- Home
- »
- Clinical Diagnostics
- »
-
Microsampling & Minimally Invasive Blood Collection Market Report 2033GVR Report cover
![Microsampling & Minimally Invasive Blood Collection Market Size, Share & Trends Report]()
Microsampling & Minimally Invasive Blood Collection Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Microsampling Devices, DBS Collection Devices, At-Home Blood Collection Devices), By Application, By End Use (Hospitals & Clinics), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-882-8
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
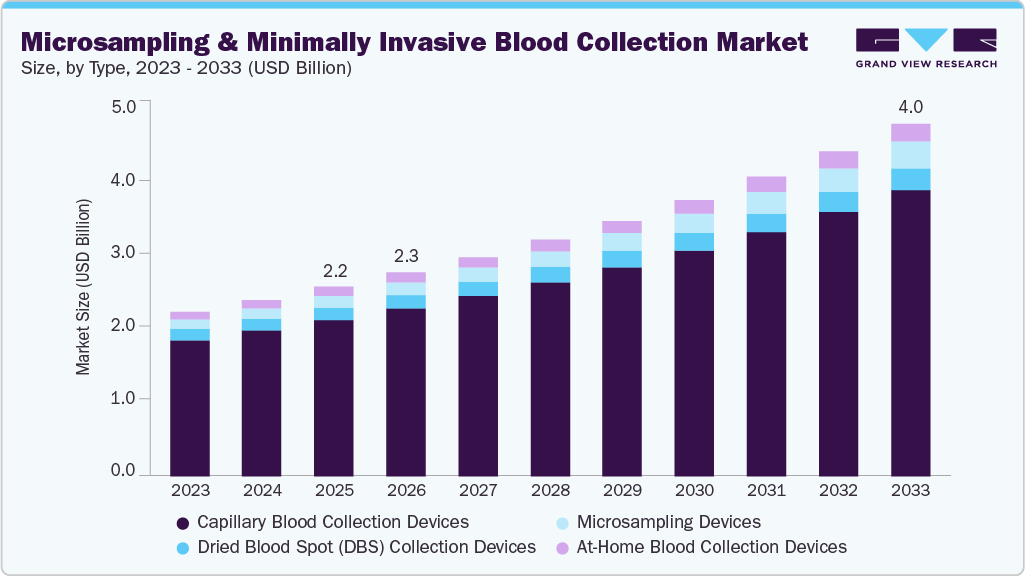
Microsampling & Minimally Invasive Blood Collection Market Summary
The global microsampling & minimally invasive blood collection market size was estimated at USD 2.16 billion in 2025 and is projected to reach USD 4.01 billion by 2033, growing at a CAGR of 8.15% from 2026 to 2033. The growth is driven by the increasing global burden of chronic conditions, rising demand for patient-centric diagnostic care, and ongoing technological advancements, supported by the expanding adoption of remote microsampling into decentralized clinical trials (DCTs).
Key Market Trends & Insights
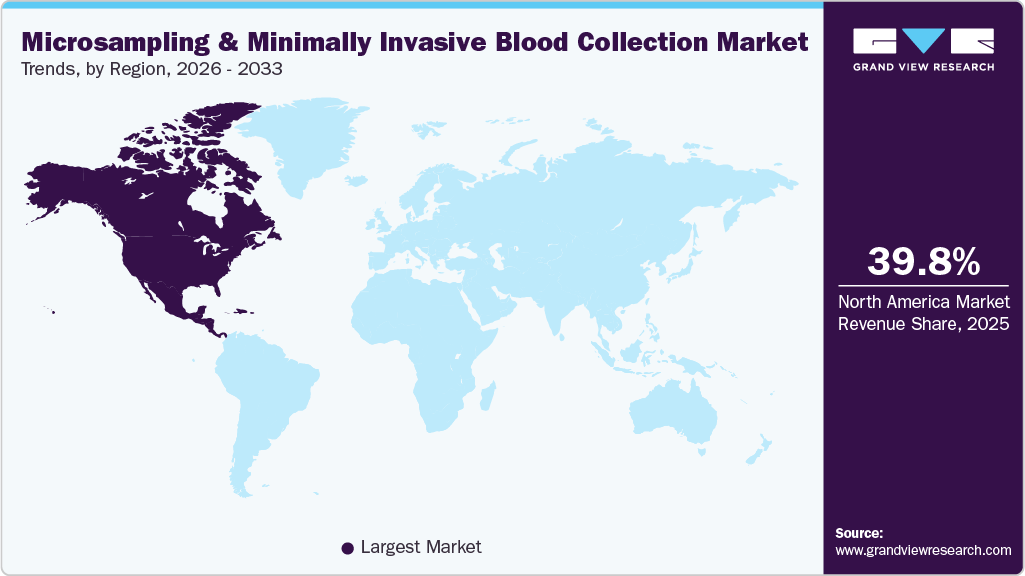
- North America dominated the microsampling & minimally Invasive blood collection market and accounted for the largest revenue share of 39.77% in 2025.
- The U.S. led the North America market and held the largest revenue share in 2025.
- Based on product, the capillary blood collection devices segment dominated the global market and accounted for the largest revenue share of 82.49% 2025.
- Based on application, the disease diagnostics segment held the largest revenue share of 45.68% in 2025.
- Based on end use, hospitals & clinics held the largest revenue share of 42.56% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 2.16 Billion
- 2033 Projected Market Size: USD 4.01 Billion
- CAGR (2026-2033): 8.15%
- North America: Largest market in 2025
Blood-based analysis continues to play a critical role in diagnostics, thereby underpinning the expansion of the industry. While traditional diagnostic approaches have largely relied on arterial blood collection in centralized laboratory settings, advancements in molecular diagnostics are now enabling accurate results from significantly smaller sample volumes. This transition is facilitating a shift toward decentralized healthcare models, with microsampling enhancing patient access, convenience, and remote testing capabilities, ultimately contributing to broader adoption and sustained market growth.The global burden of chronic diseases continues to rise, driving increased adoption of microsampling and non-invasive blood collection technologies. According to the World Health Organization (WHO), chronic diseases accounted for approximately 43 million deaths in 2021, representing nearly 75% of all non-pandemic-related deaths worldwide, including 18 million premature deaths occurring before the age of 70. This escalating disease burden is translating into sustained demand for continuous, cost-effective, and scalable diagnostic monitoring solutions. Chronic conditions necessitate frequent and longitudinal blood-based testing to enable effective disease management and early intervention. However, traditional venous sampling approaches remain resource-intensive, episodic in nature, and often pose limitations in accessibility and patient compliance.
The increasing demand for patient-centric diagnostic care is a key driver of growth in the microsampling and minimally invasive blood collection market. Healthcare systems are progressively prioritizing solutions that enhance patient comfort, convenience, and accessibility, particularly for individuals requiring frequent monitoring, such as patients with chronic diseases, as well as pediatric and geriatric populations. Microsampling technologies, which enable low-volume capillary blood collection through finger-stick methods or advanced devices such as microneedle-based systems, support at-home and decentralized testing, thereby reducing reliance on conventional clinical settings. This trend aligns with the rapid expansion of telehealth and remote care models, where self-collection kits and mail-in diagnostic services are becoming increasingly standardized. In addition, advancements such as volumetric absorptive microsampling (VAMS) and improved dried blood spot (DBS) methodologies address historical limitations like hematocrit variability, ensuring accurate and reproducible results. Consequently, microsampling is gaining traction in applications such as therapeutic drug monitoring and decentralized clinical trials, driven by benefits including faster turnaround times, lower logistical costs, and improved patient adherence.
Ongoing technological advancements are playing a critical role in accelerating the adoption of microsampling within decentralized clinical trials (DCTs). Innovations such as volumetric absorptive microsampling (VAMS), advanced dried blood spot (DBS) techniques, and next-generation microneedle-based devices have significantly enhanced sample accuracy, consistency, and ease of use. These developments address historical limitations, including variability in sample volume and hematocrit effects, thereby enabling laboratory-grade data quality from remotely collected samples. From a clinical trial perspective, the integration of remote microsampling allows participants to self-collect samples at home and ship them to centralized laboratories, reducing dependence on physical trial sites. This approach supports improved patient recruitment, particularly across geographically dispersed and underserved populations, while also enhancing retention by minimizing travel and procedural burden. Furthermore, advancements such as serum microsampling devices with remote centrifugation capabilities and controlled shipping conditions help maintain analyte stability and ensure reliable clinical and proteomic analyses. Consequently, microsampling is emerging as a key enabler of scalable and patient-centric DCT models, supporting faster trial timelines, reduced operational costs, and improved diversity in patient representation.
The adoption of microsampling in preclinical research offers significant ethical and operational advantages, particularly in toxicokinetic (TK) studies. Conventional blood collection methods often require larger sample volumes and repeated sampling, which can exacerbate toxicity responses and increase physiological stress in study animals. In contrast, microsampling techniques enable the collection of smaller blood volumes through minimally invasive approaches, thereby reducing animal distress and improving overall welfare. Approaches such as composite sampling further minimize the frequency and total volume of blood draws, aligning with the principles of reduction and refinement in animal research. In addition, microsampling enhances workflow efficiency by enabling faster sample collection and reducing handling time for both animals and technical personnel. The use of smaller gauge needles and less invasive techniques minimizes tissue damage and discomfort, supporting ethical compliance and regulatory acceptance. For instance, in rodent TK studies, microsampling facilitates serial sampling from the same animal rather than requiring larger cohorts, thereby reducing animal usage while maintaining high-quality data outputs.
Microsampling and non-invasive blood collection technologies are transforming clinical diagnostics and research by enhancing both data quality and operational efficiency. Techniques such as dried matrix microsampling, including dried blood spots (DBS), minimize blood exposure to air, thereby preserving sensitive analytes such as drugs, metabolites, and RNA. Advanced innovations, including 3D blood spheroids and pre-treated DBS cards, further improve the stability of labile compounds such as glutathione and cocaine, while volumetric absorptive microsampling (VAMS) enables precise fixed-volume collection, effectively addressing hematocrit-related variability. Additionally, devices such as HemaPEN and HemaXis enhance sample homogeneity and mitigate issues like the “coffee ring effect,” ensuring reliable and reproducible results. Beyond analytical advantages, microsampling significantly reduces logistical complexity, as dried samples can be transported at ambient temperatures, eliminating the need for costly cold-chain infrastructure. Furthermore, remote and self-sampling capabilities support at-home and decentralized collection, reducing reliance on trained phlebotomists and specialized clinical facilities. Low-volume sample requirements also minimize storage needs, enabling scalable and cost-efficient workflows across routine diagnostics and decentralized clinical trials.
While the market is experiencing strong growth, it is also subject to increasing regulatory scrutiny and a clear requirement to demonstrate performance comparable to conventional testing methods. At the same time, strategic consolidation is intensifying, with larger players acquiring specialized innovators to strengthen their capabilities and expand market presence. Success in this space will depend on the ability to navigate complex regulatory requirements while consistently proving reliability against established standards. In addition, the high cost of advanced, user-friendly microsampling devices may act as a barrier to adoption, particularly in price-sensitive markets and regions with limited reimbursement support.
Market Concentration & Characteristics
Microsampling and minimally invasive blood collection technologies represent a high level of innovation by shifting traditional venipuncture toward patient-centric, low-volume, and technology-driven approaches. These methods typically require very small blood volumes (often less than 100 µL) collected via fingerstick or microneedle-based devices, significantly reducing patient discomfort and reliance on clinical settings. A key advancement is the enablement of self-sampling and decentralized diagnostics, allowing patients to collect samples at home and send them to laboratories, thereby improving accessibility and compliance. Furthermore, innovations such as volumetric absorptive microsampling (VAMS), microneedle patches, and dried blood spot (DBS) technologies enhance accuracy, safety, and sample stability while eliminating the need for cold-chain logistics. These advancements support personalized and preventive healthcare by enabling frequent monitoring and integration with advanced analytical techniques such as mass spectrometry, ultimately improving patient outcomes and enabling scalable, data-driven diagnostics.
The market is witnessing a high level of merger and acquisition (M&A) activity, reflecting increased collaboration among manufacturers, healthcare providers, research institutions, and technology developers. These strategic initiatives are driven by the need to enhance innovation, expand product portfolios, and address growing demand for efficient, patient-friendly diagnostic solutions. Established medtech companies are actively pursuing acquisitions and partnerships to strengthen their capabilities in capillary blood collection and at-home testing. For example, acquisitions such as Trajan Scientific and Medical’s purchase of Neoteryx have enabled the integration of advanced microsampling technologies into broader diagnostic workflows. Additionally, emerging players are becoming attractive acquisition targets due to their proprietary, minimally invasive technologies. Overall, M&A activity is supporting the development of integrated, end-to-end diagnostic ecosystems and accelerating the shift toward decentralized, patient-centric care models.
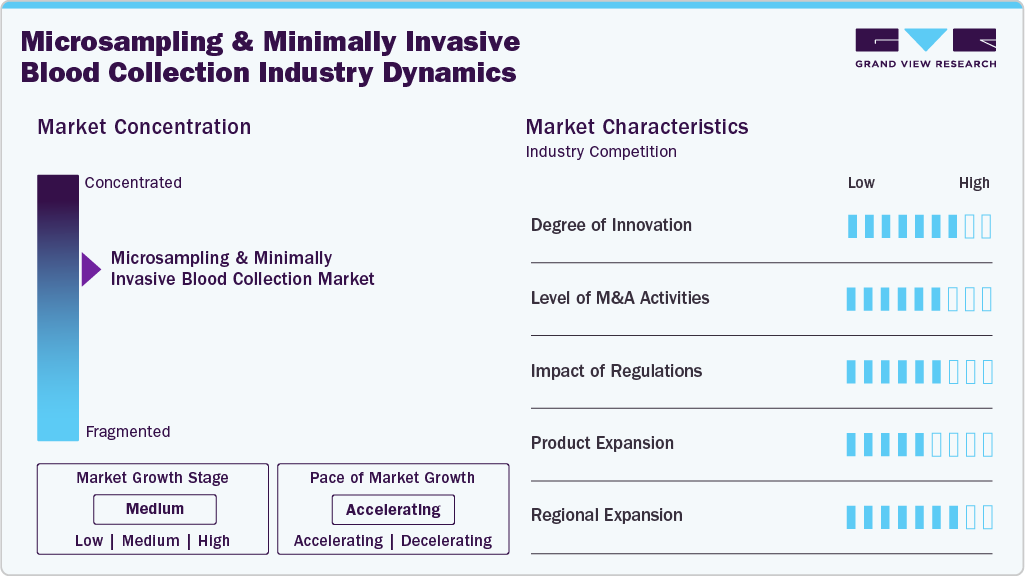
Regulatory frameworks play a critical role in shaping the development and adoption of microsampling and minimally invasive blood collection technologies. Regulatory bodies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other regional authorities require rigorous validation of device safety, performance, and quality. Compliance with standards related to biocompatibility, sterility, and analytical accuracy is essential to ensure reliable diagnostic outcomes and patient safety. Regulatory requirements also extend to decentralized clinical trials, where at-home collection devices must meet strict criteria for sample stability, storage, and validation. In addition, reimbursement policies and regional healthcare regulations significantly influence market access and commercial viability. While regulatory compliance may increase development timelines and costs, it also enhances credibility, ensures data integrity, and supports long-term adoption of these technologies.
The industry is experiencing significant product expansion, driven by increasing demand for patient-centric and decentralized diagnostic solutions. Companies are introducing innovative devices such as VAMS-based tools, capillary blood collection systems, and painless, home-use sampling devices to address the limitations of traditional venous blood collection. Both established medtech firms and emerging startups are focusing on improving sample accuracy, ease of use, and integration with digital health platforms. This expansion is further supported by the growth of telemedicine, decentralized clinical trials, chronic disease management, and at-home testing, all of which require reliable and minimally invasive sampling methods. Continuous innovation and product diversification are enhancing market competitiveness, improving patient convenience, and expanding the application scope across diagnostics, research, and healthcare delivery.
The regional expansion of the market is driven by variations in healthcare infrastructure, expenditure, and technology adoption. North America currently dominates the market, supported by strong healthcare systems, significant R&D investments, and early adoption of decentralized, patient-centric diagnostic technologies. The Asia-Pacific region is expected to witness the fastest growth, driven by increasing healthcare investments, expanding infrastructure, and rising awareness of point-of-care and home-based diagnostics across countries such as China, India, Japan, and Australia. Europe is experiencing steady growth, supported by a well-established healthcare ecosystem, high awareness of advanced diagnostic solutions, and favorable regulatory frameworks. Meanwhile, the LAMEA region (Latin America, the Middle East, and Africa) is anticipated to grow gradually over the long term, as governments and healthcare organizations continue to invest in infrastructure development, improve access to diagnostics, and promote minimally invasive, patient-focused healthcare solutions.
Product Insights
Capillary blood collection devices dominated the market and accounted for the largest revenue share of 82.49% in 2025. This segment includes key subcategories such as lancets, lancing devices, capillary tubes or microcollection tubes, and microcollection containers. It remains a critical component of diagnostic workflows due to its suitability for rapid, convenient testing across clinical, home-based, and field settings. These devices offer a simple, cost-effective alternative to traditional venipuncture, particularly in applications such as glucose monitoring, pediatric testing, and routine diagnostics. The segment’s dominance is primarily driven by the increasing demand for patient-centric healthcare, the rising prevalence of chronic conditions requiring frequent monitoring, and the growing adoption of remote and home-based diagnostic solutions. Additionally, their ability to deliver accurate, low-volume samples while minimizing logistical complexities makes them highly suitable for clinical trials, personalized medicine, and routine patient monitoring, thereby supporting widespread global adoption.
The microsampling devices segment is anticipated to grow at the fastest rate over the forecast period, driven by continuous technological innovation and increasing demand for advanced sampling solutions. Emerging devices such as Tasso and HemaPEN are designed to simplify and enhance blood collection using small sample volumes. Push-button, vacuum-assisted devices such as Tasso-M20 and Tasso-SST enable the collection of larger capillary blood volumes with integrated serum separation, making them compatible with standard clinical analyzers. Additionally, microfluidic-based sampling devices utilize advanced channel systems to precisely control small volumes and support applications such as multiplexed assays and point-of-care diagnostics. Devices like HemaPEN offer features such as quadruplicate sampling, tamper resistance, and end-to-end traceability, ensuring high sample integrity. Their ease of use, compatibility with automation, and ability to support decentralized and scalable workflows are driving rapid adoption among healthcare providers and laboratories seeking efficient and reliable sampling solutions.
Application Insight
Disease diagnostics dominated the market, accounting for a revenue share of 45.68% in 2025, and are expected to grow at the fastest rate over the forecast period. Growth in this segment is driven by the increasing adoption of minimally invasive and painless blood collection methods, which enhance patient compliance, particularly for populations requiring frequent testing, such as pediatric, geriatric, and chronic disease patients. These technologies enable the collection of high-quality, reliable samples that are compatible with automated laboratory workflows, ensuring accurate and consistent diagnostic outcomes. Furthermore, their suitability for remote and at-home testing expands access to diagnostic services, supporting early disease detection and continuous health monitoring. Collectively, these factors are contributing to strong adoption and sustained growth within the segment.
The clinical trials and Decentralized Clinical Trials (DCTs) segment is expected to witness lucrative growth over the forecast period, driven by the ability of microsampling technologies to require smaller blood volumes and reduce reliance on traditional venipuncture. These approaches significantly improve patient compliance, particularly among pediatric, geriatric, and geographically remote populations. By enabling at-home or remote sample collection, they reduce the need for site visits and associated healthcare costs. In addition, microsampling technologies offer high analytical efficiency through compatibility with automated laboratory systems and consistent sample quality, making them increasingly preferred for clinical diagnostics, therapeutic monitoring, and research applications. For instance, the use of dried blood spot sampling in remote pharmacokinetic studies has demonstrated reliable results while reducing patient burden. The rapid expansion of decentralized trials, coupled with growing demand for patient-centric solutions and advancements in bioanalytical technologies, is driving accelerated growth in this segment.
End Use Insight
Hospitals & clinics led the market, accounting for 42.56% of revenue share in 2025.This dominance is primarily driven by the high volume of patient admissions, routine diagnostic procedures, and the ongoing need for therapeutic monitoring within these settings. Hospitals and clinics benefit from well-established infrastructure, access to advanced diagnostic technologies, and the presence of skilled healthcare professionals, positioning them as the primary point of care for both acute and chronic conditions. Additionally, the rising prevalence of chronic diseases has led to increased patient visits, further driving demand for efficient sampling and diagnostic solutions. The integration of microsampling technologies into clinical workflows enhances operational efficiency, reduces turnaround times, and improves patient comfort. Furthermore, strong reimbursement frameworks and higher healthcare spending continue to reinforce the segment’s leading market position.
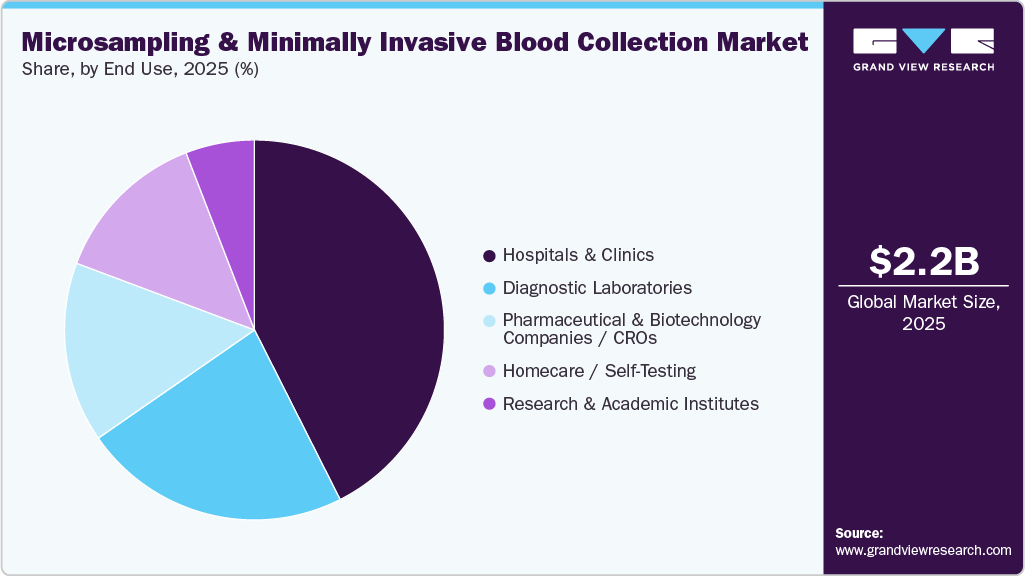
Pharmaceutical & biotechnology companies / CROs segment is anticipated to grow at the fastest rate over the forecasted period. This growth is primarily driven by the increasing number of drug development programs, rising demand for more efficient clinical trial processes, and the expanding adoption of decentralized clinical trial models. These organizations are increasingly integrating microsampling technologies to streamline pharmacokinetic and pharmacodynamic studies. In addition, CROs are leveraging these solutions to enhance trial efficiency, reduce operational costs, and accelerate study timelines. Microsampling also supports improved data quality and enables high-throughput analysis through compatibility with automated laboratory systems. The ongoing shift toward patient-centric trial designs, combined with advancements in bioanalytical techniques and supportive regulatory frameworks, is expected to further drive rapid growth in this segment.
Regional Insights
North America microsampling & minimally invasive blood collection market dominated the global industry and accounted for the largest revenue share 39.77% in 2025. The region represents a highly developed market, supported by advanced healthcare infrastructure, widespread adoption of innovative diagnostic technologies, and a strong presence of leading pharmaceutical and biotechnology companies. Additionally, the rising prevalence of chronic diseases and a well-established clinical research ecosystem are driving demand for efficient and scalable sampling solutions. Favorable regulatory frameworks, along with support from agencies such as the FDA, further facilitate the adoption of novel technologies. Moreover, increasing investments in decentralized clinical trials and digital health solutions, coupled with higher healthcare expenditure, continue to reinforce North America’s leading position in the market.
U.S. Microsampling & Minimally Invasive Blood Collection Market Trends
The microsampling & minimally invasive blood collection market in the U.S. is supported by its highly advanced healthcare system and extensive clinical research landscape. The country benefits from substantial investments in innovative diagnostic technologies and widespread adoption of patient-centric approaches, including decentralized clinical trials. Additionally, the increasing burden of chronic diseases, high healthcare expenditure, and growing preference for minimally invasive procedures continue to drive market growth.
Europe Microsampling & Minimally Invasive Blood Collection Market Trends
The microsampling & minimally invasive blood collection market in Europe is supported by a strong regulatory and research framework led by the European Medicines Agency (EMA), which promotes innovative and patient-centric clinical practices. The region benefits from well-established healthcare systems and increasing adoption of decentralized clinical trials, particularly in countries such as Germany, France, and the UK. Furthermore, the rising aging population and growing prevalence of chronic diseases are driving demand for less invasive diagnostic solutions. Strong government funding for life sciences research and active collaboration between academic institutions and biopharmaceutical companies further support the adoption of advanced microsampling technologies.
Asia Pacific Microsampling & Minimally Invasive Blood Collection Market Trends
The microsampling & minimally invasive blood collection market in Asia Pacific is driven by expanding healthcare infrastructure, increasing patient awareness, and the rising prevalence of chronic and lifestyle-related diseases. The region is witnessing growing adoption of advanced diagnostic technologies and decentralized clinical trial models, which support minimally invasive sampling approaches. In addition, Asia Pacific is emerging as a key manufacturing hub for microsampling and blood collection devices, improving product availability and affordability. Countries such as China, India, and Japan are home to a growing number of manufacturers and contract suppliers focused on cost-effective, high-quality production of innovative sampling solutions.
Latin America Microsampling & Minimally Invasive Blood Collection Market Trends
The microsampling & minimally invasive blood collection market in Latin America is driven by increasing healthcare awareness, rising prevalence of chronic and infectious diseases, and growing demand for patient-friendly diagnostic solutions. Countries such as Brazil, Mexico, and Argentina are witnessing the gradual adoption of advanced sampling technologies, particularly in urban healthcare settings and research institutions. Limited access to centralized laboratories in remote areas further highlights the need for minimally invasive and easy-to-transport sampling methods. Additionally, supportive government initiatives, increasing healthcare investments, and collaborations with global pharmaceutical and biotechnology companies are expected to accelerate market growth.
Middle East and Africa Microsampling & Minimally Invasive Blood Collection Market Trends
The microsampling & minimally invasive blood collection market in the Middle East and Africa represents a smaller but steadily emerging market, driven by ongoing healthcare modernization, increasing awareness of patient-centric diagnostics, and the rising burden of chronic and infectious diseases. Countries such as Saudi Arabia, the UAE, and South Africa are gradually adopting advanced sampling technologies across hospitals, clinics, and research centers to improve diagnostic accessibility and efficiency. Limited access to centralized laboratory infrastructure in remote areas further reinforces the need for minimally invasive and easily transportable sampling solutions. Government-led healthcare initiatives and partnerships with global industry players are expected to support gradual market expansion.
Key Microsampling & Minimally Invasive Blood Collection Company Insights
The microsampling and minimally invasive blood collection market is highly competitive and fragmented, with both established medical device companies and specialized startups competing for market share. Companies are increasingly leveraging mergers, acquisitions, and strategic partnerships to strengthen their capabilities, expand global reach, and enhance technological innovation. These collaborations enable faster product development, facilitate entry into emerging markets, and support the integration of complementary technologies into existing portfolios. Market participants are also focusing on improving analytical precision, minimizing patient discomfort, and offering cost-effective solutions to differentiate themselves. Collectively, these strategies are accelerating market adoption, supporting the growth of at-home testing and decentralized clinical trial models, and positioning innovation and strategic alliances as key drivers of competitive advantage.
Key Microsampling & Minimally Invasive Blood Collection Companies:
The following key companies have been profiled for this study on the microsampling & minimally invasive blood collection market.
- BD
- Abbott
- F. Hoffmann-La Roche Ltd
- Terumo Corporation
- PerkinElmer, Inc.
- Neoteryx
- Tasso Inc
- Seventh Sense Biosystems
- Capitainer AB
- Sarstedt AG & Co. KG
Recent Developments
-
In January 2024, Trajan Scientific and Medical entered a strategic partnership with CliniSciences to distribute its Neoteryx microsampling solutions including hemaPEN and Mitra devices across Europe. This agreement extends Trajan’s global footprint, increasing product availability in key European markets and supporting the shift toward decentralized, patient‑centric sampling.
-
In. June 2, 2023, Mitra and hemaPEN received regulatory certification under Europe’s In- Vitro Diagnostic Medical Devices Regulation and the UK MHRA, enabling decentralized, minimally invasive blood collection for healthcare, clinical trials, and home monitoring. This approval expands Trajan’s Neoteryx portfolio, enhancing patient-centric sampling and strengthening its competitive position in the microsampling market.
-
In April 2023, YourBio Health’s TAP Micro Select secured FDA 510(k) clearance for its virtually painless push‑button blood collection device, enabling high‑quality capillary whole blood sampling and enhancing accessibility for decentralized clinical trials and at‑home diagnostics. This regulatory milestone strengthens the company’s position in the market and supports broader adoption of minimally invasive sampling technologies.
Microsampling & Minimally Invasive Blood Collection Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 2.32 billion
Revenue forecast in 2033
USD 4.01 billion
Growth rate
CAGR of 8.15% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, application, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Denmark; Sweden; China; Japan; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
BD; Abbott; F. Hoffmann-La Roche Ltd; Terumo Corporation; PerkinElmer, Inc ; Neoteryx, Tasso Inc; Seventh Sense Biosystems; Capitainer AB; Sarstedt AG & Co. KG
Customization scope
Free report customization (equivalent up to 8 analyst working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Microsampling & Minimally Invasive Blood Collection Market Report Segmentation
This report forecasts revenue growth at the global, regional & country levels and provides an analysis of the latest industry trends and opportunities in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global microsampling & minimally invasive blood collection market report on the basis of product, application, end-use, and region:
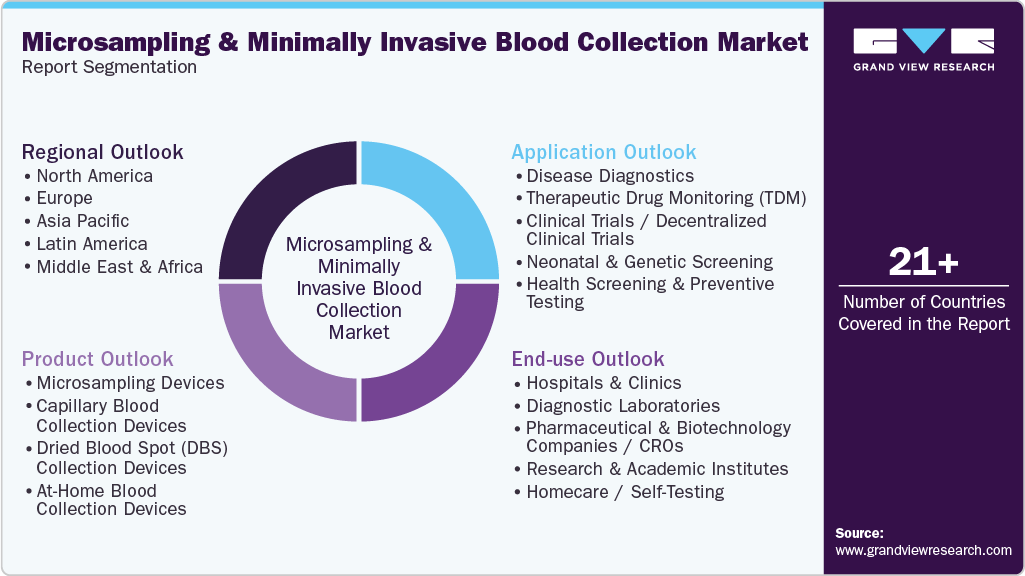
-
Product Outlook (Revenue, USD Billion, 2021 - 2033)
-
Microsampling Devices
-
Volumetric Absorptive Microsampling (VAMS) Devices
-
Push-Button Blood Collection Devices
-
Microfluidic Blood Sampling Devices
-
-
Capillary Blood Collection Devices
-
Lancets
-
Lancing Devices
-
Capillary Tubes / Microcollection Tubes
-
Microcollection Containers
-
-
Dried Blood Spot (DBS) Collection Devices
-
Dried Blood Spot (DBS) Cards
-
Dried Blood Spot (DBS) Collection Kits
-
-
At-Home Blood Collection Devices
-
Self-Collection Blood Sampling Devices
-
Remote Blood Collection Kits
-
-
-
Application Outlook (Revenue, USD Billion, 2021 - 2033)
-
Disease Diagnostics
-
Therapeutic Drug Monitoring (TDM)
-
Clinical Trials / Decentralized Clinical Trials
-
Neonatal & Genetic Screening
-
Health Screening & Preventive Testing
-
-
End-use Outlook (Revenue, USD Billion, 2021 - 2033)
-
Hospitals & Clinics
-
Diagnostic Laboratories
-
Pharmaceutical & Biotechnology Companies / CROs
-
Research & Academic Institutes
-
Homecare / Self-Testing
-
-
Regional Outlook (Revenue, USD Billion, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Norway
-
Denmark
-
Sweden
-
Rest of Europe
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
Rest of Asia Pacific
-
-
Latin America
-
Brazil
-
Argentina
-
Rest of Latin America
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Rest of Middle East & Africa
-
-
Frequently Asked Questions About This Report
b. North America dominated the microsampling & minimally invasive blood collection market with a share of 39.77% in 2025. This is attributable to presence of advanced healthcare infrastructure, widespread adoption of innovative diagnostic technologies, and a strong presence of leading pharmaceutical and biotechnology companies.
b. Some key players operating in the microsampling & minimally invasive blood collection market include BD, Abbott, F. Hoffmann-La Roche Ltd, Terumo Corporation, PerkinElmer, Inc , Neoteryx, Tasso Inc, Seventh Sense Biosystems, Capitainer AB, Sarstedt AG & Co. KG.
b. Key factors that are driving the market growth include increasing global burden of chronic conditions, rising demand for patient-centric diagnostic care, and ongoing technological advancements, supported with the expanding adoption of remote microsampling into decentralized clinical trials (DCTs).
b. The global microsampling and minimally invasive blood collection market size was estimated at USD 2.16 billion in 2025 and is expected to reach USD 2.32 billion in 2026.
b. The global microsampling & minimally invasive blood collection market is expected to grow at a compound annual growth rate of 8.15% from 2026 to 2033 to reach USD 4.01 billion by 2033.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










