- Home
- »
- Clinical Diagnostics
- »
-
Middle East Cancer And Tumor Marker Assays Market Report, 2033GVR Report cover
![Middle East Cancer And Tumor Marker Assays Market Size, Share & Trends Report]()
Middle East Cancer And Tumor Marker Assays Market (2025 - 2033) Size, Share & Trends Analysis Report By Cancer Type (Breast Cancer, Lung Cancer, Colorectal Cancer), By Biomarker Type, By End Use, By Country, And Segment Forecasts
- Report ID: GVR-4-68040-798-5
- Number of Report Pages: 130
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
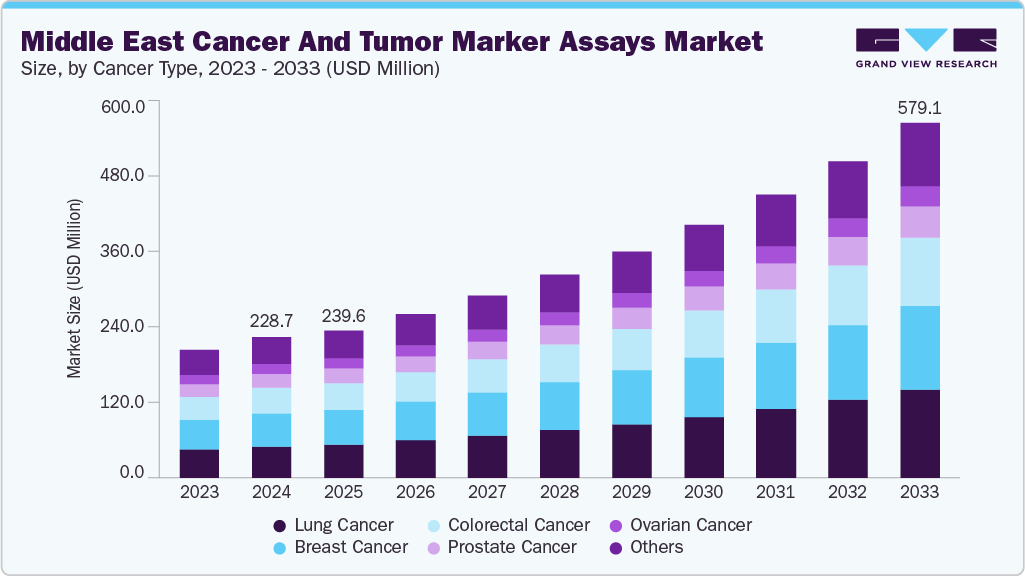
Middle East Cancer And Tumor Marker Assays Market Summary
The Middle East cancer and tumor marker assays market size was estimated at USD 228.74 million in 2024 and is expected to reach USD 579.12 million by 2033, growing at a CAGR of 11.66% from 2025 to 2033. The rising incidence of cancer in the Middle East, growing awareness through campaigns, and advances in diagnostic technologies are contributing to the market's growth. For instance, in June 2025, M42, AstraZeneca & SOPHiA GENETICS together launched a liquid biopsy initiative in the UAE. Furthermore, in a major advancement for precision oncology, M42 (UAE) collaborated with AstraZeneca (UK) and SOPHiA GENETICS (Switzerland) to bring cutting-edge liquid biopsy testing to the UAE, aimed at advancing cancer diagnosis, informing treatment decisions, and enabling faster, accurate biomarker testing across a broad range of cancers.
This strategic collaboration reflects a broader regional trend toward the adoption of precision medicine and molecular diagnostics, which is expected to significantly increase demand for tumor marker assays. Governments across the Middle East are increasing their investments in healthcare infrastructure and national cancer screening programs, enabling earlier detection and improved patient outcomes. Furthermore, as advanced immunoassay platforms become more widely available and private diagnostic laboratories expand, access to high-sensitivity biomarker tests improves. As patient awareness grows and clinicians increasingly rely on biomarker-driven treatment decisions, incorporating tumor marker assays into routine oncology workflows becomes more common, thereby accelerating market growth.
Furthermore, campaigns encourage people to undergo regular screenings and increase demand for advanced diagnostic tools, such as tumor marker assays. As awareness grows, more patients seek timely diagnostic services, prompting healthcare providers to implement biomarker-based testing solutions that enable accurate and early-stage detection and monitoring, driving market growth. For instance, in August 2023, the Qatar Red Crescent Society (QRCS) launched a fundraising campaign to provide life-saving medical care for cancer patients in northern Syria.
Additionally, increasing awareness through public health campaigns is crucial for developing the Middle East cancer and tumor marker assays market. Governments and nonprofit organizations throughout the region are increasingly launching awareness campaigns to educate the public on the importance of early detection, routine screening, and the availability of diagnostic technologies. Moreover, breast cancer awareness campaigns in Saudi Arabia, the UAE's Pink Caravan, and colorectal cancer screening programs in Qatar have all helped to improve public understanding and reduce stigma surrounding cancer testing.
Market Concentration & Characteristics
The Middle East cancer and tumor marker assays market is highly innovative, with strong adoption of next-generation technologies like liquid biopsies, multiplex biomarker panels, and AI-driven analytics. For instance, in February 2023, Sonrai Analytics (UK) partnered with Alliance Care Technologies (UAE) to bring transformational AI solutions for cancer detection and biomarker discovery to the UAE market. Furthermore, the Preferred Partnership agreement signed at the Arab Health Congress will provide UAE researchers and clinicians with access to AI-powered clinical systems to aid in the early detection of cancerous tumors and enhance patient outcomes. In addition, through this collaboration, ACT will have access to Sonrai's proprietary AI technology and expertise, allowing it to bring precision oncology to more oncologists, hospital systems, and other medical providers in the UAE.
The level of mergers and acquisitions (M&A) in the Middle East cancer and tumor marker assays market is considered medium. Global diagnostics firms and regional players are increasingly collaborating through strategic partnerships, joint ventures, and technology licensing agreements, as full-scale acquisitions are becoming less common. Benchmark reports indicate "moderate" M&A activity in biomarker-based immunoassay markets in the Middle East, with companies seeking to expand portfolios and technological capabilities. While such activity promotes access expansion and capability building, the market is not yet characterized by significant consolidation or mega-mergers.
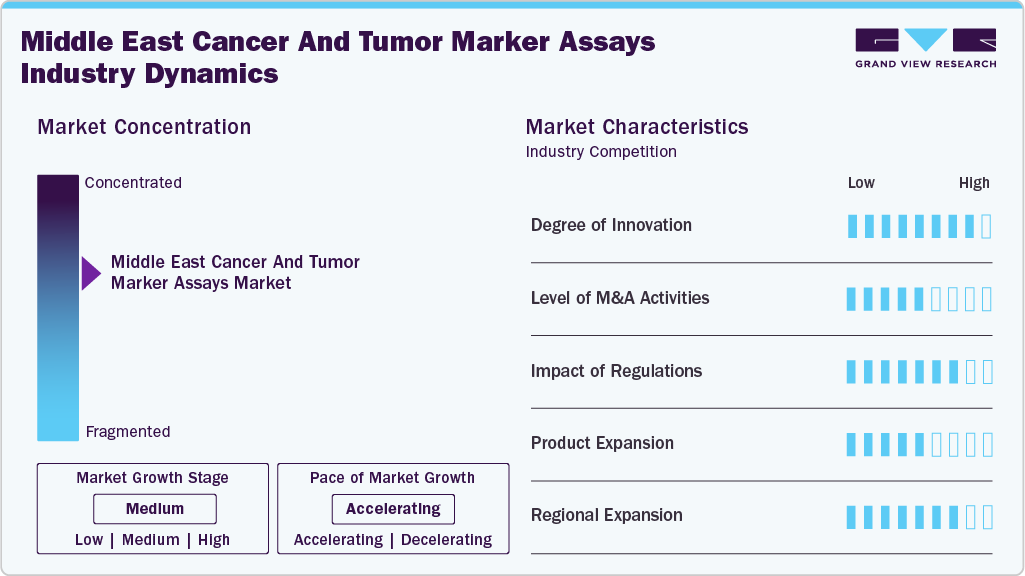
The regulatory impact on the Middle East cancer and tumor marker assays industry is significant. Middle East countries, including the UAE and Saudi Arabia, are tightening approval requirements for in vitro diagnostics to align with global standards, such as ISO accreditation and companion diagnostic frameworks. These regulatory measures cause product delays and limit market access, but they also boost trust and adoption once approvals are obtained. Thus, regulation significantly impacts the speed at which new assays are launched and adopted in the region.
The threat of substitutes for tumor marker assays in the region is medium. On the one hand, conventional diagnostic approaches, such as imaging, standard blood tests, and traditional tumor marker tests, are still widely used due to their availability and cost-effectiveness. On the other hand, the distinct value of advanced marker assays (in terms of sensitivity, earlier detection, and personalization) is gradually driving clinicians away from older substitutes. As a result, while substitution exists, it does not significantly limit the assay market-hence the medium rating.
This market has a high level of end use concentration. In the Middle East, a few large tertiary hospitals, specialized cancer centers, and reference laboratories account for the majority of advanced tumor marker assay volume and high-end technology adoption. Large hospitals in the UAE and Saudi Arabia lead the adoption of molecular and high-complexity diagnostics. The market is dominated by a few key end users rather than many small players, highlighting the importance of targeting major centers for growth.
Cancer Type Insights
Breast cancer led the Middle East cancer and tumor marker assays market with the largest share of 23.52% in 2024. This is mainly due to growing instances of breast cancer in the Middle East, developing awareness campaigns that promote early detection, and a growing acceptance of molecular diagnostics for specific tumor markers. Improvements in assay sensitivity and specificity enable better diagnostic accuracy and patient monitoring, while also facilitating personalized treatment plans. Government efforts and enhancements to healthcare infrastructure have also facilitated a better overall uptake in breast cancer tumor marker assays in the Middle East.
Lung cancer is expected to be the fastest-growing segment over the forecast period, driven by various factors. The region's high incidence of lung cancer necessitates increased testing for biomarkers and tumor markers. According to the UAE National Cancer Registry (UAE-NCR), 231 cases of lung cancer were diagnosed in the UAE in 2021, accounting for 3.9% of all cancer diagnoses. Lung cancer was the second most common cause of cancer death in both sexes, accounting for an estimated 96 (9.85%) of cancer deaths each year.
The Middle East is witnessing an increase in the use of molecular diagnostics and biomarker panels, including EGFR, ALK, and MET ex14, resulting in a higher demand for assays. Improved diagnostic infrastructure, clinician awareness of precision medicine, and government support for oncology diagnostics are accelerating the adoption of tumor marker assays in lung cancer. Finally, the availability of companion diagnostics and the push to integrate tests with targeted therapies make lung cancer assays more commercially viable in the region, resulting in the segment's rapid expansion.
Biomarker Type Insights
The genetic/genomic biomarkers segment dominated the Middle East cancer and tumor marker assays industry, with a revenue share of 66.16% in 2024, driven by the growing adoption of precision oncology workflows and increasing breakthroughs in biomarker detection. These factors, combined with government-led initiatives in several Gulf Cooperation Council (GCC) countries to integrate genomics into cancer care, have accelerated the adoption of genetic biomarkers over more traditional protein- or immunoassay-based markers. For instance, in March 2024, Saudi Arabia achieved a breakthrough in the detection of chronic disease biomarkers. Dana Al-Sulaiman of King Abdullah University of Science and Technology (KAUST) believes an affordable detection method can transform global healthcare. King Abdullah University of Science and Technology is helping patients with chronic diseases by utilizing biosensing platforms to identify biomarkers, specifically cell-free nucleic acids, including microRNAs and free DNA.
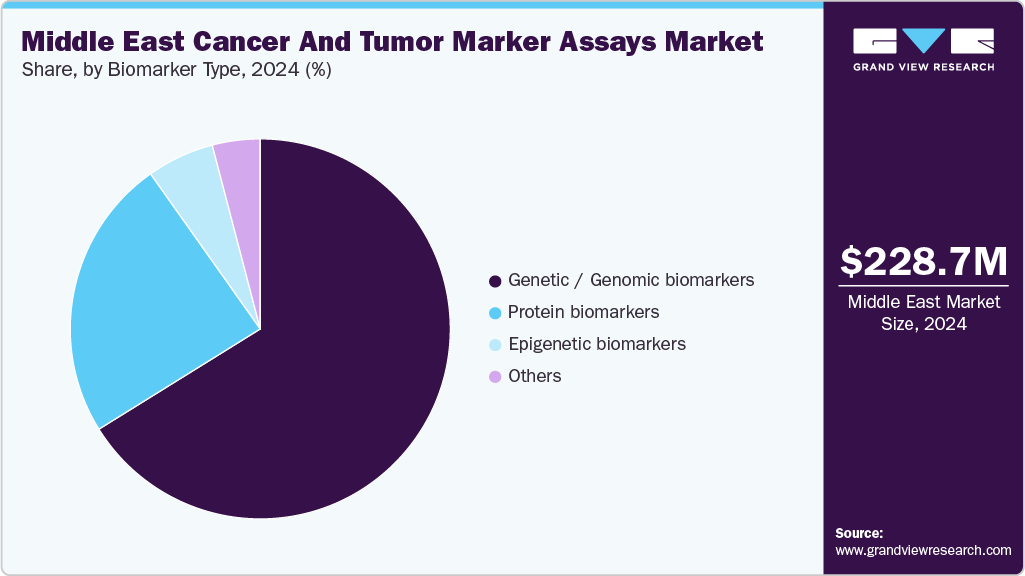
Due to several factors, the epigenetic biomarker segment is expected to be the largest growing segment in the Middle Eastern cancer and tumor marker assays market. The increasing incidence rates in the region, particularly breast cancer and colorectal cancer, have created a need for better and timelier diagnostic tests. Next-generation sequencing and bioinformatics technologies have improved the identification of epigenetic changes, which are valuable for early detection and monitoring.
Additionally, there has been an increase in government spending on healthcare-related infrastructure and cancer research, particularly in Saudi Arabia and the UAE, which promotes the development and use of epigenetic assays. In addition, as the region develops a focus on personalized medicine, the possibility that epigenetic biomarkers can provide information regarding tumor behavior and responses to treatment increases the impetus for their usefulness in tailored treatment. Overall, this can drive rapid growth for epigenetic biomarkers in the Middle East cancer diagnostics market.
End Use Insights
Hospitals and cancer centers held the largest share of the Middle East cancer and tumor marker assays market in 2024, owing to the growth of large tertiary care facilities, integrated oncology services, and government-led facility expansions. For example, the rising prevalence and the growing number of specialized multidisciplinary programs that incorporate biomarker testing into routine oncology workflows are driving up demand in hospitals. These settings typically possess the necessary infrastructure, oncology specialists, and diagnostic volumes to implement advanced assays, enabling continued growth in this segment. Large hospitals and cancer centers continue to upgrade diagnostics platforms and pursue precision medicine strategies, resulting in strong adoption of tumor marker assays and retaining the largest share.
The diagnostic laboratory segment is expected to be the fastest-growing end use channel for tumor markers and cancer assays in the Middle East, driven by the increased outsourcing of high-complexity tests, the growth of specialized reference laboratories, and the expansion of molecular diagnostic networks. Laboratories are capitalizing on rising demand for multiplex genomic panels, liquid biopsy services, and centralized testing across hospitals in the region, resulting in agile growth engines. Furthermore, as hospitals outsource more complex biomarker assays and smaller clinics rely on lab networks for quick turnaround, diagnostic labs are expanding faster and capturing a share of the growing market.
Country Insights
The Middle East cancer and tumor marker assays market is experiencing significant growth. Countries such as the UAE, Kuwait, Saudi Arabia, Qatar, and Oman are seeing an expansion of companies and their investment in biomarker diagnostic capabilities for oncology.
Saudi Arabia Cancer and Tumor Marker Assays Market Trends
The Saudi government's Vision 2030 agenda is driving the growth of the cancer and tumor marker assay market. Additionally, the demand for advanced assay platforms, such as multi-marker panels and genomic-based tumor marker tests, is increasing as large public hospital systems and specialized cancer centers invest in these technologies. The rising incidence of breast, lung, and colorectal cancers is driving earlier detection efforts, and tumor marker assays are becoming more integrated into screening and monitoring workflows. For instance, as per the Organisation for Economic Co-operation and Development 2024 report, co-funded by the Qataran Union, it stated that cancer is the fifth leading cause of mortality in Saudi Arabia, and one in 16 premature (before the age of 75) deaths (6%) will be due to cancer between 2023 and 2050. While adoption in major urban areas is high, laboratories in secondary cities and regional hospitals still have significant opportunities to improve their testing capabilities. Pricing pressure, reimbursement clarity, and clinician awareness of novel assay formats are still evolving, indicating growth with some implementation challenges.
UAE Cancer and Tumor Marker Assays Market Trends
The UAE's role as a hub for healthcare innovation and its proactive deployment of precision medicine initiatives have led to rapid growth in the tumor marker assay market. Private and public hospitals are integrating advanced diagnostic workflows, including tumor marker assays that complement molecular diagnostics and imaging, to provide faster and more personalized treatment decisions. Medical tourism, high-end infrastructure, and favorable regulatory frameworks contribute to the increased adoption of assays. Local lab networks are being improved to reduce reliance on overseas send-outs, resulting in faster turnaround times and better local access. The main challenges are cost-effectiveness for a larger population segment and expanding access beyond major urban centers to more remote areas and smaller clinics.
Kuwait Cancer and Tumor Marker Assays Market Trends
Kuwait is growing steadily in the Middle East cancer and tumor marker assays market as the healthcare system works to increase oncology diagnostic capacity. Growth is driven by rising cancer incidence, increased awareness of biomarkers among physicians and patients, and increased investment in diagnostic laboratory infrastructure. Hospitals and reference labs are expanding their service offerings by incorporating multiplex tumor marker panels, advanced assays, and traditional antigen tests. For instance, in October 2025, the Kuwait Ministry of Health (MoH) launched the Gynecological Oncology Unit at the Kuwait Cancer Control Center (KCCC) in the Al-Sabah Specialized Medical District. This initiative is part of a larger effort to improve specialized healthcare services throughout the country. The unit's primary goal is to improve diagnostic, treatment, and prevention services in gynecological oncology. The Ministry of Health's goal in implementing international medical standards is to improve women's health, reduce medical complications, and improve the quality of life for female patients. The new unit is expected to strengthen Kuwait's position as a regional leader in this medical field.
However, the market still lags behind its Gulf neighbors in terms of penetration of high-complexity testing, and regulatory and reimbursement frameworks are still catching up to enable full-scale up. As the local ecosystem matures, efforts will be made to standardize test quality, improve lab accreditation, and expand the availability of tumor marker assays in both public and private sectors.
Oman Cancer and Tumor Marker Assays Market Trends
The tumor marker assay market in Oman is growing at a slower rate than in other Gulf countries. Government initiatives to modernize healthcare infrastructure and decentralize diagnostic services are opening up new avenues for tumor marker testing, particularly as awareness of oncology and early detection grows. Due to limited local capacity, advanced assays are still sent abroad. This presents an opportunity to expand on-site assay menus and reference laboratory capabilities. Adopting advanced tumor marker testing is expected to accelerate as more public-private partnerships and training programs emerge, despite the cost and logistical challenges of upgrading smaller labs nationwide. Oman is a promising market for tumor marker assays, aiming to improve access to and localization of advanced diagnostic services.
Qatar Cancer and Tumor Marker Assays Market Trends
The cancer and tumor marker assay market in Qatar is characterized by high innovation and focused adoption, driven by national genomics programs, comprehensive cancer plans, and strong institutional research capability. Qatar's cancer centers and laboratories are implementing advanced tumor marker panels and molecular diagnostics to improve early detection, treatment monitoring, and patient stratification. The country's smaller population enables faster piloting and scaling of advanced diagnostics, leading to the earlier adoption of tumor markers in tertiary centers compared to more fragmented markets. The challenge is to expand this capability into clinical practice and provide cost-effective access beyond major centers. Qatar is expected to be a regional leader in precision oncology diagnostics as tumor marker assays are integrated into care pathways.
Key Middle East Cancer and Tumor Marker Assays Company Insights
The major market players are focused on adopting strategic initiatives, such as launches, mergers & acquisitions, and partnerships. Furthermore, several players are focusing on the development of testing services, thereby boosting the demand.
Key Middle East Cancer and Tumor Marker Assays Companies:
- F. Hoffmann-La Roche Ltd
- Thermo Fisher Scientific, Inc.
- Illumina, Inc.
- QIAGEN
- Bio-Rad Laboratories, Inc.
- Agilent Technologies, Inc.
- Sysmex Corporation
- Abbott
- bioMérieux
- Myriad Genetics, Inc.
Recent Developments
-
In May 2023, Thermo Fisher Scientific, Inc. (U.S.) collaborated with Pfizer, Inc. (U.S.) to expand localized access to next-generation sequencing (NGS)-based testing for cancer patients in international markets. The expanded localized access aims to provide faster analysis of associated genes for patients with lung and breast cancer in more than 30 countries, including the Middle East, where advanced genomic testing is limited or unavailable.
-
In June 2024, Myriad Genetics (U.S.) collaborated with GSK to improve access to HRD testing in 9 countries. Moreover, a new sponsored testing program leveraging Myriad's MyChoice HRD Plus and MyChoice CDx Plus tests is now available in Egypt, Saudi Arabia, and the United Arab Emirates. HRD testing is a valuable predictive and prognostic biomarker for patients with ovarian cancer. Myriad's MyChoice Tests identify HRD status in women with ovarian cancer. Clinicians can order this test from their local pathology lab and send samples to Myriad Genetics.
Middle East Cancer and Tumor Marker Assays Market Report Scope
Report Attribute
Details
Market size in 2025
USD 239.61 million
Revenue forecast in 2033
USD 579.12 million
Growth rate
CAGR of 11.66% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Cancer type, biomarker type, end use, country
Country scope
Saudi Arabia; UAE; Kuwait; Oman; Qatar
Key companies profiled
F. Hoffmann-La Roche Ltd; Thermo Fisher Scientific, Inc.; Illumina, Inc.; QIAGEN; Bio-Rad Laboratories, Inc.; Agilent Technologies, Inc.; Sysmex Corporation; Abbott; bioMérieux; Myriad Genetics
Customization scope
Free report customization (equivalent to 8 analyst’s working days) with purchase. Addition or alteration to country & segment scope.
Middle East Cancer And Tumor Marker Assays Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 - 2033. For this report, Grand View Research has segmented the Middle East cancer and tumor marker assays market report based on cancer type, biomarker type, end use, and country:
-
Cancer Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Breast Cancer
-
Lung Cancer
-
Colorectal Cancer
-
Prostate Cancer
-
Ovarian Cancer
-
Others
-
-
Biomarker Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Genetic/Genomic biomarkers
-
Protein biomarkers
-
Epigenetic biomarkers
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals and Cancer Centers
-
Diagnostic Laboratories
-
Others
-
-
Country Outlook (Revenue, USD Million, 2021 - 2033)
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
Rest of Middle East
-
Frequently Asked Questions About This Report
b. The Middle East cancer and tumor marker assay market size was estimated at USD 228.74 million in 2024 and is expected to reach USD 239.61 million in 2025.
b. The Middle East cancer and tumor marker assay market is expected to grow at a compound annual growth rate of 11.66% from 2025 to 2033 to reach USD 579.12 million by 2033.
b. The breast cancer held the largest share of 23.52% in 2024. This is mainly due to growing instances of breast cancer in the Middle East, developing awareness campaigns that promote early detection, and a growing acceptance of molecular diagnostics for specific tumor markers
b. Some key players operating in the Middle East cancer and tumor marker assay market include F. Hoffmann-La Roche Ltd, Thermo Fisher Scientific, Inc., Illumina, Inc., QIAGEN, Bio-Rad Laboratories, Inc., Agilent Technologies, Inc., Sysmex Corporation, Abbott, bioMérieux, Myriad Genetics
b. Key factors that are driving the Middle East cancer and tumor marker assay market are rising cancer incidence in the Middle East, growing awareness through campaigns, and advances in diagnostic technologies.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










