has streamlined complex NGS procedures, making them more accessible to clinical laboratories with limited expertise. These advancements reduce operational burdens and improve scalability, accelerating the clinical adoption of NGS in oncology. As a result, healthcare providers can efficiently detect actionable mutations, monitor disease progression, and personalize therapies with greater precision. The convergence of NGS technologies with digital health solutions and AI-driven analytics is further expected to revolutionize cancer care, supporting the ongoing shift toward precision oncology.
Market Concentration & Characteristics
The clinical oncology NGS industry is highly innovative, owing to the rapid evolution of sequencing technologies, integration of AI-driven analytics, and development of automated, high-throughput platforms. These innovations enable faster, more accurate genomic profiling and support the shift toward precision oncology and personalized treatment strategies across diverse cancer types.
The moderate increase in mergers and acquisitions (M&A) in the clinical oncology NGS industry is attributed to companies seeking to expand their technological capabilities, enhance product portfolios, and strengthen their market position. Strategic collaborations also aim to accelerate innovation, streamline R&D pipelines, and integrate complementary expertise in bioinformatics, diagnostics, and sequencing platforms to meet the growing demand for precision oncology solutions.
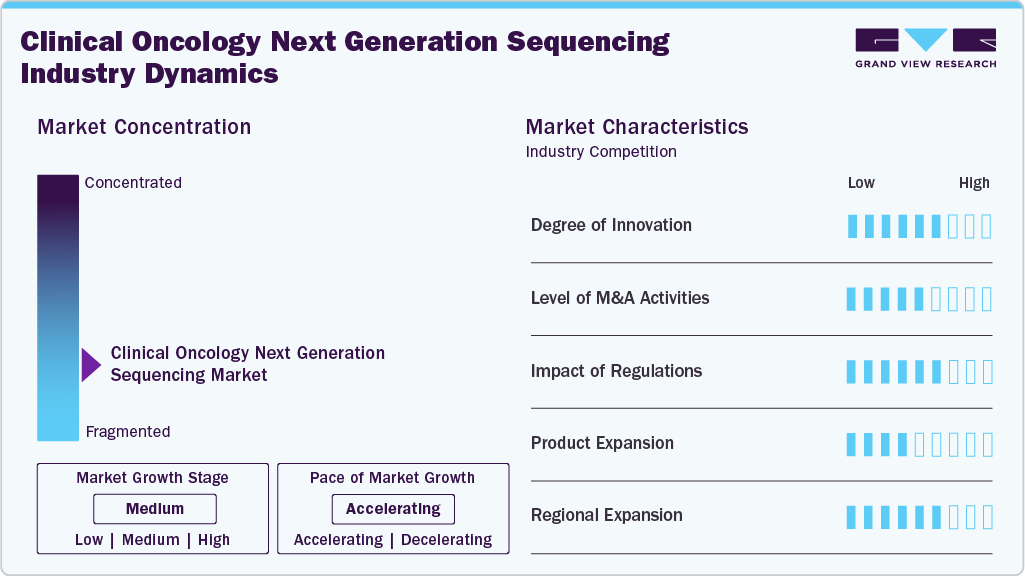
Regulations play a dual role in shaping demand within the clinical oncology NGS industry by ensuring test accuracy and patient safety while influencing market accessibility. Stringent regulatory approvals build trust and drive clinical adoption, but complex compliance requirements can delay product launches and increase costs. Harmonized global frameworks and expedited pathways, such as the FDA’s Breakthrough Device designation, are helping balance innovation with oversight, thereby supporting market growth and broader implementation in clinical settings.
Product expansion is a significant driving factor for the growth in the clinical oncology NGS industry. Introducing advanced panels, liquid biopsy tests, and companion diagnostics enables broader cancer coverage and improved clinical utility. This strategic expansion enhances diagnostic accuracy, supports personalized treatment approaches, and reinforces competitive positioning in a rapidly evolving precision oncology market.
Regional expansion is the primary demand driver in the clinical oncology NGS industry, owing to the increasing adoption of precision medicine across emerging markets, rising healthcare investments, and improved access to advanced diagnostic technologies. Companies are targeting high-growth regions such as Asia Pacific and Latin America to tap into unmet clinical needs, expand patient reach, and leverage supportive government initiatives, fueling overall market growth and accelerating the global adoption of NGS in oncology care.
Technology Insights
Based on the technology, the market is segmented into whole genome sequencing, targeted sequencing & resequencing, and whole exome sequencing. The targeted sequencing & resequencing segment held the largest market share of 64.02% in 2024 in terms of revenue, owing to the various benefits associated with this technology, such as decreased time, cost, and quantity of data analyzed during the tumor samples sequencing Furthermore, the clinical utility of targeted panels is increased due to their efficiency in detecting malignant tumors.
Whole-Genome Sequencing (WGS) is anticipated to be the fastest-growing segment with 18.39% CAGR from 2025 to 2033, owing to the usage of this technology to compare and differentiate tumor tissues from normal tissues. In addition, WGS of cancer patients enables the identification of treatments for current mutations and aids in targeting mutations in advance. This also supports analyzing cancer prognosis.
Workflow Insights
The market is segmented by workflow into NGS pre-sequencing, NGS sequencing, and NGS data analysis. The NGS sequencing segment accounted for the largest revenue share, 56.43%, in 2024 in the workflow segment, and it is expected to grow at the fastest CAGR over the forecast period. This is attributed to its central role in genomic analysis, continuous technological advancements, and increasing demand for high-throughput, accurate sequencing solutions in clinical oncology for personalized treatment and early cancer detection.
The NGS data analysis segment in the clinical oncology NGS market is expected to register a significant CAGR of 16.09% during the forecast period driven by the growing complexity of genomic data, increased need for accurate interpretation tools, and the integration of AI and machine learning to enhance clinical decision-making and streamline personalized cancer treatment planning.
Application Insights
Companion diagnostics dominated the market with a revenue share of 71.15% in 2024 and are anticipated to grow at the fastest CAGR over the forecast period. This is attributed to the rising demand for targeted cancer therapies, increased FDA approvals of biomarker-driven treatments, and the growing emphasis on precision medicine. These diagnostics enable clinicians to identify the most effective therapy based on a patient’s genetic profile, improving treatment outcomes and driving widespread adoption across oncology practices.
The screening application segment in the clinical oncology NGS market is expected to register a significant CAGR of 12.59% during the forecast period. This growth is driven by the increasing emphasis on early cancer detection, rising awareness of genetic risk factors, and the growing adoption of non-invasive screening methods such as liquid biopsy. These factors enable timely diagnosis and intervention, improving patient outcomes and reducing cancer treatment costs.
End Use Insights
Laboratories led the market with a revenue share of 63.52% in 2024, driven by their advanced infrastructure, specialized expertise in genomic testing, and high testing volumes. The increasing outsourcing of NGS services by hospitals and research institutions to certified laboratories and the availability of integrated data analysis capabilities have further solidified their dominant position in the clinical oncology NGS market.
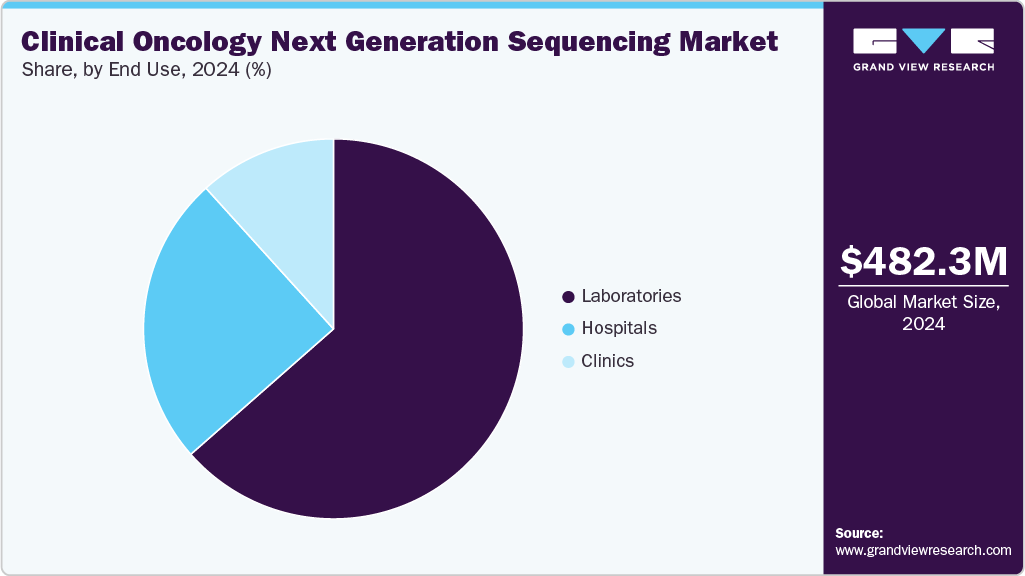
The clinics in the end use segment of the clinical oncology NGS industry are expected to register the fastest CAGR of 19.21% during the forecast period. The rising adoption of protein-based diagnostics drives this growth, the increasing use of biomarkers for disease monitoring, and the growing integration of personalized medicine in clinical settings. Hospitals are expanding their in-house capabilities to support advanced testing and targeted therapeutic development.
Regional Insights
North America clinical oncology next generation sequencing dominated the global market with the largest revenue share of 43.97% in 2024. The dominance can be attributed to the region’s advanced healthcare infrastructure, high adoption of precision medicine, and strong presence of key market players. Additionally, favorable reimbursement policies, significant investments in cancer genomics research, and early regulatory approvals for NGS-based diagnostics have accelerated clinical adoption. The region also benefits from high awareness among healthcare professionals and patients, driving demand for targeted therapies and companion diagnostics in oncology.
U.S. Clinical Oncology Next Generation Sequencing Market Trends
The U.S. clinical oncology NGS market dominated the North American market in 2024, owing to its robust healthcare infrastructure, early adoption of advanced genomic technologies, and substantial investment in cancer research. Supportive regulatory frameworks, widespread use of companion diagnostics, and a strong presence of leading NGS companies further contributed to the country’s leadership in integrating NGS into routine oncology care.
Europe Clinical Oncology Next Generation Sequencing Market Trends
The European clinical oncology NGS market is expected to grow in the forecast period, driven by increasing adoption of personalized medicine, expanding genomic research initiatives, and growing government support for cancer diagnostics. Rising cancer prevalence, along with improvements in healthcare infrastructure and access to advanced sequencing technologies, is fueling demand across the region. Additionally, collaborations between research institutions and biotechnology firms are accelerating the development of NGS-based clinical applications in oncology.
The UK clinical oncology NGS industry is expected to grow rapidly in the coming years due to strong government initiatives supporting genomics, such as the 100,000 Genomes Project, and increased investment in precision oncology research. The presence of leading academic institutions, expanding access to advanced diagnostic technologies, and growing partnerships between the public and private sectors further accelerate the integration of NGS into clinical cancer care.
The Germany clinical oncology NGS market held a substantial market share in 2024, owing to its well-established healthcare infrastructure, strong emphasis on precision medicine, and robust funding for cancer genomics research. The country's proactive regulatory environment, widespread adoption of advanced diagnostic technologies, and presence of key biotechnology and pharmaceutical companies further contributed to its leading position in the European clinical oncology NGS landscape.
Asia Pacific Clinical Oncology Next Generation Sequencing Market Trends
The clinical oncology NGS market in Asia Pacific is expected to register the fastest growth, with a CAGR of 18.28% during the forecast period. This growth is fueled by rising cancer prevalence, increasing demand for personalized medicine, and expanding healthcare infrastructure across emerging economies. Government initiatives supporting genomics research, growing awareness of early cancer detection, and increasing investments by domestic and international players are also driving market expansion. Additionally, the adoption of cost-effective NGS platforms and the rise of local laboratory services offering advanced genomic testing are accelerating clinical integration. Countries like China, India, and Japan are at the forefront of this regional growth momentum.
The China clinical oncology NGS market is expected to grow in the forecast period. This is attributed to rising cancer incidence, strong government support for genomics research, expanding local biotech companies, and increasing adoption of precision medicine in clinical practice across major healthcare institutions.
The Japan clinical oncology NGS market is witnessing significant growth over the forecast period, driven by increasing government and private funding for cancer genomics research, strong emphasis on precision medicine integration, and supportive healthcare policies. The presence of advanced sequencing infrastructure, growing collaboration between domestic biotech firms and global players, and rising demand for targeted therapies and companion diagnostics in oncology are further catalyzing market expansion.
Middle East & Africa Clinical Oncology Next Generation Sequencing Market Trends
The MEA clinical oncology NGS market is expected to grow exponentially over the forecast period. This is attributed to the rising cancer burden, increasing investments in healthcare infrastructure, and growing awareness of personalized cancer care. Government-led genomic initiatives, expanding access to advanced diagnostic technologies, and partnerships with global biotech firms are further driving the adoption of NGS-based oncology solutions across the region.
The Kuwait clinical oncology NGS market is projected to grow steadily over the forecast period. This is attributed to rising cancer prevalence, government investment in precision medicine, and increased adoption of advanced genomic diagnostics in leading healthcare institutions.
Key Clinical Oncology Next Generation Sequencing Company Insights
The clinical oncology NGS market is characterized by a high degree of consolidation, with major players such as Illumina, Inc. and Thermo Fisher Scientific dominating the global landscape due to their comprehensive sequencing platforms and robust distribution networks. Illumina continues to lead the market with its cutting-edge instruments and consumables tailored for oncology applications, while Thermo Fisher offers a wide range of solutions, including Ion Torrent systems and Oncomine assays. These companies benefit from strong brand recognition, consistent product innovation, and strategic collaborations with healthcare institutions, enabling them to maintain a competitive edge and high market share.
Other key players such as F. Hoffmann-La Roche Ltd., Agilent Technologies, and Myriad Genetics are expanding their footprint through targeted acquisitions and the development of oncology-focused genomic solutions. Roche’s AVENIO NGS platform and Myriad’s myRisk hereditary cancer test exemplify the push toward personalized treatment strategies. Agilent Technologies continues to leverage its strength in genomic sample preparation and data analysis tools, reinforcing its position in the oncology NGS ecosystem. Similarly, through its genomics services and clinical diagnostics segment, PerkinElmer contributes to the growing demand for cancer screening and genetic risk assessments, particularly in emerging markets.
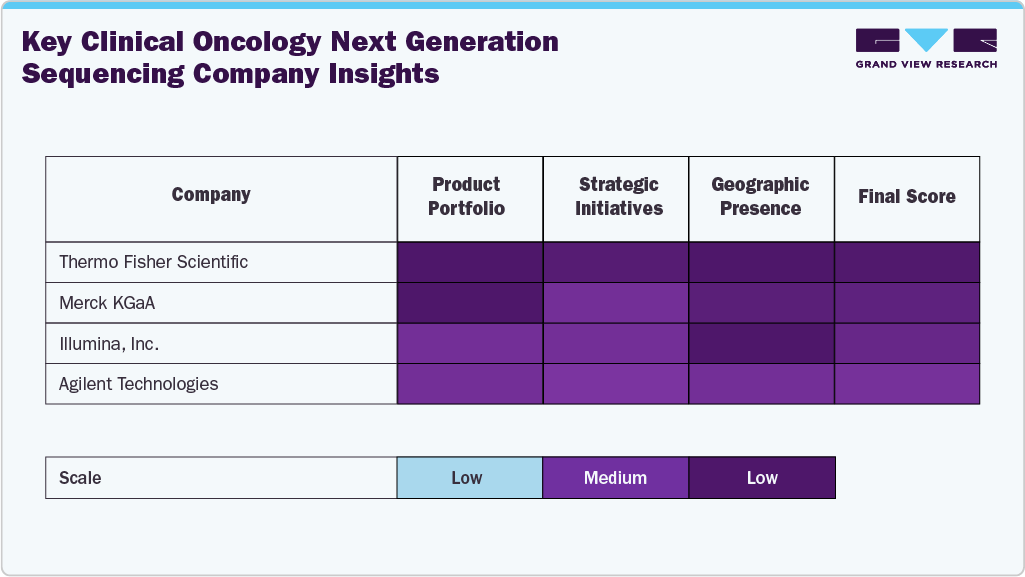
Emerging and specialized firms such as Pacific Biosciences, Oxford Nanopore Technologies Ltd., Eurofins Scientific S.E., and Qiagen N.V. are pivotal in shaping the market’s next growth phase. Pacific Biosciences and Oxford Nanopore are bringing long-read and real-time sequencing capabilities into clinical oncology, helping decode complex mutations. With their strong presence in molecular diagnostics and bioinformatics, Eurofins and Qiagen are enhancing service-based offerings to support clinical labs. As the demand for precision oncology continues to rise, these companies will likely witness accelerated growth, particularly in the companion diagnostics and liquid biopsy segments.
Key Clinical Oncology Next Generation Sequencing Companies:
The following are the leading companies in the clinical oncology next generation sequencing market. These companies collectively hold the largest market share and dictate industry trends.
- Illumina, Inc.
- Thermo Fisher Scientific
- F. Hoffmann-La Roche Ltd.
- Agilent Technologies
- Myriad Genetics
- Perkin Elmer
- Pacific Bioscience
- Oxford Nanopore Technologies Ltd.
- Eurofins Scientific S.E.
- Qiagen N.V.
Recent Developments
-
In January 2025, Tempus AI, Inc. launched its FDA-approved xT CDx, an NGS-based in vitro diagnostic featuring a 648-gene panel. The test offers comprehensive genomic profiling, including MSI and companion diagnostics for colorectal cancer, using matched tumor-normal sequencing for enhanced accuracy.
-
In August 2024, Illumina announced the launch of its NovaSeq X Series oncology menu with high-throughput versions of TruSight Oncology 500 HT and TSO 500 ctDNA v2, which deliver 40% faster run times, three‑day assay turnaround, larger batch capacity, reduced sequencing costs, and ambient‑temperature consumable shipping.
Clinical Oncology Next Generation Sequencing Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 558.6 million
Revenue forecast in 2033
USD 1,930.2 million
Growth rate
CAGR of 16.77% from 2025 to 2033
Historical data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Workflow, application, technology, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; India; China; Japan; Australia; South Korea; Thailand; Brazil; Argentina; Saudi Arabia; UAE; South Africa; Kuwait
Key companies profiled
Illumina, Inc.; Thermo Fisher Scientific; F. Hoffmann-La Roche Ltd.; Agilent Technologies; Myriad Genetics; Perkin Elmer; Pacific Bioscience; Oxford Nanopore Technologies Ltd.; Eurofins Scientific S.E.; Qiagen N.V.
Customization scope
Free report customization (equivalent to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Global Clinical Oncology Next Generation Sequencing Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and analyzes the latest industry trends in each sub-segment from 2021 to 2033. Grand View Research has segmented the global clinical oncology next generation sequencing market report based on workflow, technology, application, end use, and region.
-
Workflow Outlook (Revenue, USD Million, 2021 - 2033)
-
NGS Pre-Sequencing
-
NGS Sequencing
-
NGS Data Analysis
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Whole Genome Sequencing
-
Whole Exome Sequencing
-
Targeted Sequencing & Resequencing
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Screening
-
Sporadic Cancer
-
Inherited Cancer
-
-
Companion Diagnostics
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Clinics
-
Laboratories
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global clinical oncology NGS market size was estimated at USD 482.3 million in 2024 and is expected to reach USD 558.6 million in 2025.
b. Some key players operating in the clinical oncology NGS market include Illumina, Roche, Agilent; Thermo Fisher Scientific; Myriad Genetics; Beijing Genomics Institute (BGI); Perkin Elmer, Inc.; Qiagen NV; Partek, Inc.; Pacific Bioscience, Inc.; Foundation Medicine; Oxford Nanopore Technologies Ltd.; Paradigm Diagnostics, Inc.; Caris Life Sciences; and Eurofins Scientific.
b. Key factors that are driving the clinical oncology NGS market growth include a decrease in costs for genetic sequencing, advancements in the field of personalized medicine, growing adoption of NGS over single gene testing, and others.
b. The global clinical oncology NGS market is expected to grow at a compound annual growth rate of 16.77% from 2025 to 2033 to reach USD 1.93 billion by 2033.
b. Government support to conduct large-scale sequencing, and analysis of NGS data through BigData are some of the key factors that provide growth opportunities to the clinical oncology NGS market.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










