- Home
- »
- Clinical Diagnostics
- »
-
Infectious Respiratory Disease Diagnostics Market, 2033GVR Report cover
![Infectious Respiratory Disease Diagnostics Market Size, Share & Trends Report]()
Infectious Respiratory Disease Diagnostics Market (2026 - 2033) Size, Share & Trends Analysis By Product (Instruments, Services), By Application (COVID-19, Influenza, Tuberculosis), By Technology (Immunoassay, Microbiology, Molecular Diagnostics), By End-use, By Region, And Segment Forecasts
- Report ID: GVR-4-68039-976-4
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Infectious Respiratory Disease Diagnostics Market Summary
The global infectious respiratory disease diagnostics market size was estimated at USD 12.56 billion in 2025 and is projected to reach USD 15.70 billion by 2033, growing at a CAGR of 2.90% from 2026 to 2033. The market is being driven by several key factors, including the growing prevalence of diseases like tuberculosis and pneumonia, advancements in diagnostic technologies, and increased partnerships and collaborations between major players.
Key Market Trends & Insights
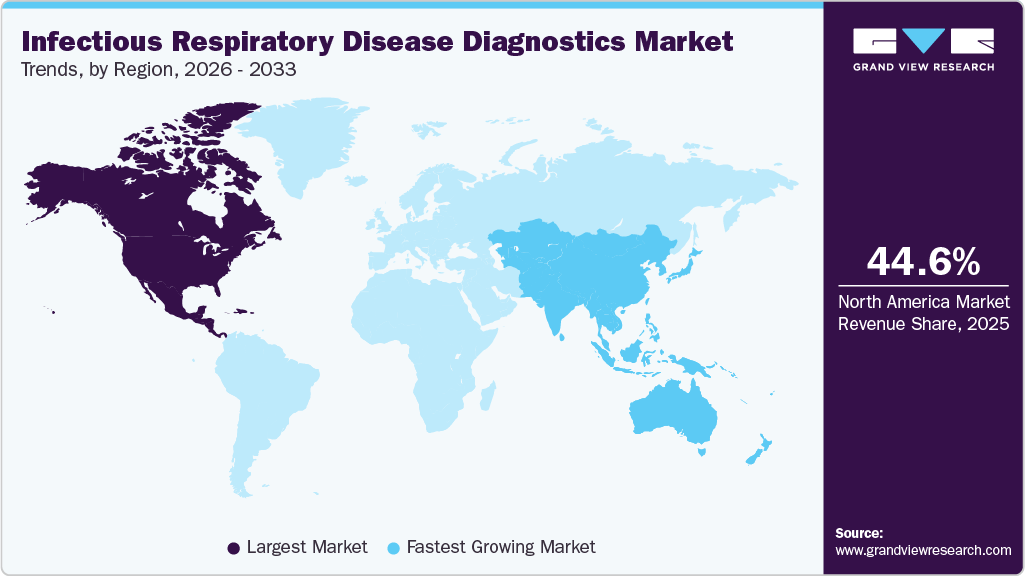
- North America infectious respiratory disease diagnostics market dominated the global market and accounted for the largest revenue share of 44.62% in 2025.
- The infectious respiratory disease diagnostics market in the U.S. held a significant share of the North American market in 2025.
- Based on product, the reagent segment dominated the global market and accounted for the largest revenue share of 65.38% 2025.
- Based on technology, the diagnostics segment accounted for 67.63% of the overall market share in 2025.
- Based on application, the COVID-19 segment dominated the global market in 2025, accounting for 89.24% of the revenue.
Market Size & Forecast
- 2025 Market Size: USD 12.56 Billion
- 2033 Projected Market Size: USD 15.70 Billion
- CAGR (2025-2033): 2.90%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
For instance, in February 2023, Thermo Fisher Scientific and Mylab Discovery Solutions announced a partnership to develop and distribute diagnostic test kits for infectious diseases such as multi-drug-resistant tuberculosis, hepatitis B virus, hepatitis C virus, HIV, and genetic analysis (HLA B27) in India. The collaboration aims to combine Thermo Fisher’s global expertise with Mylab’s local manufacturing capabilities to expand access to high-quality testing solutions.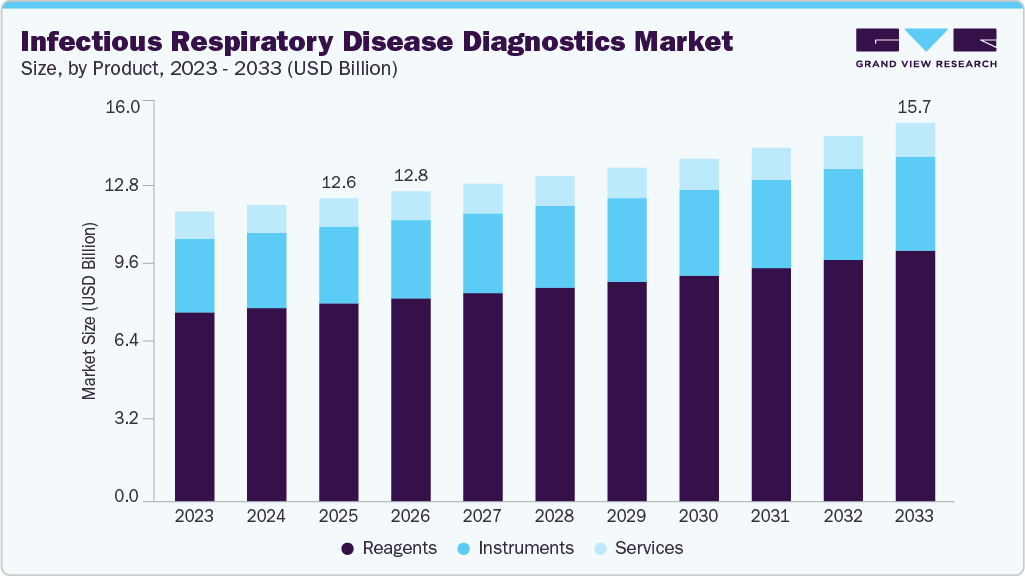
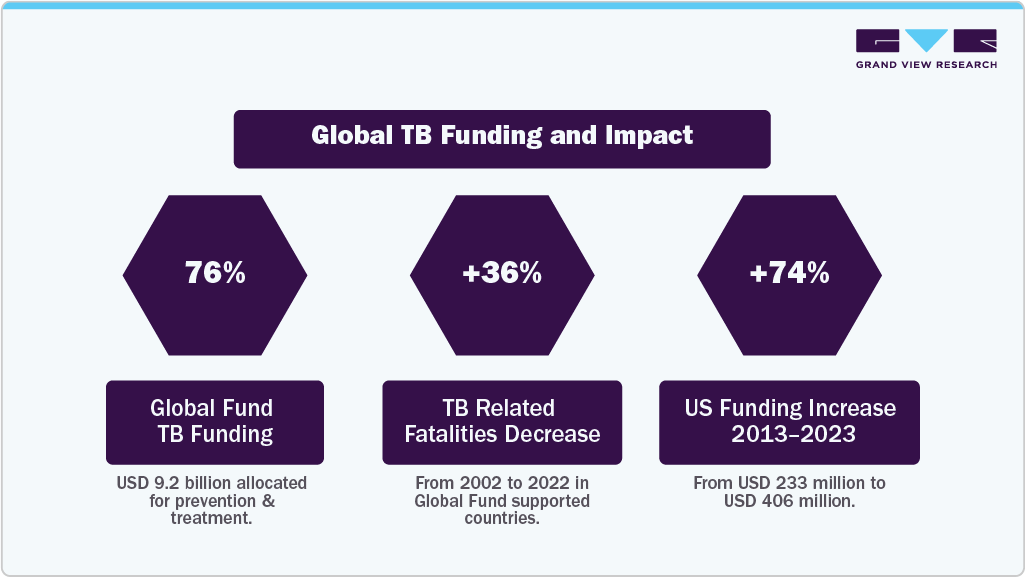
The increasing funding for TB diagnosis programs is crucial in the fight against this global disease. The current funding for prevention, diagnosis, and treatment is insufficient to meet the demand, hindering progress in reducing incidence & mortality rates. For instance, an annual investment of USD 22 billion by 2027 was set, with an additional USD 5 billion for research, and only USD 5.8 billion was allocated for TB services, falling short of the required amounts to achieve global targets. Moreover, WHO emphasizes the need for sustained & increased funding to combat TB effectively. Hence, the U.S. government has significantly increased its funding for TB initiatives, from USD 233 million in 2013 to USD 406 million in 2023. Investments in screening and preventive treatment have shown significant health & economic benefits, with a return on investment of up to USD 39 for every dollar spent.
Key global TB metrics
Metrics
Value
Estimated new TB cases (global, 2024)
10.7 million
TB incidence rate (global, 2023)
134 per 100,000 population
Actual notified (diagnosed & reported) TB cases (2024)
8.3 million
Estimated TB deaths (2024)
1.23 million
% of TB cases among people living with HIV (global, 2024)
~5.8%
Rifampicin-resistant (RR/MDR-TB) estimated new cases (2024)
~390,000
Proportion of RR/MDR-TB cases treated (2024)
~42%
Source: WHO, Grand View Research
As of August 2022, the U.S. FDA had approved 439 diagnostic tests for COVID-19 under Emergency Use Authorization (EUAs). The market is becoming increasingly competitive with the introduction of new molecular diagnostic tests for detecting SARS-CoV-2. Rapid advancements in technology, which offer greater accuracy, portability, and cost-efficiency, are expected to drive market growth significantly. In April 2020, Canon Medical Systems introduced the Aquilion Prime SP CT system, designed for the quick diagnosis of viral infectious diseases. This system features an integrated decontamination tool that uses automated UV-C technology to sanitize the equipment after each use.
The growing adoption of self-testing and point-of-care products is expected to drive market expansion further. In May 2024, Wondfo Biotech was granted Emergency Use Authorization (EUA) for Wondfo Biotech’s point-of-care assay that simultaneously tests for WELLlife COVID-19 and influenza A and B. This dual testing capability allows for rapid diagnosis in a single test, which is particularly beneficial during flu season when symptoms can overlap with those of COVID-19. Furthermore, in May 2024, BioPerfectus received approval from Brazil’s ANVISA for its dengue and malaria diagnostic kits. In collaboration with the China CDC, the company developed the Malaria Pf/Pan (HRP2/pLDH) Antigen Rapid Test Kit, supplied to Papua New Guinea, to support local efforts in managing infectious diseases. Such product approvals are likely to fuel the overall market growth in the forecast period.
Advancements in diagnostic techniques, including molecular diagnostics and point-of-care testing, have further accelerated market expansion by improving accuracy and reducing turnaround times. Governments, healthcare organizations, and key players are increasing investments in research and development to enhance diagnostic capabilities, which is also contributing to market growth. The COVID-19 pandemic played a crucial role in increasing the demand for diagnostic tools, reagents, and assays, as well as spurring innovation and product launches. Moreover, growing awareness about the importance of early detection and diagnosis in managing infectious diseases is driving both consumer demand and healthcare providers’ adoption of advanced diagnostics, further fueling market expansion.
Market Concentration & Characteristics
Innovation in infectious respiratory disease diagnostics has accelerated since 2022, with manufacturers shifting toward multiplex, rapid molecular, and syndromic testing platforms. In January 2024, QIAGEN expanded its syndromic footprint by launching the QIAstat-Dx GI2 and Meningitis/Encephalitis panels in India, complementing its respiratory panel and signaling deeper global adoption of cartridge-based multiplex PCR. The post-pandemic period also drove refinements in respiratory panels, such as Seegene’s continuous optimization of Allplex SARS-CoV-2/Flu/RSV assays through 2022-23, reflecting the industry’s push for high-sensitivity multi-target testing. These advancements have strengthened market concentration around companies with the R&D capacity to develop rapid, automated, and clinically actionable molecular solutions.
The market has seen selective but strategically important M&A aimed at strengthening point-of-care and high-throughput molecular ecosystems. In July 2024, Roche acquired LumiraDx’s point-of-care technology assets, expanding its decentralized testing capabilities and positioning itself more competitively in respiratory molecular diagnostics. Such acquisitions highlight a broader trend: large diagnostics companies are consolidating emerging POC technologies to diversify testing settings beyond central laboratories. Earlier, during 2022-23, several regional partnerships and acquisitions across Asia and Europe concentrated on expanding syndromic testing portfolios, further intensifying competitive dynamics.
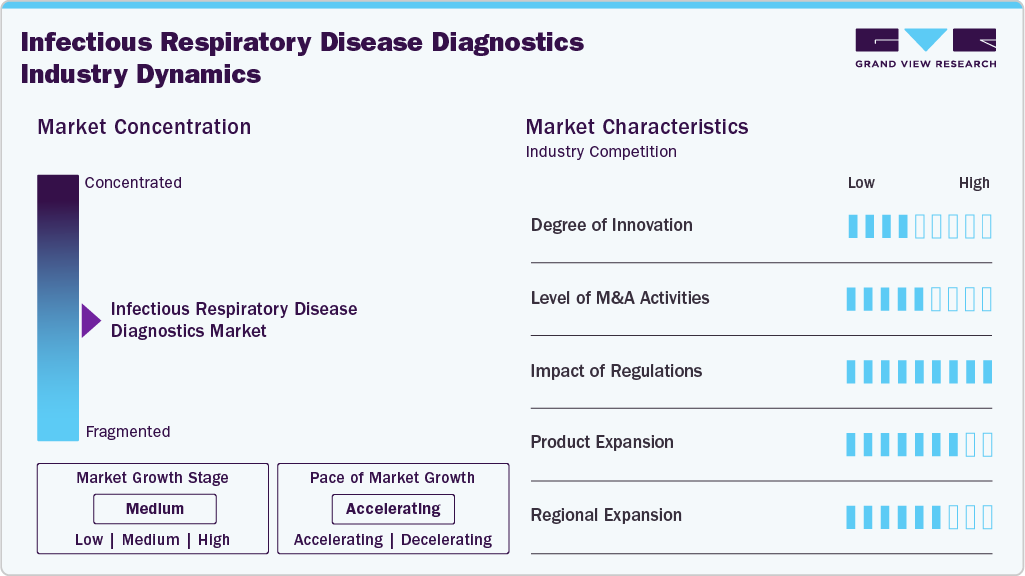
Regulation has played a defining role in shaping market entry timelines and competitive intensity. The EU In Vitro Diagnostic Regulation (IVDR), enforced in 2022 with phased transitions to 2027, substantially increased evidence requirements for respiratory assays, raising development costs and extending approval timelines. This shift favored established players with strong compliance infrastructure while slowing market access for smaller innovators. In parallel, countries such as India and the U.S. strengthened post-market surveillance norms, pushing vendors to demonstrate consistent clinical accuracy, particularly for multiplex and rapid molecular respiratory tests.
The product expansion has centered on multiplex panels, rapid molecular assays, and broader pathogen coverage. QIAGEN’s January 2024 launch of new QIAstat-Dx panels in India exemplified manufacturers’ strategies to widen their syndromic disease portfolios beyond respiratory but interconnected with respiratory workflows. bioMérieux continued growing its BioFire FilmArray respiratory menu through 2022-23, reinforcing its leadership in syndromic testing. Seegene also expanded assays targeting combined viral respiratory pathogens, addressing hospital demand for single-run multi-pathogen identification as respiratory seasons became less predictable post-COVID.
Regional expansion efforts since 2022 have increasingly targeted high-burden, high-volume markets such as India, Southeast Asia, and Latin America. QIAGEN’s India launch in January 2024 underlined how global molecular diagnostics companies are prioritizing emerging regions where respiratory disease prevalence, infrastructure modernization, and government surveillance programs create strong demand for advanced diagnostics.
Product Insights
The reagents segment dominated the market and accounted for the largest revenue share of 65.38% in 2025. Technological advancements in diagnostic devices have resulted in more sophisticated testing methods, necessitating the use of specialized reagents. Innovations in point-of-care testing and home testing kits require reagents that are not only reliable but also easy to use, driving their demand among both healthcare professionals and patients. Moreover, the COVID-19 pandemic has accelerated the production and use of reagents, with a notable increase in the development of rapid tests and related materials. This trend has prompted manufacturers to invest in research and development, leading to the introduction of new and improved reagents.
In September 2021, the U.S. FDA granted authorization for approximately 261 molecular diagnostic products, 88 serological tests, and 34 antigen tests for COVID-19 diagnosis. The instrument segment emerged as the second-largest revenue contributor in 2025, driven by the launch of innovative products designed to assist healthcare professionals in diagnosing COVID-19. For example, in March 2021, Vyaire Medical, Inc. launched AioCare, a mobile spirometry system for diagnosing respiratory diseases in the European Union, Middle East, and Australia. This product delivers hospital-grade accuracy and allows physicians to monitor patients remotely through advanced digital connectivity, making it easier to manage care at home.
The instrument segment is anticipated to grow at a considerable rate of 2.67% CAGR over the forecast period. Rising demand for rapid, accurate, and broad coverage respiratory testing continues to push healthcare systems toward fully automated, syndromic diagnostic platforms. Clinicians increasingly require tools that can quickly identify the specific pathogen causing infection, especially in settings where timely treatment decisions are critical, such as emergency departments, pediatric care, and ICU units. Moreover, the increasing availability of advanced diagnostic instruments is further expanding the market growth. For instance, the FDA-cleared BIOFIRE System, which offers panels capable of detecting viruses, bacteria, parasites, yeast, and antimicrobial resistance genes, demonstrates the market shift toward rapid syndromic diagnostics by providing broad, reliable results from a single sample.
Technology Insights
In 2025, the molecular diagnostics segment accounted for 67.63% of the overall market share. The significant market share for molecular tests is largely due to the growing demand for accurate diagnosis of COVID-19, RSV, influenza, and other respiratory infections. PCR remains the gold standard technology for amplifying DNA, making it a trusted choice for molecular diagnostics. In response to this demand, companies are developing multiplex tests that enable the simultaneous diagnosis of multiple infectious respiratory diseases using a single swab. For example, in December 2020, GENETWORx, LLC introduced an innovative test capable of detecting three respiratory viruses-SARS-CoV-2, RSV, and Influenza A & B-from a single sample.
The rapid/point-of-care diagnostics segment is anticipated to grow at the fastest rate of 3.54% CAGR over the forecast period, driven by advancements in Rapid point-of-care (POC) diagnostic innovation, which further propels the market growth. For instance, the transformative work led by Yuka Manabe and the Johns Hopkins University Center in February 2025, for innovative diagnostics for infectious diseases, is accelerating the development of fast, accurate, and user-friendly diagnostic tools that can be deployed globally. Their contributions to COVID-19 rapid tests created a technological and regulatory blueprint that is now being adapted for a wide range of infectious respiratory diseases, including influenza, RSV, and emerging pathogens.
Application Insights
The COVID-19 segment dominated the global market in 2025, accounting for 89.24% of the revenue. This dominance was driven by the increasing number of COVID-19 variants, an increase in product approvals, and heightened research and development efforts. However, with rising vaccination rates expected to lessen the severity of the disease, testing rates are likely to decline in the coming years. In response to changing market dynamics, key players are launching direct-to-consumer testing kits that allow individuals to collect samples without the need for trained personnel, thereby reducing turnaround times.
Meanwhile, the tuberculosis segment is anticipated to grow at the fastest rate of 9.74% CAGR over the forecast period. The persistent global burden of tuberculosis (TB) demonstrates the urgent need for improved diagnostic tools, driving market growth. For instance, according to an article published by Tejaswini Dharmapuri Vachaspathi (Treatment Action Group), in November 2025, TB claimed approximately 1.25 million lives, and of the 10.84 million estimated cases, 2.7 million remained undiagnosed, unreported, or untreated in 2023. This significant diagnostic gap highlights the limitations of conventional TB tests and the growing need for innovative solutions.
End-use Insights
In 2025, the diagnostic laboratories segment led the market, accounting for 38.62% of the revenue share. They are equipped with advanced technology and staffed by skilled professionals, enabling them to perform a wide array of diagnostic tests with high accuracy and reliability. This capability is essential for detecting infectious diseases and chronic conditions that require precise diagnosis. Furthermore, these laboratories offer comprehensive testing services, addressing both routine and specialized needs, and can handle large volumes of samples efficiently. The rising prevalence of diseases and an increasing focus on early diagnosis have led healthcare providers to increasingly depend on laboratory services for improved patient management. In addition, advancements in laboratory automation and artificial intelligence have enhanced the efficiency and speed of test results, further solidifying the reliance on diagnostic laboratories.
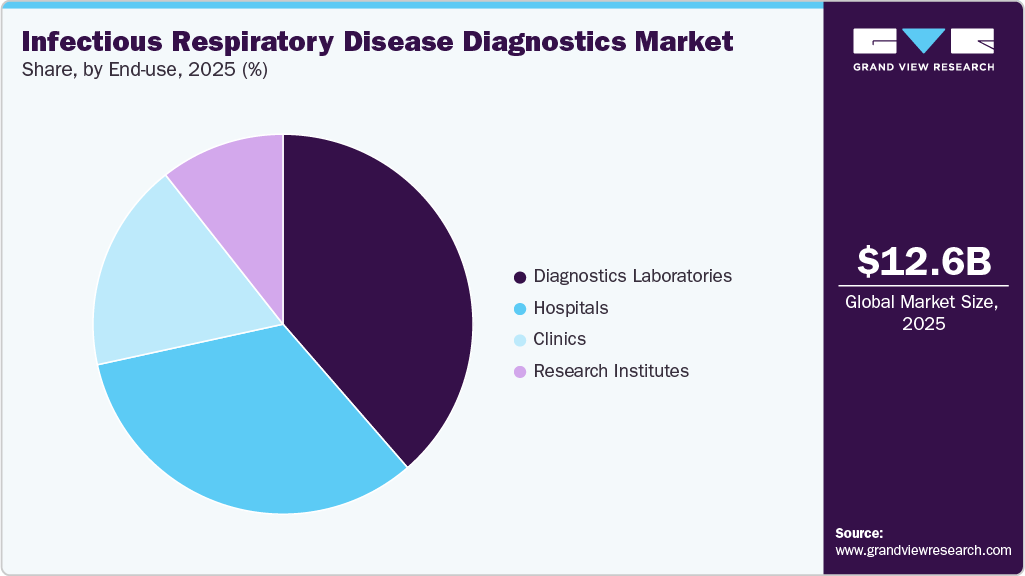
The hospitals segment is projected to experience substantial growth during the forecast period. The hospital segment plays a pivotal role in driving market growth, as hospitals increasingly depend on advanced diagnostic technologies to ensure early and accurate detection of respiratory infections. With rising patient inflows, higher disease complexity, and the need for rapid clinical decision-making, hospitals are investing heavily in advanced molecular assays, imaging systems, and rapid point-of-care diagnostic tools.
Regional Insights
North America infectious respiratory disease diagnostics market dominated the overall global market and accounted for 44.62% of the revenue share in 2025. The region benefits from advanced healthcare infrastructure and high healthcare expenditure, enabling the rapid adoption of innovative diagnostic technologies. The presence of leading biotechnology and pharmaceutical companies in the United States fosters continuous research and development, resulting in the introduction of cutting-edge diagnostic solutions.
U.S. Infectious Respiratory Disease Diagnostics Market Trends
The infectious respiratory disease diagnostics market in the U.S. held a significant share of the North American market in 2025, fueled by the presence of key players and a large end-user base. The increasing prevalence of chronic and infectious diseases, coupled with increasing public awareness about early diagnosis and preventive healthcare, drives demand for diagnostic tests.
Europe Infectious Respiratory Disease Diagnostics Market Trends
The infectious respiratory disease diagnostics market in Europe is experiencing significant growth. Europe holds a significant share of the overall market, driven by advanced healthcare systems, strong regulatory frameworks, and increasing demand for early disease detection. The region benefits from extensive research and development initiatives, leading to innovative diagnostic solutions. In addition, rising awareness of preventive healthcare further fuels market growth.
The UK infectious respiratory disease diagnostics market is likely to show significant growth owing to healthcare improvements and technological developments. The National Health Service (NHS) drives demand for accurate and efficient diagnostic solutions, particularly in response to rising chronic diseases. In addition, advancements in point-of-care testing and increased investment in medical technologies contribute to the market's growth, ensuring that the UK remains at the forefront of diagnostic advancements.
The infectious respiratory disease diagnostics market in Germany is experiencing significant growth. The country benefits from a well-established network of diagnostic laboratories and medical device manufacturers, driving the development of cutting-edge diagnostic technologies. Germany’s commitment to research and development, supported by significant government funding, fosters advancements in areas such as molecular diagnostics and point-of-care testing.
Asia Pacific Infectious Respiratory Disease Diagnostics Market Trends
The infectious respiratory disease diagnostics market in the Asia Pacific is experiencing the fastest growth, driven by significant advancements in terms of technology. Countries like China, India, and Japan are leading this expansion due to their large populations and growing demand for advanced diagnostic solutions. The region is witnessing significant advancements in technology, particularly in molecular diagnostics and point-of-care testing, which enhance the speed and accuracy of disease detection.
China infectious respiratory disease diagnostics market is growing, driven by the rapid expansion of its biotechnology and pharmaceutical industries. China is a prominent player in the market, characterized by rapid growth fueled by increasing healthcare investments and a vast population. The country has made significant strides in advancing its healthcare infrastructure, leading to a rising demand for innovative diagnostic solutions.
Latin America Infectious Respiratory Disease Diagnostics Market Trends
The infectious respiratory disease diagnostics market in Latin America is experiencing significant growth, driven by an increasing demand for healthcare services and improvements in medical infrastructure. The rising prevalence of chronic and infectious diseases, such as diabetes and COVID-19, is boosting the need for accurate and efficient diagnostic solutions.
Middle East and Africa Infectious Respiratory Disease Diagnostics Market Trends
The infectious respiratory disease diagnostics market in the MEA is expanding, driven by an increasing emphasis on healthcare improvement and disease prevention. Rapid urbanization, rising disposable incomes, and a growing burden of chronic and infectious diseases are key factors fueling demand for diagnostic solutions.
Saudi Arabia infectious respiratory disease diagnostics market growth is driven by substantial investments in healthcare infrastructure and a commitment to improving public health. The country's Vision 2033 initiative aims to enhance healthcare services, increase the availability of advanced diagnostic solutions, and promote preventive care.
Key Infectious Respiratory Disease Diagnostics Company Insights
The competitive scenario in the market is high, with key players such as Abbott, Thermo Fisher Scientific, Inc., Koninklijke Philips N.V., F. Hoffmann-La Roche Ltd., BD, bioMérieux SA, Bio-Rad Laboratories, Inc., Quidel Corporation, Siemens Healthcare GmbH, Danaher, and Qiagen holding significant positions. Prominent market participants are focusing on increasing the customer base by launching new products. For instance, in May 2024, F. Hoffman-La Roche Ltd. received FDA approval for its HPV self-testing kit, which can help identify women at risk of developing cervical cancer at an earlier stage. Furthermore, in March, SEKISUI Diagnostics obtained EUA clearance for the OSOM Flu SARS-CoV-2 Combo Test. It was approved for use in professional and home testing settings.
Key Infectious Respiratory Disease Diagnostics Companies:
The following are the leading companies in the infectious respiratory disease diagnostics market. These companies collectively hold the largest Market share and dictate industry trends.
- Abbott
- Bio-Rad Laboratories, Inc
- Siemens Healthineers AG
- bioMérieux
- BD
- QIAGEN
- QuidelOrtho Corporation
- F. Hoffmann-La Roche
- Koninklijke Philips N.V.
- Cepheid (Danaher Corporation)
- Seegene Inc.
- EliTechGroup (Bruker Corporation)
- CERTEST BIOTEC
- Thermo Fisher Scientific, Inc.
Recent Developments
-
In July 2025, BD announced the 510(k) clearance for the BD Veritor System for SARS-CoV-2 (the 510(k) will eventually replace the EUA version).
-
In April 2025, bioMérieux launched WATCHFIRE, a molecular testing solution (22-target respiratory panel) that runs on the BIOFIRE FILMARRAY TORCH and integrates with FIREWORKS software for real-time wastewater surveillance/outbreak detection.
-
In January 2025, Thermo Fisher’s TaqPath COVID-19, Flu A, Flu B, RSV Select Panel progressed from EUA/CE status to FDA 510(k) clearance, enabling broader clinical IVD adoption in the U.S.
-
In May 2024, Wondfo Biotech was granted Emergency Use Authorization (EUA) for Wondfo Biotech’s point-of-care assay that simultaneously tests for WELLlife COVID-19 and influenza A and B. This dual testing capability allows for rapid diagnosis in a single test, which is particularly beneficial during flu season when symptoms can overlap with those of COVID-19.
-
In May 2024, BioPerfectus received approval from Brazil’s ANVISA for its dengue and malaria diagnostic kits. In collaboration with the China CDC, the company developed the Malaria Pf/Pan (HRP2/pLDH) Antigen Rapid Test Kit, supplied to Papua New Guinea, to support local efforts in managing infectious diseases.
-
In May 2024, F. Hoffman-La Roche Ltd. received FDA approval for its HPV self-testing kit, which can help identify women at risk of developing cervical cancer at an earlier stage.
-
In April 2024, Cepheid announced that its Xpert HIV-1 Qual XC received prequalification from WHO, confirming its high standards of performance, quality, safety, and reliability. This in vitro nucleic acid amplification test detects HIV-1 total nucleic acids from dried blood spots and whole blood specimens, offering extended strain coverage and results up to seven to ten days before seroconversion. It was approved for early diagnosis in infants, adolescents, & adults in both POC and laboratory settings.
Infectious Respiratory Disease Diagnostics Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 12.85 billion
Revenue forecast in 2033
USD 15.70 billion
Growth rate
CAGR of 2.90% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, technology, application, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Denmark; Sweden; China; Japan; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Abbott; Bio-Rad Laboratories; Siemens Healthineers; bioMérieux; BD; QIAGEN; QuidelOrtho; F. Hoffmann-La Roche; Koninklijke Philips; Cepheid (Danaher); Seegene; EliTechGroup (Bruker); CERTEST BIOTEC; Thermo Fisher Scientific
Customization scope
Free report customization (equivalent up to 8 analyst working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Infectious Respiratory Disease Diagnostics Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends and opportunities in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global infectious respiratory disease diagnostics market report based on technology, pathogen, end-use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Instruments
-
Reagents
-
Services
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
COVID-19
-
Influenza
-
Respiratory Syncytial Virus (RSV)
-
Tuberculosis
-
Non-tuberculosis Mycobacterium Testing
-
Bacterial Pneumonia Testing
-
Other Respiratory Disease Testing
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Immunoassay
-
Molecular Diagnostics
-
Microbiology
-
Rapid/Point-of-Care Diagnostics
-
Other Technologies
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Diagnostic Laboratories
-
Clinics
-
Research Institutes
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Norway
-
Denmark
-
Sweden
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global infectious respiratory disease diagnostics market size was estimated at USD 12.56 billion in 2025 and is expected to reach USD 12.85 billion in 2026.
b. The global infectious respiratory disease diagnostics market is expected to witness a compound annual growth rate of 2.90% from 2026 to 2033 to reach USD 15.70 billion in 2033.
b. Based on product, the reagents segment dominated the market and accounted for the largest revenue share of 65.38% in 2025
b. Some key players operating in the infectious respiratory disease diagnostics market include Abbott; Thermo Fisher Scientific Inc.; Koninklijke Philips N.V.; F. Hoffmann-La Roche Ltd.; bioMérieux SA; BD; Quidel Corporation; QIAGEN; Danaher; Siemens Healthcare GmbH.
b. The major factors driving market growth are the rising incidence of infectious respiratory diseases, the rising number of product approvals, and increasing initiatives by government and non-profit organizations to improve diagnostic tests.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










