- Home
- »
- Clinical Diagnostics
- »
-
Middle East Antinuclear Antibody Test Market Report, 2033GVR Report cover
![Middle East Antinuclear Antibody Test Market Size, Share & Trends Report]()
Middle East Antinuclear Antibody Test Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Systems, Software & Services, Reagents & Assay Kits), By Technique (ELISA, Immunofluorescence Assay), By Application, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-813-2
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Middle East Antinuclear Antibody Test Summary
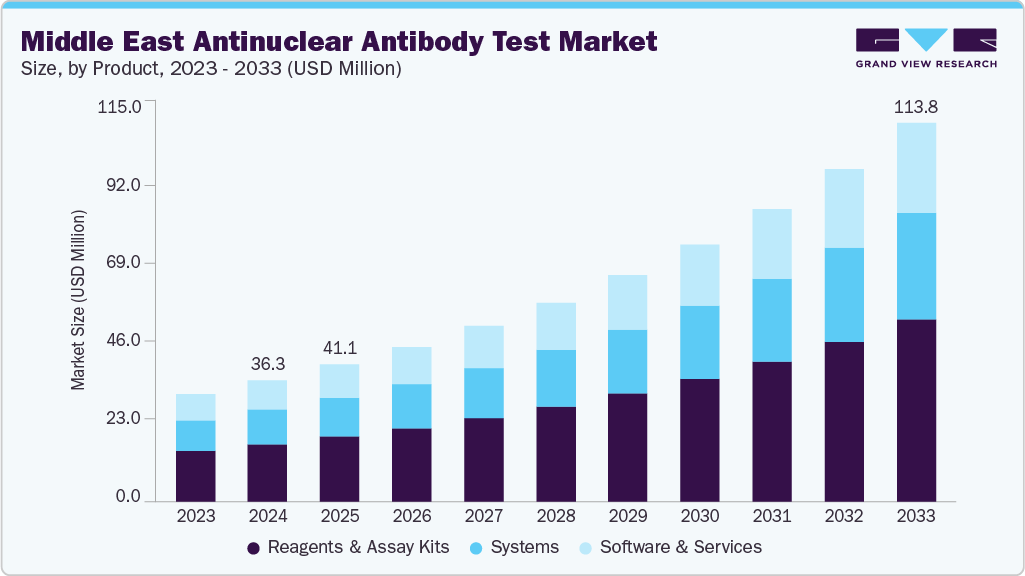
The Middle East antinuclear antibody test market size was valued at USD 36.35 million in 2024 and is projected to reach USD 113.83 million by 2033, growing at a CAGR of 13.6% from 2025 to 2033. The rising prevalence of autoimmune disorders, adoption of advanced testing technologies, increasing healthcare expenditure, and supportive government initiatives are major factors expected to propel market growth. For instance, according to the Saudi Medical Journal, in Saudi Arabia, a 2023 study of 2,258 patients with Type 1 diabetes mellitus (T1DM) found that 67.4% of those had one or more additional autoimmune disorders (thyroiditis, celiac disease, adrenal insufficiency, polyglandular syndromes) across children, adolescents, and adults.
Furthermore, given the growing epidemiologic burden, healthcare infrastructure and government policies in the Gulf and Middle East region are strongly driving diagnostics market growth. For example, Saudi Arabia is undertaking a major expansion and modernization of its healthcare system under Vision 2030, which includes substantial government investment in new hospitals and healthcare centers, as well as the promotion of public-private partnerships (PPPs). The healthcare system spans primary, secondary, and tertiary care, delivered through the Ministry of Health network alongside other government and private facilities. Key initiatives focus on digital health integration, the development of specialty clinics, and increased hospital bed capacity to accommodate population growth and rising life expectancy.
Moreover, the expansion of private diagnostic networks and the growth of specialized laboratories in major urban centers are further accelerating market development across the region. The increasing presence of private-sector players equipped with advanced laboratory infrastructure, automated immunoassay systems, and molecular testing platforms has significantly enhanced accessibility to autoimmune diagnostics. Cities such as Riyadh, Dubai, Doha, and Kuwait City have witnessed the establishment of high-capacity reference laboratories and specialist centers offering comprehensive autoimmune panels, contributing to faster turnaround times and improved clinical decision-making.
In addition, the rise of medical tourism and cross-border diagnostic demand is emerging as a significant growth catalyst. The Middle East, particularly the Gulf states and the United Arab Emirates, has established itself as a regional healthcare hub, attracting patients from Africa, South Asia, and neighboring Arab countries who seek advanced and affordable diagnostic and treatment services. This trend not only drives demand for specialized autoimmune testing but also encourages collaboration between regional healthcare providers and international diagnostic firms, promoting the transfer of technology and the adoption of global testing standards. Furthermore, the geriatric population is more prone to autoimmune disorders because aging is associated with gradual changes in the immune system, a phenomenon known as immunosenescence, so increasing the geriatric population is burgeoning market growth. Below is the tabular representation of growing geriatric populations.
Current and projected percentages in the MENA of old and oldest-old populations
Countries
Percentage of Population 65+
Percentage of Population 80+
Countries
2010
2030
2050
2010
2030
2050
Bahrain
2.1
9.0
25.4
0.3
0.6
4.3
Egypt
5.0
8.7
14.2
0.7
1.4
2.8
Iraq
3.3
3.7
6.8
0.4
0.5
1.0
Jordan
3.9
5.6
12.8
0.5
0.9
2.2
Kuwait
2.5
5.2
16.1
0.3
0.5
2.2
Lebanon
7.3
12.0
21.0
1.1
1.8
4.6
Oman
2.5
8.3
22.5
0.5
0.8
3.6
Palestine
2.7
4.0
7.1
0.3
0.6
1.2
Qatar
1.0
4.3
21.2
0.1
0.3
3.5
Saudi Arabia
3.0
6.4
15.1
0.6
0.9
2.2
Syria
3.9
7.3
12.8
0.6
1.1
2.9
UAE
0.4
6.3
28.0
0.1
0.2
4.2
Yemen
2.6
3.2
5.6
0.3
0.4
0.7
Source: International Journal on Ageing in Developing Countries, 2021, Secondary Research
Furthermore, the market outlook has also been improved by the expansion of clinical guidelines and the greater incorporation of autoimmune screening instruments, such as antinuclear antibody (ANA) testing, in routine monitoring and early detection procedures. To facilitate earlier diagnosis and intervention, doctors and specialists are increasingly recommending ANA and related antibody panels for patients presenting with nonspecific inflammatory or multisystemic symptoms. This growing clinical awareness, supported by national health authorities and medical societies, is expected to increase testing volumes in both hospital and outpatient settings substantially.
The Middle East's autoimmune diagnostics market is expected to continue growing, due to a strong ecosystem being created by these developments, which include increasing private diagnostic capacity, expanding medical tourism, and changing clinical practices.
Market Concentration & Characteristics
The antinuclear antibody testing market in the Middle East is exhibiting a moderate degree of innovation. In line with the global trends, diagnostics companies are progressively implementing increasingly sophisticated technologies, including multiplex platforms, automated immunoassays, and enhanced data interpretation software. For instance, in February 2025, Hipro Biotechnology Co., Ltd. (China) showcased cutting-edge diagnostic innovations at MedLab Middle East 2025, including the Fluorescence Immunoassay analyzer (PalmF). However, infrastructure gaps, unequal laboratory capabilities, and lower overall adoption when compared to leading Western markets somewhat limit the pace and penetration of these technologies. Because of this, even though innovation is essential and developing, it is still at a medium level rather than a high one.
The Middle East's antinuclear antibody testing niche has few mergers and acquisitions, making it a low-rate market. While there is evidence of general diagnostics sector partnerships and some acquisition activity in the larger in-vitro diagnostics (IVD) domain in the region, specific high-profile deals targeting the antinuclear antibody market are not prominent. M&A activity in this sub-segment is limited, slowing consolidation, market entry, and scale-up for specialized ANA diagnostics.
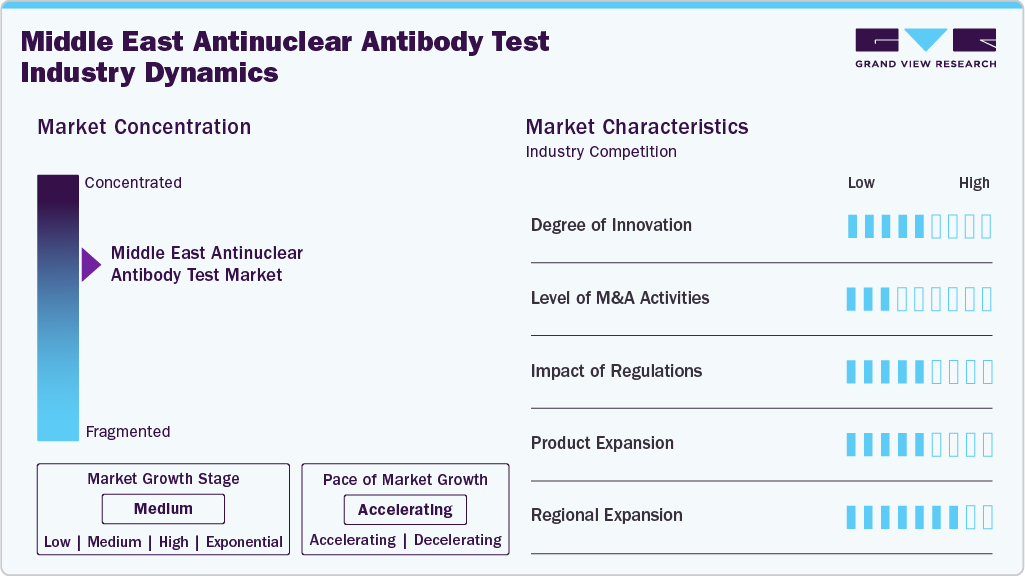
The regulatory environment in the Middle East for diagnostics, including antinuclear antibody testing, has a moderate impact. Saudi Arabia and the UAE are modernizing regulations for in-vitro diagnostics, aligning with international standards and strengthening lab accreditation frameworks. However, challenges such as cross-jurisdictional harmonisation, registration delays, and disparities in reimbursement and laboratory licensing practices persist. These factors influence the speed and ease with which new ANA assay technologies and products can be introduced, so the regulatory impact is rated medium.
In terms of product expansion, the industry is growing steadily, but it is not yet at the top tier. The demand for more comprehensive autoimmune test panels, multiplex assays, and improved reagents is growing. However, in comparison to the most advanced diagnostic markets, the expansion and adoption of novel tests vary by country, and some laboratories may continue to use more traditional manual or lower-throughput ANA methods. As a result, product expansion earns a medium rating.
The regional expansion for antinuclear antibody testing in the Middle East is rated high. Healthcare infrastructure investment in GCC countries and beyond is strong, diagnostic laboratory networks are growing, outreach into underserved markets is increasing, and awareness of autoimmune disease diagnostics in the region is rising. The opportunity for geographic expansion (both within individual countries and across national borders in the Middle East region) is significant, positioning the antinuclear antibody market to scale rapidly. Therefore, regional expansion is given a high rating.
Product Insights
The reagents & assay kits segment led the industry in 2024 with a share of 47.29% due to the growing use of high-throughput, automated diagnostic systems in clinical labs, driving the demand for reagents & kits. The segment's dominance was further aided by the rising demand for standardized and dependable testing solutions as well as the increasing prevalence of autoimmune diseases. Furthermore, ongoing product innovations by major players have improved testing efficiency and accuracy, supporting steady revenue growth. Examples of these innovations include the creation of multiplex assays and sophisticated immunofluorescence kits.
The systems segment held the second-largest market share in 2024, due to developments in laboratory automation and the incorporation of artificial intelligence into diagnostic platforms. Moreover, increasing agreements or partnerships to distribute the product and expand is burgeoning the growth of the market. For instance, in June 2022, Quotient Limited and Theradiag partnered to advance autoimmune disease diagnostics in the UAE. The two companies have collaborated to use Quotient's MosaiQ platform to improve autoimmune diagnostics, including instruments. In accordance with the new contract, Quotient will receive autoimmune reagents and multi-constituent quality controls from Theradiag in order to develop autoimmune microarrays that will be applied to the MosaiQ solution.
Technique Insights
The immunofluorescence assay segment led the market with the largest revenue share of 52.47% in 2024, as it remains the gold standard technique to diagnose pattern-specific or low-tier ANAs that other tests, such as ELISA, can miss. In addition to the specificity of the test, the introduction of automated fluorescent image readers and digital analysis reduces operator dependence and turnaround time, making the immunofluorescence assays more scalable and attractive to large labs.
Meanwhile, the ELISA segment is expected to grow at the fastest rate, due to its high sensitivity, specificity, and compatibility with automated platforms. The technique's ability to process a large number of samples with little manual intervention has made it a popular choice in diagnostic laboratories throughout the Middle East. Furthermore, the growing emphasis on early and accurate detection of autoimmune disorders has accelerated the use of ELISA-based antinuclear antibody testing kits. Also, increasing product launches in Middle East countries is boosting the growth of the market. For instance, in September 2022, Neogen Corporation (US) launched its innovative Veratox VIP assay for the detection of cashew, the second assay in Neogen’s new Veratox VIP line of enhanced quantitative ELISA products in the UAE. Moreover, the new Veratox VIP for Cashew allergen test demonstrates robust performance across sample types while maintaining the simple testing methodology of the Veratox product line.
Application Insights
The rheumatoid arthritis segment led the market with the largest revenue share of 33.67% in 2024, due to the high prevalence of rheumatoid arthritis (RA), the routine inclusion of antinuclear antibody testing in autoimmune‑panels for RA patients, and increasing awareness among physicians of overlapping autoimmune conditions.
Further, the systemic lupus erythematosus segment is expected to grow the fastest, owing to the rising incidence of systemic lupus erythematosus Middle East, more government and research initiatives focused on lupus diagnostics, and the development of more refined ANA‑based and multiplex auto‑antibody tests specifically for SLE. For instance, according to a Pubmed Central study published in 2021 for Egypt, the estimated prevalence of adult SLE is 6.1 per 100,000 population (broken down as approximately 1.2 per 100,000 in males and 11.3 per 100,000 in females).
End Use Insights
The hospitals segment led the market with the largest revenue share of 41.52% in 2024, owing to the high volume of patient visits, strong diagnostic infrastructure, and availability of skilled healthcare professionals. Hospitals in the Middle East are primary centers for autoimmune disease diagnosis and management, with comprehensive testing capabilities that include advanced antinuclear antibody testing methods such as ELISA, immunofluorescence, and multiplex assays. The growing prevalence of autoimmune disorders, rising healthcare costs, and the expansion of hospital networks, particularly in Gulf countries such as Saudi Arabia, the UAE, and Syria, have all contributed to this segment's dominance. For instance, according to the Pubmed Central studies in May 2024, osteoarthritis (12.1%) and rheumatoid arthritis (8.2%) were the subsequent most common conditions, followed by connective tissue disorders (25.6%). Furthermore, the use of automated analyzers and digital workflow solutions in hospital laboratories improves testing accuracy and operational efficiency, strengthening their market position.
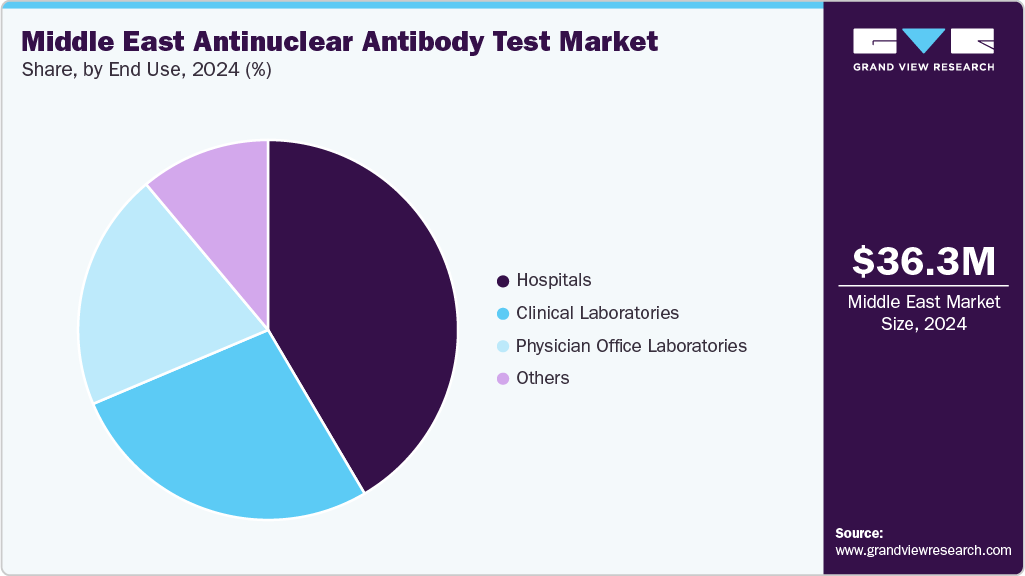
Furthermore, the clinical laboratories segment is expected to expand at a significant CAGR during the forecast period. This expansion can be attributed to the increasing trend of test outsourcing from small hospitals and clinics to specialized diagnostic laboratories outfitted with high-throughput instruments. To meet increasing diagnostic demands, clinical laboratories are focusing more on improving turnaround times, expanding test portfolios, and implementing fully automated antinuclear antibody testing platforms. Furthermore, the increasing penetration of private diagnostic chains, combined with strategic collaborations between international diagnostics providers and regional laboratories, is expected to accelerate segment growth in the Middle East.
Regional Insights
Middle East Antinuclear Antibody Test Market Trends
The market East is expanding owing to the increasing prevalence of autoimmune diseases such as systemic lupus erythematosus, rheumatoid arthritis, and Sjögren’s syndrome. Growing awareness among clinicians and patients regarding early autoimmune disease detection has spurred demand for antinuclear antibody testing across hospitals and diagnostic laboratories. In addition, the region's diagnostic accessibility and accuracy are being improved by the introduction of sophisticated immunoassay technologies and the growth of healthcare infrastructure. Governments in Gulf Cooperation Council (GCC) countries are also investing heavily in laboratory automation, digital healthcare, and clinical research, further boosting market growth. For instance, growing partnerships between international biotechnology companies and regional diagnostic centers are promoting capacity building and technology transfer. Furthermore, test standardization, turnaround time reduction, and the steady growth of the antinuclear antibody testing market throughout the Middle East are being facilitated by the increasing use of multiplex assay platforms and high-throughput ELISA.
Saudi Arabia Antinuclear Antibody Test Market Trends
The Saudi Arabia antinuclear antibody test market is growing due to rapid advancements in clinical diagnostics, expanding hospital infrastructure, and a rising burden of autoimmune disorders. The government’s continuous investment under Vision 2030 to strengthen the healthcare sector has led to increased laboratory modernization and the deployment of automated immunoassay analyzers in both public and private institutions. The use of antinuclear antibody testing for disease screening and monitoring has also increased due to a growing patient base with inflammatory and chronic illnesses. Additionally, the use of standardized testing procedures has increased, and test reliability has improved as a result of Saudi diagnostic laboratories' growing involvement in accreditation programs. Market penetration is also being aided by the rise of regional biotechnology distributors and alliances with international assay producers. The demand for biomarker-based immunoassays, such as ANA tests, is anticipated to rise steadily over the forecast period due to the nation's strategic emphasis on precision medicine and preventive healthcare.
UAE Antinuclear Antibody Test Market Trends
The UAE antinuclear antibody test market is driven by the country’s rapidly evolving healthcare system, characterized by advanced diagnostic infrastructure and high investment in biotechnology innovation. The need for antinuclear antibody testing in hospitals and private labs has increased due to the rising incidence of autoimmune diseases and growing awareness of early disease detection. The broad use of sophisticated immunoassay platforms has also been aided by government programs supporting laboratory automation and the transformation of digital health. Also, the launch of ELISA services in the country is boosting the growth of the market. For instance, the UAE's status as a regional center for medical tourism keeps increasing the number of diagnostic procedures, which helps the immunoassay industry expand. Innovative assay technologies and increased test accuracy are being made possible by strategic partnerships between global diagnostics firms and healthcare providers. Additionally, the UAE's emphasis on personalized medicine and research partnerships with academic institutions and biotech companies is strengthening biomarker discovery initiatives, which will propel the market's long-term growth.
Kuwait Antinuclear Antibody Test Market Trends
Kuwait antinuclear antibody test market is growing due to the rising incidence of autoimmune and chronic inflammatory disorders, coupled with a strong emphasis on improving diagnostic efficiency in public healthcare institutions. Automated immunoassay analyzers and ELISA platforms have become more popular as a result of government efforts to modernize labs and improve clinical diagnostic services. The need for ANA assays has also been fueled by doctors' growing understanding of the value of biomarker-based testing for early disease detection. Furthermore, greater access to high-quality diagnostic testing is being made possible by Kuwait's growing private healthcare industry, which is being bolstered by foreign collaborations and technology imports. Test integration and result tracking are getting better thanks to ongoing investments in healthcare digitization and the adoption of electronic medical records. The use of multiplex assays and high-throughput systems is anticipated to support the overall market growth trajectory in the upcoming years as clinical laboratories improve their testing capabilities and patient throughput.
Oman Antinuclear Antibody Test Market Trends
Oman antinuclear antibody test market is growing, owing to the increasing incidence of autoimmune diseases and the government’s efforts to strengthen healthcare diagnostics. Advanced immunoassay technologies, such as ELISA and multiplex platforms, are becoming more and more important as a result of the country's focus on developing hospital infrastructure and enhancing laboratory capabilities. Modernization of public and private healthcare facilities has been prompted by rising healthcare costs and the Oman Vision 2040 initiatives. Test reliability and throughput have increased due to the expanding supply of qualified laboratory staff and high-quality diagnostic reagents. Additionally, nationwide access to accurate antinuclear antibody testing is being improved through partnerships with foreign diagnostic companies and the introduction of automated testing tools. Over the forecast period, Oman's biomarker-based immunoassay segment is anticipated to grow steadily due to the growing use of preventive health screening programs and increased physician awareness of the importance of early autoimmune disorder detection.
Qatar Antinuclear Antibody Test Market Trends
Qatar antinuclear antibody test market is growing, driven by the rapid advancement of the country’s healthcare infrastructure and a strong focus on medical research and innovation. The need for sophisticated immunoassay-based testing is being driven by the increasing incidence of autoimmune diseases as well as patient awareness of the advantages of early diagnosis. The implementation of automated diagnostic technologies in hospitals and labs has been sped up by the government's ongoing investments in healthcare modernization under the Qatar National Vision 2030. Collaborations between international diagnostics manufacturers and academic research institutions are advancing the localization of immunoassay production and the discovery of techniques. Additionally, the expansion of clinical laboratories and the growing role of private healthcare providers are supporting test accessibility across the nation. The antinuclear antibody testing market in Qatar is anticipated to grow steadily as a result of the integration of digital health platforms, laboratory automation, and sophisticated assay kits, which will further improve testing efficiency.
Key Middle East Antinuclear Antibody Test Companies Insights
Key players operating in the Middle East antinuclear antibody test market are undertaking various initiatives to strengthen their presence and increase the reach of their products and services. Strategies such as expansion activities and partnerships are key in propelling the market growth.
Key Middle East Antinuclear Antibody Test Companies:
- Abbott Laboratories, Inc.
- Bio-Rad Laboratories, Inc.
- ERBA Diagnostics, Inc.
- Trinity Biotech plc
- Thermo Fisher Scientific, Inc.
- Antibodies, Inc.
- Quidel Corporation
- Zeus Scientific, Inc.
- Merck KGaA
- Revvity, Inc.
Recent Developments
- In January 2025, Anbio Biotechnology participated in the 2025 Medlab Middle East exhibition, taking place from 3rd to 6th February at the Dubai World Trade Centre. The event is one of the region's largest and most important medical and laboratory equipment trade exhibitions, bringing together top manufacturers, distributors, and healthcare professionals from all over the world.
Middle East Antinuclear Antibody Test Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 41.10 million
Revenue forecast in 2033
USD 113.83 million
Growth rate
CAGR of 13.6% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, technique, application, end use, region
Regional scope
Middle East
Country scope
Saudi Arabia; UAE; Kuwait; Oman; Qatar; Rest of Middle East
Key companies profiled
Abbott Laboratories, Inc.; Bio-Rad Laboratories, Inc.; ERBA Diagnostics, Inc.; Trinity Biotech plc; Thermo Fisher Scientific, Inc.; Antibodies, Inc.; Quidel Corporation; Zeus Scientific, Inc.; Merck KGaA; Revvity, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts’ working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Middle East Antinuclear Antibody Test Market Report Segmentation
This report forecasts revenue growth at the regional and country levels and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Middle East antinuclear antibody test market report on the basis of product, technique, application, end use, and region.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Systems
-
Software & Services
-
Reagents & Assay Kits
-
-
Technique Outlook (Revenue, USD Million, 2021 - 2033)
-
ELISA
-
Multiplex Assay
-
Immunofluorescence Assay
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Rheumatoid Arthritis
-
Systemic Lupus Erythematosus
-
Sjogren’s Syndrome
-
Scleroderma
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Physician Office Laboratories
-
Clinical Laboratories
-
Other End-uses
-
-
Regional Outlook (Revenue, USD Million, 2021- 2033)
-
Middle East
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
Rest of Middle East
-
-
Frequently Asked Questions About This Report
b. The Middle East antinuclear antibody market size was estimated at USD 36.35 million in 2024 and is expected to reach USD 41.10 million in 2025.
b. The Middle East antinuclear antibody test market is expected to grow at a compound annual growth rate of 13.6% from 2025 to 2033 to reach USD 113.83 million by 2033.
b. The reagents & assay kits segment led the antinuclear antibody market in 2024 with a share of 47.29%. mainly due to the growing use of high-throughput, automated diagnostic systems in clinical labs, driving the demand for reagents & kits
b. Some key players operating in the point-of-care influenza diagnostics market include Abbott Laboratories, Inc., Bio-Rad Laboratories, Inc., ERBA Diagnostics, Inc., Trinity Biotech plc, Thermo Fisher Scientific, Inc., Antibodies, Inc., Quidel Corporation , Zeus Scientific, Inc., Merck KGaA, and Revvity, Inc.
b. Key factors that are driving the market growth include factor such as the rising prevalence of autoimmune disorders, adoption of advanced testing technologies, increasing healthcare expenditure, and supportive government initiatives are major factors expected to propel market growth
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










