- Home
- »
- Clinical Diagnostics
- »
-
In Vitro Diagnostics Market Size, Share & Trends Report 2030GVR Report cover
![In Vitro Diagnostics (IVD) Market Size, Share & Trends Report]()
In Vitro Diagnostics (IVD) Market Size, Share & Trends Analysis Report By Product (Instruments, Reagents), By Test Location, By End-use (Hospitals, Laboratory), By Application (Diabetes, Oncology), By Technology, By Region, And Segment Forecasts, 2024 - 2030
- Report ID: 978-1-68038-080-4
- Number of Report Pages: 200
- Format: PDF, Horizon Databook
- Historical Range: 2018 - 2023
- Forecast Period: 2023 - 2030
- Industry: Healthcare
In Vitro Diagnostics Market Size & Trends
The global in vitro diagnostics (IVD) market size was estimated at USD 77.92 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 4.4% from 2024 to 2030. The growth can be attributed to increasing adoption of IVD owing to a rise in the incidence of infectious and chronic diseases. The development of automated IVD systems for laboratories and hospitals to provide efficient, accurate, and error-free diagnoses is expected to fuel market growth. The rising number of IVD products being launched by key players is also fueling market growth. For instance, in November 2023, ARUP Laboratories received a CE mark from EU-IVDR for AAV5 DetectCDx, a companion diagnostic to select the eligibility of severe hemophilia A-affected patients for BioMarin’s new gene therapy, Roctavian.
Technological advancements in terms of accuracy, portability, and cost-effectiveness are expected to be one of the high-impact rendering drivers of this market. Introduction of novel and highly accurate clinical laboratory tests is boosting the adoption of novel IVD tests worldwide. In June 2023, Toray Industries, Inc. received marketing approval from Japan’s Ministry of Health, Labour and Welfare for its Toray APOA2-iTQ used to diagnose pancreatic cancer. Moreover, in March 2023, Abbott received U.S. FDA clearance for its novel laboratory Traumatic Brain Injury (TBI) blood test in the U.S. Increasing approvals of IVD tests for life-threatening diseases are expected to create new opportunities in the untapped market.
Key players in the market undertake various strategies to strengthen their position and offer their customers diverse, technologically advanced & innovative products. New product launches and partnerships are the most prominently adopted by companies to attract more customers. For instance, in March 2023, BD received 510(k) clearance for BD Vaginal Panel on the BD COR System to detect infectious causes of vaginitis. In August 2023, the Precision Medicine Centre (PMC) formed a partnership with the Regional Molecular Diagnostic Service (RMDS) to implement genomic technology for the diagnosis of cancer in Northern Ireland.
Favorable initiatives undertaken by government and non-government bodies to improve overall healthcare services are anticipated to increase market growth. In October 2023, the WHO published the Essential Diagnostics List (EDL), a comprehensive list of IVD products that helps countries make decisions regarding diagnostic tools. It provides evidence-based recommendations and ensures the accessibility of essential products for target people. Moreover, in August 2023, the Africa CDC collaborated with the Africa Development Agency-New Partnership for Africa's Development (AUDA-NEPAD) to increase access to diagnostic tests across Africa. Such initiatives are expected to boost market growth.
A rise in the geriatric population and growth in knowledge regarding early testing have led to a surge in the number of regular check-ups, as a majority of deaths due to infections and chronic conditions occur in the population aged over 75 years. As per the Office for Budget Responsibility, UK, healthcare costs have risen exponentially, which can create economic pressure on nations with rapidly growing geriatric population. However, this expenditure is anticipated to translate positively for the IVD industry, driving market growth.
Market Characteristics
Market growth stage is medium, and the pace of the market growth is decelerating. Decreasing demand for COVID-19 tests is hampering the growth. The market is characterized by a high degree of innovation owing to increasing introduction of novel molecular diagnostics and immunoassay tests for multiple disease indications. Moreover, increasing demand for patient-centric tests is encouraging market players to develop technologically advanced products.
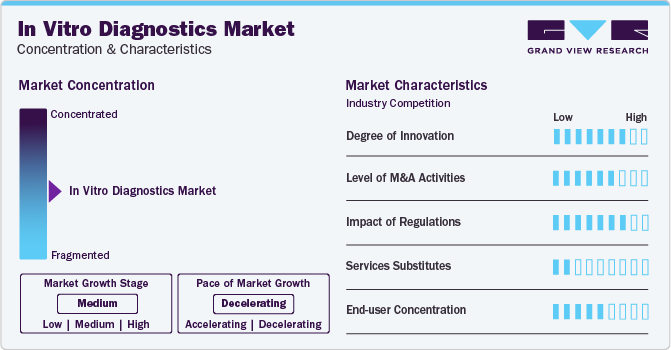
The market is also characterized by a high level of merger and acquisition (M&A) activity by the leading players. They aim to expand their businesses in developing economies to increase their market share. For instance, in August 2022, BD and LabCorp collaborated to develop, manufacture, and commercialize cytometry-based companion diagnostics for cancer & other diseases.
The in vitro diagnostics market is subject to increasing regulatory scrutiny. For instance, in July 2023, the U.S. FDA announced a renewed attempt to regulate laboratory-developed tests (LDTs) as medical devices. Later, in September 2023, the U.S. FDA proposed that LDT manufacturers would need to comply with medical device statutes and regulations. FDA is planning to enforce this in phases starting in 2024 to 2028.
The low threat of substitute products and services, such as high-cost imaging methods, is supporting the market expansion. Using imaging tools to monitor health conditions to some extent can hamper the growth. However, IVD tests overcome the threat of substitutes due to their accuracy and cost-effectiveness.
End-user concentration is a significant factor in the in vitro diagnostics market. The availability of IVD tests at different healthcare facilities, such as hospitals, laboratories, nursing homes, physician offices, and home-based settings, increases the market reach of novel IVD products. Moreover, increasing demand for rapid and self-tests creates new market opportunities for players in different end-use applications.
Product Insights
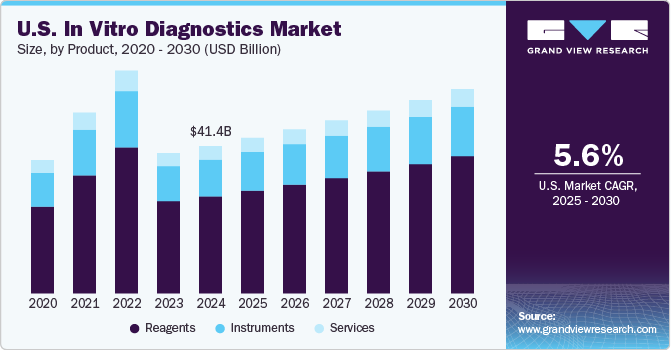
The reagents segment held the largest share of 65.88% of the overall revenue in 2023. The segment is expected to retain its dominance growing at the fastest CAGR from 2024 to 2030 owing to the extensive R&D initiatives undertaken by major players to develop novel reagents and test kits. For instance, in February 2023, BD received the market approval for the BD Onclarity HPV Assay to be used with the ThinPrep Pap Test in the U.S. The increasing R&D activities to enable faster cancer detection and precision medicine are allowing companies to focus on niche profitable areas in the IVD business. For instance, in March 2023, QIAGEN partnered with Servier to develop a companion diagnostic test for TIBSOVO, indicated for treating the blood cancer acute myeloid leukemia.
The growth in precision medicine is projected to enhance the overall demand for such novel reagents and consumables. The instruments segment held the second-largest share in 2023. Increasing approvals for novel IVD instruments are anticipated to drive segment growth. For instance, in April 2023, bioMérieux SA submitted 510(k) application to the U.S. FDA for VITEK REVEAL, rapid AST system. Players are aligning their instrument launches in line with the increasing genetic test requirement globally. For instance, in March 2022, Ion Torrent Genexus Dx Integrated Sequencer was introduced in the market by Thermo Fisher Scientific for research as well as diagnostic purposes.
Technology Insights
The immunoassay segment accounted for the largest revenue share in 2023. Increasing incidence of chronic & communicable diseases and rising need for early diagnosis are among the key factors leading to an increase in demand for immunological methods, including different types of Enzyme-Linked Immunosorbent Assays (ELISAs). Moreover, key players are focused on R&D pertaining to development of new immunological diagnostic instruments and tests for IVD applications. For instance, in October 2023, Sysmex Corporation and Fujirebio Holdings, Inc. collaborated to enhance their R&D, production, clinical development, and marketing activities in immunoassay.
The coagulation segment is expected to grow at the fastest CAGR from 2024 to 2030. The segment can be attributed to the increasing prevalence of cardiovascular diseases, blood-related disorders, and autoimmune diseases. Moreover, instruments are getting updated and handheld coagulation analyzers, such as Xprecia stride coagulation analyzer, are anticipated to enhance the overall workflow of detection. Some of the key players offering instruments and coagulation tests are Abbott, Siemens Healthcare GmbH, and Beckman Coulter, Inc.
End-use Insights
The hospitals segment held the largest revenue share in 2023 owing to a rise in the rate of hospitalizations that require support from faster diagnostics. Moreover, the ongoing development of healthcare infrastructure and favorable initiatives taken by government bodies are anticipated to enhance the existing hospital facilities. Thus, the demand for hospital-based IVD tests is increasing. Most IVD devices are purchased by hospitals and used in significant volumes. In 2023, there are over 6,129 hospitals in the U.S. that require constant aid from IVD for critical decision-making, as IVD tests provide faster and more accurate results.
In December 2023, the American Hospital Association wrote a letter to FDA not to apply device regulations on hospital’s LDTs. The homecare segment is expected to grow at a high CAGR from 2024 to 2030 due to the rising geriatric population and increasing demand for home care IVD devices. There is a growing need for novel molecular diagnostic and immunoassay platforms that can assist patients in conducting self-tests. For instance, in February 2023, the initial over-the-counter (OTC) home diagnostic test that can distinguish and identify influenza A and B, commonly referred to as the flu, and SARS-CoV-2, the virus causing COVID-19, was given an EUA by the FDA.
Test Location Insights
The others (lab-based tests) segment accounted for the largest revenue share in 2023. A large number of test analyses at one time and the higher accuracy of laboratory-based tests make them more reliable compared to PoC and home tests, giving segment a competitive edge over the other two segments. Moreover, the availability of tests that allow for sample collection at home and sending it to the laboratories for testing makes testing highly convenient for patients.
The homecare segment is expected to grow at a high CAGR from 2024 to 2030 due to the increasing reliability of these tests and patient-centric approaches of manufacturers. These tests have been an important source in mitigating the spread of the SARS-CoV-2. Significant actions have been taken by government bodies globally to ensure access to affordable and efficient homecare tests. In April 2023, Llusern Scientific, a University of South Wales spinoff, developed the PoC molecular diagnostic platform, Lodestar Dx platform, for panel testing of urinary tract infections.
Application Insights
The infectious diseases segment dominated the market in 2023. The outbreak of the COVID-19 pandemic increased the segment share significantly in recent years. Moreover, key players are introducing novel testing products to improve access to high-quality, innovative laboratory services for patients & healthcare providers. For instance, in February 2023, BD received EUA from the U.S. FDA for a new molecular diagnostic combination test for SARS-CoV-2, Influenza A+B, and Respiratory Syncytial Virus (RSV). Such initiatives by key players to strengthen their presence are expected to drive market growth.
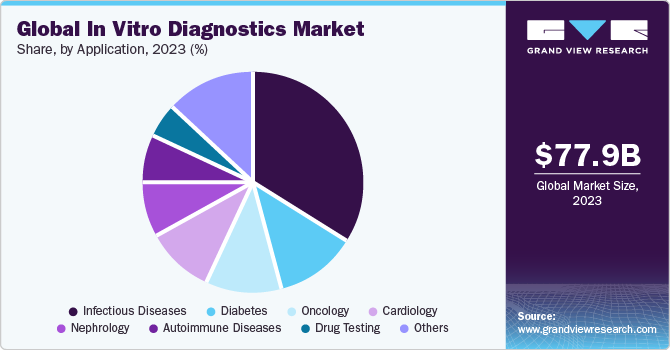
The oncology segment is anticipated to grow at the fastest CAGR from 2024 to 2030. Increasing incidence of cancer coupled with a high mortality rate is increasing the demand for cancer biomarker tests at an early stage. Moreover, increasing approval of novel tests, R&D activities, and favorable initiatives undertaken by regulatory bodies are expected to drive industry growth. For instance, in June 2023, the U.S. FDA announced a pilot program that gives chance to manufacturers to submit validation and performance data for LDTs for cancer. Moreover, in April 2023, Biocartis Group NV & APIS Assay Technologies Ltd. collaborated to manufacture and commercialize a breast cancer subtyping test on Idylla, a molecular diagnostics platform.
Regional Insights
North America dominated the market and accounted for a share of 42.28% in 2023. The region is estimated to retain its leading market position throughout the forecast period. The market in this region is collectively driven by factors, such as the rising incidence of chronic diseases, presence of strong players, increasing number of novel test launches, and supportive government funding. For instance, in January 2023, BD and CerTest Biotec received EUA from the U.S. FDA for a PCR test for Mpox virus detection in the U.S. Moreover, the increasing requirement for genetic testing for personalized health care, such as that for diabetes and cancer, is expected to drive market growth in North America.
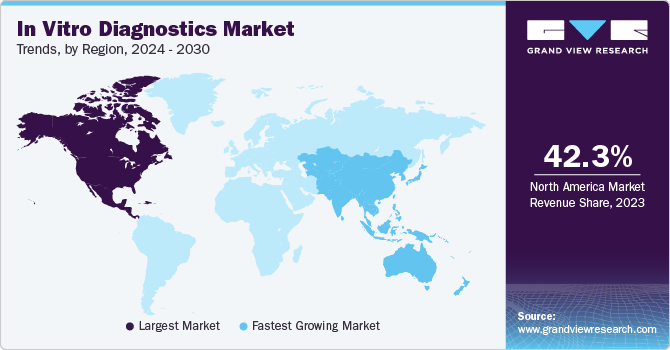
Asia Pacific is anticipated to exhibit significant growth from 2024 to 2030. The regional market of Asia Pacific is expected to be driven by a number of factors, some of which include the presence of stabilizing economies, rapidly growing middle-class population, supportive government policies, and rapid urbanization across the region. For instance, in October 2023, Fapon and Halodoc partnered to increase the in vitro diagnostic product sales and services in Indonesia. Moreover, leading players are collaborating with regional players to expand their reach in developing countries in Asia Pacific.
Key Companies & Market Share Insights
Some of the key players operating in the in vitro diagnostics market include F. Hoffmann-La Roche Ltd.; Abbott; Quest Diagnostics Inc.; and Danaher. Market players are adopting various strategies, such as new product launches, mergers & acquisitions, and partnerships, to strengthen their product portfolios and offer diverse technologically advanced & innovative products.
Llusern Scientific, Biocartis Group NV, ARUP Laboratories, Veracyte, and Exact Sciences Corp are some of the emerging market participants in the market. Emerging companies are actively involved in developing novel and accurate IVD testing products to improve overall health services. Moreover, these companies are collaborating with research institutes, government bodies, and global leaders to increase the range of their products in potential markets.
Key In Vitro Diagnostics (IVD) Companies:
- Abbott
- bioMérieux SA
- QuidelOrtho Corporation
- Siemens Healthineers AG
- Bio-Rad Laboratories, Inc.
- Qiagen
- Sysmex Corporation
- Charles River Laboratories
- Quest Diagnostics Incorporated
- Agilent Technologies, Inc.
- Danaher Corporation
- BD
- F. Hoffmann-La Roche Ltd.
Recent Developments
-
In December 2023, ARUP Laboratories and Medicover collaborated to provide diagnostic and healthcare services in Europe. ARUP Laboratories has developed AAV5 DetectCDx in collaboration with BioMarin Pharmaceutical Inc. to select therapies for severe hemophilia A patients
-
In November 2023, Veracyte joined Illumina to develop molecular tests for decentralized IVD applications. Companies are focusing on the development of Prosigna breast cancer and Percepta nasal swab tests of Veracyte
-
In October 2023, Promega Corporation announced its plan to develop and commercialize companion diagnostics kits with GSK Plc to identify cancer patients with MSI-H solid tumors
-
In February 2023, Unilabs announced investing over USD 200 million in Siemens Healthineers' technology and acquiring more than 400 laboratory analyzers to strengthen its laboratory infrastructure
-
In February 2023, F. Hoffmann-La Roche Ltd. collaborated with Janssen Biotech Inc. to develop companion diagnostics for targeted therapies. Companion diagnostic technologies include digital pathology, NGS, PCR, immunoassays, and immunohistochemistry
In Vitro Diagnostics (IVD) Market Report Scope
Report Attribute
Details
Market size value in 2024
USD 78.42 billion
Revenue forecast in 2030
USD 101.58 billion
Growth rate
CAGR of 4.4% from 2024 to 2030
Actual data
2018 - 2023
Forecast period
2024 - 2030
Quantitative units
Revenue in USD billion and CAGR from 2024 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Products, technology, application, end use, test location, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; UK; Germany; France; Spain; Italy; Russia; Denmark; Sweden; Norway; Japan; China; India; South Korea; Australia; Thailand; Singapore; Brazil; Mexico; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Abbott; bioMérieux SA; QuidelOrtho Corp.; Siemens Healthineers AG; Bio-Rad Laboratories, Inc.; Qiagen; Sysmex Corp.; Charles River Laboratories; Quest Diagnostics Inc.; Agilent Technologies, Inc.; Danaher Corp.; BD; F. Hoffmann-La Roche Ltd.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global In Vitro Diagnostics Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global in vitro diagnostics (IVD) market report based on product, technology, application, end-use, test location, and region:
-
Product Outlook (Revenue, USD Million, 2018 - 2030)
-
Instruments
-
Reagents
-
Services
-
-
Technology Outlook (Revenue, USD Million, 2018 - 2030)
-
Immunoassay
-
Instruments
-
Reagents
-
Services
-
-
Hematology
-
Instruments
-
Reagents
-
Services
-
-
Clinical Chemistry
-
Instruments
-
Reagents
-
Services
-
-
Molecular Diagnostics
-
Instruments
-
Reagents
-
Services
-
-
Coagulation
-
Instruments
-
Reagents
-
Services
-
-
Microbiology
-
Instruments
-
Reagents
-
Services
-
-
Others
-
Instruments
-
Reagents
-
Services
-
-
-
Application Outlook (Revenue, USD Million, 2018 - 2030)
-
Infectious Diseases
-
Diabetes
-
Oncology
-
Cardiology
-
Nephrology
-
Autoimmune Diseases
-
Drug Testing
-
Others
-
-
Test Location Outlook (Revenue, USD Million, 2018 - 2030)
-
Point of Care
-
Home-care
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2018 - 2030)
-
Hospitals
-
Laboratory
-
Home-care
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
UK
-
Germany
-
France
-
Spain
-
Italy
-
Russia
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
Singapore
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global in vitro diagnostics market size was valued at USD 77.92 billion in 2023 and is expected to reach USD 78.42 billion in 2024.
b. The global in vitro diagnostics market is expected to grow at a compound annual growth rate of 4.4% from 2024 to 2030 to reach USD 101.58 billion by 2030.
b. Reagents dominated the in vitro diagnostics market with a market share of 65.88% in 2023 owing to a rise in the number of R&D initiatives related to reagents, an increase in the demand for self-test, and Point-of-Care (POC) products.
b. Major players in the IVD market are Abbott; bioMérieux SA; Bio-Rad Laboratories, Inc.; Siemens Healthineers; Qiagen; Quidel Corporation; F. Hoffmann-La Roche Ltd; Sysmex Corporation; and Becton Dickinson and Company.
b. Key factors that are driving the IVD market growth include the use of technologically advanced devices for early detection of diseases, the development of automated IVD systems, the increasing prevalence of diseases, and the growing geriatric population.
b. The coronavirus pandemic led to many pharmaceutical companies launching novel rapid diagnostic kits for the detection of the virus, with federal agencies around the globe enabling their early launch, which accelerated the market growth.
Table of Contents
Chapter 1 In Vitro Diagnostics Market: Methodology and Scope
1.1 Market Segmentation
1.1.1 Estimates And Forecast Timeline
1.2 Research Methodology
1.3 Information Procurement
1.3.1 Purchased Database
1.3.2 GVR’s Internal Database
1.3.3 Secondary Sources
1.3.4 Primary Research
1.3.5 Details Of Primary Research
1.4 Information Or Data Analysis
1.4.1 Data Analysis Models
1.5 Market Formulation & Validation
1.6 Model Details
1.6.1 Commodity Flow Analysis
1.6.1.1 Approach 1: Commodity Flow Approach
1.6.1.2 Approach 2: Country-wise market estimation using bottom-up approach
1.7 Global Market: CAGR Calculation
1.8 Research Assumptions
1.9 List of Secondary Sources
1.10 List of Primary Sources
1.11 Objectives
1.11.1 Objective 1
1.11.2 Objective 2
1.12 List Of Abbreviations
Chapter 2 In Vitro Diagnostics Market: Market Definitions
Chapter 3 In Vitro Diagnostics Market: Executive Summary
3.1 Market Summary
Chapter 4. In Vitro Diagnostics Market: Product Estimates & Trend Analysis
4.1. Segment Dashboard
4.2. In Vitro Diagnostics Market: Product Movement Analysis, USD Million, 2023 & 2030 (Volume Analysis)
4.3. Instruments
4.3.1. Instruments Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold)
4.4. Reagents
4.4.1. Reagents Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Tests Sold)
4.5. Services
4.5.1. Services Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
Chapter 5. In Vitro Diagnostics Market: Technology Estimates & Trend Analysis
5.1. Segment Dashboard
5.2. In Vitro Diagnostics Market: Technology Movement Analysis, USD Million, 2023 & 2030 (Volume Analysis)
5.3. Immunoassay
5.3.1. Immunoassay Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.3.2. Instruments
5.3.2.1. Instruments Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.3.3. Reagents
5.3.3.1. Reagents Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.3.4. Services
5.3.4.1. Services Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.4. Hematology
5.4.1. Hematology Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.4.2. Instruments
5.4.2.1. Instruments Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.4.3. Reagents
5.4.3.1. Reagents Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.4.4. Services
5.4.4.1. Services Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.5. Clinical Chemistry
5.5.1. Clinical Chemistry Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.5.2. Instruments
5.5.2.1. Instruments Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.5.3. Reagents
5.5.3.1. Reagents Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.5.4. Services
5.5.4.1. Services Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.6. Molecular Diagnostics
5.6.1. Molecular Diagnostics Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.6.2. Instruments
5.6.2.1. Instruments Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.6.3. Reagents
5.6.3.1. Reagents Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.6.4. Services
5.6.4.1. Services Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.7. Coagulation
5.7.1. Coagulation Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.7.2. Instruments
5.7.2.1. Instruments Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.7.3. Reagents
5.7.3.1. Reagents Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.7.4. Services
5.7.4.1. Services Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.8. Microbiology
5.8.1. Microbiology Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.8.2. Instruments
5.8.2.1. Instruments Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.8.3. Reagents
5.8.3.1. Reagents Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.8.4. Services
5.8.4.1. Services Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.9. Others
5.9.1. Others Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.9.2. Instruments
5.9.2.1. Instruments Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.9.3. Reagents
5.9.3.1. Reagents Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
5.9.4. Services
5.9.4.1. Services Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
Chapter 6. In Vitro Diagnostics Market: Application Estimates & Trend Analysis
6.1. Segment Dashboard
6.2. In Vitro Diagnostics Market: Application Movement Analysis, USD Million, 2023 & 2030
6.3. Infectious Disease
6.3.1. Infectious Disease Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
6.4. Diabetes
6.4.1. Diabetes Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
6.5. Oncology/Cancer
6.5.1. Oncology/Cancer Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
6.6. Cardiology
6.6.1. Cardiology Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
6.7. Nephrology
6.7.1. Nephrology Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
6.8. Autoimmune Diseases
6.8.1. Autoimmune Diseases Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
6.9. Drug Testing
6.9.1. Drug Testing Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
6.10. Others
6.10.1. Others Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
Chapter 7. In Vitro Diagnostics Market: Test Location Estimates & Trend Analysis
7.1. Segment Dashboard
7.2. In Vitro Diagnostics Market: Test Location Movement Analysis, USD Million, 2023 & 2030
7.3. Point of Care
7.3.1. Point of Care Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
7.4. Home-care
7.4.1. Home-care Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
7.5. Others
7.5.1. Others Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
Chapter 8. In Vitro Diagnostics Market: End Use Estimates & Trend Analysis
8.1. Segment Dashboard
8.2. In Vitro Diagnostics Market: End Use Movement Analysis, USD Million, 2023 & 2030
8.3. Hospitals
8.3.1. Hospitals Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
8.4. Laboratory
8.4.1. Laboratory Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
8.5. Home-care
8.5.1. Home-care Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
8.6. Others
8.6.1. Others Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Million)
Chapter 9. In Vitro Diagnostics Market: Regional Estimates & Trend Analysis
9.1. In Vitro Diagnostics Market Share, By Region, 2023 & 2030, USD Million
9.2. North America
9.2.1. North America In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.2.2. U.S.
9.2.2.1. Key Country Dynamics
9.2.2.2. Regulatory Landscape/Reimbursement Scenario
9.2.2.3. Competitive Insights
9.2.2.4. U.S. In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.2.3. Canada
9.2.3.1. Key Country Dynamics
9.2.3.2. Regulatory Landscape/Reimbursement Scenario
9.2.3.3. Competitive Insights
9.2.3.4. Canada In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.3. Europe
9.3.1. Europe In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.3.2. UK
9.3.2.1. Key Country Dynamics
9.3.2.2. Regulatory Landscape/Reimbursement Scenario
9.3.2.3. Competitive Insights
9.3.2.4. U.K. In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million)
9.3.3. Germany
9.3.3.1. Key Country Dynamics
9.3.3.2. Regulatory Landscape/Reimbursement Scenario
9.3.3.3. Competitive Insights
9.3.3.4. Germany In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.3.4. France
9.3.4.1. Key Country Dynamics
9.3.4.2. Regulatory Landscape/Reimbursement Scenario
9.3.4.3. Competitive Insights
9.3.4.4. France In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.3.5. Italy
9.3.5.1. Key Country Dynamics
9.3.5.2. Regulatory Landscape/Reimbursement Scenario
9.3.5.3. Competitive Insights
9.3.5.4. Italy In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.3.6. Spain
9.3.6.1. Key Country Dynamics
9.3.6.2. Regulatory Landscape/Reimbursement Scenario
9.3.6.3. Competitive Insights
9.3.6.4. Spain In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.3.7. Sweden
9.3.7.1. Key Country Dynamics
9.3.7.2. Regulatory Landscape/Reimbursement Scenario
9.3.7.3. Competitive Insights
9.3.7.4. Sweden In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.3.8. Denmark
9.3.8.1. Key Country Dynamics
9.3.8.2. Regulatory Landscape/Reimbursement Scenario
9.3.8.3. Competitive Insights
9.3.8.4. Denmark In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.3.9. Norway
9.3.9.1. Key Country Dynamics
9.3.9.2. Regulatory Landscape/Reimbursement Scenario
9.3.9.3. Competitive Insights
9.3.9.4. Norway In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.3.10. Russia
9.3.10.1. Key Country Dynamics
9.3.10.2. Regulatory Landscape/Reimbursement Scenario
9.3.10.3. Competitive Insights
9.3.10.4. Russis In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.4. Asia Pacific
9.4.1. Asia Pacific In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.4.2. China
9.4.2.1. Key Country Dynamics
9.4.2.2. Regulatory Landscape/Reimbursement Scenario
9.4.2.3. Competitive Insights
9.4.2.4. China In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.4.3. Japan
9.4.3.1. Key Country Dynamics
9.4.3.2. Regulatory Landscape/Reimbursement Scenario
9.4.3.3. Competitive Insights
9.4.3.4. Japan In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.4.4. India
9.4.4.1. Key Country Dynamics
9.4.4.2. Regulatory Landscape/Reimbursement Scenario
9.4.4.3. Competitive Insights
9.4.4.4. India In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.4.5. South Korea
9.4.5.1. Key Country Dynamics
9.4.5.2. Regulatory Landscape/Reimbursement Scenario
9.4.5.3. Competitive Insights
9.4.5.4. South Korea In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.4.6. Australia
9.4.6.1. Key Country Dynamics
9.4.6.2. Regulatory Landscape/Reimbursement Scenario
9.4.6.3. Competitive Insights
9.4.6.4. Australia In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.4.7. Thailand
9.4.7.1. Key Country Dynamics
9.4.7.2. Regulatory Landscape/Reimbursement Scenario
9.4.7.3. Competitive Insights
9.4.7.4. Thailand In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.4.8. Singapore
9.4.8.1. Key Country Dynamics
9.4.8.2. Regulatory Landscape/Reimbursement Scenario
9.4.8.3. Competitive Insights
9.4.8.4. Singapore In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.5. Latin America
9.5.1. Latin America In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.5.2. Brazil
9.5.2.1. Key Country Dynamics
9.5.2.2. Regulatory Landscape/Reimbursement Scenario
9.5.2.3. Competitive Insights
9.5.2.4. Brazil In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.5.3. Mexico
9.5.3.1. Key Country Dynamics
9.5.3.2. Regulatory Landscape/Reimbursement Scenario
9.5.3.3. Competitive Insights
9.5.3.4. Mexico In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.5.4. Argentina
9.5.4.1. Key Country Dynamics
9.5.4.2. Regulatory Landscape/Reimbursement Scenario
9.5.4.3. Competitive Insights
9.5.4.4. Argentina In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.6. Middle East and Africa
9.6.1. Middle East and Africa In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million)
9.6.2. Saudi Arabia
9.6.2.1. Key Country Dynamics
9.6.2.2. Regulatory Landscape/Reimbursement Scenario
9.6.2.3. Competitive Insights
9.6.2.4. Saudi Arabia In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.6.3. UAE
9.6.3.1. Key Country Dynamics
9.6.3.2. Regulatory Landscape/Reimbursement Scenario
9.6.3.3. Competitive Insights
9.6.3.4. UAE In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.6.4. South Africa
9.6.4.1. Key Country Dynamics
9.6.4.2. Regulatory Landscape/Reimbursement Scenario
9.6.4.3. Competitive Insights
9.6.4.4. South Africa In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
9.6.5. Kuwait
9.6.5.1. Key Country Dynamics
9.6.5.2. Regulatory Landscape/Reimbursement Scenario
9.6.5.3. Competitive Insights
9.6.5.4. Kuwait In Vitro Diagnostics Market Estimates and Forecasts, 2018 - 2030 (USD Million) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
Chapter 10. Competitive Landscape
10.1. Recent Developments & Impact Analysis by Key Market Participants
10.2. Company Categorization
10.3. Global Company Market Share Analysis, 2023
10.4. Company Heat Map Analysis
10.5. Strategy Mapping
10.5.1. Expansion
10.5.2. Mergers & Acquisition
10.5.3. Partnerships & Collaborations
10.5.4. New Product Launches
10.5.5. Research And Development
10.6. Company Profiles
10.6.1. QIAGEN
10.6.1.1. Participant’s Overview
10.6.1.2. Financial Performance
10.6.1.3. Product Benchmarking
10.6.1.4. Recent Developments
10.6.2. BD
10.6.2.1. Participant’s Overview
10.6.2.2. Financial Performance
10.6.2.3. Product Benchmarking
10.6.2.4. Recent Developments
10.6.3. bioMérieux SA
10.6.3.1. Participant’s Overview
10.6.3.2. Financial Performance
10.6.3.3. Product Benchmarking
10.6.3.4. Recent Developments
10.6.4. F. Hoffmann-La Roche, Ltd.
10.6.4.1. Participant’s Overview
10.6.4.2. Financial Performance
10.6.4.3. Product Benchmarking
10.6.4.4. Recent Developments
10.6.5. QuidelOrtho Corporation
10.6.5.1. Participant’s Overview
10.6.5.2. Financial Performance
10.6.5.3. Product Benchmarking
10.6.5.4. Recent Developments
10.6.6. Abbott
10.6.6.1. Participant’s Overview
10.6.6.2. Financial Performance
10.6.6.3. Product Benchmarking
10.6.6.4. Recent Developments
10.6.7. Agilent Technologies, Inc.
10.6.7.1. Participant’s Overview
10.6.7.2. Financial Performance
10.6.7.3. Product Benchmarking
10.6.7.4. Recent Developments
10.6.8. Siemens Healthineers AG
10.6.8.1. Participant’s Overview
10.6.8.2. Financial Performance
10.6.8.3. Product Benchmarking
10.6.8.4. Recent Developments
10.6.9. Bio-Rad Laboratories, Inc.
10.6.9.1. Participant’s Overview
10.6.9.2. Financial Performance
10.6.9.3. Product Benchmarking
10.6.9.4. Recent Developments
10.6.10. Danaher
10.6.10.1. Participant’s Overview
10.6.10.2. Financial Performance
10.6.10.3. Product Benchmarking
10.6.10.4. Recent Developments
10.6.11. Sysmex Corporation
10.6.11.1. Participant’s Overview
10.6.11.2. Financial Performance
10.6.11.3. Product Benchmarking
10.6.11.4. Recent Developments
10.6.12. Charles River Laboratories
10.6.12.1. Participant’s Overview
10.6.12.2. Financial Performance
10.6.12.3. Product Benchmarking
10.6.12.4. Recent Developments
10.6.13. Quest Diagnostics Incorporated
10.6.13.1. Participant’s Overview
10.6.13.2. Financial Performance
10.6.13.3. Product Benchmarking
10.6.13.4. Recent Developments
List of Tables
Table 1 List of Secondary Sources
Table 2 List of Abbreviations
Table 3 Regulatory Framework
Table 4 Global In Vitro Diagnostics Market, By Region, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million Number of instruments installed in thousands, Number of reagents sold in million
Table 5 Global In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 6 Global In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 7 Global In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 8 Global In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 9 Global In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 10 North America In Vitro Diagnostics Market, By Country, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 11 North America In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 12 North America In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 13 North America In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 14 North America In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 15 North America In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 16 U.S. In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 17 U.S. In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 18 U.S. In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 19 U.S. In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 20 U.S. In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 21 Canada In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 22 Canada In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 23 Canada In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 24 Canada In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 25 Canada In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 26 Europe In Vitro Diagnostics Market, By Country, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 27 Europe In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 28 Europe In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 29 Europe In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 30 Europe In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 31 Europe In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 32 UK In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 33 UK In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 34 UK In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 35 UK In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 36 UK In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 37 Germany In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 38 Germany In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 39 Germany In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 40 Germany America In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 41 Germany In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 42 France In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 43 France In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 44 France In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 45 France In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 46 France In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 47 Spain In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 48 Spain In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 49 Spain In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 50 Spain In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 51 Spain In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 52 Italy In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 53 Italy In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 54 Italy In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 55 Italy In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 56 Italy In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 57 Russia In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 58 Russia In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 59 Russia In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 60 Russia In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 61 Russia In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 62 Asia Pacific In Vitro Diagnostics Market, By Country, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 63 Asia Pacific In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 64 Asia Pacific In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 65 Asia Pacific In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 66 Asia-Pacific In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 67 Asia-Pacific In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 68 China In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 69 China In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 70 China In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 71 China In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 72 China In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 73 Japan In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 74 Japan In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 75 Japan In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 76 Japan In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 77 Japan In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 78 India In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 79 India In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 80 India In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 81 India In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 82 India In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 83 South Korea In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 84 South Korea In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 85 South Korea In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 86 South Korea In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 87 South Korea In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 88 Singapore In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 89 Singapore In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 90 Singapore In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 91 Singapore In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 92 Singapore In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 93 Australia In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 94 Australia In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 95 Australia In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 96 Australia In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 97 Australia In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 98 Latin America In Vitro Diagnostics Market, By Country, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 99 Latin America In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 100 Latin America In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 101 Latin America In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 102 Latin America In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 103 Latin America In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 104 Brazil In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 105 Brazil In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 106 Brazil In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 107 Brazil In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 108 Brazil In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 109 Mexico In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 110 Mexico In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 111 Mexico In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 112 Mexico In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 113 Mexico In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 114 Argentina In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 115 Argentina In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 116 Argentina In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 117 Argentina In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 118 Argentina In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 119 MEA In Vitro Diagnostics Market, By Country, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 120 MEA In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 121 MEA In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 122 MEA In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 123 MEA In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 124 MEA In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 125 South Africa In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 126 South Africa In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 127 South Africa In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 128 South Africa In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 129 UAE In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 130 UAE In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 131 UAE In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 132 UAE In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 133 UAE In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 134 UAE In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
Table 135 Saudi Arabia In Vitro Diagnostics Market, By Product, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 136 Saudi Arabia In Vitro Diagnostics Market, By Technology, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 137 Saudi Arabia In Vitro Diagnostics Market, By Application, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 138 Saudi Arabia In Vitro Diagnostics Market, By End-use, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in
Table 139 Saudi Arabia In Vitro Diagnostics Market, By Test Location, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in million
List of Figures
Fig. 1 In Vitro Diagnostics market segmentation
Fig. 2 Market research process
Fig. 3 Data triangulation techniques
Fig. 4 Primary research pattern
Fig. 5 Market research approaches
Fig. 6 Value-chain-based sizing & forecasting
Fig. 7 QFD modeling for market share assessment
Fig. 8 Market formulation & validation
Fig. 9 In vitro diagnostic market snapshot
Fig. 10 Parent market outlook, 2023
Fig. 11 Penetration and growth prospect mapping
Fig. 12 In vitro diagnostic market driver impact
Fig. 13 Global population by age group, 2016 & 2027
Fig. 14 Global elderly population
Fig. 15 New cancer cases by year
Fig. 16 In vitro diagnostic market restraint impact
Fig. 17 Market entry strategy
Fig. 18 SWOT analysis, by factor (political & legal, economic, and technological)
Fig. 19 Porter’s five forces analysis
Fig. 20 Consumer behavior analysis
Fig. 21 Strategy mapping
Fig. 22 IVD market: Product outlook and key takeaways
Fig. 23 IVD market: Products movement analysis
Fig. 24 Instruments market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands
Fig. 25 Reagents market estimates and forecast, 2018 - 2030 (USD Million), Number of reagents sold in millions
Fig. 26 Services market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 27 IVD market: Technology outlook and key takeaways
Fig. 28 IVD Market: Technology movement analysis
Fig. 29 Immunoassay market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 30 Instruments market for immunoassay estimates and forecast, 2018 - 2030 (USD Million)
Fig. 31 Reagents market for immunoassay estimates and forecast, 2018 - 2030 (USD Million)
Fig. 32 Services market for immunoassay estimates and forecast, 2018 - 2030 (USD Million)
Fig. 33 Hematology market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 34 Instruments market for hematology estimates and forecast, 2018 - 2030 (USD Million)
Fig. 35 Reagents market for hematology estimates and forecast, 2018 - 2030 (USD Million)
Fig. 36 Services market for hematology estimates and forecast, 2018 - 2030 (USD Million)
Fig. 37 Clinical chemistry market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 38 Instruments market for clinical chemistry estimates and forecast, 2018 - 2030 (USD Million)
Fig. 39 Reagents market for clinical chemistry estimates and forecast, 2018 - 2030 (USD Million)
Fig. 40 Services market for clinical chemistry estimates and forecast, 2018 - 2030 (USD Million)
Fig. 41 Molecular diagnostics market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 42 Instruments market for molecular diagnostics estimates and forecast, 2018 - 2030 (USD Million)
Fig. 43 Reagents market for molecular diagnostics estimates and forecast, 2018 - 2030 (USD Million)
Fig. 44 Services market for molecular diagnostics estimates and forecast, 2018 - 2030 (USD Million)
Fig. 45 Coagulation market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 46 Instruments market for coagulation estimates and forecast, 2018 - 2030 (USD Million)
Fig. 47 Reagents market for coagulation estimates and forecast, 2018 - 2030 (USD Million)
Fig. 48 Services market for coagulation estimates and forecast, 2018 - 2030 (USD Million)
Fig. 49 Microbiology market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 50 Instruments market for microbiology estimates and forecast, 2018 - 2030 (USD Million)
Fig. 51 Reagents market for microbiology estimates and forecast, 2018 - 2030 (USD Million)
Fig. 52 Services market for microbiology estimates and forecast, 2018 - 2030 (USD Million)
Fig. 53 Others market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 54 Instruments others market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 55 Reagents others market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 56 Services market for other technology estimates and forecast, 2018 - 2030 (USD Million)
Fig. 57 IVD market: Application outlook and key takeaways
Fig. 58 IVD market: Application movement analysis
Fig. 59 Infectious diseases market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 60 Diabetes market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 61 Oncology market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 62 Cardiology market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 63 Nephrology market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 64 Autoimmune diseases market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 65 Drug testing market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 66 Others market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 67 IVD market: End-use outlook and key takeaways
Fig. 68 IVD treatment market: End-use movement analysis
Fig. 69 Hospitals market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 70 Laboratory market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 71 Home care market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 72 Other market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 73 IVD market: test location outlook and key takeaways
Fig. 74 IVD treatment market: Test location movement analysis
Fig. 75 Point-of-care market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 76 Homecare market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 77 Others market estimates and forecast, 2018 - 2030 (USD Million)
Fig. 78 IVD Market: Regional outlook and key takeaways
Fig. 79 IVD market: Regional movement analysis
Fig. 80 North America
Fig. 81 North America, SWOT
Fig. 82 North America. market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 83 U.S. key country dynamics
Fig. 84 U.S. market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 85 Canada key country dynamics
Fig. 86 Canada market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 87 Europe
Fig. 88 Europe, SWOT
Fig. 89 Europe market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 90 UK key country dynamics
Fig. 91 UK market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 92 Germany key country dynamics
Fig. 93 Germany market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 94 Spain key country dynamics
Fig. 95 Spain market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 96 France key country dynamics
Fig. 97 France market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 98 Italy key country dynamics
Fig. 99 Italy market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 100 Russia key country dynamics
Fig. 101 Russia market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 102 Denmark key country dynamics
Fig. 103 Denmark market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 104 Sweden key country dynamics
Fig. 105 Sweden market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 106 Norway key country dynamics
Fig. 107 Norway market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 108 Asia Pacific
Fig. 109 Asia Pacific, SWOT
Fig. 110 Asia Pacific market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 111 Japan key country dynamics
Fig. 112 Japan market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 113 China key country dynamics
Fig. 114 China. market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 115 India key country dynamics
Fig. 116 India. market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 117 South Korea key country dynamics
Fig. 118 South Korea market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 119 Singapore key country dynamics
Fig. 120 Singapore market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 121 Thailand key country dynamics
Fig. 122 Thailand market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 123 Australia key country dynamics
Fig. 124 Australia market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 125 Latin America
Fig. 126 Latin America, SWOT
Fig. 127 Latin America market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 128 Brazil key country dynamics
Fig. 129 Brazil market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 130 Mexico key country dynamics
Fig. 131 Mexico market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 132 Argentina key country dynamics
Fig. 133 Argentina market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 134 Middle East & Africa
Fig. 135 Middle East & Africa, SWOT
Fig. 136 MEA market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 137 South Africa key country dynamics
Fig. 138 South Africa market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 139 United Arab Emirates key country dynamics
Fig. 140 United Arab Emirates market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 141 Saudi Arabia key country dynamics
Fig. 142 Saudi Arabia market estimates and forecast, 2018 - 2030 (USD Million), Number of instruments installed in thousands, Number of reagents sold in millions
Fig. 143 Ansoff matrix
Fig. 144 Competition categorization
Fig. 145 Company market share analysis, 2023
Fig. 146 Global mass spectrometry company market share analysis, 2023
Fig. 147 North America mass spectrometry company market share analysis, 2023
Fig. 148 Europe mass spectrometry company market share analysis, 2023
Fig. 149 Asia Pacific mass spectrometry company market share analysis, 2023
Fig. 150 Global immunoassay company market share analysis, 2023
Fig. 151 North America immunoassay company market share analysis, 2023
Fig. 152 Europe immunoassay company market share analysis, 2023
Fig. 153 Asia Pacific immunoassay company market share analysis, 2023
Fig. 154 Global PCR company market share analysis, 2023
Fig. 155 North America PCR company market share analysis, 2023
Fig. 156 Europe PCR company market share analysis, 2023
Fig. 157 Asia Pacific PCR company market share analysis, 2023
Fig. 158 Company market position analysis
Fig. 159 Company market position analysis
Fig. 160 Company market position analysis
Fig. 161 Company profiles
Fig. 162 Regional network mapWhat questions do you have? Get quick response from our industry experts. Request a Free ConsultationMarket Segmentation
- In Vitro Diagnostics Product Outlook (Revenue, USD Million, 2018 - 2030) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
- Instruments
- Reagents
- Software
- Molecular Diagnostics Technology Outlook (Revenue, USD Million, 2018 - 2030)
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- In Vitro Diagnostics Application Outlook (Revenue, USD Million, 2018 - 2030)
- Infectious Diseases
- Diabetes
- Oncology
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Other applications
- In Vitro Diagnostics Test Location Outlook (Revenue, USD Million, 2018 - 2030)
- Point of Care
- Home-care
- Others
- In Vitro Diagnostics End Use Outlook (Revenue, USD Million, 2018 - 2030)
- Hospitals
- Laboratory
- Home Care
- Others
- In Vitro Diagnostics Regional Outlook (Revenue, USD Million, 2018 - 2030) (Volume, Number of Instruments Sold) (Volume, Number of Tests Sold)
- North America
- North America In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- North America In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- North America In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- North America In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- North America In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- U.S.
- U.S. In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- U.S. In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- U.S. In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- U.S. In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- U.S. In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- U.S. In Vitro Diagnostics Market, By Product
- Canada
- Canada In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Canada In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Canada In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Canada In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Canada In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Canada In Vitro Diagnostics Market, By Product
- North America In Vitro Diagnostics Market, By Product
- Europe
- Europe In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Europe In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Europe In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Europe In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Europe In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- UK
- UK In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- UK In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- UK In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- UK In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- UK In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- UK In Vitro Diagnostics Market, By Product
- Germany
- Germany In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Germany In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Germany In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Germany In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Germany In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Germany In Vitro Diagnostics Market, By Product
- France
- France In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- France In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- France In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- France In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- France In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- France In Vitro Diagnostics Market, By Product
- Spain
- Spain In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Spain In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Spain In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Spain In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Spain In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Spain In Vitro Diagnostics Market, By Product
- Italy
- Italy In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Italy In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Italy In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Italy In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Italy In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Italy In Vitro Diagnostics Market, By Product
- Russia
- Russia In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Russia In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Russia In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Russia In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Russia In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Russia In Vitro Diagnostics Market, By Product
- Denmark
- Denmark In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Denmark In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Denmark In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Denmark In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Denmark In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Denmark In Vitro Diagnostics Market, By Product
- Sweden
- Sweden In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Sweden In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Sweden In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Sweden In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Sweden In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Sweden In Vitro Diagnostics Market, By Product
- Norway
- Norway In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Norway In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Norway In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Norway In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Norway In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Norway In Vitro Diagnostics Market, By Product
- Europe In Vitro Diagnostics Market, By Product
- Asia Pacific
- Asia Pacific In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Asia Pacific In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Asia Pacific In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Asia Pacific In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Asia Pacific In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Japan
- Japan In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Japan In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Japan In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Japan In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Japan In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Japan In Vitro Diagnostics Market, By Product
- China
- China In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- China In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- China In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- China In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- China In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- China In Vitro Diagnostics Market, By Product
- India
- India In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- India In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- North America In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- India In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- India In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- India In Vitro Diagnostics Market, By Product
- South Korea
- South Korea In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- South Korea In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- South Korea In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- South Korea In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- South Korea In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- South Korea In Vitro Diagnostics Market, By Product
- Australia
- Australia In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Australia In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Australia In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Australia In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Australia In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Australia In Vitro Diagnostics Market, By Product
- Thailand
- Thailand In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Thailand In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Thailand In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Thailand In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Thailand In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Thailand In Vitro Diagnostics Market, By Product
- Singapore
- Singapore In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Singapore In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Singapore In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Singapore In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Singapore In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Singapore In Vitro Diagnostics Market, By Product
- Asia Pacific In Vitro Diagnostics Market, By Product
- Latin America
- Latin America In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Latin America In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Latin America In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Latin America In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Latin America In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Brazil
- Brazil In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Brazil In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Brazil In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Brazil In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- North America In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Brazil In Vitro Diagnostics Market, By Product
- Mexico
- Mexico In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Mexico In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Mexico In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Mexico In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Mexico In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Mexico In Vitro Diagnostics Market, By Product
- Argentina
- Argentina In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Argentina In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Argentina In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Argentina In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Argentina In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Argentina In Vitro Diagnostics Market, By Product
- Latin America In Vitro Diagnostics Market, By Product
- Middle East and Africa (MEA)
- Middle East and Africa (MEA) In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Middle East and Africa (MEA) In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Middle East and Africa (MEA) In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Middle East and Africa (MEA) In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Middle East and Africa (MEA) In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- South Africa
- South Africa In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- South Africa In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- South Africa In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- South Africa In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- South Africa In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- South Africa In Vitro Diagnostics Market, By Product
- Saudi Arabia
- Saudi Arabia In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Saudi Arabia In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Saudi Arabia In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Saudi Arabia In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Saudi Arabia In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Saudi Arabia In Vitro Diagnostics Market, By Product
- UAE
- UAE In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- UAE In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- UAE In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- UAE In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- UAE In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- UAE In Vitro Diagnostics Market, By Product
- Kuwait
- Kuwait In Vitro Diagnostics Market, By Product
- Instruments
- Reagents
- Services
- Kuwait In Vitro Diagnostics Market, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Coagulation
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Kuwait In Vitro Diagnostics Market, By Application
- Infectious Disease
- Diabetes
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- Others
- Kuwait In Vitro Diagnostics Market, By Test Location
- Point of Care
- Home-care
- Others
- Kuwait In Vitro Diagnostics Market, By End Use
- Hospitals
- Laboratory
- Home Care
- Others
- Kuwait In Vitro Diagnostics Market, By Product
- Middle East and Africa (MEA) In Vitro Diagnostics Market, By Product
- North America
Report content
Qualitative Analysis
- Industry overview
- Industry trends
- Market drivers and restraints
- Market size
- Growth prospects
- Porter’s analysis
- PESTEL analysis
- Key market opportunities prioritized
- Competitive landscape
- Company overview
- Financial performance
- Product benchmarking
- Latest strategic developments
Quantitative Analysis
- Market size, estimates, and forecast from 2018 to 2030
- Market estimates and forecast for product segments up to 2030
- Regional market size and forecast for product segments up to 2030
- Market estimates and forecast for application segments up to 2030
- Regional market size and forecast for application segments up to 2030
- Company financial performance
What questions do you have? Get quick response from our industry experts. Request a Free ConsultationResearch Methodology
A three-pronged approach was followed for deducing the in vitro diagnostics market estimates and forecasts. The process has three steps: information procurement, analysis, and validation. The whole process is cyclical, and steps repeat until the estimates are validated. The three steps are explained in detail below:
Information procurement: Information procurement is one of the most extensive and important stages in our research process, and quality data is critical for accurate analysis. We followed a multi-channel data collection process for in vitro diagnostics market to gather the most reliable and current information possible.
- We buy access to paid databases such as Hoover’s and Factiva for company financials, industry information, white papers, industry journals, SME journals, and more.
- We tap into Grand View’s proprietary database of data points and insights from active and archived monitoring and reporting.
- We conduct primary research with industry experts through questionnaires and one-on-one phone interviews.
- We pull from reliable secondary sources such as white papers and government statistics, published by organizations like WHO, NGOs, World Bank, etc., Key Opinion Leaders (KoL) publications, company filings, investor documents, and more.
- We purchase and review investor analyst reports, broker reports, academic commentary, government quotes, and wealth management publications for insightful third-party perspectives.
Analysis: We mine the data collected to establish baselines for forecasting, identify trends and opportunities, gain insight into consumer demographics and drivers, and so much more. We utilized different methods of in vitro diagnostics market data depending on the type of information we’re trying to uncover in our research.
-
Market Research Efforts: Bottom-up Approach for estimating and forecasting demand size and opportunity, top-down Approach for new product forecasting and penetration, and combined approach of both Bottom-up and Top-down for full coverage analysis.
-
Value-Chain-Based Sizing & Forecasting: Supply-side estimates for understanding potential revenue through competitive benchmarking, forecasting, and penetration modeling.
-
Demand-side estimates for identifying parent and ancillary markets, segment modeling, and heuristic forecasting.
-
Qualitative Functional Deployment (QFD) Modelling for market share assessment.
Market formulation and validation: We mine the data collected to establish baselines for forecasting, identify trends and opportunities, gain insight into consumer demographics and drivers, and so much more. We utilize different methods of data analysis depending on the type of information we’re trying to uncover in our research.
-
Market Formulation: This step involves the finalization of market numbers. This step on an internal level is designed to manage outputs from the Data Analysis step.
-
Data Normalization: The final market estimates and forecasts are then aligned and sent to industry experts, in-panel quality control managers for validation.
-
This step also entails the finalization of the report scope and data representation pattern.
-
Validation: The process entails multiple levels of validation. All these steps run in parallel, and the study is forwarded for publishing only if all three levels render validated results.
In Vitro Diagnostics Market Categorization:
The in vitro diagnostics market was categorized into six segments, namely product (Instruments, Reagents, Services), application (Infectious Disease, Diabetes, Oncology, Cardiology, Nephrology, Autoimmune Disease, Drug Testing), technology ( Immunoassay, Hematology, Clinical Chemistry, Molecular Diagnostics, Coagulation, Microbiology, ), end-use (Hospitals, Laboratories, Home Care), test location (Point of Care, Home Care), and region (North America, Europe, Asia Pacific, Latin America, Middle East & Africa).
Segment Market Methodology:
The in vitro diagnostics market was segmented into product, application, technology, end-use, test location and regions. The demand at a segment level was deduced using a funnel method. Concepts like the TAM, SAM, SOM, etc., were put into practice to understand the demand. We at GVR deploy three methods to deduce market estimates and determine forecasts. These methods are explained below:
Market research approaches: Bottom-up
-
Demand estimation of each product across countries/regions summed up to from the total market.
-
Variable analysis for demand forecast.
-
Demand estimation via analyzing paid database, and company financials either via annual reports or paid database.
-
Primary interviews for data revalidation and insight collection.
Market research approaches: Top-down
-
Used extensively for new product forecasting or analyzing penetration levels.
-
Tool used invoice product flow and penetration models Use of regression multi-variant analysis for forecasting Involves extensive use of paid and public databases.
-
Primary interviews and vendor-based primary research for variable impact analysis.
Market research approaches: Combined
- This is the most common method. We apply concepts from both the top-down and bottom-up approaches to arrive at a viable conclusion.
Regional Market Methodology:
The in vitro diagnostics market was analyzed at a regional level. The globe was divided into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa, keeping in focus variables like consumption patterns, export-import regulations, consumer expectations, etc. These regions were further divided into twenty countries, namely, the U.S.; Canada; Germany; the UK.; France; Italy; Spain; Russia; China; Japan; India; Australia; South Korea; Singapore; Brazil; Mexico; Argentina; South Africa; Saudi Arabia; UAE.
All three above-mentioned market research methodologies were applied to arrive at regional-level conclusions. The regions were then summed up to form the global market.
In vitro diagnostics market companies & financials:
The in vitro diagnostics market was analyzed via companies operating in the sector. Analyzing these companies and cross-referencing them to the demand equation helped us validate our assumptions and conclusions. Key market players analyzed include:
-
BIOMÉRIEUX SA, BioMérieux SA was founded in 1963 and headquartered in France. The company is a subsidiary of Institut Mérieux, which provides in vitro diagnostics, especially for infectious diseases. Moreover, the company produces equipment, chemical substances, and computer programs utilized by medical laboratories to detect various types of infections, including tuberculosis, sepsis, hospital-acquired infections, AIDS, and cardiovascular and cancer-related illnesses. The business operates 19 production facilities and 20 R&D centers. It has a presence in 43 nations and distributes its goods in 160 nations, and the business has a global workforce of 13,000. Considering the last couple of years, the business showcased a strong commercial performance, with a 10.5% increase in overall sales. This can be attributed to the company's extensive installed base of equipment worldwide, which enables it to cater to a larger consumer base.
-
BIO-RAD LABORATORIES, INC, Bio-Rad Laboratories, Inc. is headquartered in the U.S. and was established in 1952. The company specializes in manufacturing products and systems that are utilized to separate complex biological and chemical materials. Again, the company analyzes, identifies, and purifies their respective components. Additionally, the organization operates in two segments such as clinical diagnostics and life sciences. Under the clinical diagnostics segment, Bio-Rad Laboratories designs, manufactures, supports and sells test systems, informatics systems, test kits, and quality controls to clinical laboratories worldwide. On the other hand, the life sciences segment is involved in creating and discovering tools necessary for biological processes. Bio-Rad Laboratories, Inc. produces reagents, laboratory instruments, and apparatus used in biopharmaceutical production, research techniques, and food testing regimes. Key applications of Bio-Rad products include hospitals, laboratories, research institutions, biotechnology companies, and pharmaceutical firms. The company has direct distribution channels in around 35 countries and has a workforce of over 8,000 employees globally. Moving forward, the company is looking for expansion in the near future.
-
BECTON, DICKINSON AND COMPANY, Becton, Dickinson and Company, founded in 1897, is a leading medical technology company operating worldwide. The company specializes in manufacturing and marketing a diverse range of medical devices, diagnostic products, and laboratory equipment, along with medical supplies. BD operates through three business segments: BD Diagnostics, BD Medical, and BD Biosciences. BD Diagnostics offers a wide range of products and instruments for collecting and safely transporting diagnostic specimens. Furthermore, it provides reagent systems and instruments that accurately detect various healthcare-associated infections, infectious diseases, and cancer. Additionally, the segment offers fully automated molecular systems that provide rapid results, require less hands-on time, and are user-friendly. BD has a global presence, with offices in around 50 countries and employs over 65,000 employees worldwide.
-
SIEMENS HEALTHINEERS AG, Siemens Healthcare GmbH, a Germany-based healthcare company, specializes in providing cutting-edge healthcare solutions, including in vitro and in vivo diagnostics. In addition, the company offers imaging and therapy systems, audiology solutions, clinical products, and healthcare IT solutions. The company was established in 2015 as a separate entity and provides medical accessories, medical electronics solutions, OEM components, and refurbished medical imaging and therapy systems. Its vast range of products and solutions includes therapeutic drug monitoring, cardiology, organ transplant, urology, infectious disease, diabetes, women's health, surgery, oncology, neurology, and growth disorders. The company has a global workforce of about 54,000 employees and also provides technical, maintenance, and professional consulting services.
-
QIAGEN, Qiagen is a holding company that provides samples and assay technologies for various areas such as academic research, applied testing, molecular diagnostics, and pharmaceutical research. The company markets more than 500 products, including consumable kits and automation systems. Qiagen products for molecular diagnostics cover relevant areas of healthcare such as early disease diagnosis, establishment of diagnosis, diagnostic testing for suitable treatment determination, and point-of-need testing. The company's customers include healthcare providers, pharmaceutical and biotechnology companies, government or industrial customers, and researchers. It was incorporated in 1984 and is headquartered in Germany, employing over 5,610 people globally.
-
QUIDEL CORPORATION, Quidel Corporation was established in 1979 and headquartered in the U. S. The company specializes in developing and manufacturing point-of-care diagnostic solutions for contagious diseases such as group A streptococcal infections and influenza. The company mainly aims to develop and market diagnostic solutions. These testing solutions are mainly developed to cure women’s health, gastrointestinal diseases, and infectious diseases. Moreover, the company is proficient in selling its commodities to distributors and end-users. It has supply chain channels in the U. S. markets to distribute products in the local market. The company employs more than 1600 people in its worldwide offices.
-
F. HOFFMANN-LA ROCHE LTD., F. Hoffmann-La Roche Ltd., a Switzerland-based healthcare company, operates in two divisions - pharmaceuticals and diagnostics. One of its segments, Roche Diagnostics, consists of Roche Professional Diagnostics (RPD), Roche Molecular Diagnostics (RMD), Roche Tissue Diagnostics, and Roche Diabetes Care. Roche Diagnostics offers a range of products such as blood glucose meters, point-of-care testing devices, high-throughput analyzers for commercial and hospital diagnostic laboratories, and reagents and instruments for life sciences research. RMD, a segment of Roche Diagnostics, is involved in the development, manufacturing, and distribution of tests based on PCR technology and medical diagnostic products, platforms, and services. RMD's automated platforms and diagnostic tests focus on areas including virology, genomics and oncology, blood screening tests, HPV and CT/NG, and microbiology. The company caters to various clients such as physicians, researchers, laboratories, hospitals, patients, and blood banks. With a global workforce of 101,465 employees, Roche is a leading name in the healthcare industry.
-
SYSMEX CORPORATION, Sysmex Corporation is a Japanese medical devices company that was established in 1968. The company primarily focuses on the field of in vitro diagnostics, manufacturing and developing laboratory test drugs, clinical laboratory equipment, and other products associated with laboratory testing. Sysmex operates through three major divisions: Diagnostic business, life sciences business, and other businesses. The Diagnostic business division is responsible for producing and selling instruments, reagents, and laboratory systems that cater to hematology, hemostasis, immunochemistry, clinical chemistry, and urinalysis fields. Sysmex has a strong presence both in domestic as well as international markets, such as Europe, the Americas, and Asia Pacific. The company currently employs more than 9,000 people worldwide.
-
CHARLES RIVER LABORATORIES, Charles River Laboratories is headquartered in the U.S.. It was founded in 1947 and is a leading provider of clinical and preclinical laboratory services. Its clientele includes biotechnology industries, government agencies, and pharmaceutical and medical device companies. The company specializes in basic research, clinical support, and process manufacturing and offers a diverse range of products, such as biomarkers, cellular therapeutics, biosimilars, and vaccines. Its services are not limited to healthcare and extend to industries such as agrochemicals, veterinary medicines, dairy, food & beverage, home & beauty, medical devices, nutraceuticals, and dietary supplements. With 65 facilities spanning 16 countries, the company has a global presence and employs over 12,400 people.
-
QUEST DIAGNOSTICS, Quest Diagnostics, an American-based company that offers clinical laboratory tests, is a publicly traded corporation. It was established as an independent entity known as Quest Diagnostics in 1997. The company has its headquarters in the United States and administrative offices in Lyndhurst, Collegeville, and Pennsylvania. In the field of pharmacy and other services, it was ranked at the top by Fortune magazine in 2013. Quest Diagnostics conducts more than 150 million tests annually and has formed partnerships with various clinics and hospitals around the world. It currently operates over 6,850 patient and retail access points and serves approximately 50% of the hospitals in the United States. The firm employs mergers and acquisitions as one of the primary strategies to expand its reach.
Value chain-based sizing & forecasting
Supply Side Estimates
-
Company revenue estimation via referring to annual reports, investor presentations, and Hoover’s.
-
Segment revenue determination via variable analysis and penetration modeling.
-
Competitive benchmarking to identify market leaders and their collective revenue shares.
-
Forecasting via analyzing commercialization rates, pipelines, market initiatives, distribution networks, etc.
Demand side estimates
-
Identifying parent markets and ancillary markets
-
Segment penetration analysis to obtain pertinent
-
revenue/volume
-
Heuristic forecasting with the help of subject matter experts
-
Forecasting via variable analysis
In Vitro Diagnostics Market Report Objectives:
-
Understanding market dynamics (in terms of drivers, restraints, & opportunities) in the countries.
-
Understanding trends & variables in the individual countries & their impact on growth and using analytical tools to provide high-level insights into the market dynamics and the associated growth pattern.
-
Understanding market estimates and forecasts (with the base year as 2022, historic information from 2018 to 2021, and forecast from 2023 to 2030). Regional estimates & forecasts for each category are available and are summed up to form the global market estimates.
In Vitro Diagnostics Market Report Assumptions:
-
The report provides market value for the base year 2022 and a yearly forecast till 2030 in terms of revenue/volume or both. The market for each of the segment outlooks has been provided on region & country basis for the above-mentioned forecast period.
-
The key industry dynamics, major technological trends, and application markets are evaluated to understand their impact on the demand for the forecast period. The growth rates were estimated using correlation, regression, and time-series analysis.
-
We have used the bottom-up approach for market sizing, analyzing key regional markets, dynamics, & trends for various products and end-users. The total market has been estimated by integrating the country markets.
-
All market estimates and forecasts have been validated through primary interviews with the key industry participants.
-
Inflation has not been accounted for to estimate and forecast the market.
-
Numbers may not add up due to rounding off.
-
Europe consists of EU-8, Central & Eastern Europe, along with the Commonwealth of Independent States (CIS).
-
Asia Pacific includes South Asia, East Asia, Southeast Asia, and Oceania (Australia & New Zealand).
-
Latin America includes Central American countries and the South American continent
-
Middle East includes Western Asia (as assigned by the UN Statistics Division) and the African continent.
Primary Research
GVR strives to procure the latest and unique information for reports directly from industry experts, which gives it a competitive edge. Quality is of utmost importance to us, therefore every year we focus on increasing our experts’ panel. Primary interviews are one of the critical steps in identifying recent market trends and scenarios. This process enables us to justify and validate our market estimates and forecasts to our clients. With more than 8,000 reports in our database, we have connected with some key opinion leaders across various domains, including healthcare, technology, consumer goods, and the chemical sector. Our process starts with identifying the right platform for a particular type of report, i.e., emails, LinkedIn, seminars, or telephonic conversation, as every report is unique and requires a differentiated approach.
We send out questionnaires to different experts from various regions/ countries, which is dependent on the following factors:
-
Report/Market scope: If the market study is global, we send questionnaires to industry experts across various regions, including North America, Europe, Asia Pacific, Latin America, and MEA.
-
Market Penetration: If the market is driven by technological advancements, population density, disease prevalence, or other factors, we identify experts and send out questionnaires based on region or country dominance.
The time to start receiving responses from industry experts varies based on how niche or well-penetrated the market is. Our reports include a detailed chapter on the KoL opinion section, which helps our clients understand the perspective of experts already in the market space.
What questions do you have? Get quick response from our industry experts. Request a Free ConsultationShare this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





