- Home
- »
- Medical Devices
- »
-
Insulin Delivery Devices Market Size, Industry Report, 2033GVR Report cover
![Insulin Delivery Devices Market Size, Share & Trends Report]()
Insulin Delivery Devices Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Reusable Insulin Pens, Disposable Insulin Pens), By End Use (Hospitals & Clinics, Homecare), By Region, And Segment Forecasts
- Report ID: GVR-1-68038-991-3
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size, 2025$19.3BMarket Estimate, 2026$20.9BMarket Forecast, 2033$35.2BCAGR, 2026 - 20337.8%Insulin Delivery Devices Market Summary
The global insulin delivery devices market size was estimated at USD 19.3 billion in 2025 and is expected to reach USD 35.2 billion by 2033, growing at a CAGR of 7.8% from 2026 to 2033. The growth is largely attributed to the rising global prevalence of diabetes, which has heightened the demand for advanced and user-friendly solutions for diabetes management.

Key Market Trends & Insights
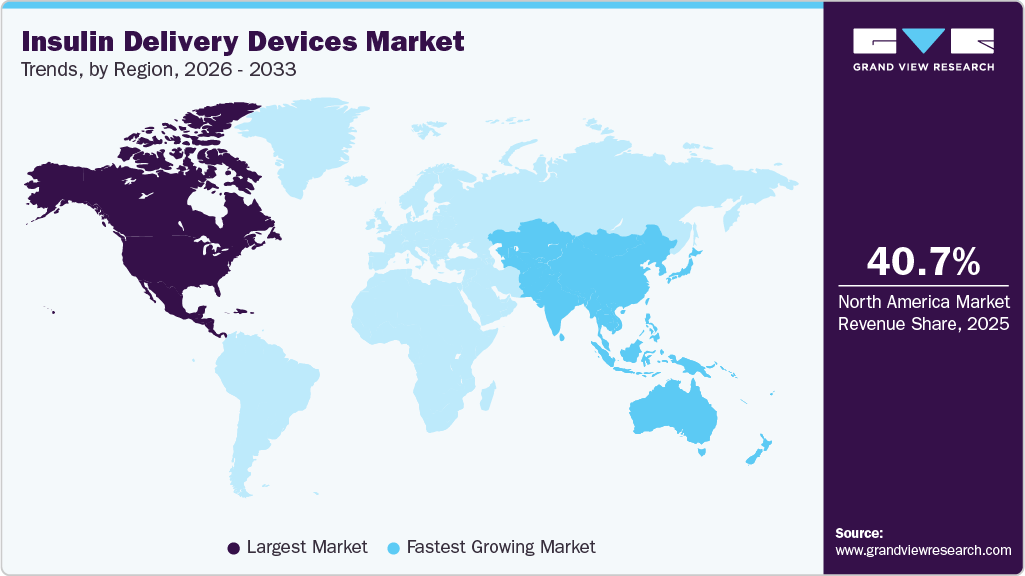
- North America's insulin delivery devices market held the largest share of 40.7% of the global market in 2025.
- The Asia Pacific insulin delivery devices industry is expected to grow significantly over the forecast period.
- By product, the insulin pens segment held the highest market share of 43.7% in 2025.
- By end use, the homecare segment held a leading market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 19.3 Billion
- 2033 Projected Market Size: USD 35.2 Billion
- CAGR (2026-2033): 7.8%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Technological advancements play a crucial role in the market growth, with the development of devices that are more effective, less invasive, and tailored to meet patients' individual needs. Furthermore, integrating digital technology with insulin delivery devices is expected to boost market growth. This includes the rise of Continuous Glucose Monitoring (CGM) systems that work in tandem with insulin delivery devices to provide real-time glucose monitoring and automated insulin adjustments.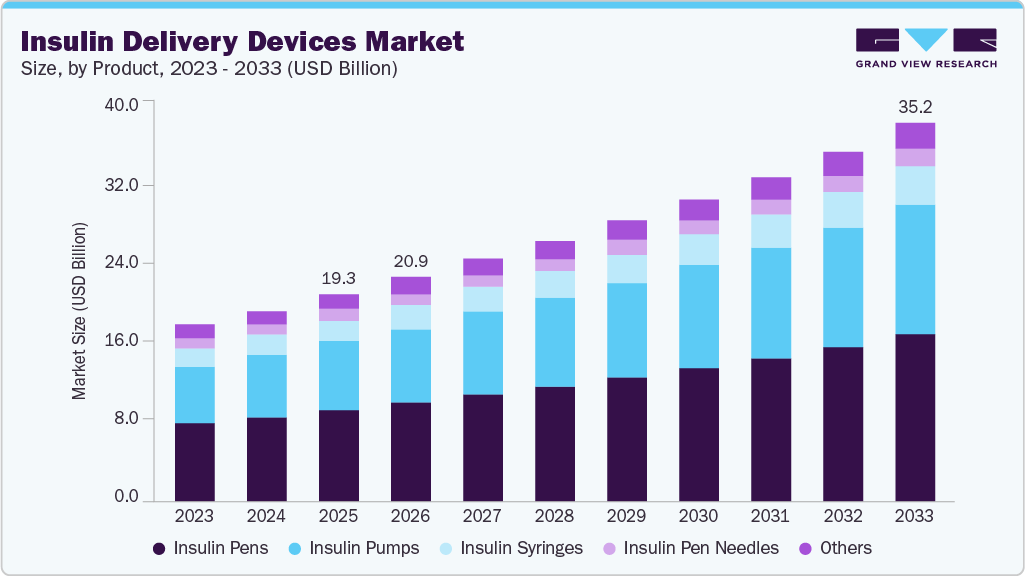
Technological advancements in diabetes management have significantly transformed insulin delivery methods, evolving from traditional injections to more advanced, minimally invasive techniques. Initially, insulin administration faced challenges in maintaining consistent blood glucose levels. However, the advent of insulin pens facilitated easier self-administration, reducing discomfort. The development of insulin pumps allowed for continuous insulin infusion, enhancing glucose control. A pivotal advancement was the introduction of Continuous Glucose Monitoring (CGM) devices, providing real-time glucose data, essential for informed decision-making. This set the stage for Artificial Pancreas Systems (APS), which integrate CGM data with insulin pumps for automated delivery, closely mimicking natural pancreatic function.
Bionic technology, blending biological and artificial processes, has been instrumental in advancing insulin delivery. It underpins devices such as APS, utilizing algorithms, sensors, and automated pumps for precise blood sugar regulation, minimizing manual intervention. These innovations offer tailored, user-friendly solutions, notably improving patient outcomes. Modern insulin pumps, equipped with CGM integration, predictive algorithms, and smartphone connectivity, exemplify significant progress in device functionality, offering accurate and adaptable insulin management. These features not only mitigate risks associated with blood glucose extremes but also empower users with finer control over their insulin dosing, responding dynamically to glucose level changes.
The increasing adoption of connected diabetes management solutions is transforming the way insulin therapy is monitored and administered, with a strong focus on improving patient adherence and clinical outcomes. For instance, in December 2024, a study published by MDPI highlighted that the use of smart insulin pens integrated with continuous glucose monitoring (CGM) in pregnant women with type 1 diabetes, demonstrating improved glycemic control and increased time-in-range without severe hypoglycemia. The system enabled automatic tracking of insulin dose timing and quantity, along with real-time data sharing through a connected mobile application. This development highlights the growing role of connected insulin pen technologies in enhancing adherence and optimizing diabetes management outcomes.
Furthermore, the industry is witnessing a shift towards wearable devices, such as insulin pumps and insulin patch pumps, which offer continuous insulin infusion in a convenient and discreet manner. For instance, in January 2024, PharmaSens, a Switzerland-based company specializing in medical devices, has recently applied for FDA approval in the United States for its 'niia essential' insulin patch pump system. This application represents a pivotal step for PharmaSens in the evolution of insulin pump technologies. Furthermore, the company achieved ISO 13485 certification in November 2023, encompassing the design, development, manufacturing, and distribution of insulin infusion pumps and their accessories. PharmaSens has positioned its product as focused on patient needs and ease of use. Furthermore, in January 2024, Embecta Corp. declared that it had submitted a 510(k) premarket application to the U.S. Food and Drug Administration (FDA) for its unique disposable insulin delivery system.
"This patch pump is intended for people who require insulin to manage diabetes and is informed by the unique needs of people with type 2 diabetes and their healthcare providers, we worked with them to address the needs of those who may require more daily insulin and are looking for a simplified and convenient option for automated insulin delivery that offers the ease of use and discretion of a patch pump, along with a larger 300U insulin reservoir."
- Colleen Riley, Chief Technology Officer, embecta.
The development of smart insulin pens, which can track and share dosage data, represents another significant trend, as does the progression towards bionic and artificial pancreas systems that more closely mimic the natural insulin delivery of a healthy pancreas. For instance, in July 2025, Byram Healthcare highlighted advancements in diabetes technology, noting that automated insulin delivery (AID) systems such as patch pumps and integrated devices significantly improved glycemic control, with clinical studies showing reductions in HbA1c levels and increased time-in-range. These systems combine insulin delivery with real-time glucose monitoring and smartphone connectivity, enhancing data tracking and patient management. This development reflects the broader trend toward connected, intelligent diabetes care solutions that mimic natural pancreatic function.
The rising geriatric population is fueling the industry. These devices, such as insulin pens, pumps, and smart insulin devices, cater specifically to the needs of older adults by simplifying the insulin administration process, which can be particularly challenging due to age-related factors such as reduced vision, skill, and cognitive function. The ease of use, dosing accuracy, and the ability to integrate with digital health platforms for monitoring and reminders ensure better adherence to treatment regimens, thereby improving glycemic control & reducing the risk of complications. Given the increasing prevalence of diabetes among older adults globally, the growth potential within this demographic is substantial.
Market Dynamics
The rising prevalence of diabetes is a major factor which drives the growth of the insulin delivery devices market, due to the increasing global diabetic population that continues to boost the demand for effective insulin administration solutions. Factors such as sedentary lifestyles, unhealthy dietary habits, obesity, and a growing aging population have significantly contributed to the increase in incidence of both type 1 and type 2 diabetes. According to a research paper, at least an estimated 150 to 200 million people worldwide require insulin therapy for their health, while over 800 million people live with diabetes globally. This creates a strong demand for advanced insulin delivery devices such as insulin pens, pumps, patch pumps, and smart connected systems. In addition, growing awareness regarding diabetes management, technological advancements in minimally invasive and user-friendly devices, and the increasing preference for home-based care are further accelerating market growth.
Stringent regulatory requirements act as a significant restraint for the insulin delivery devices market, due to several factors. Manufacturers must comply with complex approval processes and rigorous quality standards before commercializing products. Regulatory authorities such as the U.S. Food and Drug Administration and the European Medicines Agency require extensive clinical testing, safety validation, cybersecurity assessments for connected devices, and continuous post-market surveillance to ensure patient safety and device reliability. These lengthy and costly approval procedures increase product development timelines and operational expenses, particularly for small and emerging companies. In addition, frequent updates to regulatory guidelines and reimbursement policies create further challenges for manufacturers seeking rapid product launches and global market expansion.
Market Concentration & Characteristics
The industry is characterized by a moderate to high level of industry concentration, with a few major global players dominating the scene. These include companies such as Novo Nordisk, Sanofi, Eli Lilly, Medtronic, and Insulet Corporation. These companies have established strong brand recognition, extensive product lines, and significant global footprints. The market is driven by the rising prevalence of diabetes worldwide, increasing awareness about diabetes management, and the continuous need for more effective and convenient insulin delivery methods.
Innovation is a cornerstone of the industry. Companies are continually researching and developing new technologies to make insulin delivery more efficient, less painful, and more user-friendly. For instance, in March 2026, Sequel Med Tech announced that its twiist automated insulin delivery (AID) system is now widely available in the U.S., marking a significant innovation in the industry. The system utilizes the Tidepool Loop algorithm embedded in a compact tubed pump and is FDA-cleared for individuals aged 6 and above with type 1 diabetes, enabling real-time, automated insulin adjustments.
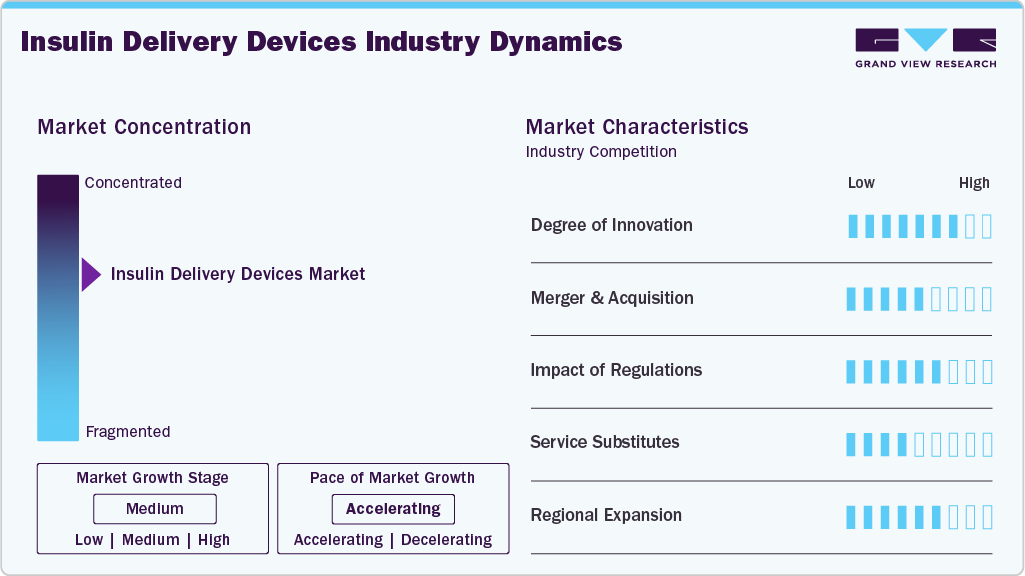
The industry has witnessed a steady level of mergers and acquisitions (M&A) activities as companies strive to enhance their product portfolios, expand their market reach, and leverage new technologies. M&A activities enable larger companies to quickly adopt innovative technologies developed by smaller firms and startups, thereby accelerating the pace of innovation within the industry. For instance, on August 4, 2025, TecMed AG completed the acquisition of Ypsomed Diabetes Care AG from Ypsomed Holding AG for approximately USD 506.8 million (CHF 420), marking a significant consolidation in the insulin delivery devices market. The transaction included insulin pump technologies, subsidiaries, and patch pump development activities, strengthening TecMed’s position in insulin infusion systems. This development highlights ongoing M&A-driven portfolio expansion and specialization aimed at accelerating innovation and market competitiveness in diabetes care.
Regulatory approvals are crucial in the insulin delivery devices industry. The process can be lengthy and requires substantial evidence of safety and efficacy. Regulations can significantly impact the time-to-market of new devices. However, regulatory bodies like the FDA in the United States and the EMA in Europe have been working towards streamlining the approval process for diabetes management devices, recognizing their critical role in patient care. In February 2024, Insulet Corporation announced the attainment of the CE mark approval in accordance with the European Medical Device Regulation. This approval is for the enhanced compatibility with the Abbott FreeStyle Libre 2 Plus sensor of Insulet’s Omnipod 5 Automated Insulin Delivery System, designed for use by individuals as young as two years old who are living with type 1 diabetes.
Product approvals are a key driver for growth and competition in the market. Companies are in a constant race to get their new and improved devices approved by regulatory authorities. These approvals not only validate the safety and efficacy of the devices but also often bring about significant improvements in diabetes management for patients. For instance, Sanofi (India) has obtained marketing approval from the CDSCO for its diabetes medication, Soliqua, available in pre-filled pens in March 2023. Designed for adults with type 2 diabetes and obesity who haven't seen sufficient control with other therapies, Soliqua enhances blood sugar levels alongside diet and exercise. It's administered once daily in a set combination (10-40 and 30-60) of insulin glargine and lixisenatide.
The global nature of diabetes as a health issue means that there is a demand for insulin delivery devices across the world. However, market penetration varies by region, influenced by factors such as healthcare infrastructure, regulatory environment, and economic conditions. Companies are continuously exploring opportunities to expand into emerging markets, where the prevalence of diabetes is growing rapidly, alongside efforts to consolidate their presence in established markets. For instance, in early 2023, Novo Nordisk disclosed an investment exceeding 16 billion Danish kroner (approximately 2.1 billion euros) aimed at enlarging its existing production facility in Chartres, France, in November 2023. This expansion is intended to cater to both present and future product lines targeting severe chronic conditions.
Product Insights
Insulin pens captured the largest market share of 43.7% in 2025. The dominance of insulin pens is primarily driven by their superior convenience, ease of use, and dosing accuracy compared to traditional vial-and-syringe methods, making them highly suitable for daily self-administration. Their portable, discreet design reduces pain during injection and minimizes social stigma, which significantly improves patient comfort and adherence to insulin therapy. Additionally, the increasing preference for home-based diabetes management and the availability of both reusable and prefilled pen options further support widespread adoption, contributing to the segment’s leading market share. For instance, as per data released by MSF-SA Association in May 2024, a survey conducted by MSF and T1International involving over 400 individuals using insulin across 38 countries revealed that 82% of respondents who had experience with both insulin pens and traditional syringes expressed a preference for insulin pens. The preference was attributed to several factors, including the ease and safety of pen usage, accurate dosing, reduced pain during administration, and decreased social stigma when using pens in public settings.
The insulin pumps segment is expected to grow at the fastest CAGR during the forecast period. The growth is primarily due to the increasing adoption of advanced automated insulin delivery systems integrated with continuous glucose monitoring (CGM), which significantly improve glycemic control and reduce diabetes-related complications. These systems enable real-time insulin adjustments, enhancing treatment precision and reducing the burden of manual dosing, making them highly preferred among patients requiring intensive insulin therapy. These compact, wearable gadgets deliver insulin through a tiny tube placed beneath the skin, replicating the natural function of a healthy pancreas. They offer a steady supply of basal insulin throughout the day and additional doses, known as boluses, to match mealtime requirements. A study published by the American Diabetes Association in October 2023 revealed a growing trend in insulin pump usage over time, with variations noted based on gender, insurance coverage, and racial/ethnic backgrounds. Those using insulin pumps showed improved A1C levels, increased adoption of Continuous Glucose Monitoring (CGM), and reduced occurrences of diabetic ketoacidosis and severe hypoglycemia. Individuals utilizing insulin pumps and CGM experienced fewer acute events than those using only insulin pumps.
End Use Insights
The home care segment captured the largest revenue share of 48.2% in 2025. This segment plays a significant role in the insulin delivery devices market, providing patients with the convenience of managing their diabetes at home. Key market players offer a range of insulin delivery devices, such as insulin pens, syringes, pumps, and CGM systems. These devices enable patients to administer insulin doses accurately and regularly monitor their blood glucose levels without frequent visits to healthcare facilities. In addition, advancements in diabetes technology, including automated insulin delivery systems and telehealth-enabled care, are enabling patients to achieve better glycemic outcomes outside clinical environments. The widespread adoption of remote patient monitoring is also accelerating this trend globally. According to the article published by Medixine Ltd. In October 2024, over 76.7 million patients were monitored remotely using connected health devices in 2023, with diabetes management being one of the key use cases.
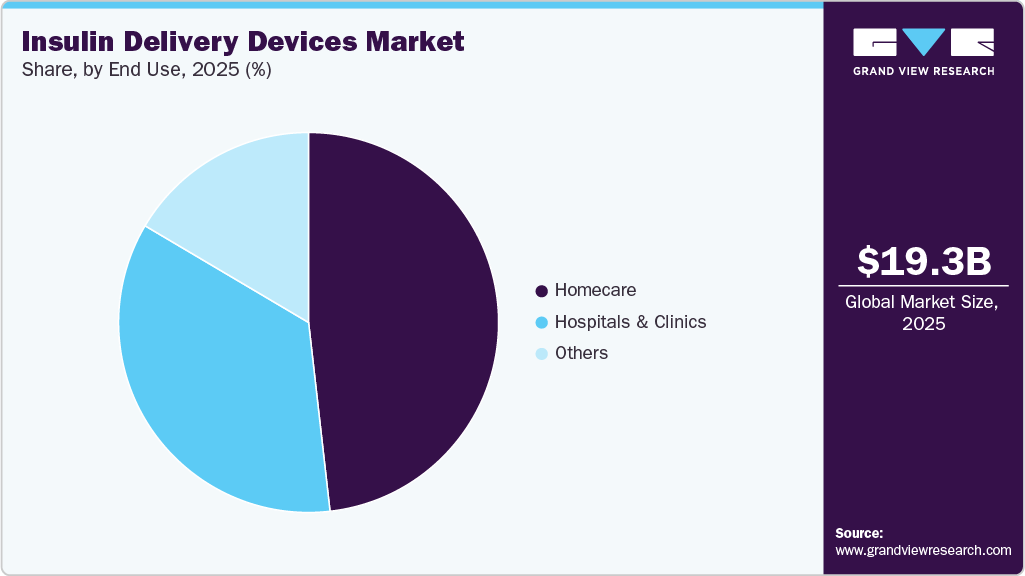
The hospitals and clinics segment is expected to witness the fastest growth rate of 8.3% during the forecast period. Hospitals play a crucial role in the insulin delivery market, serving as hubs for diagnosing, treating, and managing diabetes. In hospital settings, various insulin delivery methods are employed to cater to patients' diverse needs. These include the use of insulin pumps, insulin pens, syringes, and other delivery devices. For instance, research conducted by the American Medical Association in February 2024 revealed that the utilization of home insulin pumps was considered safe for children hospitalized in non-intensive care units at a children's hospital, especially for hypoglycemia and hyperglycemia. These pumps administer insulin continuously, replicating the natural pattern of insulin secretion observed in a healthy pancreas. In addition, hospitals are increasingly adopting integrated diabetes management systems, including continuous glucose monitoring (CGM) and automated insulin delivery technologies, to improve patient outcomes and reduce complications.
Regional Insights
North America insulin delivery devices market held the largest share of 40.7% in 2025 and is expected to maintain its dominance during the forecast period. Some key factors contributing to the region’s growth are the increasing geriatric population, the growing burden of diabetes due to lifestyle changes, the rising prevalence of obesity, and high treatment costs. Some of the key players, such as Abbott, Medtronic, and Dexcom, have their operational headquarters in North America, which is contributing to the market growth.
U.S. Insulin Delivery Devices Market Trends
The insulin delivery devices market in the U.S. held the largest market share in 2025 in the North America region. The high per capita income and increasing healthcare spending are among the key factors likely to drive market growth. For instance, in February 2026, the Centers for Disease Control and Prevention reported that approximately 40.1 million people in the U.S. are living with diabetes, representing nearly 1 in every 8 individuals. The report also highlighted that more than 1 in 4 adults with diabetes remain undiagnosed, indicating a significant gap in disease awareness and management. In addition, over 115 million adults in the U.S. have prediabetes, emphasizing the expanding patient pool driving demand for advanced insulin delivery devices.
Europe Insulin Delivery Devices Market Trends
The insulin delivery devices market in Europe held a significant market share in 2025. The increasing prevalence of diabetes and the rising awareness about diabetes management & control are some key factors expected to propel market growth over the forecast period. For instance, in 2025, the International Diabetes Federation (IDF) Diabetes Atlas reported that approximately 66 million adults in Europe are living with diabetes, representing nearly 1 in 10 adults, indicating a continued rise from earlier estimates. The report also highlighted that around 34% of cases remain undiagnosed, reflecting a significant unmet need for effective diabetes management solutions. Moreover, diabetes-related healthcare expenditure in Europe reached USD 193 billion in 2024, accounting for nearly 19% of global spending, emphasizing the strong economic burden driving demand for advanced insulin delivery devices.
The UK insulin delivery devices market is expected to exhibit the fastest growth rate during the forecast period owing to factors such as rising burden of diabetes, well-established healthcare infrastructure, and growing healthcare expenditure on diabetes disease and its treatment. For instance, in year 2024-2025, the NHS initiated a 5-year nationwide rollout plan to make hybrid closed-loop insulin delivery systems available to people with type 1 diabetes, starting from April 2024. These systems integrate insulin pumps with continuous glucose monitoring (CGM) to automate insulin delivery and improve glycemic control. This large-scale deployment further reinforces the UK’s leadership in adopting innovative insulin delivery solutions.
The insulin delivery devices market in France is closely linked to the country's diabetes landscape. Diabetes stands out as the most prevalent chronic condition, fully covered by France’s Statutory Health Insurance (SHI), with the number of covered patients doubling over the last decade. According to WHO, France has approximately one million diagnosed cases of diabetes. The prevalence of diabetes across all age groups in France is surging due to a rising obese population, unhealthy dietary habits, and sedentary lifestyles. In addition, around 0.26 million individuals are estimated to be diagnosed with type 1 diabetes.
Germany insulin delivery devices market is experiencing significant growth, largely driven by the growing diabetes prevalence and country's aging population. Diabetes poses a significant health challenge for healthcare systems in Germany. The prevalence of type 1 & 2 diabetes in the adult population is high, with several patients remaining undiagnosed. The growing aging population and unhealthy lifestyle choices are expected to increase the prevalence of type 2 diabetes in the coming decade.
Asia Pacific Insulin Delivery Devices Market Trends
The insulin delivery devices market in Asia Pacific is projected to grow at the fastest CAGR of 8.3% during the forecast period. The Asia Pacific market is majorly driven by China and Japan. China dominated the overall insulin delivery devices market in terms of revenue share. This can be attributed to the presence of major key players and the increasing prevalence of diabetes. In addition, technological advancements in insulin delivery devices, the increasing geriatric population, and the growing prevalence of lifestyle diseases, such as obesity, are some key factors anticipated to promote the insulin delivery devices market growth. Moreover, in December 2025, the International Diabetes Federation reported that the Western Pacific region continues to account for the largest global share of diabetes cases, exceeding 215 million adults, with countries like China contributing significantly to this burden. The report also highlighted a steady rise in prevalence driven by urbanization, aging populations, and lifestyle changes. This growing disease burden is accelerating demand for advanced insulin delivery devices across Asia Pacific.
China insulin delivery devices market is expected to experience the highest growth rate in the Asia Pacific region, driven by an increasing number of diabetes cases and rising costs associated with diabetes treatment and management. The country’s rapidly aging population, coupled with rising urbanization and sedentary lifestyles, has significantly contributed to the growing prevalence of diabetes, thereby increasing the demand for efficient insulin administration solutions. In addition, the Chinese government is actively promoting healthcare reforms and digital health initiatives, which are supporting the adoption of advanced diabetes management technologies such as insulin pumps, smart insulin pens, and continuous glucose monitoring (CGM)-integrated systems. Favorable reimbursement policies and increasing healthcare investments are further improving patient access to these devices, particularly in urban areas.
The insulin pen market in Japan is anticipated to expand at a CAGR of 18.8% from 2026 to 2033. One of the primary growth drivers is Japan’s rapidly aging population, which has led to a higher prevalence of diabetes, particularly type 2 diabetes among elderly individuals who require convenient and easy-to-use insulin delivery solutions. Insulin pens, known for their accuracy, portability, and ease of administration compared to traditional syringes, are increasingly preferred by both patients and healthcare providers. In addition, Japan’s well-established healthcare infrastructure and favorable reimbursement policies are supporting widespread adoption of advanced insulin delivery devices. The government’s focus on improving chronic disease management and reducing long-term healthcare costs has further accelerated the use of insulin pens and other connected diabetes technologies. Technological advancements, such as smart insulin pens with dose-tracking and connectivity features, are also gaining traction, enhancing patient adherence and treatment outcomes.
India's insulin delivery devices market is expected to grow owing to the increasing prevalence of diabetes disorder. The International Diabetes Federation (IDF) has projected that the global diabetic population will increase from 425 million in 2017 to around 629 million by 2045. Estimates from the Indian Council of Medical Research India Diabetes (ICMR—INDIAB) suggest that approximately 62.4 million individuals in India are living with diabetes. In addition, it's worth noting that there is a decline in diabetes prevalence after the age of 65, which can be attributed to early-stage deaths resulting from comorbid conditions.
Latin America Insulin Delivery Devices Market Trends
The insulin delivery devices market in Latin America is experiencing growth due to the increasing incidence of Type 1 and Type 2 diabetes. Latin American countries have a high prevalence of diabetes, and Mexico is recognized for having many patients with diabetes as type-2 diabetes is becoming prevalent. The genetic predisposition to type-2 diabetes and the steadily increasing incidence of obesity have been major contributors to the rise in type-2 diabetes cases over the past 40 years. Approximately 10% of people worldwide currently live with diabetes. The Latin America insulin delivery devices market is primarily driven by Brazil, Mexico, Argentina, and Colombia. Growing investments by market players in the region, proximity to North America, and free-trade agreements with major countries, such as the U.S., Canada, Japan, & several European countries, are among the factors anticipated to boost the Latin American market during the forecast period.
The Brazil insulin delivery devices market is witnessing consistent growth, primarily driven by the increasing burden of diabetes, fueled by urbanization, aging population, and lifestyle-related risk factors such as obesity and physical inactivity. The country’s large patient pool is creating sustained demand for effective and convenient insulin administration methods. A key trend in the market is the growing preference for insulin pens over traditional syringes, as they offer greater ease of use, improved dosing accuracy, and better patient compliance. At the same time, insulin pumps are gradually gaining traction, particularly among patients requiring intensive insulin therapy, due to their ability to provide continuous and precise insulin delivery.
Middle East & Africa (MEA) Insulin Delivery Devices Market Trends
The insulin delivery devices market in the Middle East & Africa (MEA) is expected to witness rapid growth due to the increased prevalence of diabetes and a rapidly aging population, as well as growing demand for home-based monitoring devices. The region is witnessing rapid urbanization and lifestyle transitions, including sedentary habits, unhealthy diets, and rising obesity rates, all of which significantly contribute to the growing burden of type 2 diabetes. This expanding patient pool is directly increasing the demand for efficient and long-term insulin management solutions. Moreover, improving healthcare infrastructure and increasing healthcare expenditure across countries such as Saudi Arabia, the UAE, and South Africa are enabling better access to advanced diabetes care technologies. Governments and healthcare organizations are also launching initiatives to enhance diabetes awareness, early diagnosis, and treatment adherence, which further supports device adoption.
The Saudi Arabia insulin delivery devices market is evolving rapidly, driven by factors such as a high prevalence of diabetes, changing lifestyles, and a strong government focus on healthcare modernization. The increasing incidence of diabetes linked to rising obesity rates, sedentary habits, and dietary patterns continues to expand the demand for effective insulin delivery solutions. A key market trend is the shift from traditional syringes to more advanced and user-friendly options such as insulin pens and insulin pumps, which offer greater convenience, improved dosing accuracy, and better patient adherence. Insulin pens are widely preferred for their ease of use and affordability, while insulin pumps are gradually gaining traction among patients requiring intensive insulin therapy.
Key Insulin Delivery Devices Companies Insights
The insulin delivery devices market is a dynamic and rapidly evolving segment within the medical device industry, primarily driven by the increasing prevalence of diabetes globally. This market includes a range of devices designed to administer insulin to diabetic patients, aiming to maintain their blood glucose levels. As diabetes management shifts towards more patient-friendly, efficient, and less invasive methods, the demand for innovative insulin delivery devices has surged.
Key Insulin Delivery Devices Companies:
The following key companies have been profiled for this study on the insulin delivery devices market.
- Novo Nordisk A/S
- Sanofi
- Eli Lilly and Company
- Ypsomed AG
- Medtronic
- Insulet Corporation
- B. Braun SE
- Owen Mumford Ltd.
- Tandem Diabetes Care, Inc.
- BD (Becton, Dickinson and Company)
Competitive Benchmarking
Operating Strategies
Competitive Edge
Weaknesses
Mature Players: Novo Nordisk A/S
- Mature players focus on expanding integrated diabetes management portfolios through insulin pens, pumps, patch pumps, and smart connected delivery systems while strengthening partnerships with digital health and CGM providers.
- These companies emphasize continuous R&D, regulatory approvals, product lifecycle extensions, and geographic expansion through strategic initiatives and advanced manufacturing capabilities.
- Established players maintain a strong competitive advantage through broad product portfolios, global distribution networks, advanced smart insulin technologies, strong reimbursement coverage, and high brand recognition among healthcare providers and diabetic patients.
- High R&D and regulatory compliance costs, cybersecurity concerns associated with connected devices, and pricing pressure from reimbursement authorities and low-cost competitors can impact profitability and product adoption.
Emerging Players: Insulet Corporation
- Emerging players adopt innovation-driven and niche-focused strategies, targeting wearable insulin delivery systems, AI-enabled diabetes management platforms, and cost-effective smart insulin devices.
- Their agility, faster innovation cycles, and focus on user-friendly, connected, and affordable insulin delivery technologies enable them to rapidly address evolving patient and home care needs.
- Limited financial resources, smaller commercial networks, and challenges in obtaining regulatory approvals and reimbursement coverage restrict large-scale market penetration and global expansion.
Recent Developments
-
In March 2026, diaTribe reported that Medtronic (MiniMed) announced the upcoming launch of its Flex insulin pump, a compact, screenless tubed device that is approximately half the size of its current 780G system while using the same advanced automated insulin delivery (AID) algorithm. The device can be controlled via iPhone or Android smartphones and is compatible with the Simplera Sync CGM and Abbott Laboratories’s Instinct CGM, enhancing connectivity and real-time diabetes management.
-
In February 2026, Portal Diabetes received U.S. FDA Breakthrough Device designation for its implantable Portal insulin pump, an innovative insulin delivery system designed to function as a next-generation artificial pancreas. The device aims to deliver insulin directly within the body, enabling more precise and automated glucose control compared to traditional external pumps.
-
In August 2023, Novo Nordisk A/S and Inversago Pharma reached an agreement wherein Novo Nordisk will acquire Inversago for a sum that could reach up to 1.075 billion US dollars in cash, contingent upon the fulfillment of specific development and commercial benchmarks. Based in Montreal, Inversago Pharma is a privately held company specializing in the development of therapies targeting the CB1 receptor, aimed at potentially treating obesity, diabetes, and related metabolic disorder complications.
Insulin Delivery Devices Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 20.9 billion
Revenue forecast in 2033
USD 35.2 billion
Growth rate
CAGR of 7.8% from 2026 to 2033
Actual period
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Novo Nordisk A/S; Sanofi; Eli Lilly and Company; Ypsomed AG; Medtronic; Insulet Corporation; B. Braun SE; Owen Mumford Ltd.; Tandem Diabetes Care, Inc.; BD (Becton, Dickinson and Company)
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Insulin Delivery Devices Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global insulin delivery devices market report on the basis of product, end use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Insulin Pens
-
Reusable Insulin Pens
-
Disposable Insulin Pens
-
-
Insulin Pumps
-
Tubed Pumps
-
Reusable Device/Components
-
Consumables
-
-
Tubeless Insulin Pumps
-
Reusable Device/Components
-
Consumables
-
-
-
Insulin Pen Needles
-
Standard Pen Needles
-
Safety Pen Needles
-
-
Insulin Syringes
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals & Clinics
-
Homecare
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Delivered Customizations
This report has been delivered with the following In-depth customizations
Client Request
Customization Delivered
Value Adds
Diabetes Prevalence & Patient Adoption Analysis
Developed a tailored analysis of the insulin delivery devices market focused on rising diabetes prevalence, patient adoption trends, and demand patterns for insulin pens, pumps, smart insulin pens, pumps, smart insulin devices, and patch pumps across key regions. The study incorporated assessment of demographic trends, homecare adoption, digital diabetes management, and patient preference for connected and minimally invasive delivery systems.
Enables stakeholders to understand evolving patient needs, identify high-growth device categories, evaluate adoption trends across diabetes populations, and align commercialization strategies with the increasing demand for convenient and technology-enabled insulin delivery solutions.
Competitive Landscape and Product Benchmarking
Conducted a detailed competitive landscape assessment of the insulin delivery devices market, including analysis of key companies, product portfolios, strategic initiatives, and innovation pipelines. The benchmarking was done for the types of insulin delivery devices, such as insulin pens, and insulin pumps.
Enables stakeholders to evaluate competitor positioning, identify product differentiation opportunities, benchmark pricing, and technology trends, and develop effective market entry and expansion strategies based on competitive intelligence and evolving product demand patterns.
Technology Innovation & Competitive Landscape Assessment
Delivered a customized evaluation of technological advancements in insulin delivery devices, including smart insulin pens, wearable pumps, CGM-integrated systems, and AI-enabled diabetes management platforms. The analysis also assessed competitive positioning, strategic partnerships and other key initiatives of market participants.
Provides actionable insights into emerging technology trends, competitive benchmarking, and future innovation opportunities, helping clients identify commercially attractive segments and strengthen product development and expansion strategies.
Frequently Asked Questions About This Report
Some key players operating in the insulin delivery devices market include Medtronic, Novo Nordisk A/S, Sanofi, Eli Lilly and Company, Ypsomed AG, Insulet Corporation, B. Braun SE, Owen Mumford Ltd., BD (Becton, Dickinson and Company), and Tandem Diabetes Care, Inc.
Key factors driving the insulin delivery devices market include the increasing prevalence of diabetes worldwide, advancements in insulin administration technology, growing awareness among patients about the benefits of automated insulin delivery devices over traditional syringe methods, and the convenience and ease of use offered by them.
The global insulin delivery devices market size was estimated at USD 19.3 billion in 2025 and is expected to reach USD 20.9 billion in 2026.
The global insulin delivery devices market is expected to grow at a compound annual growth rate of 7.8% from 2026 to 2033 to reach USD 35.2 billion by 2033.
The insulin pens segment dominated the insulin delivery devices market with the highest share in 2025. This is attributable to the increasing prevalence of diabetes, their superior convenience, ease of use, and dosing accuracy compared to traditional vial-and-syringe methods, making them highly suitable for daily self-administration.
About the Author(s)
Medical Devices Research Team
Healthcare · Medical DevicesThis report was authored by the medical devices Research Team at Grand View Research - comprising two research analysts, one senior research analyst, and one industry expert - with specialized expertise in the medical devices segment of the healthcare industry. All findings are based on proprietary healthcare databases, executive interviews, and regulatory analysis, subject to internal peer review prior to publication.
Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









