- Home
- »
- Medical Devices
- »
-
Europe Foot And Ankle Devices Market Size Report, 2033GVR Report cover
![Europe Foot And Ankle Devices Market Size, Share & Trends Report]()
Europe Foot And Ankle Devices Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Foot & Ankle Trauma, Foot & Ankle Reconstruction, Foot & Ankle Orthobiologics, Foot & Ankle Soft Tissue Repair), By End-use, By Country, And Segment Forecasts
- Report ID: GVR-4-68040-825-9
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Europe Foot And Ankle Devices Market Summary
The Europe foot and ankle devices market size was estimated at USD 1,067.3 million in 2025 and is projected to reach USD 1,716.9 million by 2033, growing at a CAGR of 6.2% from 2026 to 2033. The rising incidence of musculoskeletal disorders, including fractures, deformities, and degenerative conditions, in addition to an aging population with expanding orthopedic needs, is driving the European industry.
Key Market Trends & Insights
- Germany's foot and ankle devices market dominated the global market in 2025 and accounted for the largest revenue share of 23.2%.
- Spain foot and ankle devices market is anticipated to register the fastest growth rate during the forecast period.
- In terms of product, the foot and ankle (F&A) trauma segment held the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1,067.3 Million
- 2033 Projected Market Size: USD 1,716.9 Million
- CAGR (2026-2033): 6.2%
Treatment options are growing owing to developments in minimally invasive surgical methods, innovative implant designs, and the use of 3D-printed patient-specific implants. In October 2024, an AFAS-ESSKA update highlighted Europe’s growing role in advancing sports-injury treatment, underscored by its collaboration with MIFAS to support international arthroscopy training. The initiative strengthened Europe’s influence in ankle and foot education as faculty from France and Spain led global clinical and cadaveric training programs.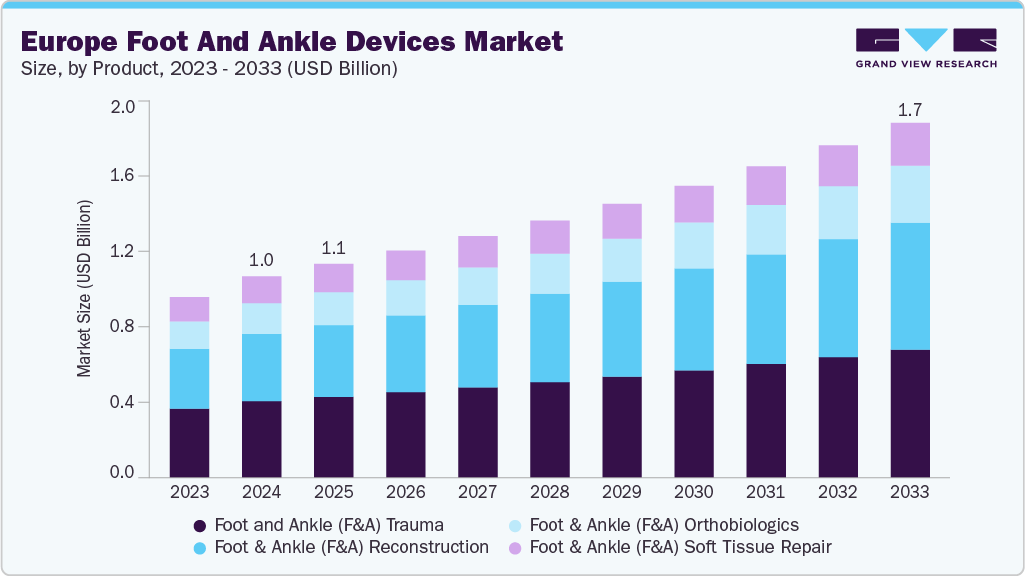
The European foot and ankle devices market is driven by aging-related musculoskeletal disorders, rising sports-related injuries, and increased awareness of foot and ankle health. Growth is supported by innovations such as minimally invasive procedures, 3D-printed implants, and advanced devices, along with favorable reimbursement policies that promote early diagnosis and adoption of treatment solutions.
Rising Incidence of Musculoskeletal Disorders
The demand for devices in Europe is largely driven by the increasing prevalence of musculoskeletal disorders. As people age, conditions such as osteoporosis, hallux valgus, and anklebone instability become more common, necessitating supportive implants and surgical procedures. Hospitals and clinics are expanding their treatment capabilities as part of this demographic trend that emphasizes orthopedic care. In May 2024: European Journal of Pediatrics reported 687,753 foot and ankle events among 416,137 children and young people (CYP) aged 0-18 years in England between 2015 and 2021. Consultations were highest in males aged 10-14 years, with musculoskeletal issues (34%) and unspecified pain (21%) being the most common.
Increasing Number of Sports Injuries and Road Traffic Accidents
The prevalence of injuries requiring orthopedic intervention has increased due to growing awareness of ankle health and increased participation in sports and physical activities. Access to advanced therapies is being made easier by supportive reimbursement policies and coverage provided by healthcare systems in a number of European nations. These factor driver demand growth for both clinical and elective procedures by promoting prompt diagnosis, intervention, and adoption of innovative devices. In November 2024, an article published in Sensors reviewed wearable technology used to assess clinical outcomes in foot and ankle disorders, noting that recent studies increasingly validate wearables for tracking gait, balance, and post-treatment recovery.
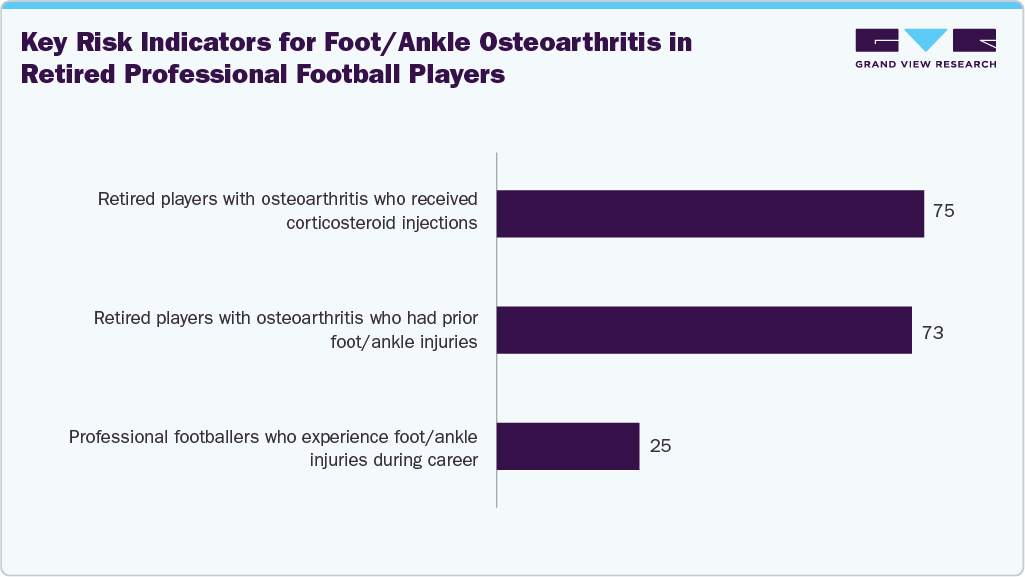
In October 2025, an article published in Rheumatology reported that 73% of retired UK male footballers with osteoarthritis had a history of injuries, while 75% had received corticosteroid injections during their careers. The study highlighted that about 25% of players experience such injuries during active play, underscoring a strong link between early-career joint damage and later-life osteoarthritis.
Chronic Conditions And Foot & Ankle Burden
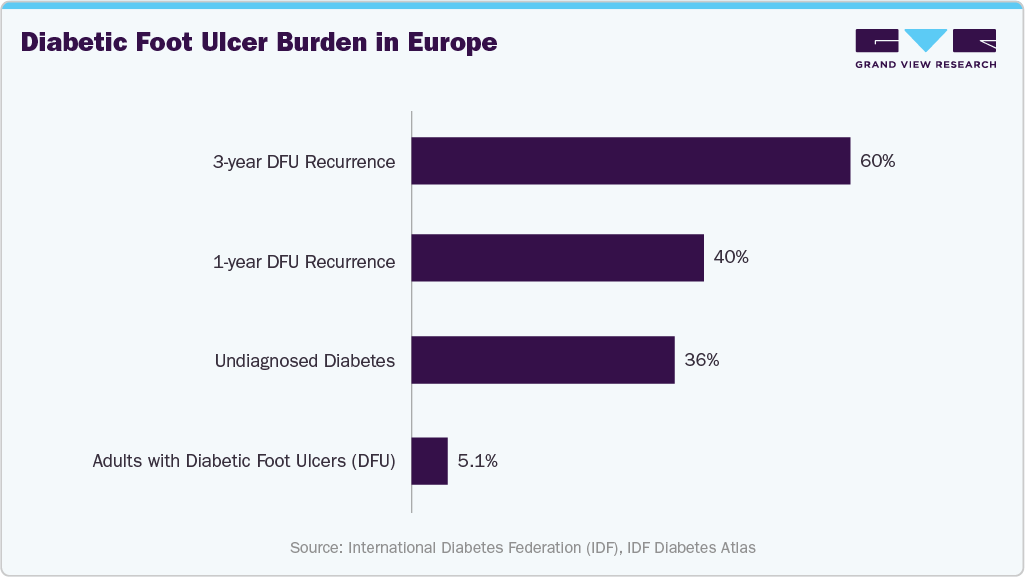
Chronic diseases also contribute significantly to foot and ankle treatment demand. Diabetic foot ulcers (DFU) affect approximately 5.1% of adults with diabetes in Europe, with 40% experiencing recurrence within one year and nearly 60% within three years. These complications drive demand for specialized interventions, surgical management, and preventive care solutions.
Market Concentration & Characteristics
The development of patient-specific 3D-printed devices, bioresorbable materials, and advanced implants is driving the high level of innovation in the European foot and ankle devices industry. The use of minimally invasive surgical techniques and robotic-assisted procedures is expanding, improving patient recovery and accuracy. In March 2025, Orthofix received CE Mark for the TrueLok Elevate Transverse Bone Transport System, enabling its clinical use across Europe. The system supports limb preservation and complex foot and ankle reconstruction procedures.
The European market for foot and ankle devices sees a moderate number of mergers and acquisitions, which happen frequently to strengthen regional distribution networks, expand product portfolios, or acquire specialized technologies. In September 2024, Stryker Corporation strengthened its foot and ankle portfolio by acquiring 4WEB Medical’s Osteotomy Truss System (OTS) and Ankle Truss System (ATS).
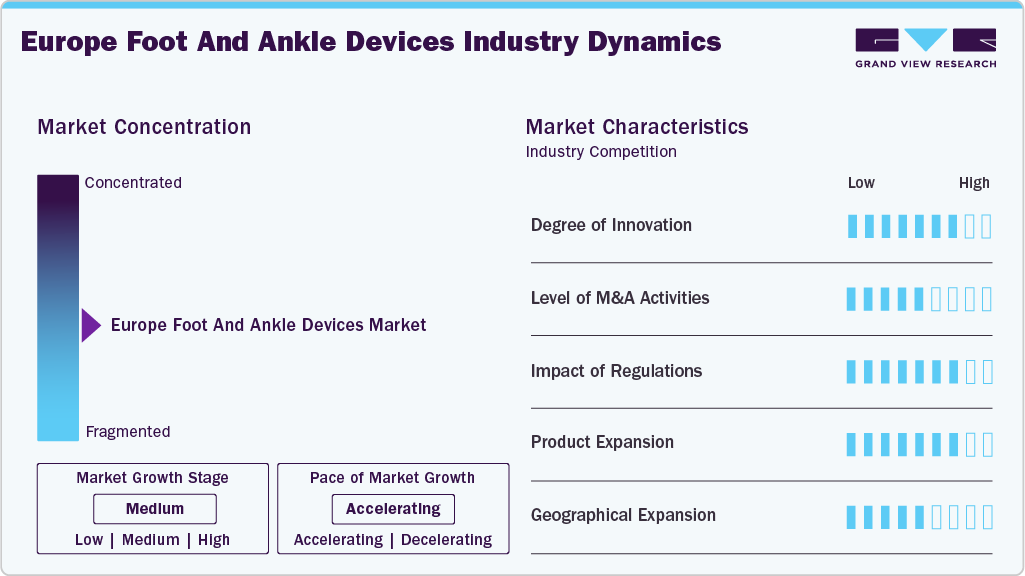
The industry for foot and ankle devices is significantly shaped by regulatory compliance. Devices approved with the European CE mark are able to meet stringent safety, performance, and clinical evidence requirements. To comply with regulatory requirements, businesses must conduct rigorous clinical trials and adhere to medical device directives, which influence the pace at which products are introduced and encourage investment in advanced research and documentation.
Key players are actively expanding their portfolios by launching innovative implants, fixation systems, and biologics. The introduction of modular implants, minimally invasive tools, and hybrid solutions that combine orthobiologics with traditional hardware allows companies to address a broader range of conditions and surgical needs, enhancing their presence across hospitals and specialized orthopedic centers.
Several companies are targeting expansion across European countries to increase their market penetration. By establishing localized sales and distribution networks, forming partnerships with leading orthopedic hospitals, and providing surgeon training programs, market players introduce advanced foot and ankle devices to new regions while addressing country-specific clinical needs and regulatory requirements.
Product Insights
By product, the foot and ankle (F&A) trauma segment dominated the market in 2025 with a revenue share of 37.9%. This dominance is driven by the high incidence of fractures, dislocations, and acute injuries in both the elderly and active populations. In November 2025, a Cureus article reported that a major UK trauma centre seldom used early weight-bearing after ankle fixation, with most patients starting only after six weeks.
The foot & ankle (F&A) reconstruction segment is anticipated to witness the fastest growth over the forecast period, fueled by rising cases of deformities, degenerative conditions, and post-traumatic complications. In May 2025, ESSKA introduced the second edition of its Ankle Ligament Pathology certification module, expanding specialised training for European surgeons. The updated programme focuses on improving competency in treating ligament injuries through refreshed coursework and hands-on cadaveric skills assessment.
End Use Insights
By end use, the hospitals segment dominated the market in 2025 with a revenue share of 66.0% and is anticipated to grow at the fastest CAGR over the forecast period. This dominance is attributed to the high volume of complex anklebone procedures performed in hospital settings, including trauma surgeries, reconstructions, and revision cases. Hospitals benefit from access to advanced surgical infrastructure, specialized orthopedic teams, and comprehensive post-operative care, which collectively support strong demand for a wide range of devices.
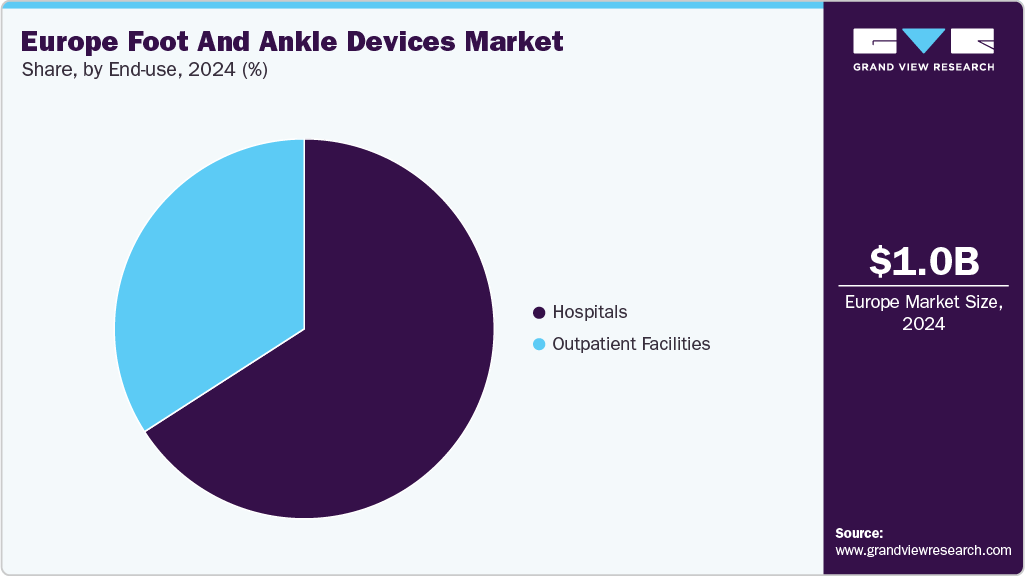
The outpatient facilities segment is anticipated to witness significant growth over the forecast period, driven by the increasing shift toward minimally invasive procedures and same-day surgeries. Advances in portable surgical tools, streamlined fixation systems, and enhanced pain management protocols enable more procedures to be safely performed in outpatient settings. The growing patient preference for shorter hospital stays and cost-effective treatment options is further driving the adoption of devices in ambulatory care centers across Europe.
Country Insights
Germany Foot And Ankle Devices Market Trends
Germany foot and ankle devices market led with a revenue share of 23.2% in 2025. The market is driven by an aging population with a high prevalence of musculoskeletal disorders, well-established healthcare infrastructure, and advanced orthopedic surgical capabilities. The adoption of innovative implants, minimally invasive procedures, and patient-specific solutions further supports market growth, making Germany a key hub for device demand in Europe. In May 2025, Thetis Medical reported that Germany recorded an average incidence of Achilles tendon ruptures at 7.77 cases per 100,000 person-years between 1991 and 2015.
Spain Foot And Ankle Devices Market Trends
Spain foot and ankle devices market is anticipated to register a significant growth rate during the forecast period. This growth is driven by increasing awareness of foot and ankle health, rising sports-related injuries, and expanding access to advanced orthopedic care. Investments in modern hospitals and outpatient surgical centers, along with the adoption of minimally invasive and 3D-printed patient-specific implants, are facilitating higher procedure volumes and accelerating market expansion in the country. In October 2024, Reports (MDPI) published an article discussing the management of chronic plantar plate tears of the second metatarsophalangeal joint.
Key Europe Foot And Ankle Devices Company Insights
Key participants in the Europe foot and ankle devices market are focusing on developing innovative business growth strategies in the form of product portfolio expansions, partnerships & collaborations, mergers & acquisitions, and business footprint expansions.
Key Europe Foot And Ankle Devices Companies:
- Stryker Corporation
- Johnson & Johnson (De PuySynthes)
- Zimmer Biomet
- Smith+Nephew
- Bioventus LLC
- Enovis Corp.
- Acumed LLC
- Arthrex, Inc.
- Anika Therapeutics, Inc
- Orthofix, Inc.
- Globus Medical
- CONMED Corporation
Recent Developments
-
In June 2025, Foot Innovate partnered with Voom Medical Devices to advance minimally invasive bunion surgery education and share clinical innovations. The partnership enables Foot Innovate to connect surgeons with cutting-edge solutions and support ongoing professional education
-
In March 2025, Orthofix received the European CE Mark for its TrueLok Elevate Transverse Bone Transport System, enabling its clinical use across Europe. The system began limited rollout in key markets, with early procedures performed in the UK and Germany, supporting advanced limb-preservation and complex foot and ankle reconstruction needs in the region.
-
In November 2024, Osteotec secured exclusive rights to distribute Novastep’s validated foot and ankle surgical solutions in the UK and Ireland, expanding its orthopedic portfolio in the region.
Europe Foot And Ankle Devices Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1,129.5 million
Revenue forecast in 2033
USD 1,716.9 million
Growth rate
CAGR of 6.2% from 2026 to 2033
Actual data
2021 - 2025
Forecast data
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, end use, country
Country scope
Germany; UK; Spain; Italy; France; Norway; Denmark; Sweden
Key companies profiled
Stryker Corporation; Johnson & Johnson (De PuySynthes); Zimmer Biomet; Smith+Nephew; Bioventus LLC; Enovis Corp.; Acumed LLC; Arthrex, Inc.; Anika Therapeutics, Inc; Orthofix; Inc.; Globus Medical; CONMED Corporation
Customization scope
Free report customization (equivalent up to 8 analyst working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Europe Foot And Ankle Devices Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country level and provides an analysis of industry trends in each of the sub segments from 2021 to 2033. For this study, Grand View Research, Inc. has segmented the Europe foot and ankle devices market report on the basis of product, end use and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Foot & Ankle (F&A) Trauma
-
Ankle Trauma
-
Fibula fixation and syndesmosis repair systems
-
Talus and calcaneus trauma plates
-
Pilon fracture fixation sets
-
Intramedullary nails
-
-
Foot & Ankle (F&A) Reconstruction
-
Hallux Valgus / Bunion Correction
-
Hammertoe Correction
-
Midfoot Reconstruction
-
Hindfoot & Ankle Recon
-
Total Ankle Replacement (TAR)
-
Toe Joint Replacement
-
-
Foot & Ankle (F&A) Orthobiologics
-
Biologics
-
Wedges
-
-
Foot & Ankle (F&A) Soft Tissue Repair
-
Lateral ligament reconstruction
-
Achilles tendon repairs
-
Spring ligament and deltoid repair
-
Plantar plate repair systems
-
Flexor/extensor tendon augmentation
-
Soft tissue augmentation scaffolds
-
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Outpatient Facilities
-
-
Country Outlook (Revenue, USD Million, 2021 - 2033)
-
Germany
-
UK
-
Spain
-
Italy
-
France
-
Denmark
-
Norway
-
Sweden
-
Frequently Asked Questions About This Report
b. The Europe foot and ankle devices market size was estimated at USD 1,067.3 million in 2025 and is expected to reach USD 1,129.5 million in 2026.
b. The Europe foot and ankle devices market is expected to grow at a compound annual growth rate of 6.2% from 2026 to 2033 to reach USD 1,716.9 million by 2033.
b. Germany dominated the foot and ankle devices market with a revenue share of 23.2% in 2025. The market is driven by an aging population with a high prevalence of musculoskeletal disorders, well-established healthcare infrastructure, and advanced orthopedic surgical capabilities.
b. Some key players operating in the Europe foot and ankle devices market include Stryker Corporation, Johnson & Johnson (De PuySynthes), Zimmer Biomet, Smith+Nephew, Bioventus LLC, Enovis Corp., Acumed LLC, Arthrex, Inc., Anika Therapeutics, Inc, Orthofix, Inc., Globus Medical, and CONMED Corporation
b. Key factors that are driving the Europe foot and ankle devices market growth include rising incidence of musculoskeletal disorders, including fractures, deformities, and degenerative conditions, and an aging population with expanding orthopedic needs.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










