- Home
- »
- Biotechnology
- »
-
Lentiviral Vector Market Size, Share, Industry Report, 2033GVR Report cover
![Lentiviral Vector Market Size, Share & Trends Report]()
Lentiviral Vector Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (1st-Generation, 2nd-Generation, 3rd-Generation), By Indication (HIV, B-Thalassemia), By End Use (Academic and Research Institute, CROs & CMOs), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-884-2
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Lentiviral Vector Market Summary
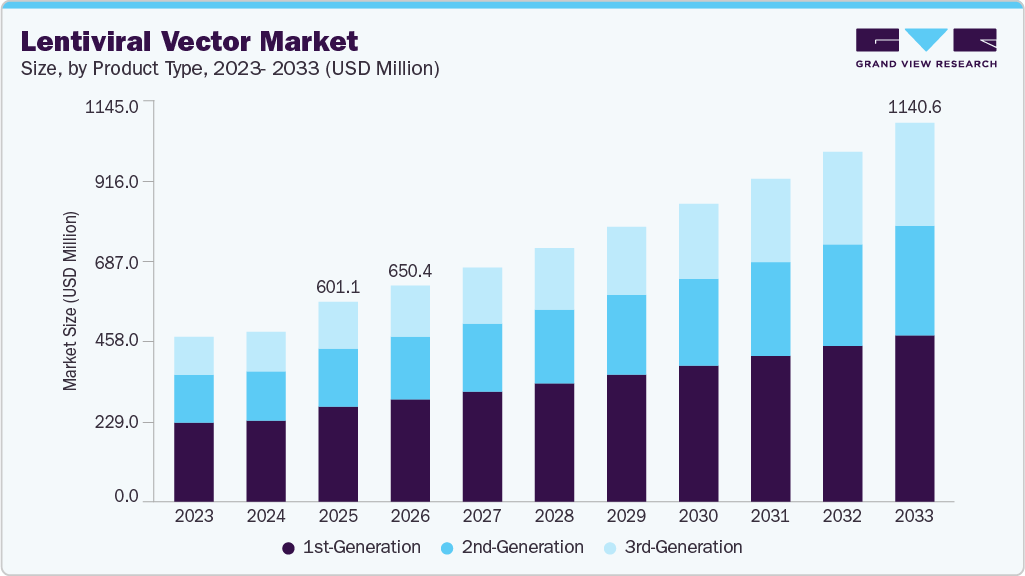
The global lentiviral vector market size was valued at USD 601.1 million in 2025 and is projected to reach USD 1,140.6 million by 2033, growing at a CAGR of 8.4% from 2026 to 2033. Growth is driven by rising demand for gene and cell therapies, increasing clinical research, and advancements in vector manufacturing. However, high costs and regulatory complexities may hinder market expansion.
Key Market Trends & Insights
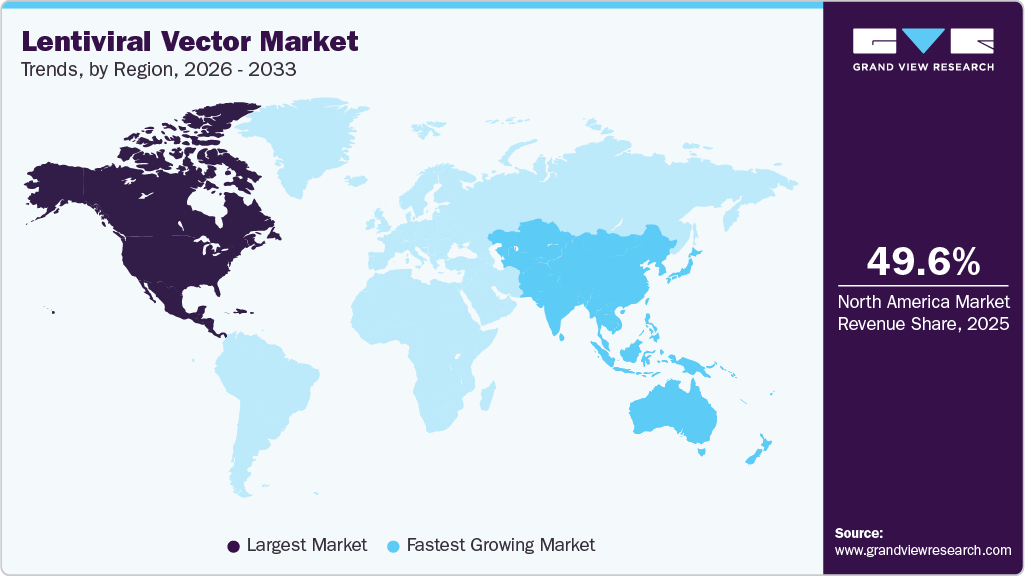
- The North America lentiviral vector industry held the largest global market share of 49.6% in 2025.
- The lentiviral vector industry in the U.S. is expected to grow over the forecast period due to the expanding aging population.
- Based on product, the 1st-generation segment dominated the market with a share of 47.5% in 2025.
- Based on the indication, the HIV segment dominated the market in 2025.
- Based on end use, the pharmaceutical and biotechnology companies segment dominated the market in 2025.
Market Size & Forecast
- 2025 Market Size: USD 601.1 Billion
- 2033 Projected Market Size: USD 1,140.6 Billion
- CAGR (2026-2033): 8.4%
- North America: Largest market in 2025
- Asia Pacific: Fastest-growing market
Growth of CAR-T and Advanced Immunotherapies
The rapid growth of CAR-T cell therapy is a key driver of the lentiviral vector market. These vectors are essential for modifying T-cells to target cancer, and the increasing number of approved and pipeline therapies for leukemia and lymphoma is significantly boosting demand. Since each treatment requires patient-specific cell engineering, the need for scalable, high-quality vector production continues to grow.
FDA-Approved CAR T-cell Therapies
CAR T-Cell Therapy
Approved Use(s)
Abecma (ide-cel)
Multiple myeloma
Aucatzyl (obe-cel)
B-cell ALL (adult)
Breyanzi (liso-cel)
Follicular lymphoma
Large B-cell lymphoma
Mantle cell lymphoma
Chronic lymphocytic leukemia
Carvykti (cilta-cel)
Multiple myeloma
Kymriah (tisa-cel)
B-cell ALL (pediatric/young adult)
Diffuse Large B-cell Lymphoma
Follicular lymphoma
Tecartus (brexu-cel)
B-cell ALL (adult)
Mantle cell lymphoma
Yescarta (axi-cel)
Large B-cell lymphoma
Follicular lymphoma
Source: National Cancer Institute, Secondary Research, Primary Interviews, Grand View Research
In addition, expanding research into solid tumors and autoimmune diseases is further increasing reliance on lentiviral vectors. Rising clinical trials, robust biopharma investments, and collaborations with CDMOs are accelerating the commercialization of therapies, making lentiviral vectors a critical component of advanced immunotherapy manufacturing.
Technological Advancements in Vector Design
Technological advancements in lentiviral vector design, particularly self-inactivating (SIN) vectors, are a key market driver. These innovations improve safety by reducing insertional mutagenesis risk and enhancing transduction efficiency, making vectors more suitable for clinical use across gene therapy applications.
Moreover, improvements in vector optimization and scalable manufacturing are increasing production efficiency and reducing costs. These advancements support broader adoption in research and commercial settings, strengthening the role of lentiviral vectors in cell therapy and driving market growth.
Market Concentration & Characteristics
The lentiviral vector industry demonstrates a high level of innovation, driven by advances in vector design, safety, and manufacturing. Improvements such as self-inactivating vectors and enhanced transduction efficiency support wider use in gene therapy and cell therapy, while scalable production technologies are enabling commercialization and market growth. For instance, in June 2024, Charles River Laboratories partnered with the Gates Institute in the U.S. to advance GMP lentiviral vector manufacturing for CAR-T therapies targeting hematological cancers and clinical development.
The level of M&A activity is moderate to high in the market, driven by the need to secure manufacturing capacity and access specialized technologies. Companies are actively acquiring vector production firms and forming strategic partnerships to strengthen their capabilities in advanced therapies. These transactions are focused on scaling production, improving supply chain reliability, and accelerating time-to-market, reflecting the growing commercial importance of lentiviral vectors.
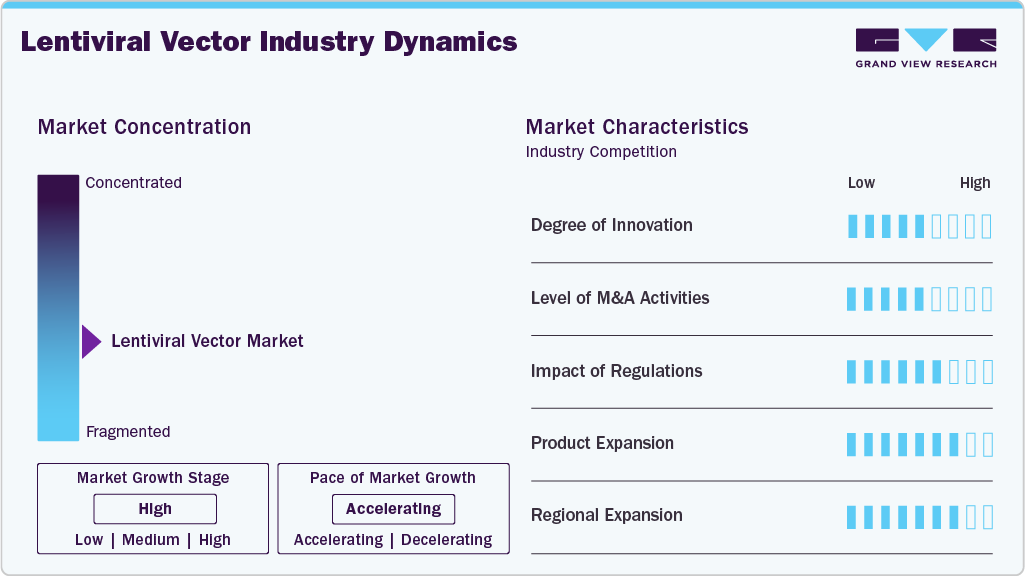
Regulations significantly influence the industry, with agencies such as the U.S. Food and Drug Administration and the European Medicines Agency enforcing strict safety, quality, and efficacy standards. While these requirements increase development costs and timelines, supportive policies and accelerated pathways for gene therapy are helping drive investment and commercialization, balancing safety with market growth.
Product expansion in the lentiviral vector market is focused on diversifying offerings and enhancing application-specific solutions. Companies are introducing specialized vector variants, ready-to-use kits, and integrated development-to-manufacturing services to cater to evolving research and clinical needs. This expansion is enabling greater flexibility, improved workflow efficiency, and broader adoption across emerging therapeutic applications.
Regional expansion is growing as companies establish facilities and partnerships across North America, Europe, and Asia-Pacific. This is driven by rising demand, the need for localized production, and increasing biotech investments in emerging markets, enabling broader market reach and growth.
Product Insights
The 1st-generation segment led the lentiviral vector market with a 47.5% share in 2025, driven by its cost-effectiveness, simpler design, and widespread use in research and early-stage gene therapy applications. Besides, its well-established protocols and ease of production have supported its extensive adoption across academic and biotechnology laboratories. The segment continues to benefit from consistent demand in preclinical studies and vector development workflows.
The 3rd-generation segment is expected to be the fastest-growing segment, driven by its advanced safety features and improved transduction efficiency. Its growing adoption of clinical and commercial gene therapy applications is further supported by increasing demand for high-quality, scalable vector systems. In addition, ongoing innovations in vector design and manufacturing technologies are accelerating its uptake across biotechnology and pharmaceutical companies.
Indication Insights
The HIV segment dominated the lentiviral vector industry with the largest revenue share of 39.2% in 2025, driven by the extensive use of lentiviral vectors derived from Human Immunodeficiency Virus (HIV) for gene delivery applications. Their high efficiency in stable gene integration and strong performance in both in vitro and in vivo systems have supported widespread adoption.
The Beta thalassemia segment is expected to grow at the fastest CAGR over the forecast period, driven by increasing adoption of gene therapies and rising research focus on curative treatments. Moreover, the strong pipeline of lentiviral vector-based therapies and growing clinical success rates are accelerating segment growth.
End Use Insights
The pharmaceutical and biotechnology companies segment dominated the lentiviral vector market with a 50.8% revenue share in 2025 and is projected to register the fastest CAGR over the forecast period. This is supported by strong pipeline momentum, increased funding in gene therapy programs, and expanding manufacturing capabilities. Ongoing strategic collaborations and growing focus on commercialization are further reinforcing segment expansion.
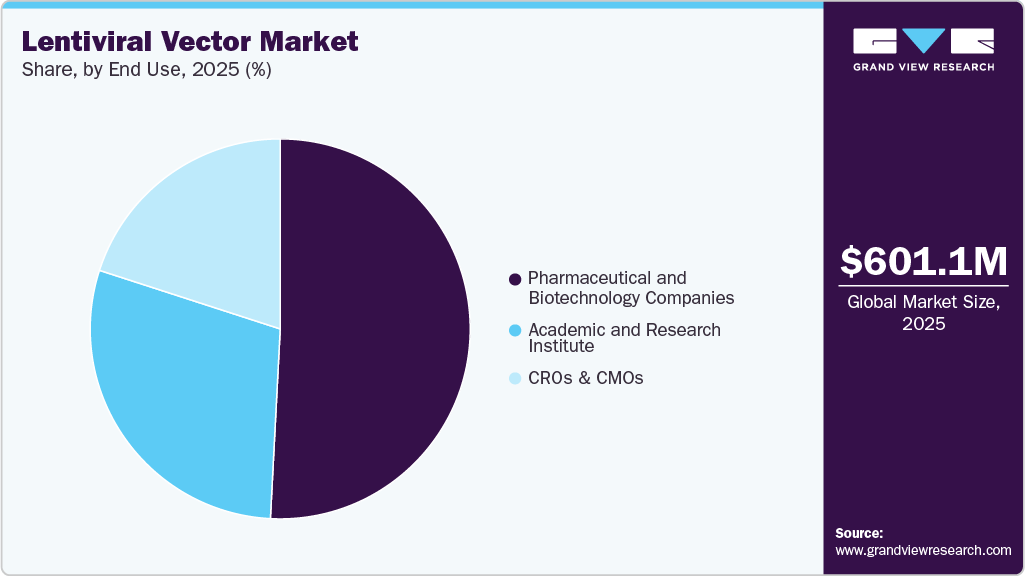
CROs & CMOs are expected to witness significant growth from 2026 to 2033, supported by the shift toward outsourced development and production models. Their ability to offer flexible capacity, regulatory expertise, and advanced manufacturing infrastructure is increasing their relevance in the value chain. Expanding long-term service agreements and integrated end-to-end solutions are further accelerating their market uptake.
Regional Insights
The lentiviral vector industry in North America dominated the global market and accounted for a 49.6% share in 2025, due to strong gene therapy pipelines, advanced manufacturing infrastructure, and supportive regulatory pathways. High R&D funding and the presence of major biotech firms and CDMOs drive rapid clinical development and commercialization, sustaining regional dominance.
U.S. Lentiviral Vector Market Trends
The U.S. lentiviral vector industry represents the most advanced market, driven by a strong pipeline of gene and cell therapies, extensive funding, and a well-defined regulatory pathway. The presence of leading biotech companies, academic research centers, and large-scale CDMOs supports high demand for lentiviral vectors and continuous innovation. For instance, in January 2023, Vector BioMed launched in the U.S., aiming to address lentiviral vector supply bottlenecks by providing scalable, high-quality vector manufacturing and end-to-end solutions for clinical development and commercialization.
Europe Lentiviral Vector Market Trends
The lentiviral vector industry in Europe is experiencing steady growth, supported by a robust regulatory framework and increasing investment in advanced therapies. Key countries such as Germany, the UK, and France benefit from strong academic research and collaborative initiatives, driving consistent demand for lentiviral vectors. For instance, in April 2023, Yposkesi launched the LentiSure platform in Europe, enhancing lentiviral vector production efficiency, scalability, and consistency to support CAR-T development and commercialization with improved yields and optimized manufacturing processes.
The UK lentiviral vector market is supported by a robust research ecosystem and a favorable regulatory environment for advanced therapies. Strong collaboration between academia and industry, along with government-backed initiatives in gene therapy, is driving steady adoption of lentiviral vectors.
The lentiviral vector market in Germany is characterized by its strong biopharmaceutical manufacturing base and emphasis on quality and compliance. The country benefits from advanced research infrastructure and increasing participation in clinical trials, supporting consistent demand for lentiviral vectors.
Asia Pacific Lentiviral Vector Market Trends
The lentiviral vector industry in the Asia Pacific is anticipated to witness the fastest growth at a CAGR of 10.8% from 2026 to 2033, fueled by expanding biotech industries, rising government support, and increasing clinical trials. Growing local manufacturing capabilities and improving regulatory systems are accelerating adoption and reducing reliance on imports.
The China lentiviral vector market is rapidly expanding, driven by a booming biotech sector, significant government funding, and increasing clinical trial activity. The country is focusing on building domestic manufacturing capabilities, which is strengthening its position in the market.
The lentiviral vector market in Japan is witnessing growth due to progressive regulatory reforms and accelerated approval pathways for regenerative medicine. Government support and rising investment in gene therapy are encouraging local development and adoption of lentiviral vector technologies.
Middle East & Africa Lentiviral Vector Market Trends
The lentiviral vector industry in the Middle East & Africa is gradually developing, supported by healthcare investments and growing interest in advanced therapies, particularly in the UAE and Saudi Arabia. However, limited local production keeps the region reliant on imports, and partnerships help bridge the gaps.
The Kuwait lentiviral vector market is at an early stage, supported by improving healthcare infrastructure and gradual interest in advanced therapies. While reliance on imports remains high, increased investment and regional collaboration are expected to support future growth.
Key Lentiviral Vector Company Insights
The lentiviral vector market is led by key players such as Thermo Fisher Scientific Inc., Merck KGaA, Lonza, and Oxford Biomedica, driven by strong global presence, advanced manufacturing capabilities, and strategic collaborations. Their integrated service offerings across development and commercialization further strengthen market positioning.
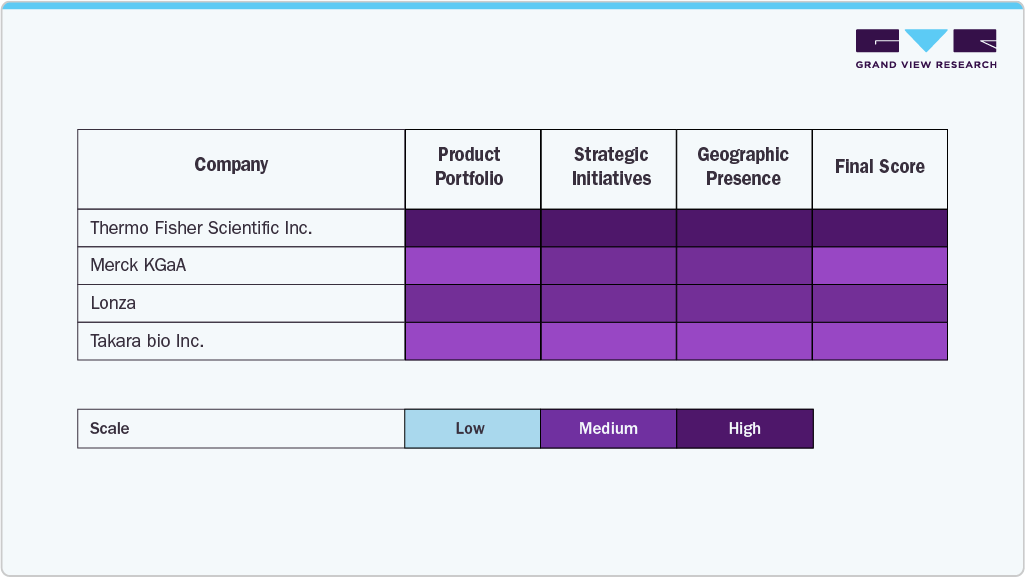
Other players, including Takara Bio Inc., Sino Biological Inc., OriGene Technologies Inc., Vector Biolabs, FinVector Oy, and Sirion-Biotech GmbH (part of Revvity), enhance competition through specialized technologies and portfolio expansion.
Key Lentiviral Vector Companies:
The following key companies have been profiled for this study on the lentiviral vector market
- Thermo Fisher Scientific Inc.
- Merck KGaA
- OriGene Technologies Inc.
- FinVector Oy
- Sino Biological Inc.
- Oxford Biomedica
- Lonza
- Vector Biolabs
- Takara Bio Inc.
- Sirion-Biotech GmbH (Revvity)
Recent Developments
- In July 2025, ViroCell Biologics partnered with AvenCell Therapeutics across the U.K. and U.S. to advance retroviral vector manufacturing for allogeneic CAR-T therapies targeting hematological cancers.
- In May 2025, QIAGEN expanded its digital Β-Thalassemiaportfolio globally with lentiviral solutions, enhancing cell and gene therapy quality control through precise analytics, standardized workflows, and regulatory-compliant manufacturing support capabilities.
- In July 2023, BioPharm International highlighted globally that stable producer cell line advancements improved lentiviral vector manufacturing scalability, consistency, and cost-efficiency, while addressing safety, yield, and process complexity challenges in production.
Lentiviral Vector Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 650.4 million
Revenue forecast in 2033
USD 1,140.6 million
Growth rate
CAGR of 8.4% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, indication, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia, UAE; Kuwait
Key companies profiled
Thermo Fisher Scientific Inc.; Merck KGaA; OriGene Technologies Inc.; FinVector Oy; Sino Biological Inc.; Oxford Biomedica; Lonza; Vector Biolabs; Takara Bio Inc.; Sirion-Biotech GmbH (Revvity)
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Global Lentiviral Vector Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For the purpose of this report, Grand View Research has segmented the global lentiviral vector market report on the basis of product, indication, end use, and region.
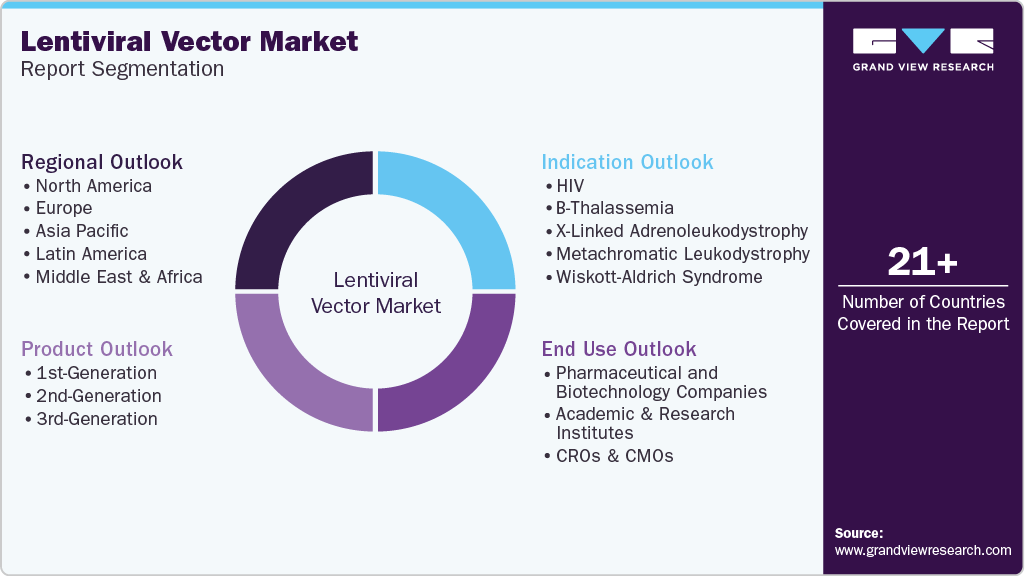
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
1st-Generation
-
2nd-Generation
-
3rd-Generation
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
HIV
-
Β-Thalassemia
-
X-Linked Adrenoleukodystrophy
-
Metachromatic Leukodystrophy
-
Wiskott-Aldrich Syndrome
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical and Biotechnology Companies
-
Academic & Research Institutes
-
CROs & CMOs
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
MEA
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global lentiviral vector market size was estimated at USD 601.1 million in 2025 and is expected to reach USD 650.4 million in 2026.
b. The global lentiviral vector market is expected to grow at a compound annual growth rate of 8.35% from 2026 to 2033 to reach USD 1,140.6 million by 2033.
b. North America lentiviral vector market dominated the global market and accounted for a 49.6% share in 2025, due to strong gene therapy pipelines, advanced manufacturing infrastructure, and supportive regulatory pathways.
b. Some key players operating in the lentiviral vector market include Thermo Fisher Scientific Inc.; Merck KGaA; OriGene Technologies Inc.; FinVector Oy; Sino Biological Inc.; Oxford Biomedica; Lonza; Vector Biolabs; Takara Bio Inc.; Sirion-Biotech GmbH (Revvity)
b. Growth is driven by rising demand for gene and cell therapies, increasing clinical research, and advancements in vector manufacturing. However, high costs and regulatory complexities may hinder market expansion.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










