- Home
- »
- Medical Devices
- »
-
Neurovascular Microcatheter Market, Industry Report, 2033GVR Report cover
![Neurovascular Microcatheter Market Size, Share, & Trends Report]()
Neurovascular Microcatheter Market (2026 - 2033) Size, Share, & Trends Analysis Report By Type (Single-Lumen Microcatheters, Dual-Lumen Balloon Microcatheters), By Application (Diagnostic Delivery, Therapeutic Embolization Delivery), By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-888-1
- Number of Report Pages: 200
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Neurovascular Microcatheter Market Summary
The global neurovascular microcatheter market size was valued at USD 703.98 million in 2025 and is projected to reach USD 1,224.73 million by 2033, growing at a CAGR of 7.07% from 2026 to 2033. The industry is driven by the rising prevalence of neurovascular aneurysms, which is increasing demand for minimally invasive procedures such as coiling and flow diversion, where precise catheter navigation is essential.
Key Market Trends & Insights
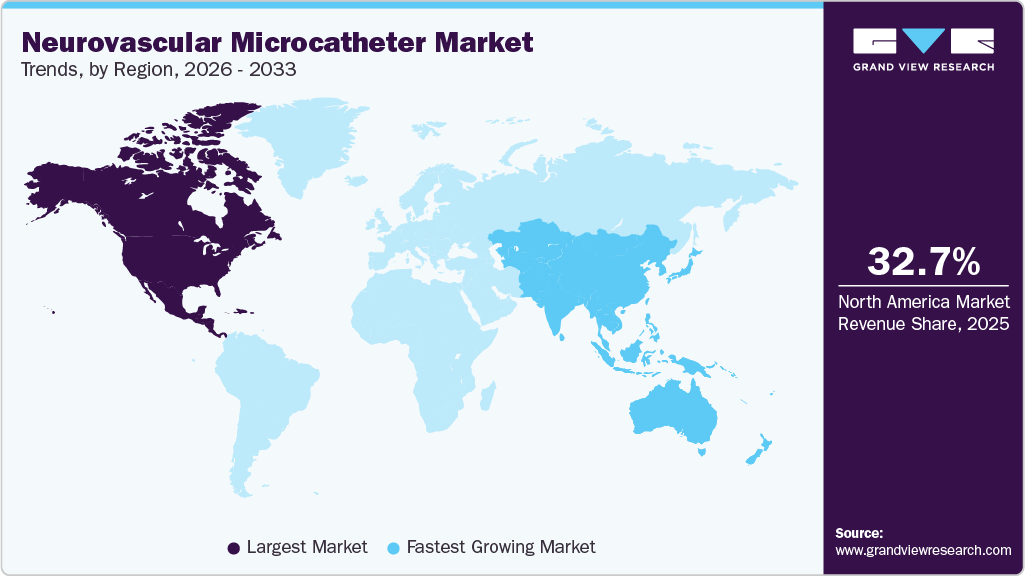
- North America dominated the neurovascular microcatheter market with the largest revenue share of 32.71% in 2025.
- The neurovascular microcatheter market in the U.S. accounted for the largest market revenue share in North America in 2025.
- By type, the single-lumen microcatheters segment led the market with the largest revenue share in 2025.
- By application, the therapeutic embolization delivery segment led the market with the largest revenue share in 2025.
- By end use, the hospitals segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 703.98 Million
- 2033 Projected Market Size: USD 1,224.73 Million
- CAGR (2026-2033): 7.07%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing region
In addition, the growing incidence of ischemic stroke is a major factor, as these devices are widely used in mechanical thrombectomy procedures to restore blood flow quickly and effectively. The expanding global burden of stroke, particularly among aging populations, further supports market growth. Moreover, the rising cases of hypertension, a key risk factor for both aneurysms and strokes, are contributing to a larger patient pool requiring neurovascular interventions, thereby boosting the demand for advanced microcatheter technologies and supporting overall market expansion globally.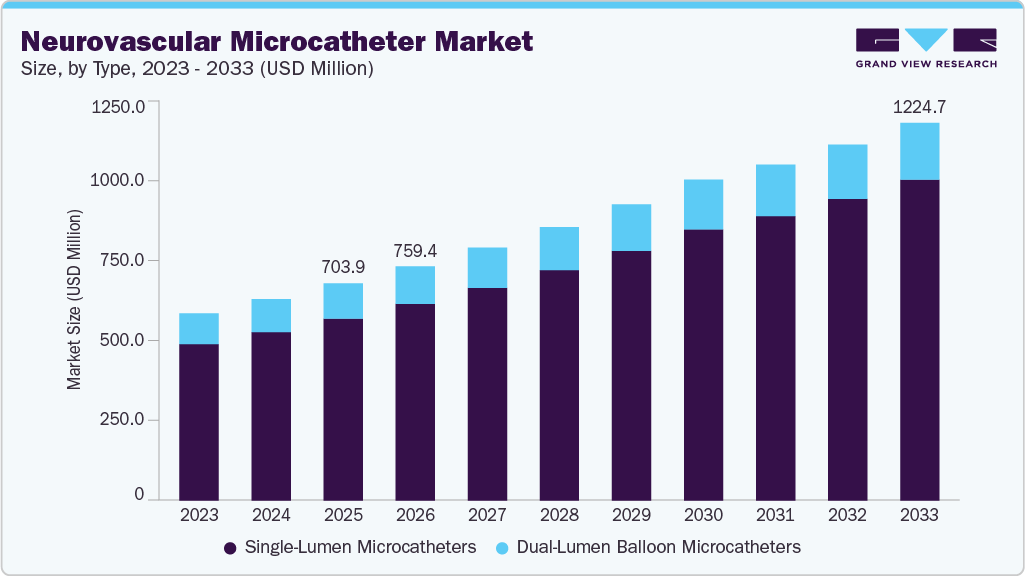
The rising prevalence of neurovascular aneurysms is growing the market for neurovascular microcatheters, as it significantly increases the demand for minimally invasive treatment procedures. Cerebral aneurysms, if left untreated, can rupture and lead to severe complications such as hemorrhagic stroke, prompting early diagnosis and timely intervention. This has led to an increasing number of endovascular procedures, including coil embolization and flow diversion, where microcatheters are essential for precise navigation through complex cerebral vessels. According to the Hereditary Brain Aneurysm Support 2025, unruptured brain aneurysms, localized bulges or swellings in the brain’s blood vessels, are relatively common, affecting about 3% of the UK population, or roughly 2 million people. While most aneurysms remain asymptomatic and do not cause problems, a small number, between 3,000 and 5,000 cases each year, rupture, with approximately half of these resulting in fatal outcomes. In addition, advancements in imaging technologies and increasing awareness among patients and healthcare providers have improved early detection rates. The shift toward minimally invasive techniques, due to reduced recovery time and lower procedural risks, is further accelerating adoption.
"Brain aneurysms typically produce no symptoms but can burst without warning with catastrophic consequences. The reality is that most brain aneurysms will never rupture, but we currently have no way of identifying the minority that will. We are making great strides in understanding clinical risk factors but our ability to understand the genetics behind their development and understand why they rupture remains surprisingly limited for a condition that potentially impacts 3% of the UK population."- Professor Diederik Bulters, Consultant Neurosurgeon at UHS, Honorary Professor of Neurovascular Surgery at the University of Southampton, and Chief Investigator of the ROAR Study.
The rising prevalence of stroke is growing the market for neurovascular microcatheters, as it significantly increases the need for timely and effective treatment interventions. Ischemic strokes, which account for the majority of cases, require procedures such as mechanical thrombectomy to restore blood flow to the brain quickly. Neurovascular microcatheters play a vital role in these procedures by enabling precise navigation through complex cerebral arteries and supporting accurate device delivery. The increasing incidence of stroke, driven by aging populations and lifestyle-related risk factors such as hypertension, diabetes, and obesity, is leading to a higher number of interventional procedures. In addition, growing awareness about early stroke management and improved healthcare infrastructure are further boosting the adoption of neurovascular microcatheters, thereby driving market growth.
Technological advancements are driving the neurovascular microcatheter market by significantly improving the performance, safety, and precision of endovascular procedures. Innovations such as enhanced flexibility, smaller diameters, improved trackability, and hydrophilic coatings enable microcatheters to navigate complex, tortuous cerebral vessels with greater ease and accuracy. In 2025, Researchers at EPFL developed an extremely small, highly flexible neurovascular microcatheter that can navigate complex, branching blood vessels with ease. Uniquely, the device is driven by natural blood flow, allowing it to travel quickly and safely through intricate arterial pathways, improving navigation and reducing the risk during neurovascular procedures.
“Catheters eliminate concerns about device removal after use, and don’t have limited payloads. At the same time, many blood vessels lie beyond a traditional catheter’s reach,” Sakar explains. “That’s why we have developed and tested MagFlow: an ultraminiaturized magnetic microcatheter - twice as small as benchmark microcatheters - that minimizes contact with vessel walls by hitching a ride on the blood stream’s own kinetic energy.” - head of the MicroBioRobotic Systems Laboratory in EPFL’s School of Engineering
The development of dual-lumen, steerable microcatheters further enables better control and targeted delivery of therapeutic devices during procedures such as coil embolization and thrombectomy. In addition, integration with advanced imaging systems and real-time navigation technologies enhances procedural outcomes and reduces risks. These continuous improvements are increasing physician confidence, expanding the scope of minimally invasive treatments, and driving the adoption of neurovascular microcatheters across healthcare settings.
Market Concentration & Characteristics
The neurovascular microcatheter market is growing due to increasing demand for minimally invasive neurovascular procedures and continuous technological advancements. It is characterized by a few well-established global players alongside emerging regional companies, creating a competitive, innovative-focused environment. Leading manufacturers are investing heavily in research and development to introduce advanced microcatheters with improved flexibility, trackability, and safety features. High entry barriers, including stringent regulatory requirements and extensive clinical validation requirements, limit new entrants. Companies are also adopting strategies such as partnerships, product launches, and geographic expansion to strengthen their market presence. In addition, the rising burden of neurovascular disorders is further encouraging innovation and shaping the competitive landscape of the market.
The degree of innovation is significantly impacting the industry by enhancing device performance, safety, and clinical outcomes. Continuous advancements in materials, design, and engineering, such as greater flexibility, smaller diameters, hydrophilic coatings, and improved trackability, enable microcatheters to navigate complex, tortuous cerebral vessels with greater precision. Innovations such as dual-lumen and steerable microcatheters also enable more controlled, targeted delivery of therapeutic devices during procedures. These improvements reduce procedural risks, shorten operation times, and increase success rates, encouraging wider adoption among healthcare professionals. Furthermore, ongoing innovation is expanding the scope of minimally invasive neurovascular treatments, strengthening physician confidence and driving competitive differentiation among manufacturers, thereby accelerating overall market growth.
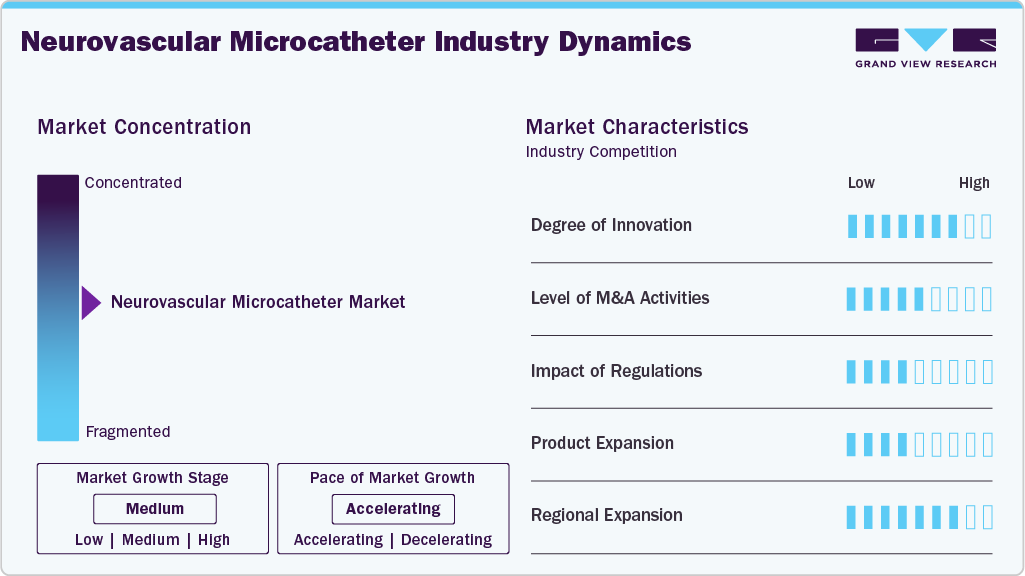
The impact of regulations is notable, as stringent approval processes and compliance requirements directly influence product development, commercialization timelines, and market entry. Regulatory bodies require extensive clinical testing and safety validation to ensure device efficacy, thereby increasing development costs and prolonging time-to-market for new products. While these strict standards can limit the entry of new players, they also ensure high-quality and reliable devices, enhancing patient safety and physician confidence. In addition, evolving regulatory frameworks and guidelines for medical devices push manufacturers to continuously improve product quality and adhere to international standards. Companies that successfully navigate these regulations gain a competitive advantage, while delays or non-compliance can hinder market growth and product adoption.
The level of mergers and acquisitions is influencing the market for neurovascular microcatheter by strengthening the capabilities of key players and accelerating technological advancement. Through acquisitions, companies gain access to innovative product portfolios, proprietary technologies, and specialized expertise, thereby expanding their neurovascular offerings. These strategic moves also help firms expand their geographic presence and strengthen their distribution networks, enabling broader market reach. In addition, mergers and acquisitions support faster product development and commercialization by combining resources and R&D capabilities. This consolidation trend increases competition among leading players while also driving innovation, improving product quality, and ultimately contributing to the overall growth and evolution of the industry.
For instance, in January 2026, Boston Scientific Corporation and Penumbra, Inc. announced that they had entered into a definitive agreement in which Boston Scientific would acquire Penumbra through a combination of cash and stock. The deal valued Penumbra at USD 374 per share, representing an overall enterprise value of approximately USD 14.5 billion.
Type Insights
Single-lumen microcatheters dominated the market in 2025 primarily due to their widespread use in routine neurovascular procedures such as embolization, aneurysm treatment, and targeted drug delivery. These microcatheters are preferred because of their simple design, superior flexibility, and excellent navigability through small and tortuous cerebral vessels, which is critical in neurovascular interventions. In addition, single-lumen devices are more cost-effective than dual-lumen systems and are compatible with a wide range of guidewires and embolic materials, making them the standard choice for most minimally invasive neurovascular procedures. Their established clinical performance, ease of use, and broad application range across diagnostic and therapeutic procedures have resulted in single-lumen microcatheters holding the largest share of the market.
Dual-lumen balloon microcatheters hold a significant share of the neurovascular microcatheter market due to their ability to perform multiple functions simultaneously during complex neurovascular procedures. These devices contain two separate lumens, allowing physicians to inflate a balloon for vessel occlusion while simultaneously delivering embolic agents, coils, or medications through the second lumen. This dual functionality improves procedural efficiency, reduces the need for catheter exchanges, and enhances precision during procedures such as aneurysm coiling, arteriovenous malformation embolization, and stroke interventions. As neurovascular procedures become more complex and minimally invasive techniques continue to advance, the demand for dual-lumen balloon microcatheters has increased, enabling them to capture a significant and growing share of the market.
Application Insights
The therapeutic embolization delivery segment dominated the neurovascular microcatheter market in 2025, due to the increasing number of embolization procedures performed for conditions such as brain aneurysms, arteriovenous malformations, tumors, and hemorrhage control. Therapeutic embolization requires precise delivery of coils, liquid embolic, and other embolic agents directly into targeted blood vessels, making the delivery of microcatheters essential in these procedures. The growing preference for minimally invasive endovascular treatments and the rising prevalence of stroke, vascular malformations, and neurovascular disorders have significantly increased the volume of embolization procedures globally.
The diagnostic delivery segment is expected to grow at the fastest CAGR over the forecast period, due to the increasing demand for advanced imaging, angiography, and real-time intra-procedural diagnostics in neurovascular interventions. Diagnostic microcatheters are widely used for vascular mapping, contrast media delivery, and evaluation of blood vessel structure before therapeutic procedures such as embolization or thrombectomy. The rising adoption of image-guided minimally invasive procedures, growing focus on early diagnosis of neurovascular diseases, and increasing use of combined diagnostic-therapeutic procedures are major factors driving segment growth.
End Use Insights
Hospitals dominated the industry in 2025 due to the high volume of complex neurovascular procedures performed in hospital settings, including aneurysm coiling, embolization, thrombectomy, and arteriovenous malformation treatments. Hospitals are equipped with advanced imaging systems, catheterization laboratories, hybrid operating rooms, and specialized neurointerventional teams, which are essential for performing minimally invasive neurovascular procedures. In addition, hospitals handle emergency cases such as acute ischemic stroke and intracranial hemorrhage, where microcatheters are frequently used for rapid intervention. The availability of reimbursement support, skilled professionals, and advanced infrastructure further contributes to the large number of procedures performed in hospitals, thereby enabling this segment to hold the largest market share.
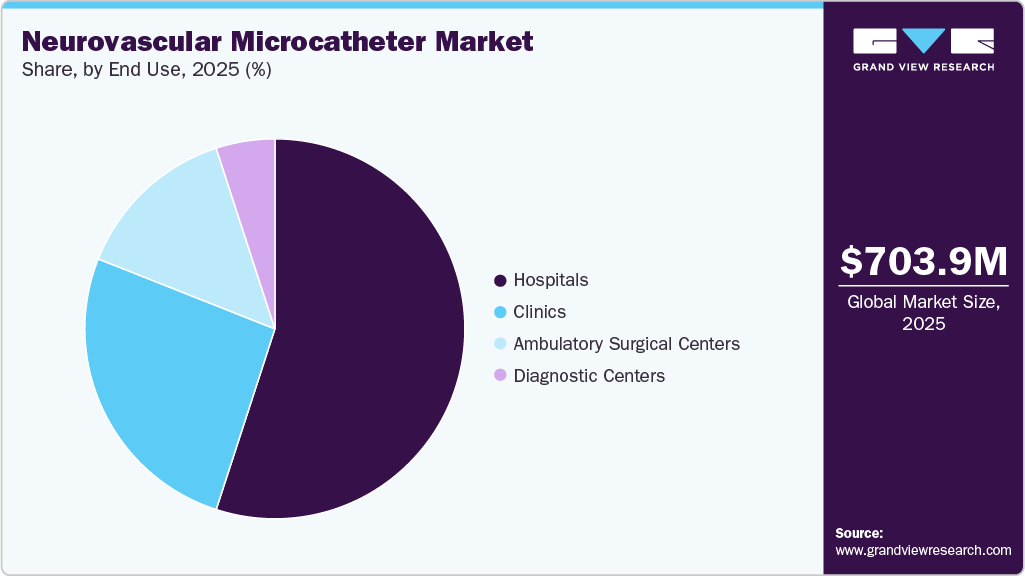
Ambulatory Surgical Centers (ASCs) are expected to grow at the fastest CAGR during the forecast period, driven by the shift toward outpatient and minimally invasive procedures. ASCs offer cost-effective treatment, shorter hospital stays, faster patient turnaround time, and reduced risk of hospital-acquired infections compared to traditional hospital settings. Technological advancements in microcatheters and imaging systems have made many neurovascular and endovascular procedures safer and more suitable for outpatient settings, driving the adoption in ASCs.
Regional Insights
North America region held the largest revenue share of over 32.71% in 2025. The neurovascular microcatheter industry in North America is growing due to its advanced healthcare infrastructure, high adoption of minimally invasive procedures, and strong presence of leading medical device companies. The region has a high prevalence of neurovascular disorders such as stroke and cerebral aneurysms, which is increasing the demand for effective interventional treatments.
In 2025, the Brain Aneurysm Foundation noted that approximately 1 in 50 individuals have an unruptured brain aneurysm, and over 30,000 cases of rupture occur each year in the United States. About half of these ruptures result in death, while nearly two-thirds of those who survive experience lasting neurological impairments. Continuous investments in research and development, along with early access to innovative products, are further supporting market expansion. Moreover, the availability of skilled healthcare professionals and specialized neurointerventional centers is driving the widespread adoption of neurovascular microcatheters across North America.
U.S. Neurovascular Microcatheter Market Trends
The neurovascular microcatheter industry in the U.S. is driving growth due to its highly advanced healthcare system, strong adoption of minimally invasive procedures, and ongoing technological innovation. The country has a high burden of neurovascular disorders, particularly stroke and brain aneurysms, which is increasing the demand for effective endovascular treatments. A large portion of the population is affected by unruptured aneurysms, with a significant number of rupturing cases occurring annually, often leading to severe outcomes. According to established estimates cited by NIH in December 2025, ischemic stroke, comprising nearly 85% of all strokes, remains a leading cause of death and disability worldwide. Annually, about 795,000 individuals in the U.S. experience a new or recurrent stroke. This has created a strong need for precise and reliable devices such as microcatheters. In addition, the presence of leading medical device companies, well-established hospitals, and skilled neurointerventional specialists further supports the widespread adoption and development of neurovascular microcatheters in the U.S.
Europe Neurovascular Microcatheter Market Trends
The neurovascular microcatheter industry in Europe is driving the growth due to its well-established healthcare systems, increasing adoption of minimally invasive procedures, and growing focus on early diagnosis and treatment of neurovascular disorders. The region is witnessing a rising prevalence of conditions such as stroke and cerebral aneurysms, which is increasing the demand for advanced endovascular interventions. According to the National Library of Medicine 2025 study, Europe recorded a notable reduction in ischemic stroke incidence, with Western Europe showing reductions of approximately 44.6% in men and 45.05% in women between 1991 and 2021, compared to Central Europe (24.4% in men and 32.39% in women) and Eastern Europe (26.8% in men and 28.02% in women). Meanwhile, Eastern Europe continues to report the highest incidence rates, highlighting regional disparities. This variation and continued disease burden are driving increased market demand for neurovascular microcatheters, as demand for effective endovascular treatment solutions remains high across Europe.
In addition, strong government support, healthcare funding, and favorable regulatory frameworks are encouraging the adoption of innovative medical devices. The presence of skilled healthcare professionals, specialized neurointerventional centers, and ongoing research further supports the widespread use of neurovascular microcatheters, driving overall market growth across Europe.
The UK neurovascular microcatheter market is growing due to the country’s strong healthcare infrastructure and increasing adoption of minimally invasive neurovascular procedures. A rising burden of conditions such as stroke and brain aneurysms is driving the demand for effective endovascular treatments. The ROAR-DNA Project is a UK-based, multicenter research initiative led by the University Hospital Southampton NHS Foundation Trust and the University of Southampton. Brain aneurysms, which are abnormal bulges in cerebral blood vessels, affect around 3% of the UK population, equivalent to more than 2 million individuals. However, most remain stable and show no symptoms; about 1% rupture unexpectedly, causing subarachnoid hemorrhage, a serious and often life-threatening type of stroke. Each year in the UK, approximately 3,000 to 5,000 aneurysm ruptures occur, with nearly half of these cases resulting in death.
The presence of advanced imaging technologies and specialized stroke care centers supports accurate diagnosis and timely intervention. In addition, ongoing government initiatives, public health programs, and investments in healthcare services are encouraging the adoption of innovative medical devices, further driving the expansion of the market in the UK.
The neurovascular microcatheter market in Germany is witnessing a highly advanced healthcare system, strong clinical infrastructure, and a high volume of neurovascular procedures. The country has a well-established network of specialized stroke centers that treat many patients annually, driving demand for advanced endovascular treatments. In addition, the increasing prevalence of stroke and cerebral aneurysms is further boosting the need for precise and reliable microcatheters.
-
In 2025, the National Library of Medicine reported that approximately 1.4% of the population in Germany in 2022 was living with the long-term consequences of a stroke that had occurred within the previous 10 years (1.3% in women and 1.6% in men). The prevalence increases steadily with age, rising from 1.2% in women and 2.3% in men aged 60-64 to 8.3% and 9.8%, respectively, among those aged 90-94.
Moreover, strong collaboration between research institutions and medical device companies supports continuous innovation and early adoption, positioning Germany as a key contributor to the expansion of the neurovascular microcatheters in Europe.
Asia Pacific Neurovascular Microcatheter Market Trends
The neurovascular microcatheter industry in the Asia Pacific is growing due to the region’s expanding healthcare infrastructure and increasing burden of neurovascular disorders. A rising incidence of stroke and cerebral aneurysms, particularly in densely populated countries such as China and India, is driving the demand for advanced endovascular treatments. In addition, improving access to diagnostic imaging, growing healthcare expenditure, and the expansion of specialized stroke care centers are supporting higher procedural volumes. Government initiatives, increasing awareness about early diagnosis, and rising investments in medical technologies are further encouraging adoption. As a result, the large patient population and ongoing healthcare developments are driving significant growth in the Asia Pacific.
The India neurovascular microcatheter market is growing, driven by increasing demand for advanced neurovascular treatments and expanding healthcare capabilities. The country is experiencing a rising incidence of stroke and cerebral aneurysms, largely driven by hypertension, diabetes, and lifestyle changes, which fuel the need for effective endovascular procedures.
In 2025, according to the National Library of Medicine analysis based on 112 studies, it was concluded that hypertension was highly prevalent in India, affecting approximately 27.2% of the population overall. The burden was even higher in the northern region, where prevalence reached about 33%. This highlighted a significant, regionally variable public health challenge, underscoring the need for targeted prevention, early diagnosis, and effective management to reduce associated cardiovascular and neurovascular risks.
In addition, the growing adoption of minimally invasive techniques, the expansion of multispecialty hospitals, and improved access to advanced imaging technologies are supporting higher procedural volumes. Government initiatives to enhance healthcare accessibility and rising healthcare spending are further encouraging market growth. Furthermore, a large patient pool and improved availability of skilled neurointerventional specialists are contributing to the increasing adoption of neurovascular microcatheters across India.
The neurovascular microcatheter market in Japan is growing due to the country’s rapidly aging population, which is increasing the incidence of neurovascular disorders such as stroke and cerebral aneurysms. With one of the highest proportions of elderly individuals globally, there is a rising demand for effective and minimally invasive treatment options. In 2025, according to the World Economic Forum, Japan's Ministry of Internal Affairs and Communications reported that, as of 2024, the country had a record 36.25 million people aged 65 and over, accounting for 29.3% of the total population. This share is projected to rise to 34.8% in 2040 and 36.3% in 2045. Japan's proportion of elderly residents is already far higher than second-ranked Martinique (25.3%), making it the world's most aged society.
In addition, Japan’s advanced healthcare infrastructure and strong adoption of innovative medical technologies support the widespread use of neurovascular procedures. Moreover, continuous advancements in medical devices and a focus on improving patient outcomes are driving the adoption of neurovascular microcatheters in Japan.
Latin America Neurovascular Microcatheter Market Trends
The neurovascular microcatheter industry in Latin America is growing due to improving healthcare infrastructure and increasing awareness of neurovascular disorders. The region is witnessing a rising incidence of stroke and cerebral aneurysms, which is driving the demand for effective and minimally invasive treatment options. Expanding access to healthcare services, along with the gradual adoption of advanced imaging technologies and endovascular procedures, is supporting higher procedural volumes. In 2025, the National Library of Medicine mentioned that in 2019, approximately 1,975 individuals were identified with a history of stroke, representing a prevalence of 2.1% in Brazil, equivalent to around 4.4 million people living with the condition. A higher proportion of cases was observed among women (52.9%), individuals of brown ethnicity (48.8%), and those residing in urban areas (78.1%). The average age of affected individuals was about 63.8 years, while the mean age at diagnosis was nearly 53.1 years, indicating that stroke often occurs in mid to late adulthood.
Middle East & Africa Neurovascular Microcatheter Market Trends
The neurovascular microcatheter industry in the Middle East & Africa (MEA) region is contributing to the growth through ongoing improvements in healthcare infrastructure and rising investment in specialized medical services. Countries such as Saudi Arabia and the United Arab Emirates are expanding advanced hospital facilities and strengthening their neurology and neurosurgery capabilities, enabling greater adoption of minimally invasive procedures, such as endovascular treatments. In addition, the increasing burden of neurological conditions, including stroke and cerebral aneurysms, is driving demand for precise and flexible microcatheters used in diagnosis and treatment. Across the region, governments are enhancing healthcare systems through long-term development strategies such as UAE Vision 2031, Saudi Vision 2030, and Qatar National Vision 2030, all of which emphasize the adoption of advanced medical technologies and the expansion of specialized healthcare services.
Key Neurovascular Microcatheter Company Insights
The market is highly competitive, with key players holding significant market share. Major companies are pursuing various organic and inorganic strategies, such as new product development, collaborations, acquisitions, mergers, and regional expansion, to address unmet customer needs.
Key Neurovascular Microcatheter Companies:
The following key companies have been profiled for this study on the neurovascular microcatheter market.
- Stryker
- Medtronic
- MicroVention (Terumo)
- Balt
- Johnson & Johnson (Cerenovus)
- Penumbra (Boston Scientific Corporation)
- Cook Medical
- Merit Medical
- Asahi Intecc
- Integer Holdings
- Acandis GmbH (Lauxera Capital Partners)
- phenox GmbH (Wallaby Medical)
Recent Developments
-
In 2025, Penumbra Inc. announced the launch of the Access25 delivery microcatheter, a single-lumen device that was designed to support physicians in navigating the neurovasculature and facilitating the deployment of its 0.020-inch coil platform.
-
In May 2025, Terumo Corporation’s neurovascular division announced the commercial availability of the SOFIA 88 Neurovascular Support Catheter, which expanded its stroke treatment portfolio with a large-bore device. The catheter was engineered to improve reliability, procedural flexibility, and physician control during interventions. The launch also built on the established clinical performance of the SOFIA catheter line, which had supported over 500,000 procedures across more than 170 countries over the past decade.
-
In June 2024, MicroVention Inc. announced that the LVIS EVO Intraluminal Support Device became commercially available in the United States for treating wide-neck intracranial aneurysms. The device had already been introduced in Europe in 2019, with more than 12,000 units distributed. It also represented the first fully visible coil-assist intracranial stent to enter the U.S. market.
Neurovascular Microcatheter Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 759.44 million
Revenue forecast in 2033
USD 1,224.73 million
Growth rate
CAGR of 7.07% from 2026 to 2033
Actual Data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; U.K.; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait.
Key companies profiled
Stryker Corporation; Medtronic plc; MicroVention; Balt; Johnson & Johnson; Penumbra, Inc. (Boston Scientific Corporation); Cook Medical; Merit Medical Systems; Asahi Intecc Co., Ltd.; Integer Holdings Corporation; Acandis GmbH; phenox GmbH
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Neurovascular Microcatheter Market Report Segmentation
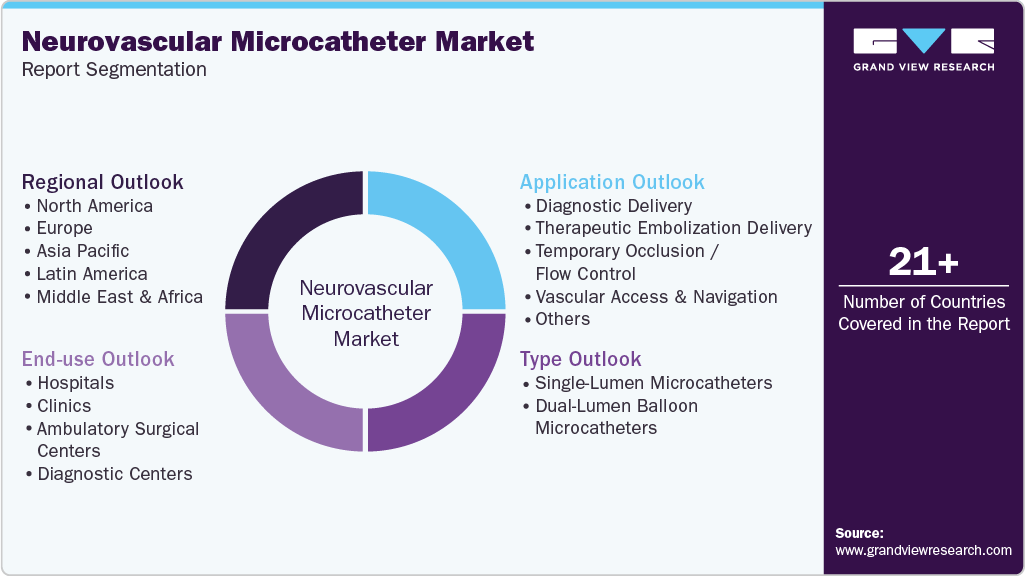
This report forecasts global, regional, and country-level revenue growth and analyzes the latest industry trends and opportunities across sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global neurovascular microcatheter market report on the basis of type, application, end use, and region:
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Single-Lumen Microcatheters
-
Dual-Lumen Balloon Microcatheters
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Diagnostic Delivery
-
Therapeutic Embolization Delivery
-
Temporary Occlusion / Flow Control
-
Vascular Access & Navigation
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Clinics
-
Ambulatory Surgical Centers
-
Diagnostic Centers
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global neurovascular microcatheter market size was estimated at USD 703.98 million in 2025 and is expected to reach USD 759.44 million in 2026.
b. The global neurovascular microcatheter market is expected to grow at a compound annual growth rate of 7.07% from 2026 to 2033, reaching USD 1,224.73 million by 2033.
b. North America dominated the neurovascular microcatheter market with the largest revenue share in 2025.
b. Some key players operating in the neurovascular microcatheter market include Stryker, Medtronic, MicroVention (Terumo), Balt, Johnson & Johnson (Cerenovus), Penumbra (Boston Scientific Corporation), Cook Medical, Merit Medical, Asahi Intecc, Integer Holdings, Acandis GmbH (Lauxera Capital Partners), and phenox GmbH (Wallaby Medical).
b. The neurovascular microcatheter market is driven by the increasing prevalence of neurological disorders such as stroke and aneurysms, the growing adoption of minimally invasive and endovascular procedures, and continuous technological advancements in catheter design that improve safety and performance.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










