- Home
- »
- Medical Devices
- »
-
Hemorrhagic & Ischemic Stroke Treatment Devices Market, 2033GVR Report cover
![Hemorrhagic And Ischemic Stroke Treatment Devices Market Size, Share & Trends Report]()
Hemorrhagic And Ischemic Stroke Treatment Devices Market (2026 - 2033) Size, Share & Trends Analysis Report By Treatment Type (Aneurysm & Hemorrhagic Stroke Interventions, Ischemic Stroke Interventions, Cerebral Angioplasty & Vascular Remodeling), By End Use, And Regional Segment Forecasts
- Report ID: GVR-4-68040-843-2
- Number of Report Pages: 220
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Hemorrhagic And Ischemic Stroke Treatment Devices Market Summary
The global hemorrhagic and ischemic stroke treatment devices market size was estimated at USD 3.80 billion in 2025 and is projected to reach USD 6.24 billion by 2033, growing at a CAGR of 6.4% from 2026 to 2033. The rising global burden of stroke, aging populations, and increasing prevalence of risk factors such as hypertension and diabetes.
Key Market Trends & Insights
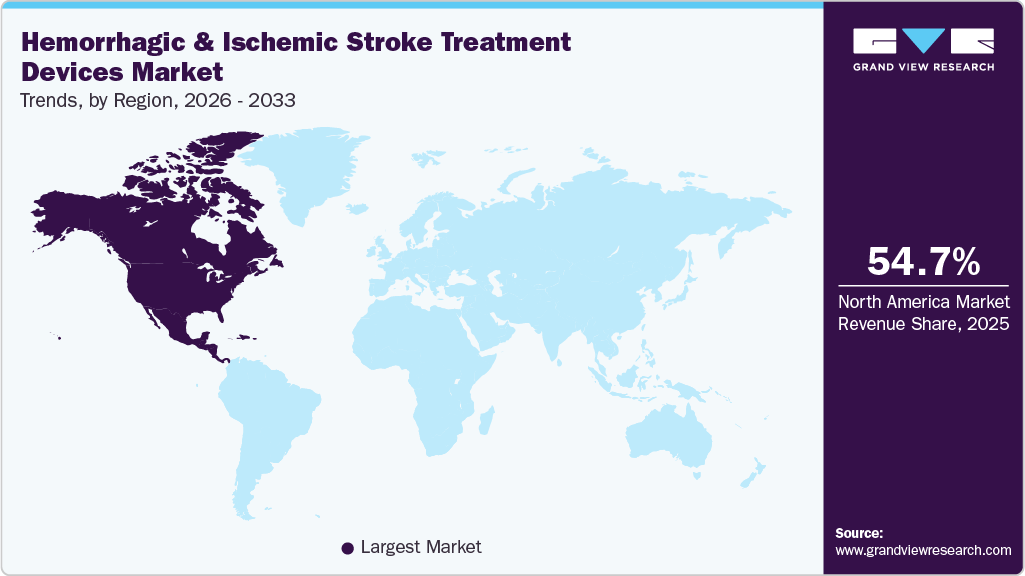
- North America dominated the hemorrhagic & ischemic stroke treatment devices market with the largest revenue share of 54.75% in 2025.
- The hemorrhagic & ischemic stroke treatment devices market in the U.S. accounted for the largest market revenue share in North America in 2025.
- By treatment type, the aneurysm & hemorrhagic stroke interventions segment led the market with the largest revenue share in 2025.
- By end use, the hospitals segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 3.80 Billion
- 2033 Projected Market Size: USD 6.24 Billion
- CAGR (2026-2033): 6.4%
- North America: Largest market in 2025
Growing adoption of minimally invasive endovascular procedures, along with improvements in device safety, precision, and clinical outcomes, is accelerating demand. In addition, the expansion of comprehensive stroke centers and the development of faster treatment pathways are supporting the wider utilization of advanced stroke treatment technologies.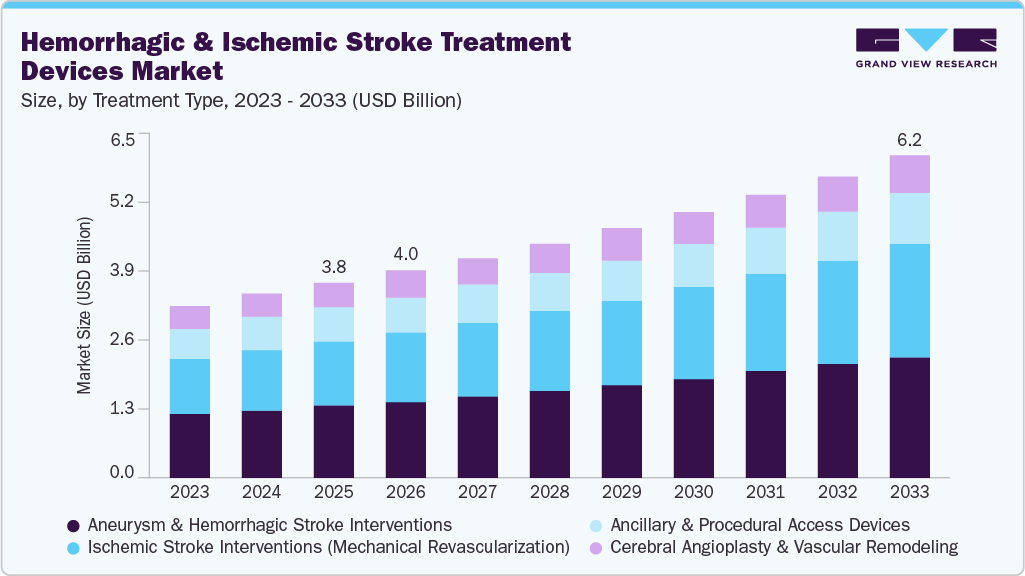
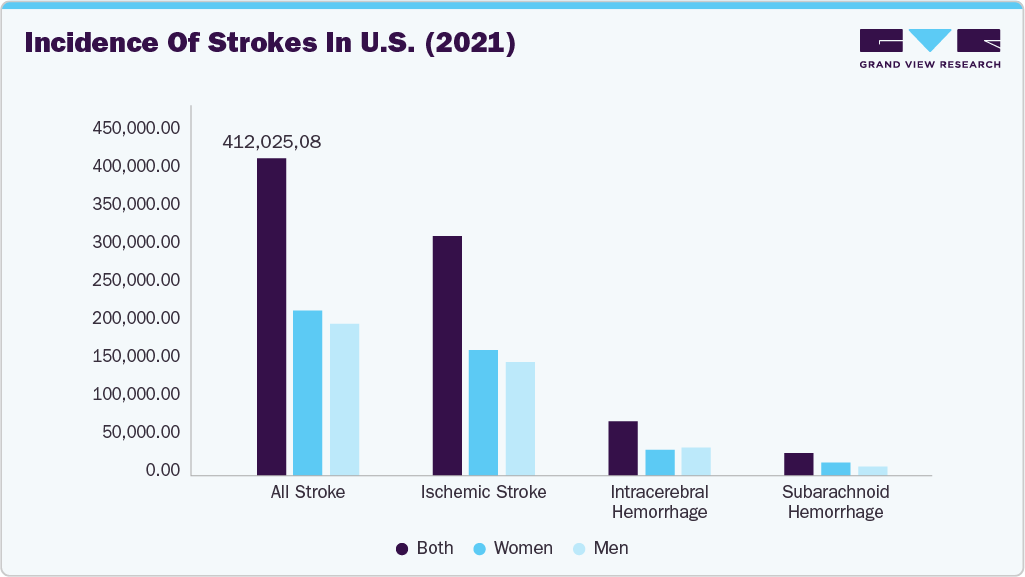
The rising incidence of hemorrhagic strokes is expected to significantly increase demand for hemorrhagic stroke treatment devices over the coming years. According to the Global Stroke Fact Sheet 2025 published by the World Stroke Organization, more than 12 million new stroke cases occur annually, and nearly one in four people worldwide will experience a stroke during their lifetime. The report indicates that intracerebral hemorrhage (ICH) accounts for 28.8% and subarachnoid hemorrhage (SAH) for 5.8% of all incident strokes, underscoring the substantial hemorrhagic stroke burden. The growing prevalence of this disease is anticipated to be a key factor supporting market growth during the forecast period.
The increasing focus on minimally invasive treatment strategies, including early minimally invasive evacuation of intracerebral hemorrhage (ICH) and endoscopic hematoma removal, is expected to be a key driver of market growth over the forecast period. Furthermore, ongoing clinical trials are supporting the development of technologies that improve procedural safety and clinical outcomes. For instance, in April 2024, NICO Corporation reported positive results from the ENRICH (Early MiNimally-invasive Removal of ICH) clinical trial, showing that early minimally invasive parafascicular surgery (MIPS) using its BrainPath and Myriad systems was safe and demonstrated superior outcomes compared with guideline-based medical management alone, the current standard of care for ICH.
The increasing volume of clinical trials conducted by research institutions, academic centers, and industry players is a key driver of the hemorrhagic stroke treatment devices market. These trials generate clinical evidence that validates the safety and effectiveness of minimally invasive and next-generation technologies, supporting regulatory approvals and clinician confidence. In addition, they facilitate innovation, expand treatment indications, and accelerate the adoption and commercialization of advanced devices, thereby driving sustained market growth.
Some of the notable clinical trials focusing on hemorrhagic stroke treatment devices are highlighted as follows:
Study Title
Conditions
Interventions
Sponsor
Enrollment
Primary Completion Date
Non-invasive Intracranial Pressure Monitoring to Improve Emergency Care in Brazil's Public Health System
Traumatic Brain Injury (TBI) Patients|Stroke Hemorrhagic|Stroke Ischemic
DEVICE: non-invasive intracranial pressure monitoring
Braincare USA Corp
300
12/31/2027
Evaluating Functional Outcomes of 3D-Printed Splints in Post-Stroke Upper Limb Recovery
Stroke|Stroke Rehabilitation|Hemiparesis of the Upper Limb Following Stroke
DEVICE: 3D Printed splint|DEVICE: conventional splint
King Saud University
100
9/30/2027
Post Market Clinical Follow-up of CODMAN CERTAS Programmable Valve
Hydrocephalus|Hydrocephalus in Children|NPH (Normal Pressure Hydrocephalus)|IIH - Idiopathic Intracranial Hypertension|Brain Tumor|Post-Traumatic Hydrocephalus|Hemorrhagic Stroke
DEVICE: CODMAN CERTAS Plus Programmable Valve
Integra LifeSciences Corporation
158
8/31/2027
Accelerated rTMS vs. Sham for Stroke Apathy
Apathy|Stroke|Stroke Sequelae|Stroke/Brain Attack|Stroke/ Cerebrovascular Accident (Ischemic or Hemorrhagic)|Motivation|Abulia
DEVICE: MagVenture MagPro Transcranial Magnetic Stimulation (TMS) System (Active)|DEVICE: MagVenture MagPro Transcranial Magnetic Stimulation (TMS) System (Sham)|OTHER: Brainsight Neuronavigation System
Medical University of South Carolina
40
6/30/2027
Source: ClinicalTrials.gov
The rapid advancement of neurovascular device technology is expected to play a crucial role in driving the market expansion. Industry participants are focusing on developing and commercializing next-generation solutions that improve procedural accuracy, safety, and treatment effectiveness. In addition, innovations such as enhanced device design, improved materials, and better imaging compatibility are helping clinicians manage complex cases more efficiently, further supporting market growth.
Market Concentration & Characteristics
The hemorrhagic and ischemic stroke treatment devices market exhibits a high degree of innovation, driven by continuous advancements in neurovascular engineering and minimally invasive technologies. Manufacturers are focusing on improved device designs, advanced biomaterials, and surface modifications to enhance safety, precision, and clinical outcomes. Innovation is further supported by clinical research activity and the rapid translation of trial results into commercial products, enabling more effective and patient-specific approaches to stroke treatment. For instance, in July 2025, Boston Scientific Corporation announced the launch of the Neuroform EZ Stent System in the U.S. and Europe. This fourth-generation intracranial aneurysm stent is designed to be used in combination with endovascular coiling for the treatment of wide-necked aneurysms. The company stated that commercial rollout would begin immediately across both regions.
The hemorrhagic and ischemic stroke treatment devices market shows a moderate to high level of merger and acquisition activity, as established players seek to strengthen their neurovascular portfolios and expand technological capabilities. Acquisitions are primarily focused on gaining access to innovative minimally invasive technologies, proprietary device platforms, and clinical pipelines. These strategic deals also help companies accelerate market entry, broaden geographic presence, and enhance competitiveness in a rapidly evolving stroke care landscape. In February 2025, Stryker announced the completion of its acquisition of Inari Medical, Inc. Inari is recognized for its innovative, non-thrombolytic solutions for the removal of venous thromboembolism (VTE) clots. This acquisition enhances Stryker’s peripheral vascular portfolio and strengthens its position in the fast-growing VTE treatment market.

Regulatory frameworks have a significant impact on the hemorrhagic and ischemic stroke treatment devices market by shaping product development, approval timelines, and clinical adoption. Stringent requirements for safety, clinical evidence, and post-market surveillance, as mandated by authorities such as the European Union Medical Device Regulation and other global regulators, increase development costs but ensure the production of high-quality and reliable devices. Furthermore, clear regulatory pathways and support for innovation to accelerate the adoption of advanced, minimally invasive stroke treatment technologies, influencing overall market growth.
The end-use concentration of the hemorrhagic and ischemic stroke treatment devices market is highly uneven toward tertiary care hospitals and comprehensive stroke centers, where advanced neurointerventional procedures are routinely performed. These settings account for the majority of device utilization due to the availability of specialized infrastructure, trained neurointerventionists, and 24/7 emergency services. While ambulatory and secondary hospitals are gradually increasing adoption, complex stroke interventions remain largely concentrated in high-volume, specialized centers.
Treatment Type Insights
The aneurysm & hemorrhagic stroke interventions segment dominates the market in 2025. The market is driven by the high clinical urgency and complexity associated with intracranial bleeding. Strong demand for devices such as coils, flow diverters, stents, and embolization systems support segment leadership, as these interventions are lifesaving and require specialized endovascular solutions. In addition, growing adoption of minimally invasive aneurysm repair techniques and improved clinical outcomes reinforced the segment’s dominant position within the overall market.
The ischemic stroke interventions (Mechanical Revascularization) segment is anticipated to grow at the fastest CAGR over the forecast period, driven by expanding clinical evidence supporting mechanical thrombectomy for large-vessel occlusions. The increasing availability of thrombectomy-capable stroke centers, wider treatment windows, and advancements in stent retrievers and aspiration systems are accelerating the adoption of this treatment. In addition, growing physician expertise and favorable guideline updates are enabling broader patient eligibility, further supporting the rapid growth of this segment.
End Use Insights
Hospitals segment dominated in 2025 in the hemorrhagic & ischemic stroke treatment devices market, due to their central role in acute stroke management and emergency care delivery. Hospitals-particularly tertiary and comprehensive stroke centers-are equipped with advanced imaging, neurointerventional suites, and multidisciplinary stroke teams required for complex procedures such as mechanical thrombectomy and aneurysm repair. The availability of 24/7 emergency services, increased patient inflow, and access to skilled specialists further reinforced hospitals as the primary end sers of stroke treatment devices.
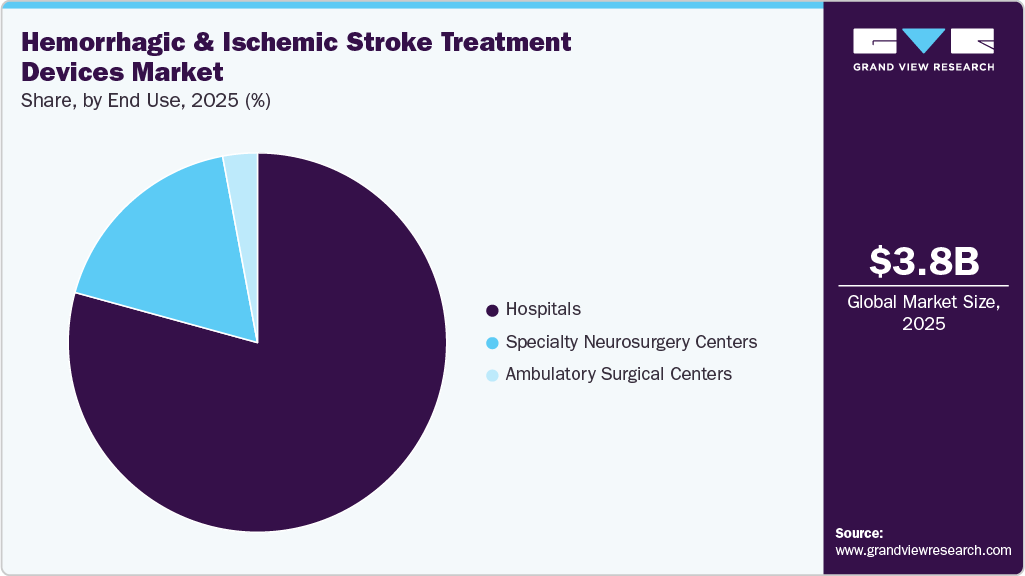
Ambulatory Surgical Centers (ASCs) segment is expected to be the fastest-growing end use segment over the forecast period. This growth is driven by the gradual shift of selected neurovascular and follow-up interventions to outpatient settings, supported by advancements in minimally invasive technologies and shorter recovery times. ASCs benefit from lower procedural costs, improved operational efficiency, and an increasing preference among physicians for streamlined care models. As patient selection improves and regulatory support expands, ASCs are positioned to handle less complex stroke-related interventions, accelerating their adoption trajectory.
Regional Insights
North America hemorrhagic & ischemic stroke treatment devices market held the largest share and accounted for 54.75% of global revenue in 2025. This can be attributed to the high stroke burden, aging demographics, advanced healthcare infrastructure, and rapid adoption of innovative technologies. Rising prevalence of hypertension, diabetes, and obesity, particularly among the elderly population, continues to elevate the incidence of both ischemic and hemorrhagic strokes, boosting demand for mechanical thrombectomy systems, aspiration catheters, stent retrievers, flow diverters, and embolization devices.
U.S. Hemorrhagic & Ischemic Stroke Treatment Devices Market Trends
The U.S. hemorrhagic & ischemic stroke treatment devices market is driven by high stroke prevalence, an aging population, and rising comorbidities such as hypertension, diabetes, and obesity, which increase the risk of both ischemic and hemorrhagic strokes. Expansion of primary, thrombectomy-capable, and comprehensive stroke centers, supported by tele stroke networks and standardized care programs. Continuous technological advancements, favorable Medicare/insurance reimbursement, and growing public and physician awareness of stroke symptoms further accelerate device adoption, driving strong market growth in the U.S.
Europe Hemorrhagic & Ischemic Stroke Treatment Devices Market Trends
The European hemorrhagic and ischemic stroke treatment devices market is shaped by a shift toward integrated stroke care networks, where rapid diagnosis, inter-hospital coordination, and access to advanced neurointerventional devices are prioritized. Demand is rising for minimally invasive solutions, such as stent retrievers, aspiration systems, flow diverters, and embolization devices, driven by the growing adoption of mechanical thrombectomy and endovascular therapies. A strong emphasis on clinical evidence, safety, and post-market surveillance within the European Union's regulatory framework is encouraging innovation while standardizing device quality across countries.
Hemorrhagic and ischemic stroke treatment devices market in the UK is witnessing a growing emphasis on faster treatment pathways and optimized acute stroke response, aligned with national goals to lower stroke-related disability. Utilization of minimally invasive neurointerventional devices, including thrombectomy and aneurysm management systems, is increasing as clinical confidence and expertise expand. Ongoing investment in specialized stroke centers and clinician training is strengthening the adoption of advanced technologies. In parallel, outcome-focused care models and innovation initiatives within the National Health Service are supporting the gradual integration of next-generation stroke treatment devices across the country.
The France hemorrhagic and ischemic stroke treatment devices market is characterized by a push toward enhanced emergency response systems and improved access to cutting-edge interventions. There is a growing uptake of endovascular and minimally invasive technologies, such as thrombectomy devices and aneurysm coils, as clinicians seek better outcomes with reduced procedural risk. Supportive government policies and investment in regional stroke networks are facilitating quicker diagnosis and device deployment across urban and rural hospitals. The focus on clinical research collaborations and outcome measurement is driving innovation and encouraging the adoption of advanced stroke treatment devices throughout the French healthcare landscape.
Asia Pacific Hemorrhagic & Ischemic Stroke Treatment Devices Market Trends
The Asia Pacific hemorrhagic and ischemic stroke treatment devices market is expanding rapidly, driven by rising incidence of stroke, increasing healthcare investments, and greater awareness of acute stroke care. Countries across the region are adopting advanced endovascular and minimally invasive devices, such as mechanical thrombectomy systems and aneurysm treatment tools, to improve clinical outcomes. Enhanced government initiatives and infrastructure development in emerging markets are helping bridge gaps in emergency response and specialized care. Furthermore, collaborations between local healthcare providers and global device manufacturers are facilitating technology transfer and training, thereby accelerating market growth and increasing access to innovative stroke treatment solutions.
The China hemorrhagic and ischemic stroke treatment devices market is witnessing significant growth as stroke prevalence rises and demand for rapid, effective interventions increases. Adoption of advanced neurointerventional technologies, including thrombectomy and aneurysm management systems, is expanding, particularly in major urban medical centers. Government support for healthcare modernization and improved emergency care pathways is enhancing access to innovative treatment devices nationwide.
Latin America Hemorrhagic & Ischemic Stroke Treatment Devices Market Trends
Hemorrhagic and ischemic stroke treatment devices market in the Latin America is experiencing significant growth as awareness of stroke care improves and healthcare infrastructure strengthens across key countries. There is increasing adoption of minimally invasive neurointerventional devices, although uptake varies between urban centers and rural regions. Investments in training and specialized stroke units are enabling clinicians to deliver timely care, while partnerships with global device manufacturers facilitate access to advanced treatment technologies.
MEA Hemorrhagic & Ischemic Stroke Treatment Devices Market Trends
Hemorrhagic and ischemic stroke treatment devices market in the MEA is evolving significantly as countries strengthen acute care capabilities and stroke-specific infrastructure. The use of advanced, minimally invasive neurointerventional devices is increasing in major hospitals, while their penetration in peripheral regions remains limited. Increasing focus on physician training, awareness programs, and healthcare modernization is gradually improving access to timely stroke treatment and supporting market development across the region.
Key Hemorrhagic & Ischemic Stroke Treatment Devices Company Insights
The hemorrhagic and ischemic stroke treatment devices market is largely dominated by a small group of established global manufacturers, including Medtronic, Stryker, and Johnson & Johnson, which collectively hold a significant portion of the total market revenue. Their strong position is supported by comprehensive neurovascular portfolios, proven clinical outcomes, and a wide international presence. Furthermore, emerging and regional companies are increasing their footprint by offering specialized and cost-efficient stroke treatment solutions.
Key Hemorrhagic And Ischemic Stroke Treatment Devices Companies:
The following key companies have been profiled for this study on the hemorrhagic and ischemic stroke treatment devices market.
- Medtronic
- Terumo Corporation
- Balt
- Scientia Vascular, Inc.
- MicroPort Scientific Corporation
- Johnson & Johnson (MedTech)
- phenox GmbH (Wallaby Medical)
- Acandis GmbH
- B. Braun SE
- Integra LifeSciences
- Infinity Neuro LLC
- Penumbra, Inc.
- Boston Scientific Corporation
- Stryker
- Peter LAZIC GmbH
- Vesalio Inc.
Recent Developments
-
In December 2025, Penumbra Inc. received CE Mark approval for its MIDWAY and ACCESS25 delivery catheters. These catheters are now commercially available across Europe, further strengthening Penumbra’s neuro access platform.
-
In October 2025, Penumbra launched the SwiftSET Coil, an advanced complex embolization coil designed for adaptive vessel occlusion. The device features a unique shape configuration that improves vessel wall apposition, allowing smooth deployment and natural conformity within tight anatomical spaces. This design enables dense and controlled occlusion, particularly in small and challenging vessels.
-
In July 2025, Stryker announced the commercial launch of the Surpass Elite Flow Diverter across the U.S., South Korea, and Europe. This next-generation device represents a significant advancement in flow diversion therapy, combining an enhanced implant design with a proprietary surface modification to improve clinical performance and reduce thrombin generation.
Hemorrhagic And Ischemic Stroke Treatment Devices Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 4.04 billion
Revenue forecast in 2033
USD 6.24 billion
Growth rate
CAGR of 6.4% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Treatment type, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Norway; Italy; Denmark; Sweden; Spain; China; Japan; India; South Korea; Australia; Thailand; Brazil; Argentina; KSA; UAE; South Africa; Kuwait
Key companies profiled
Medtronic; MicroVention, Inc. (Terumo Corporation); Balt; Scientia Vascular, Inc.; MicroPort Scientific Corporation; Johnson & Johnson (MedTech); phenox GmbH (Wallaby Medical); Acandis GmbH; B. Braun SE; Integra LifeSciences; Infinity Neuro LLC; Penumbra Inc.; Boston Scientific Corporation; Stryker; Peter LAZIC GmbH; Vesalio Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
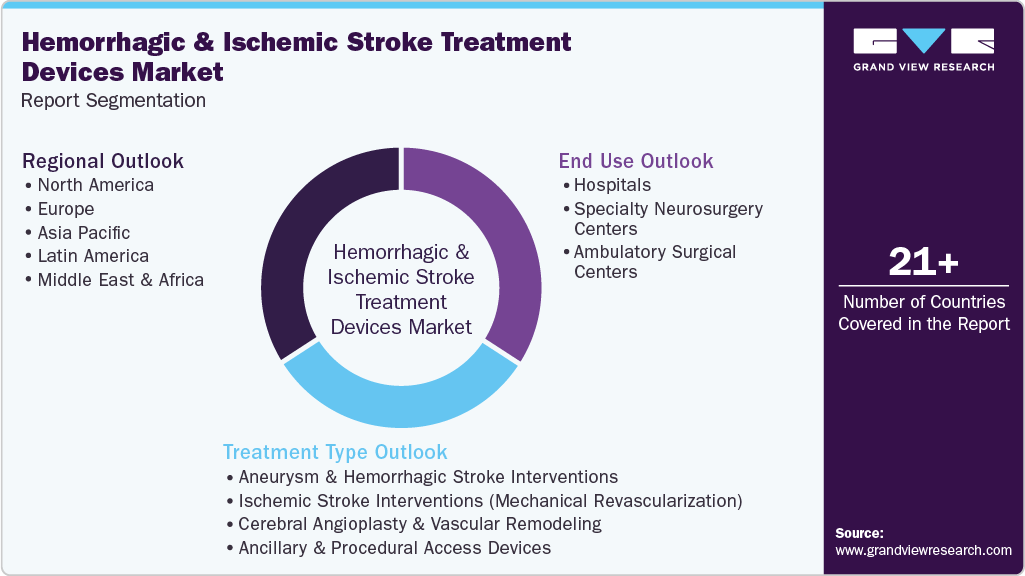
Global Hemorrhagic And Ischemic Stroke Treatment Devices Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis on the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global hemorrhagic and ischemic stroke treatment devices market report based on treatment type, end use, and region:
-
Treatment Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Aneurysm & Hemorrhagic Stroke Interventions
-
By Indication
-
Intracranial Aneurysm
-
cSDH (MMA Embolization)
-
AVM & AVF Embolization
-
-
By Device
-
Embolization Coiling Systems
-
Bare Platinum Coils
-
Coated / Bioactive Coils
-
-
Flow Diversion Systems
-
Standard Flow-Diverting Stents (e.g., Pipeline)
-
Next-Generation / Surface-Modified Flow Diverters
-
-
Intrasaccular Flow Disruption Devices
-
Woven EndoBridge (WEB)
-
Contour Neurovascular System
-
Luna Aneurysm Embolization System
-
-
Liquid Embolization Agents
-
Non-Adhesive Liquid Embolics (Onyx, Squid, PHIL)
-
Adhesive Liquid Embolics (NBCA)
-
-
-
-
Ischemic Stroke Interventions (Mechanical Revascularization)
-
Stent Retriever Systems
-
Aspiration-Based Thrombectomy Systems
-
-
Cerebral Angioplasty & Vascular Remodeling
-
Balloon Angioplasty Systems
-
Intracranial & Carotid Stenting Systems
-
Coil-Assisted Adjunctive Devices (Balloon-assisted and stent-assisted)
-
-
Ancillary & Procedural Access Devices
-
Guiding & Distal Access Catheters
-
Microcatheters
-
Access Sheaths & Wires
-
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Specialty Neurosurgery Centers
-
Ambulatory Surgical Centers
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Northeast
-
Connecticut
-
Maine
-
Massachusetts
-
New Hampshire
-
Rhode Island
-
Vermont
-
New Jersey
-
New York
-
Pennsylvania
-
-
Midwest
-
Illinois
-
Indiana
-
Iowa
-
Kansas
-
Michigan
-
Minnesota
-
Missouri
-
Nebraska
-
North Dakota
-
Ohio
-
South Dakota
-
Wisconsin
-
-
South
-
Alabama
-
Arkansas
-
Delaware
-
Florida
-
Georgia
-
Kentucky
-
Louisiana
-
Maryland
-
Mississippi
-
North Carolina
-
Oklahoma
-
South Carolina
-
Tennessee
-
Texas
-
Virginia
-
West Virginia
-
-
West
-
Alaska
-
Arizona
-
California
-
Colorado
-
Hawaii
-
Idaho
-
Montana
-
Nevada
-
New Mexico
-
Oregon
-
Utah
-
Washington
-
Wyoming
-
-
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Sweden
-
Denmark
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global hemorrhagic and ischemic stroke treatment devices market size was valued at USD 3.80 billion in 2025 and is expected to reach USD 4.04 billion in 2026.
b. The global hemorrhagic and ischemic stroke treatment devices market is projected to grow at a compound annual growth rate (CAGR) of 6.42% from 2026 to 2033 to reach USD 6.24 billion by 2033.
b. The hemorrhagic & ischemic stroke treatment devices market in North America held the largest share and accounted for 54.75% of global revenue in 2025. This can be attributed to the high stroke burden, aging demographics, advanced healthcare infrastructure, and rapid adoption of innovative technologies.
b. Some of the key players in hemorrhagic and ischemic stroke treatment devices market are Medtronic, MicroVention, Inc. (Terumo Corporation), Balt, Scientia Vascular, Inc., MicroPort Scientific Corporation, Johnson & Johnson (MedTech), phenox GmbH (Wallaby Medical), Acandis GmbH, B. Braun SE, Integra LifeSciences, Infinity Neuro LLC, Penumbra, Inc., Boston Scientific Corporation, Stryker, Peter LAZIC GmbH, and Vesalio Inc., among others.
b. Key factors that are driving the hemorrhagic & ischemic stroke treatment devices market growth include rising global incidence of stroke and increasing awareness of early intervention benefits. Ischemic stroke, accounting for the majority of cases, continues to fuel demand for mechanical thrombectomy devices, stent retrievers, and aspiration systems, as these technologies have demonstrated strong clinical outcomes when used within appropriate treatment windows. Meanwhile, the growing diagnosis of intracranial aneurysms and vascular malformations is supporting demand for hemorrhagic stroke treatment devices such as flow diverters, coils, and embolization systems.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










