- Home
- »
- Medical Devices
- »
-
GVR Report cover
![Coronary Heart Disease Diagnostic Imaging Devices Market Size, Share & Trends Report]()
Coronary Heart Disease Diagnostic Imaging Devices Market (2026 - 2033) Size, Share & Trends Analysis Report By Modality (Computed Tomography, X-rays, Ultrasound, Magnetic Resonance Imaging, Nuclear Medicine), By Region (North America, Europe, Asia Pacific, Middle East & Africa), And Segment Forecasts
- Report ID: GVR-4-68040-891-5
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
Coronary Heart Disease Diagnostic Imaging Devices Market Summary
The global coronary heart disease diagnostic imaging devices market size was estimated at USD 4.67 billion in 2025 and is projected to reach USD 7.73 billion by 2033, growing at a CAGR of 6.5% from 2026 to 2033. Growth is driven by the rising prevalence of cardiovascular diseases, increasing adoption of advanced imaging technologies such as CT, MRI, and nuclear imaging, and growing awareness of early diagnosis.
Key Market Trends & Insights
- North America's coronary heart disease diagnostic imaging devices market held the largest share of 39.5% of the global market in 2025.
- The Asia Pacific coronary heart disease diagnostic imaging devices industry is expected to grow at the fastest CAGR over the forecast period.
- By modality, the X rays segment held the largest market share of 26.3% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 4.67 Billion
- 2033 Projected Market Size: USD 7.73 Billion
- CAGR (2026-2033): 6.5%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
In addition, aging populations and lifestyle-related risk factors are fueling demand for accurate and non-invasive diagnostic solutions. Technological advancements, including AI integration and improved imaging accuracy, are further supporting market expansion, enabling better clinical outcomes and enhancing the efficiency of cardiovascular disease management worldwide.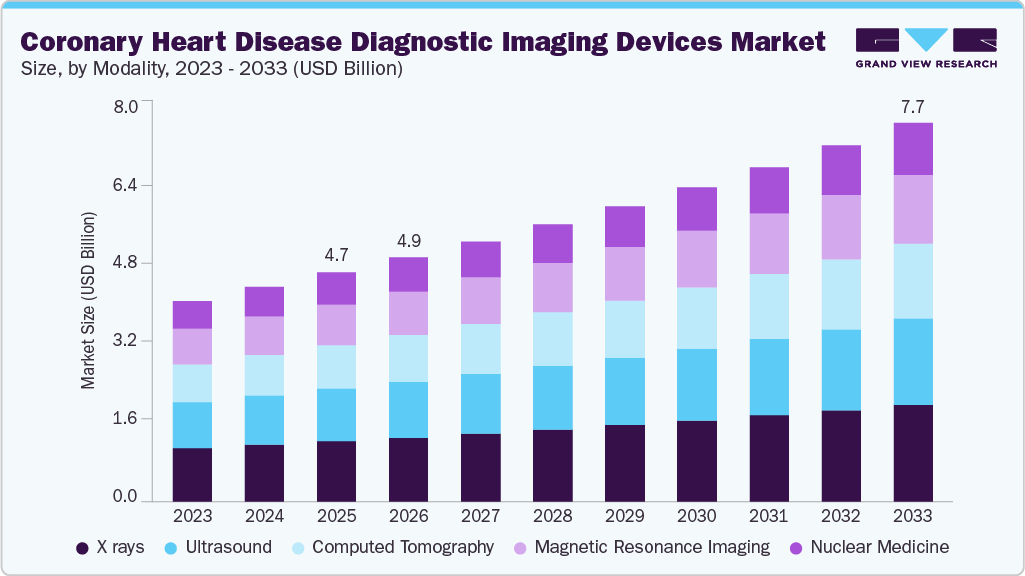
One of the main factors driving the market expansion for coronary heart disease diagnostic imaging devices is the rising incidence of coronary artery diseases (CAD) worldwide. With nearly 19.8 million deaths per year, cardiovascular diseases continue to be the leading cause of death globally, with coronary heart disease accounting for a sizable portion of these deaths. Coronary artery disease (CAD) is primarily caused by atherosclerosis, which leads to the narrowing or blockage of coronary arteries. The demand for diagnostic imaging modalities such as computed tomography (CT), magnetic resonance imaging (MRI), echocardiography, and coronary angiography have directly increased due to the increasing number of patients at risk.
Moreover, demographic changes, especially population aging, have led to a marked increase in the prevalence of CAD. The incidence of coronary conditions is higher in older populations because they are more vulnerable to atherosclerotic changes. For instance, compared to younger populations, prevalence rates are significantly higher among those over 65, underscoring age as a crucial risk factor. Furthermore, a greater number of patients have chronic cardiovascular conditions because of longer life expectancies worldwide, which has increased the use of diagnostic imaging technologies over time.
Estimated Number of Coronary Artery Disease Patients by Region
Region/Country
Estimated Number of CAD Patients
Insights
North America
~20-30 million
High diagnosis rates & aging population
Europe
~40-50 million
High prevalence due to aging population
Asia-Pacific
~150-180 million
High population + rising lifestyle diseases
Latin America
~15-25 million
Increasing burden due to urbanization
Middle East & Africa
~10-20 million
Rapidly growing burden; limited access to care
Source: National Library of Medicine, PMC
Another additional factor contributing to the growing prevalence of coronary artery diseases is lifestyle-related risk factors. The incidence of CAD has dramatically increased in both developed and developing countries due to sedentary lifestyles, poor eating habits, smoking, obesity, and the rising incidence of diabetes and hypertension. Ischemic heart disease is a major contributor to cardiovascular diseases, which account for a significant portion of overall mortality in developing nations such as India. These variables not only raise the incidence of the disease but also cause it to manifest earlier, increasing the number of patients in need of a diagnostic assessment.

Demand is further being driven primarily by the increasing prevalence of chronic cardiovascular disorders, especially coronary artery disease. Early and accurate diagnosis is becoming increasingly important as the prevalence of cardiovascular diseases rises. This is expected to allow for prompt clinical intervention and slow the progression of the disease. When it comes to identifying arterial blockages, evaluating cardiac function, and directing treatment choices for ailments such as heart attacks, coronary artery disease, and atrial fibrillation, diagnostic imaging modalities like computed tomography (CT), magnetic resonance imaging (MRI), echocardiography, and coronary angiography are essential. The trend toward preventive and precision-based cardiovascular care is reflected in the growing dependence on these imaging technologies.
According to the data reported in May 2024, cardiovascular diseases (CVDs) remain the leading cause of disability and premature mortality in Europe, accounting for over 42.5% of annual deaths, which translates to approximately 10,000 deaths per day. The burden is disproportionately higher among men, who are nearly 2.5 times more likely to die from CVDs than women. Additionally, younger populations aged 30-69 in Eastern Europe and Central Asia face nearly a fivefold higher risk compared to those in Western Europe. These alarming statistics underscore the urgent need for widespread screening and early diagnosis, thereby accelerating the adoption of advanced diagnostic imaging systems across healthcare settings.
Furthermore, improved detection rates of coronary artery disease, including asymptomatic and early-stage cases, have resulted from improvements in awareness and screening procedures. Since CAD frequently advances silently, more patients who were previously undiagnosed have been found thanks to improved screening programs. Because of this, early diagnosis and risk assessment are increasingly dependent on non-invasive and minimally invasive imaging technologies. The use of imaging devices in hospitals, diagnostic facilities, and specialized cardiac clinics has increased drastically as a result of this.
Overall, there is a persistent need for sophisticated diagnostic imaging solutions due to the increased incidence of coronary artery diseases brought on by aging populations, lifestyle modifications, and better detection. It is anticipated that the demand for precise, effective, and easily accessible imaging technologies likely to rise as healthcare systems place a greater emphasis on early diagnosis and preventive care, underscoring their critical role in controlling the worldwide burden of coronary heart disease.
Market Concentration & Characteristics
The coronary heart disease diagnostic imaging devices market is moderately concentrated, with a few major players such as GE HealthCare, Siemens Healthineers AG, and Koninklijke Philips N.V. holding significant shares. The market is technology-driven, relying on advanced imaging modalities such as CT, MRI, X-ray, and nuclear imaging. It requires high capital investment and strong regulatory compliance. Growth is supported by rising cardiovascular disease prevalence and demand for early diagnosis. Moreover, continuous innovation, including AI integration and non-invasive imaging solutions, is shaping competitive dynamics and expanding market opportunities globally.
The coronary heart disease diagnostic imaging devices industry is undergoing rapid innovation, driven by a shift toward non-invasive technologies. While traditional angiography is effective, it carries higher risks and costs. Advanced CT-based imaging, particularly coronary CT angiography, enables safer diagnosis and reduces unnecessary procedures. Clinical research from the University of Galway demonstrated that high-resolution cardiac CT scans achieved 99.1% feasibility in detecting coronary blockages, highlighting its transformative potential. Furthermore, AI integration is enhancing diagnostic accuracy, improving consistency, and streamlining workflows, thereby supporting wider adoption of advanced imaging solutions in cardiovascular care.
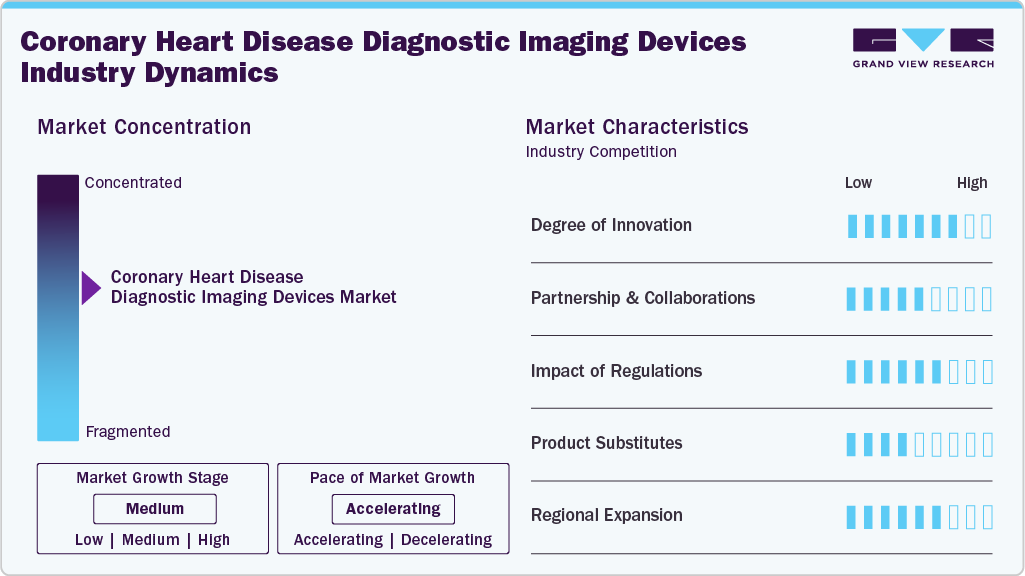
Partnerships and collaborations are essential in the coronary heart disease diagnostic imaging devices industry, helping companies expand technological capabilities and strengthen market presence. For instance, in February 2025, Royal Philips partnered with Myocardial Solutions to advance AI-enabled cardiac MRI solutions. This collaboration combines advanced MRI acquisition methods with AI-driven analytics to support earlier detection of heart disease, enhance diagnostic precision, and accelerate clinical decision-making, ultimately improving patient outcomes and workflow efficiency.
Regulatory approval plays a crucial role in shaping the coronary heart disease diagnostic imaging devices industry by ensuring safety, efficacy, and quality of imaging systems. Strict guidelines from authorities such as the U.S. Food and Drug Administration and European Medicines Agency govern product approvals and commercialization. While these regulations enhance patient safety and clinical reliability, they can also increase time-to-market and development costs for manufacturers. Compliance requirements drive continuous innovation and quality improvements. In addition, evolving regulatory frameworks for AI-based imaging solutions are influencing adoption, ensuring accuracy, data security, and standardized performance across advanced diagnostic technologies.
Product substitutes in the coronary heart disease diagnostic imaging devices industry include alternative diagnostic approaches that reduce reliance on advanced imaging systems. Traditional methods such as electrocardiograms (ECG), stress tests, and blood biomarkers are commonly used for initial assessment of coronary conditions. Additionally, invasive procedures such as coronary angiography remain substitutes for certain imaging modalities despite higher risks. Emerging non-imaging techniques, including wearable cardiac monitoring devices and digital health tools, are also gaining traction. While these substitutes may offer cost advantages and accessibility, they often lack the detailed anatomical insights provided by advanced imaging technologies, limiting their effectiveness in comprehensive diagnosis and treatment planning.
Regional expansion in the coronary heart disease diagnostic imaging devices market is accelerating across key global regions, driven by increasing healthcare investments and demand for advanced diagnostics. Companies are strengthening their presence through infrastructure development and strategic expansion. For instance, in March 2026, Fujifilm Sonosite, Inc. enhanced diagnostic capabilities in Tamil Nadu by deploying advanced imaging systems at Porunai Hospitals, improving access and clinical efficiency. Similarly, in September 2025, Esaote S.p. A expanded its operations across Europe through multiple subsidiaries, reinforcing its research, production, and service capabilities to support growing regional demand.
Modality Insights
The X ray segment held the largest revenue share of 26.3% in 2025, driven by its established role as the gold standard for diagnosing and managing coronary artery disease. Coronary angiography delivers high diagnostic accuracy of approximately 97.6%, enabling timely interventions and improved outcomes. In addition, X-ray systems support both diagnosis and treatment by guiding procedures such as angioplasty and stenting, enhancing workflow efficiency.
Technological advancements are further strengthening the adoption of X-ray imaging systems. Recent studies highlight improvements in radiation management, image processing, and procedural techniques, which are helping to enhance safety and efficiency. In January 2025, a study published in the Indian Heart Journal evaluated radiation exposure in coronary angiography using trans-radial versus trans-femoral approaches. The study, conducted on 910 patients, found that the trans-radial approach was associated with higher radiation exposure and longer fluoroscopy time compared to the femoral approach. These findings highlight the ongoing need for procedural optimization and radiation management in X-ray-based cardiac imaging. Moreover, emerging AI and image-guided technologies are improving vessel visualization, device tracking, and procedural precision, enabling better clinical outcomes and supporting broader adoption in complex cardiac interventions.
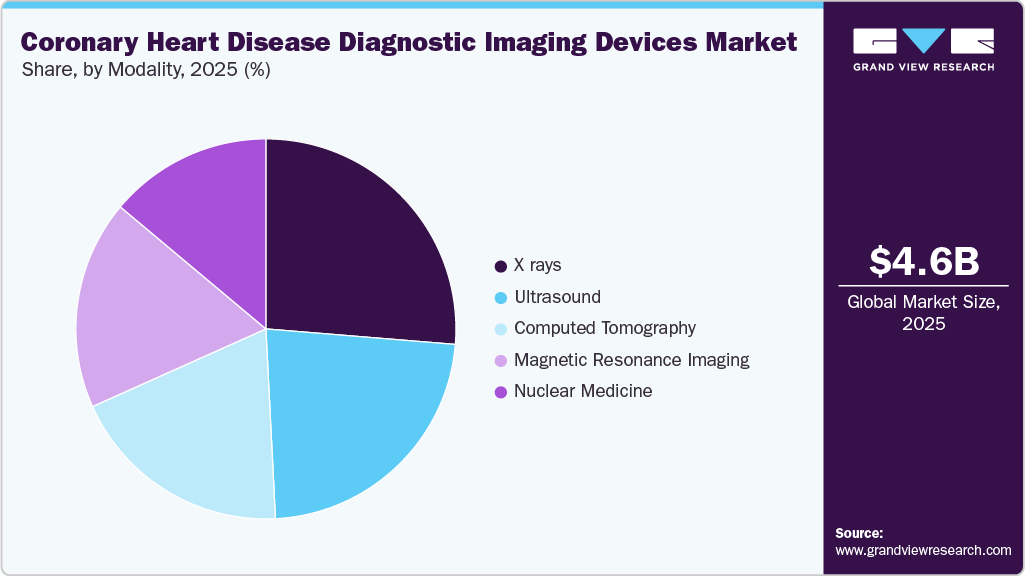
The computed tomography (CT) segment is projected to witness the fastest CAGR during the forecast period, driven by the increasing preference for non-invasive diagnostic approaches. Unlike traditional invasive coronary angiography, which involves higher risks and costs, CT-based imaging enables safe and effective exclusion of significant coronary disease, minimizing unnecessary procedures. A clinical evaluation by the National Library of Medicine in October 2025 highlighted the growing use of coronary CT angiography in pre-procedural assessments. Additionally, a 2024 study by the University of Galway demonstrated 99.1% feasibility in detecting coronary blockages, underscoring its potential to transform cardiac diagnostics.
Regional Insights
North America coronary heart disease diagnostic imaging devices market dominated the global industry with a share of 39.5% in 2025, driven by strong healthcare infrastructure, and early adoption of advanced imaging technologies. The region benefits from widespread availability of modalities such as CT, MRI, echocardiography, and nuclear imaging, along with favorable reimbursement systems and strong clinical guidelines promoting early cardiovascular screening. One of the primary drivers of market growth in North America is the high prevalence and mortality associated with cardiovascular diseases. According to the report published by Centers for Disease Control and Prevention (CDC) in October 2024, cardiovascular disease causes a death approximately every 34 seconds in the U.S., highlighting the urgent need for accurate and timely diagnosis. In Canada as well, heart disease remains one of the leading causes of death, with millions of adults affected by ischemic heart disease and related conditions. This significant patient pool drives consistent demand for diagnostic imaging technologies used in screening, risk stratification, and disease monitoring.
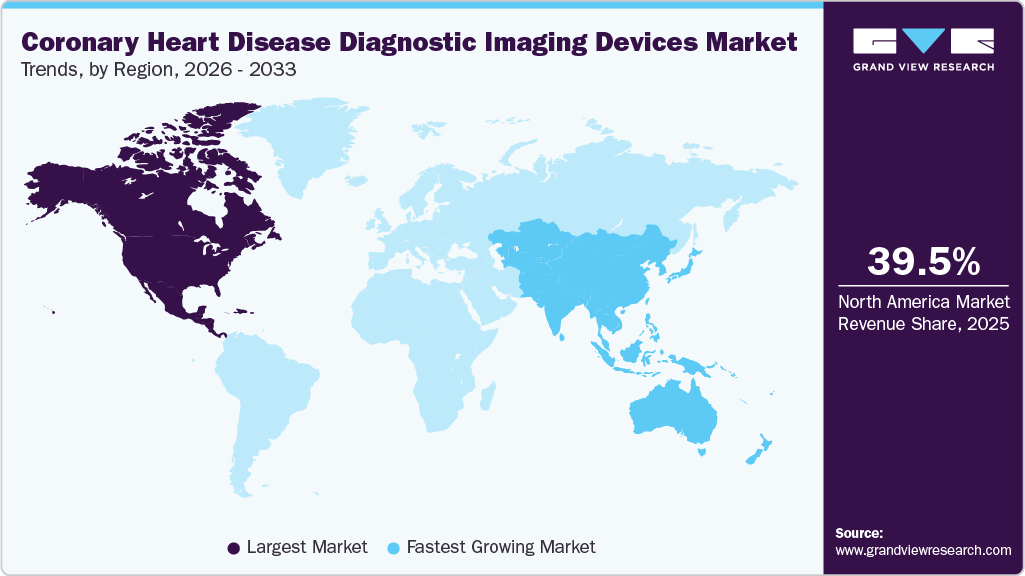
Another major growth factor is the advanced healthcare infrastructure and high adoption of imaging technologies. North America has a significantly higher density of imaging systems such as CT and MRI scanners compared to other regions, enabling faster diagnosis and improved patient outcomes. For instance, the U.S. has substantially more MRI and CT scanners per million populations than Canada, reflecting the region’s strong technological penetration and investment in diagnostic capabilities. Hospitals and cardiac centers increasingly rely on these modalities for both routine screening and emergency cardiac assessments, particularly for acute coronary syndromes.
U.S. Coronary Heart Disease Diagnostic Imaging Devices Market Trends
In 2025, the U.S. accounted for the largest share of the coronary heart disease diagnostic imaging devices market in the North America. Several major drivers are accelerating market growth in the U.S., anchored primarily in the high prevalence and health burden of CHD and the evolving role of advanced diagnostics in early detection and risk stratification. In addition, innovations enhancing clinical performance are influencing adoption trends; for example, in September 2024, the U.S. Food and Drug Administration (FDA) approved GE Healthcare’s Flyrcado radiotracer for PET myocardial perfusion imaging, offering improved diagnostic accuracy and workflow efficiency compared to standard techniques and expanding PET’s clinical use in difficult‑to‑image patients like those with high BMI or women. These developments reflect how technological breakthroughs and regulatory approvals are shaping the competitive landscape by enabling providers to deliver more precise and accessible coronary diagnostics.
Europe Coronary Heart Disease Diagnostic Imaging Devices Market Trends
Coronary heart disease diagnostic imaging devices market in Europe accounted for a considerable share in 2025. Across the region, coronary heart disease continues to be a major contributor to morbidity and mortality, placing a significant strain on public health systems as well as economic resources. The market is also shaped by artificial intelligence (AI) reimbursement breakthroughs, workflow digitization, regulatory reinforcement, and a renewed focus on appropriate imaging utilization. Between 2024 and 2026, Europe has moved decisively from pilot adoption toward scaled, system-level integration of advanced imaging technologies. According to OECD report published in 2025, cardiovascular diseases (CVDs) were responsible for affecting approximately 62 million deaths in the European Union, with CHD and cerebrovascular diseases accounting for over a third of these deaths. The aging demographic profile, persistent lifestyle‑related risk factors (such as smoking, obesity, and hypertension), and high healthcare costs reinforce the need for accurate, timely, and cost‑effective CHD diagnostics. These epidemiological realities form a foundational driver of demand for advanced diagnostic imaging in Europe.
Germany coronary heart disease diagnostic imaging devices market dominated with the largest revenue share of 23.4% in 2025. Several pivotal factors are accelerating growth in Germany’s CHD diagnostic imaging devices market. Public health policy changes have directly influenced imaging utilization as reimbursement for coronary CT angiography (CCTA) under statutory insurance in Germany since January 2025 has made non‑invasive CHD assessment more economically attractive for providers, potentially shifting volumes from invasive angiography to CT‑based diagnostics. In addition, clinical consensus and guideline updates supporting advanced functional imaging such as cardiac MRI for CHD diagnostics are encouraging hospitals to invest in multimodality imaging capabilities. Moreover, collaborative research partnerships aimed at enhancing heart imaging approaches including international consortia based in Germany are promoting innovation and adoption of next‑generation imaging modalities.
Coronary heart disease diagnostic imaging devices market in UK held the second largest market share in 2025. An ageing population with a high prevalence of chronic cardiovascular conditions has substantially increased demand for high‑precision imaging solutions such as CT angiography and echocardiography. Additionally, NHS and private healthcare providers are investing in cutting‑edge imaging systems to reduce diagnosis times and enhance clinical outcomes. For instance, advanced AI‑powered imaging technologies such as HeartFlow analysis are being adopted across multiple NHS hospitals, speeding up CHD diagnosis while reducing the need for invasive procedures, and saving millions of pounds in healthcare costs. This initiative has been implemented in at least 56 hospitals in England with a significant impact on patient management and NHS efficiency.
Italy coronary heart disease diagnostic imaging devices market is anticipated to witness a significant CAGR of 9.0% during the forecast period. The market is growing due to the rising prevalence of cardiovascular diseases in Italy and the increasing focus on early diagnosis and preventive care. Hospitals are adopting advanced imaging devices like AI-enabled CT and MRI systems to improve accuracy and efficiency. For instance, Italian researchers developed AI software called “THAITI” to enhance cardiac MRI image quality and reduce reliance on operator skill. These factors collectively drive the growth of market across the country.
Asia Pacific Coronary Heart Disease Diagnostic Imaging Devices Market Trends
The APAC region is expected to grow at the fastest CAGR during the forecast period. The growth is also driven by its large population base and rising demand for early and accurate cardiac diagnosis. Growing popularity towards non-invasive imaging modalities such as CT, MRI, and nuclear imaging to improve clinical outcomes and reduce reliance on invasive procedures. The rising aging populations, urbanization, sedentary lifestyles, and rising incidence of diabetes and hypertension contributes to the increasing prevalence of cardiovascular diseases across region. This epidemiological shift is significantly increasing the demand for diagnostic imaging technologies that enable early detection and management of coronary artery disease. The broader diagnostic imaging ecosystem in the region is expanding rapidly as healthcare systems respond to this burden, with chronic diseases identified as a key factor driving imaging demand.
Coronary heart disease diagnostic imaging devices market in China accounted for the largest share in the Asia Pacific in 2025, due to leading global healthcare technology companies actively competing for market share by supplying advanced imaging systems used in the diagnosis and management of coronary artery disease. Key players in this space include Koninklijke Philips, Fujifilm Sonosite, Inc., Shimadzu Corporation, and Shenzhen Mindray Bio-Medical Electronics Co., Ltd. These companies offer a wide range of diagnostic imaging technologies such as high‑resolution CT scanners, MRI systems, and ultrasound devices that provide critical insights into cardiac anatomy and function. Competition in China centers on product performance, imaging precision, integration of artificial intelligence (AI) for faster and more accurate interpretation, and strong local support services that enable hospitals and specialty cardiac centers maximize clinical utility.
Japan coronary heart disease diagnostic imaging devices market held the second largest market share in the Asia Pacific region. Key competitors such as GE Healthcare, Shimadzu Corporation, Koninklijke Philips, Siemens Healthineers AG, and Fujifilm Sonosite, Inc., are actively competing for contracts with hospitals, specialty heart centers, and diagnostic imaging facilities. These companies offer a broad portfolio that includes high‑resolution CT scanners, MRI systems, and ultrasound platforms all of which are essential for the accurate and early detection of coronary artery disease and other cardiac conditions. Competition is centered on innovation in imaging clarity, workflow automation, AI‑enabled analysis, and comprehensive service and maintenance support, as providers seek solutions that reduce scan times, improve diagnostic confidence, and integrate with digital healthcare systems.
Coronary heart disease diagnostic imaging devices market in India is witnessing strong growth, supported by multiple key factors. The rising prevalence of cardiovascular diseases is increasing the need for accurate and early diagnostic solutions. Healthcare facilities are increasingly adopting advanced CT and MRI systems to improve cardiac imaging and enable timely intervention. Additionally, efforts to bridge clinical skill gaps are supporting market expansion. For instance, in February 2025, Philips and Medtronic partnered to train over 300 clinicians in multi-modality imaging, enhancing diagnostic accuracy and strengthening cardiac care capabilities across the country.
Latin America Coronary Heart Disease Diagnostic Imaging Devices Market Trends
coronary heart disease diagnostic imaging device market in Latin America is witnessing demand due to an aging population, changing lifestyles leading to increased cardiovascular risk factors and improving healthcare infrastructure. Countries including Brazil, Mexico, and Argentina are vital contributors to this growth, focusing on early detection and management of heart diseases. Cultural factors such as preference for non-invasive diagnostic procedures influence market trends in Latin America.
Estimated Prevalence of Key CHD Risk Factors in Latin America
Risk Factors
Estimated Prevalence
Apo B / Apo A-1 Imbalance
42.0%
Tobacco Use
48.1%
Diabetes Mellitus
9.5%
Arterial Hypertension
29.1%
High Waist/Hip Ratio
48.6%
Depression
28.9%
Permanent Stress
6.8%
Lack of Regular Exercise
22.0%
Alcohol Consumption
19.4%
Low Fruit & Vegetable Intake
15.0%
Source: Global Heart Journal
Brazil coronary heart disease (CHD) diagnostic imaging device market is expanding due to several distinct growth drivers. Cardiovascular diseases remain the leading cause of death in Brazil, accounting for approximately 374,000 deaths annually, highlighting the substantial clinical need for early diagnosis and monitoring of coronary conditions. Despite gradual declines in mortality rates over time, ischemic heart disease continues to represent a major proportion of cardiovascular-related deaths, with notable regional disparities in disease outcomes. This persistent disease burden is creating sustained demand for diagnostic imaging modalities such as CT, MRI, nuclear imaging, and echocardiography, particularly in urban healthcare centers.
Middle East and Africa Coronary Heart Disease Diagnostic Imaging Devices Market Trends
Coronary heart disease diagnostic imaging devices Market in MEA is expected to grow at the lucrative growth, driven by rising healthcare investments, expansion of private hospital networks, and increasing adoption of advanced imaging and AI-enabled diagnostics. Major regional platforms such as Arab Health and World Health Expo (WHX) have become catalysts for technology diffusion, partnerships, and localization strategies. In February 2025, United Imaging Healthcare reinforced its growing footprint in MEA at Arab Health 2025 in Dubai, unveiling next-generation MRI, PET/CT, radiotherapy, angiography, and CT platforms. Flagship systems such as ultra-high-field MRI, digital PET/CT, precision radiotherapy, and AI-powered CT solutions highlighted the region’s shift toward high-end, data-driven imaging. During the event, United Imaging formalized strategic collaborations across Turkey, Saudi Arabia, Kuwait, Jordan, Kenya, and Malaysia, supporting advanced molecular imaging, oncology diagnostics, and AI-assisted workflows. Installations such as whole-body ultra-high-field MRI in Turkey and digital PET/CT systems in Kuwait and Jordan reflect growing regional demand for precision medicine and theranostics.
The coronary heart disease (CHD) diagnostic imaging device market in South Africa is influenced by the country’s growing burden of cardiovascular diseases and increasing shift toward non-communicable disease management. Cardiovascular diseases account for approximately 17-18% of total deaths in South Africa, meaning nearly one in six deaths is attributed to heart disease or related conditions. South Africa’s medical imaging market is evolving amid a complex mix of workforce shortages, uneven infrastructure distribution, and accelerating adoption of advanced technologies. As of October 2024, the country faces a critical radiology capacity gap, with approximately one radiologist per 100,000 people, compared to around 13 per 100,000 in Europe. An estimated 700 active radiologists serve the country, with roughly 60% working in the private sector and the remainder in academic institutions. Annual training output of around 20 new radiologists per year is sufficient to maintain existing numbers but inadequate to support market expansion, particularly as skilled professionals continue to emigrate.
Key Coronary Heart Disease Diagnostic Imaging Devices Company Insights
Some of the key players operating in the industry include GE Healthcare, Koninklijke Philips, Siemens Healthineers AG, Fujifilm Sonosite. Company’s key strategies include understanding the strengths and weaknesses of major market participants, anticipating future market trends, opportunities, and challenges, and making proactive decisions based on insights into emerging technologies and changing consumer preferences.
Key Coronary Heart Disease Diagnostic Imaging Devices Companies:
The following key companies have been profiled for this study on the coronary heart disease diagnostic imaging devices market.
- GE Healthcare
- Koninklijke Philips
- Siemens Healthineers AG
- Fujifilm Sonosite, Inc.
- Canon Medical Systems Corporation
- Samsung Medison Co. Ltd
- Shimadzu Corporation
- Shenzhen Mindray Bio-Medical Electronics Co., Ltd
- Arineta Ltd
- Esaote SpA
Recent Developments
-
In March 2026, Philips received FDA 510(k) clearance for its AI-powered SmartHeart, which automates cardiac MRI planning in under 30 seconds. It improves workflow consistency, reduces operator variability, and enhances accessibility. The solution also minimizes breath-holds, boosts patient comfort, and supports faster, more accurate cardiac imaging for improved clinical efficiency and outcomes.
-
In February 2026, GE HealthCare received U.S. FDA 510(k) clearance and CE mark for Allia Moveo, expanding its clinical use. Allia Moveo is a mobile, AI-enabled imaging system that provides real-time high-resolution X-ray guidance for minimally invasive cardiovascular procedures, improving efficiency and procedural safety.
-
In November 2025, Canon Medical Systems received FDA 510(k) clearance for its Alphenix 4D CT with Aquilion ONE/INSIGHT Edition. This enables diagnosis, planning, treatment, and verification in one room, eliminating the need for patient transfer between departments.
-
In May 2024, Samsung acquired 100% shares of Sonio SAS, a fetal ultrasound AI software company. Sonio has developed IT solutions and AI assistant features for obstetrics and gynecology ultrasound that assist medical professionals in the evaluation and documentation of ultrasound exams.
Coronary Heart Disease Diagnostic Imaging Devices Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 4.98 billion
Revenue forecast in 2033
USD 7.73 billion
Growth rate
CAGR of 6.5% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Modality and region
Regional scope
North America; Europe; Asia Pacific; Latin America; and MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait.
Key companies profiled
GE Healthcare; Koninklijke Philips; Siemens Healthineers AG; Fujifilm Sonosite Inc.; Canon Medical Systems Corporation; Samsung Medison Co. Ltd; Shimadzu Corporation; Shenzhen Mindray Bio-Medical Electronics Co., Ltd; Arineta Ltd; Esaote SpA
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options Global Coronary Heart Disease Diagnostic Imaging Devices Market Report Segmentation
This report forecasts revenue growth at global, regional & country levels and provides an analysis on the industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global coronary heart disease diagnostic imaging devices market report based on modality and region:
-
Modality Outlook (Revenue, USD Million, 2021 - 2033)
-
Computed Tomography
-
X rays
-
Ultrasound
-
Magnetic Resonance Imaging
-
Nuclear Medicine
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










