- Home
- »
- Pharmaceuticals
- »
-
Hereditary Angioedema Therapeutics Market Report, 2033GVR Report cover
![Hereditary Angioedema Therapeutics Market Size, Share & Trends Report]()
Hereditary Angioedema Therapeutics Market (2026 - 2033) Size, Share & Trends Analysis Report By Treatment (C1-Inhibitor Concentrates, Bradykinin B2 Receptor Antagonist), By End Use, By Region, And Segment Forecasts
- Report ID: GVR-3-68038-156-6
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Hereditary Angioedema Therapeutics Market Summary
The global hereditary angioedema therapeutics market size was estimated at USD 3.60 billion in 2025 and is projected to reach USD 8.18 billion by 2033, growing at a CAGR of 10.43% from 2026 to 2033. The global hereditary angioedema therapeutics market is expanding due to increasing diagnosis rates and greater clinical awareness of rare genetic disorders.
Key Market Trends & Insights
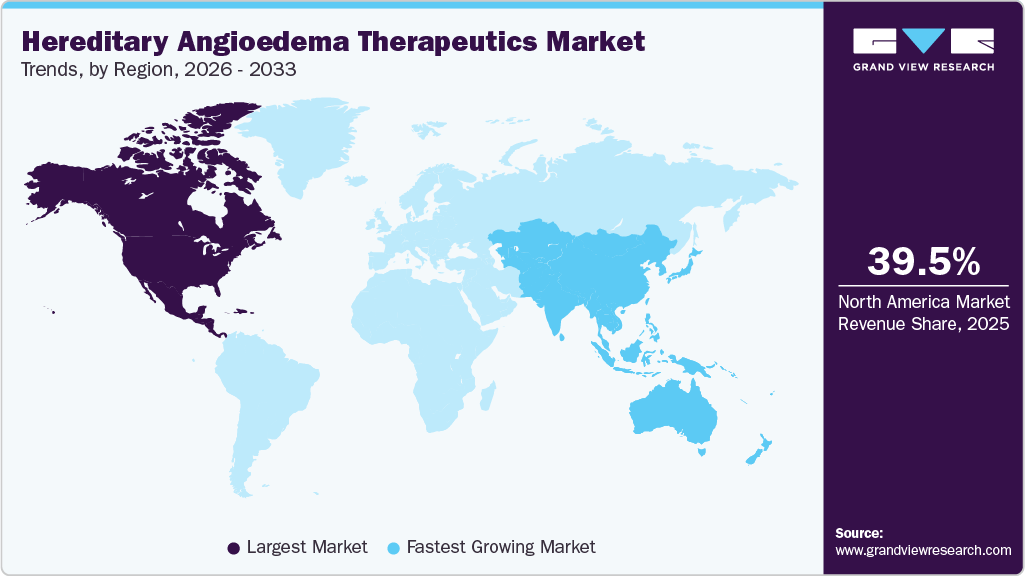
- North America hereditary angioedema therapeutics market held the largest share of 39.50% of the global market in 2025.
- The hereditary angioedema therapeutics industry in the U.S. is expected to grow significantly over the forecast period.
- By treatment, the kallikrein inhibitor segment held the highest market share of 46.14% in 2025.
- By end use, the on-demand segment held the highest market share of 60.77% in 2025.
- By route of administration, the intravenous segment held the highest market share of 54.58% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 3.60 Billion
- 2033 Projected Market Size: USD 8.18 Billion
- CAGR (2026-2033): 10.43%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Improved access to specialized testing has enabled earlier identification of patients, which supports timely treatment initiation. The availability of targeted therapies that address underlying disease pathways has strengthened treatment outcomes and patient confidence. Pharmaceutical innovation has introduced convenient administration options, including subcutaneous and oral formulations, which improve adherence. Rising prevalence reporting across developed and emerging regions continues to create sustained demand for effective therapies.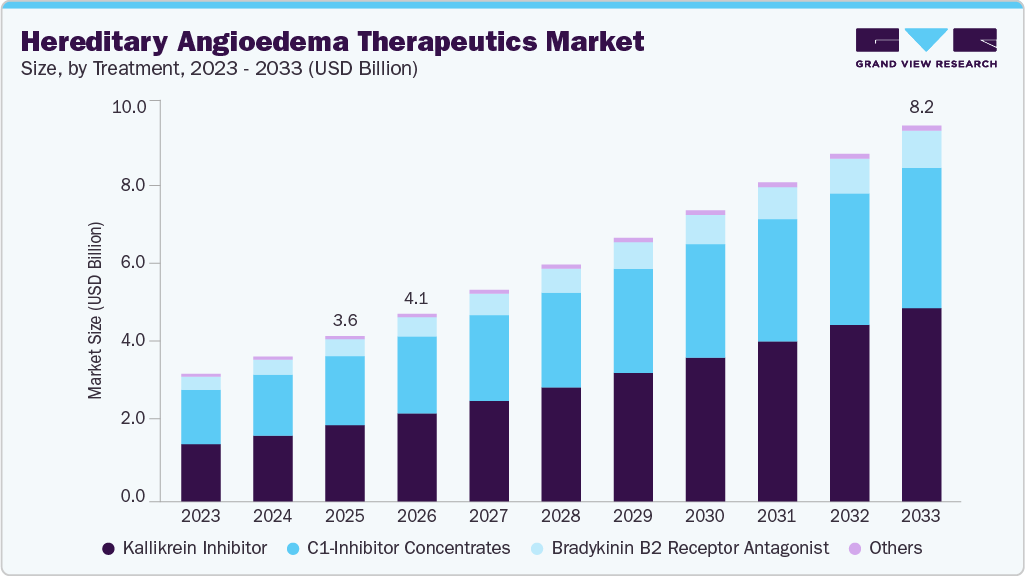
Advancements in biologics and novel drug classes represent another major growth driver for the market. Long-acting prophylactic therapies have reduced attack frequency and improved quality of life for patients, which has increased adoption among physicians. Oral kallikrein inhibitors and gene-targeted approaches under development are expanding the future treatment landscape. Strong clinical trial activity and regulatory approvals have accelerated commercialization timelines for innovative therapies. For instance, December 2025, HCPLive reported that the U.S. Food and Drug Administration had approved three new hereditary angioedema treatments in 2025 over three months, including garadacimab gxii, Andembry, in June, sebetralstat, Ekterly, in July, and donidalorsen, Dawnzera, in August, in a disease affecting about 1 in 50,000 people globally and nearly 6,000 in the United States. Prior to these approvals, 9 therapies were available, increasing to 12 by 2026, reflecting the expansion of oral and injectable options. Strategic collaborations between biotechnology firms and larger pharmaceutical companies have further enhanced research capabilities and global distribution reach.
Healthcare infrastructure improvements and the expansion of specialty care networks are further contributing to market growth. Increased involvement of immunologists and rare disease specialists has strengthened disease management standards and treatment utilization. Patient advocacy initiatives and education programs have encouraged treatment uptake and long-term therapy compliance. Favorable reimbursement coverage in several regions has improved patient access to high-cost biologic treatments. For instance, in December 2025, Rare Disease Advisor reported that the U.S. Food and Drug Administration had approved berotralstat, marketed as ORLADEYO, as the first oral plasma kallikrein inhibitor prophylactic therapy for hereditary angioedema in children aged 2 to 11 years, extending prior approval for patients 12 years and older. The pediatric approval was based on the APeX P trial involving 29 patients aged 2 to 11 years, 48.3 percent female, with a mean age of 8 years and a mean onset at 2 years, demonstrating sustained reductions in monthly attack rates and a peak median concentration of 3.9 hours, range 0.9 to 6.0 hours. Growing investment in rare disease research continues to support innovation and competitive market expansion.
Market Concentration & Characteristics
The hereditary angioedema therapeutics market demonstrates a high degree of innovation, driven by advances in molecular biology and targeted drug development. Companies are focusing on therapies that inhibit the kallikrein-bradykinin pathway to achieve improved disease control. Long-acting biologics, monoclonal antibodies, and oral small-molecule inhibitors have significantly transformed treatment approaches. Pipeline research includes gene-silencing therapies and next-generation prophylactic treatments with extended dosing intervals. Continuous clinical development supports differentiated product positioning and lifecycle expansion. Innovation remains essential for maintaining competitive advantage in a rare disease market with evolving patient needs.
Barriers to entry in this market are substantial due to the complexity of rare disease drug development and high research costs. Clinical trials require specialized patient populations, which increases recruitment challenges and timelines. Manufacturing biologics demands advanced infrastructure, regulatory expertise, and quality control capabilities. Established companies benefit from strong intellectual property portfolios and brand recognition among specialists. Pricing pressures and reimbursement negotiations create additional challenges for new entrants. These factors collectively limit the number of competitors entering the hereditary angioedema therapeutics space.
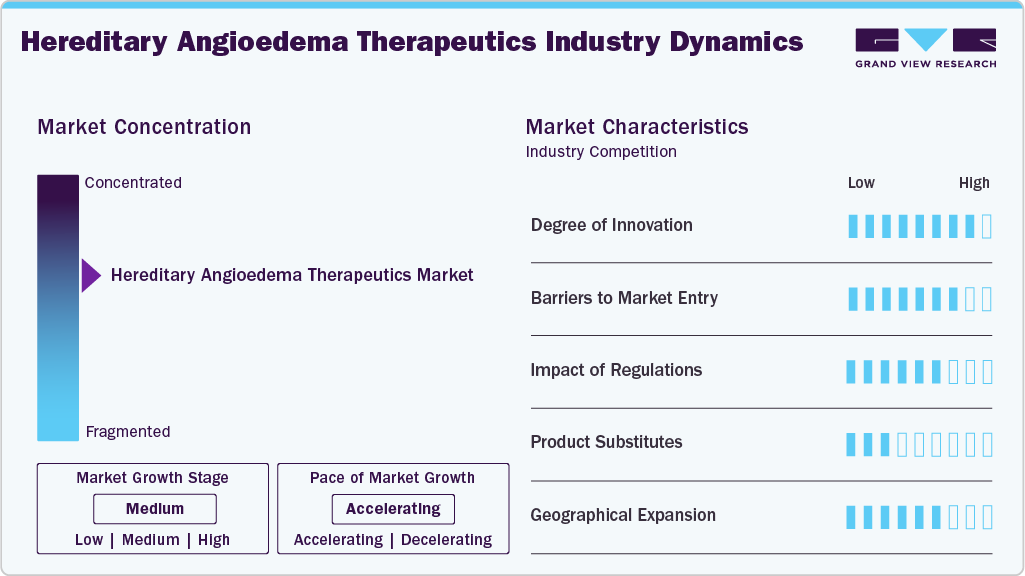
Regulatory frameworks strongly influence the hereditary angioedema therapeutics market through strict safety and efficacy requirements. Orphan drug designation pathways provide incentives that encourage innovation in rare disease treatment development. Approval processes involve detailed clinical evidence demonstrating reduction in attack frequency and severity. Post-marketing surveillance and pharmacovigilance requirements add ongoing compliance responsibilities for manufacturers. Variations in regulatory standards across regions influence launch timelines and commercialization strategies. Companies must maintain regulatory alignment to sustain market access and long-term product adoption.
Product substitution risk in this market remains relatively low due to the specialized nature of hereditary angioedema treatments. Acute therapies and prophylactic treatments serve distinct clinical purposes, which reduces direct interchangeability. Plasma-derived C1-Inhibitor concentrates and recombinant therapies continue to compete with newer targeted drugs. Emerging oral treatments may shift prescribing preferences toward more convenient administration routes. Physicians typically select therapies based on efficacy, safety profile, and patient-specific factors rather than price alone. The limited availability of alternative treatment classes supports stable demand for approved therapies.
Geographical expansion represents a key characteristic of market growth as companies seek to increase patient reach beyond established regions. North America and Europe currently dominate due to advanced healthcare systems and higher diagnosis rates. Emerging markets in Asia-Pacific and Latin America offer untapped opportunities with improving rare disease awareness. Companies are investing in regional partnerships, distribution networks, and local clinical studies to strengthen presence. Expansion strategies also include regulatory approvals across multiple jurisdictions to broaden market penetration. Increasing healthcare access in developing economies is expected to support long-term global growth.
Treatment Insights
The kallikrein inhibitor segment dominated the market with the largest revenue share of 46.14% in 2025, driven by strong clinical efficacy in reducing the frequency and severity of hereditary angioedema attacks and increasing physician confidence in targeted therapies. These agents act on the kallikrein bradykinin pathway, a central mechanism responsible for swelling episodes, thereby supporting improved disease control and faster symptom resolution. The availability of both oral and injectable formulations has enhanced treatment flexibility and patient convenience, contributing to improved adherence and broader adoption across diverse patient populations. Growing preference for prophylactic therapy among individuals with recurrent attacks has further strengthened demand. For instance, June 2025, KalVista Pharmaceuticals presented new data at the European Academy of Allergy and Clinical Immunology Congress 2025 from the KONFIDENT-S analysis of sebetralstat, involving 35 participants and 382 attacks, reporting a median 6 minute treatment time and 1.3 hour symptom relief, alongside adherence rates below 44 percent over 12 months. Ongoing innovation is expected to reinforce segment growth.
The bradykinin B2 receptor antagonist segment is projected to grow at a CAGR of 9.95% over the forecast period, fueled by increasing demand for rapid acting therapies that effectively manage acute hereditary angioedema attacks. These treatments provide targeted symptom relief by blocking bradykinin activity, which is responsible for vascular permeability and swelling episodes. Rising awareness among healthcare professionals regarding early intervention has supported greater utilization of on demand therapies. Improved accessibility of self-administration options has enhanced patient convenience and reduced emergency care dependence. Expanding clinical experience and treatment guidelines recommending timely acute management are further contributing to segment growth. For instance, September 2024, Clinical and Translational Allergy reported on oral bradykinin B2 receptor antagonists for hereditary angioedema, noting that deucrictibant had completed phase 2 trials for both on demand treatment and long term prophylaxis, with median baseline VAS 3 scores ranging from 24.33 to 27.00 at 10 mg, 20 mg, and 30 mg doses, with symptom reduction assessed every 30 minutes up to 48 hours, and analyses using least square mean differences, confidence intervals, and mixed effect model repeated measures in the modified intent to treat population, with nominal p values reported. Continued innovation in formulation development and delivery methods is expected to strengthen adoption across diverse patient populations.
End Use Insights
The on-demand segment dominated the market with the largest revenue share of 60.77% in 2025 due to the critical need for immediate treatment during acute hereditary angioedema attacks. Rapid symptom relief remains essential to prevent complications and reduce hospitalization risk, which supports strong adoption of on-demand therapies. Physicians continue to recommend rescue medications as a standard component of disease management across all patient groups. Availability of self-administered injectable treatments has improved patient independence and treatment accessibility outside clinical settings. Increasing diagnosis rates and better patient education regarding early attack recognition have further strengthened the segment demand. For instance, September 2025, KalVista Pharmaceuticals announced that the European Commission and the Swiss Agency for Therapeutic Products had approved EKTERLY, sebetralstat, the first and only oral on demand treatment for acute hereditary angioedema attacks in adults and adolescents aged 12 years and older, based on the phase 3 KONFIDENT trial that randomized 136 patients across 66 clinical sites in 20 countries and demonstrated significantly faster symptom relief and attack resolution versus placebo, with marketing authorizations covering 27 European Union member states plus Iceland, Liechtenstein, and Norway, supported by 10 years of orphan market exclusivity, with launch in Germany planned for the fourth quarter of 2025 and Switzerland availability anticipated in the second half of 2026. Continued advancements in fast-acting formulations are expected to maintain segment growth.
The prophylaxis segment is projected to grow at a CAGR of 11.61% over the forecast period, owing to increasing preference for preventive treatment strategies that reduce the frequency and severity of hereditary angioedema attacks. Long term disease control improves patient quality of life and supports consistent daily functioning, which encourages adoption among individuals with recurrent symptoms. Availability of long-acting biologics and emerging oral therapies has enhanced convenience and adherence compared to earlier treatment options. Physicians are increasingly recommending prophylactic therapy for patients with moderate to severe disease burden to minimize emergency interventions. Expanding clinical evidence demonstrating sustained efficacy and safety continues to strengthen confidence in preventive approaches. For instance, in August 2025, Ionis Pharmaceuticals announced that the U.S. Food and Drug Administration had approved DAWNZERA, donidalorsen, as the first and only RNA-targeted prophylactic treatment for hereditary angioedema in adults and pediatric patients 12 years and older, administered at 80 mg via subcutaneous autoinjector once every four weeks or eight weeks. In the Phase 3 OASIS HAE study, Q4W dosing reduced monthly attack rates by 81 percent versus placebo over 24 weeks and by 87 percent from the second dose, with approximately 90 percent reduction in moderate to severe attacks, while the OASISplus extension showed a 94 percent mean reduction after one year and a 62 percent reduction over 16 weeks in switch patients, with 84 percent preferring the therapy. Ongoing innovation focused on extended dosing intervals and targeted mechanisms is expected to support segment expansion.
Route of Administration Insights
The intravenous segment dominated the market, accounting for 54.58% of revenue in 2025, driven by the widespread use of intravenous C1-Inhibitor concentrates therapies for both acute treatment and prophylaxis in the management of hereditary angioedema. These therapies provide rapid systemic absorption and predictable therapeutic response, which supports strong physician confidence in clinical settings. Hospital-based administration and specialist supervision contribute to treatment reliability, particularly for severe cases. Established clinical experience with intravenous products has reinforced their continued utilization across major markets. Availability of well-known branded therapies with proven efficacy further strengthens segment demand. Continued use in emergency care and high-risk patient populations is expected to sustain segment dominance over the forecast period.
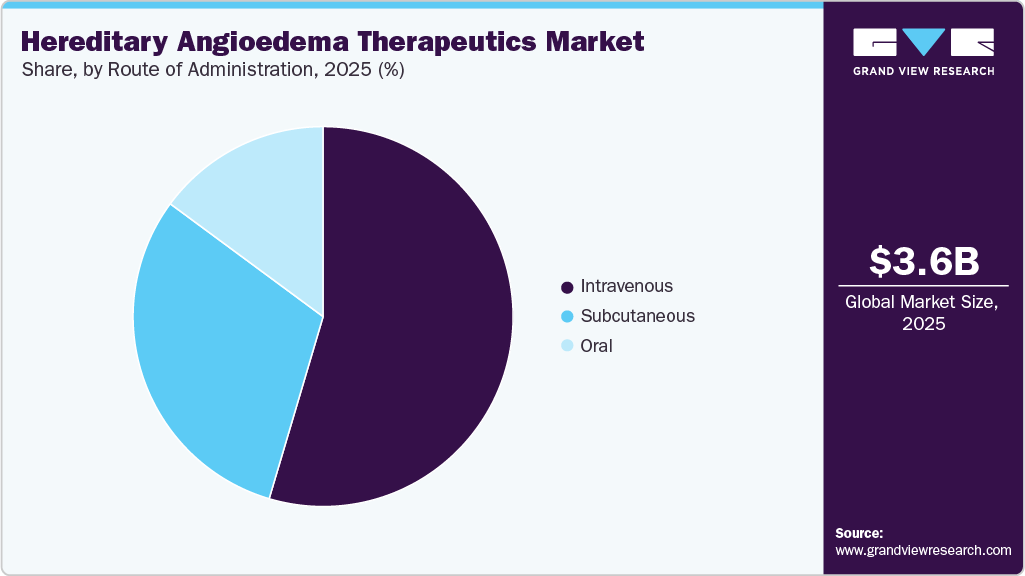
The oral segment is projected to grow at a CAGR of 15.88% over the forecast period, driven by increasing patient preference for convenient, noninvasive treatment options in hereditary angioedema management. Oral therapies reduce the need for injections or hospital visits, thereby improving adherence and the overall treatment experience. Advancements in small-molecule drug development have enabled effective targeting of the kallikrein pathway through oral formulations. Physicians are showing growing interest in prescribing oral prophylactic therapies for long-term disease control. Expanding clinical evidence supporting the safety and efficacy of these treatment options is strengthening confidence in them. Ongoing pipeline innovation focused on improved dosing convenience and sustained therapeutic response is expected to accelerate segment growth.
Regional Insights
North America held the largest hereditary angioedema therapeutics market share of 39.50% in 2025 due to advanced healthcare infrastructure and strong presence of leading biopharmaceutical companies. High disease awareness among clinicians supports early diagnosis and timely treatment initiation. Availability of multiple approved therapies provides physicians with diverse treatment options tailored to patient needs. Favorable reimbursement coverage improves patient access to premium biologic therapies. Active research programs and clinical trial activity strengthen regional leadership in innovation. Established specialty care centers contribute to consistent treatment adoption across the region.
U.S. Hereditary Angioedema Therapeutics Market Trends
The U.S. represents the largest share within North America, supported by high diagnosis rates and extensive specialty treatment networks. Strong commercial presence of major pharmaceutical companies accelerates product availability and market penetration. Physicians have access to advanced biologics, prophylactic therapies, and emerging oral treatments. Patient advocacy organizations enhance awareness and encourage adherence to long-term treatment plans. Significant investment in rare disease research drives continuous therapeutic innovation. Insurance coverage for specialty drugs supports sustained market demand.
Europe Hereditary Angioedema Therapeutics Market Trends
Europe hereditary angioedema therapeutics market shows steady growth driven by structured healthcare systems and increasing rare disease awareness. Regional clinical guidelines support standardized diagnosis and treatment approaches across multiple countries. Access to both plasma-derived and recombinant therapies strengthens treatment availability. Collaborative research initiatives among academic institutions and pharmaceutical companies encourage innovation. Expanding patient registries improve disease tracking and management efficiency. Growth in specialty care centers supports broader therapy adoption throughout the region.
The UK hereditary angioedema therapeutics market is supported by well-established diagnostic pathways and strong presence of specialized immunology centers. Physicians increasingly adopt long-term prophylactic therapies to reduce disease burden and hospital visits. Access to advanced biologics improves patient outcomes and treatment satisfaction. Growing awareness among healthcare professionals enhances early detection rates. Ongoing clinical research activities contribute to the introduction of innovative treatment options. Structured patient management programs encourage adherence and long-term therapy utilization.
The Germany hereditary angioedema therapeutics market represents a key market in Europe due to advanced healthcare services and strong pharmaceutical industry participation. High diagnostic accuracy supports timely identification of hereditary angioedema cases. Physicians demonstrate strong adoption of innovative biologics and targeted therapies. Presence of specialized treatment centers improves disease management standards. Clinical research collaborations strengthen access to emerging therapies and clinical trials. Stable reimbursement mechanisms support consistent patient access to treatment.
The France hereditary angioedema therapeutics market shows steady growth with increasing focus on rare disease management programs. Specialized reference centers contribute to improved diagnosis and coordinated care. Access to modern biologic therapies enhances treatment effectiveness and patient quality of life. Physician awareness initiatives support early intervention strategies. Research collaborations between hospitals and biotechnology companies encourage therapeutic innovation. Expanding patient education efforts contribute to improved treatment adherence.
Asia-Pacific Hereditary Angioedema Therapeutics Market Trends
Asia Pacific hereditary angioedema therapeutics market is expected to register the fastest CAGR of 12.65% over the forecast period due to improving healthcare infrastructure and rising disease awareness. Large population base creates significant opportunities for diagnosis expansion and treatment adoption. Increasing presence of multinational pharmaceutical companies supports product availability across major countries. Growing investment in clinical research strengthens regional therapeutic development. Expanding specialty care services improve patient access to advanced therapies. Economic development across emerging countries supports long-term market growth potential.
The Japan hereditary angioedema therapeutics market represents a mature market within Asia Pacific with strong diagnostic capabilities and advanced treatment access. Physicians adopt targeted therapies and prophylactic biologics to improve patient outcomes. Established pharmaceutical research environment encourages innovation and clinical trial participation. High healthcare standards support consistent disease management practices. Patient monitoring programs enhance adherence to prescribed therapies. Market growth remains supported by continuous introduction of advanced therapeutic options.
The China hereditary angioedema therapeutics market is experiencing rapid growth in the due to expanding healthcare infrastructure and increasing physician awareness. Improved diagnostic capabilities enable identification of previously undiagnosed patient populations. Rising presence of international pharmaceutical companies strengthens access to innovative therapies. Growing investment in biotechnology research supports domestic drug development initiatives. Urban healthcare centers are adopting advanced treatment protocols for rare diseases. Increasing patient education contributes to improved therapy utilization rates.
Latin America Hereditary Angioedema Therapeutics Market Trends
Latin America shows emerging growth potential in the hereditary angioedema therapeutics market with improving access to specialty healthcare services. Awareness initiatives among healthcare professionals support earlier diagnosis and treatment adoption. Expansion of pharmaceutical distribution networks improves therapy availability in major urban regions. Increasing collaboration with global pharmaceutical companies supports access to innovative treatments. Economic development in key countries contributes to healthcare spending growth. Market expansion remains gradual with improving disease recognition across the region.
The Brazil hereditary angioedema therapeutics market lead the Latin American market due to its large population and expanding healthcare infrastructure. Growing awareness among physicians supports improved diagnosis of hereditary angioedema cases. Availability of advanced biologics in major medical centers enhances treatment options. Increasing participation in clinical research activities strengthens therapeutic adoption. Expansion of specialty clinics contributes to improved patient management. Rising healthcare investment supports continued market growth potential.
Middle East & Africa Hereditary Angioedema Therapeutics Market Trends
The Middle East and Africa region demonstrates gradual growth in the hereditary angioedema therapeutics market with improving healthcare facilities in selected countries. Increasing physician awareness supports identification of rare disease cases. Expansion of pharmaceutical company presence enhances access to modern therapies. Urban healthcare centers are adopting advanced treatment approaches for hereditary angioedema management. Growing medical education initiatives contribute to better disease understanding. Market development remains uneven across countries with varying healthcare resources.
The Saudi Arabia hereditary angioedema therapeutics market represents a key market within the Middle East due to improving healthcare infrastructure and rising focus on specialized medical care. Advanced hospitals provide access to biologic therapies for hereditary angioedema management. Increasing awareness among healthcare professionals supports early diagnosis and treatment. Expansion of specialty clinics contributes to improved patient monitoring and therapy adherence. Presence of international pharmaceutical companies strengthens treatment availability. Ongoing investment in healthcare services supports future market growth.
Key Hereditary Angioedema Therapeutics Company Insights
Takeda Pharmaceutical Company Limited and CSL hold significant market presence through plasma-derived and recombinant C1-Inhibitor concentrates therapies supported by broad distribution networks. BioCryst Pharmaceuticals, Inc. and KalVista Pharmaceuticals, Inc. focus on oral kallikrein inhibitors that improve treatment convenience and patient adherence. Pharming and Sanofi continue expanding biologic treatment access while strengthening lifecycle strategies for existing products. Ionis Pharmaceuticals and Arrowhead Pharmaceuticals, Inc. are developing RNA-targeted therapies aimed at long-acting disease control. Pharvaris B.V., Attune Pharmaceuticals, and Adverum Biotechnologies, Inc. contribute with innovative pipeline candidates. Competitive dynamics are shaped by ongoing innovation, expanding prophylactic therapy adoption, and active clinical research programs.
Key Hereditary Angioedema Therapeutics Companies:
The following key companies have been profiled for this study on the hereditary angioedema therapeutics market.
- BioCryst Pharmaceuticals, Inc.
- Ionis Pharmaceuticals
- CSL
- Attune Pharmaceuticals
- Adverum Biotechnologies, Inc.
- KalVista Pharmaceuticals, Inc.
- Takeda Pharmaceutical Company Limited
- Pharming
- Sanofi
- Pharvaris B.V.
- Arrowhead Pharmaceuticals, Inc.
Recent Developments
-
In August 2025, Ionis Pharmaceuticals announced that the U.S. Food and Drug Administration had approved DAWNZERA, donidalorsen, as the first and only RNA targeted prophylactic therapy for hereditary angioedema in patients aged 12 years and older. The 80 mg dose was administered every four or eight weeks. In Phase 3 OASIS HAE, Q4W dosing reduced monthly attack rates by 81 percent over 24 weeks and 87 percent from the second dose, with 94 percent reduction after one year and 62 percent reduction in switch patients, with 84 percent preference.
-
In June 2025, BioCryst Pharmaceuticals presented new hereditary angioedema data at the European Academy of Allergy and Clinical Immunology meeting, highlighting ORLADEYO, berotralstat, in the APeX-P study of pediatric patients aged 2 to 11 years with a median age of 8 years, range 3 to 11. Across four weight based cohorts receiving 150 mg, 108 mg, 96 mg, and 78 mg once daily, treated attacks declined 86 percent, from 22 during 12 weeks standard of care to 3 over 12 weeks, with monthly rates decreasing from 1.28 to 0.38 and sustained through 48 weeks.
-
In April 2025, CSL Behring announced the launch of ANDEMBRY, garadacimab, in Japan after approval on February 20, 2025 for prevention of acute hereditary angioedema attacks in patients aged 12 years and older. The therapy was administered as a 400 mg loading dose followed by 200 mg once monthly via pre filled pen. National Health Insurance pricing was listed on April 16, 2025, with launch on April 18, 2025, addressing about 430 diagnosed patients and an estimated 2,500 nationally.
Hereditary Angioedema Therapeutics Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 4.09 billion
Revenue forecast in 2033
USD 8.18 billion
Growth rate
CAGR of 10.43% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Treatment, end use, route of administration, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key company profiled
BioCryst Pharmaceuticals, Inc.; Ionis Pharmaceuticals; CSL; Attune Pharmaceuticals; Adverum Biotechnologies, Inc.; KalVista Pharmaceuticals, Inc.; Takeda Pharmaceutical Company Limited; Pharming, Sanofi; Pharvaris B.V.; Arrowhead Pharmaceuticals, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Hereditary Angioedema Therapeutics Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global hereditary angioedema therapeutics market based on treatment, end use, route of administration, and region:
-
Treatment Outlook (Revenue, USD Million, 2021 - 2033)
-
C1-Inhibitor Concentrates
-
Bradykinin B2 Receptor Antagonist
-
Kallikrein Inhibitor
-
Others
-
-
End use Outlook (Revenue, USD Million, 2021 - 2033)
-
Prophylaxis
-
On-demand
-
-
Route of Administration Outlook (Revenue, USD Million, 2021 - 2033)
-
Intravenous
-
Subcutaneous
-
Oral
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global hereditary angioedema therapeutics market size was estimated at USD 3.3 billion in 2022 and is expected to reach USD 3.5 billion in 2023.
b. The global hereditary angioedema therapeutics market is expected to grow at a compound annual growth rate of 7.4% from 2023 to 2030 to reach USD 5.8 billion by 2030.
b. The C1-esterase inhibitor segment accounted for the largest revenue share of 52.2% in 2022. The dominance of the segment is attributed to the use of the products in both on-demand and prophylactic treatment of HAE.
b. A few of the key players operating in the hereditary angioedema therapeutic market include BioCryst Pharmaceuticals, Inc.; Ionis Pharmaceuticals; CSL; Attune Pharmaceuticals; Adverum Biotechnologies, Inc.; KalVista Pharmaceuticals, Inc.; Takeda Pharmaceutical Company Limited; Pharming.
b. A significant number of patients being misdiagnosed for the condition has generated a vast patient pool with substantial medical needs. This is likely to be a key driving factor for the hereditary angioedema therapeutic market.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










